Abstract
Background. Glioblastoma multiforme (GBM) is the most aggressive brain tumor malignancy in adults, accounting for nearly 50% of all gliomas. Current medications for GBM frequently lead to drug resistance.
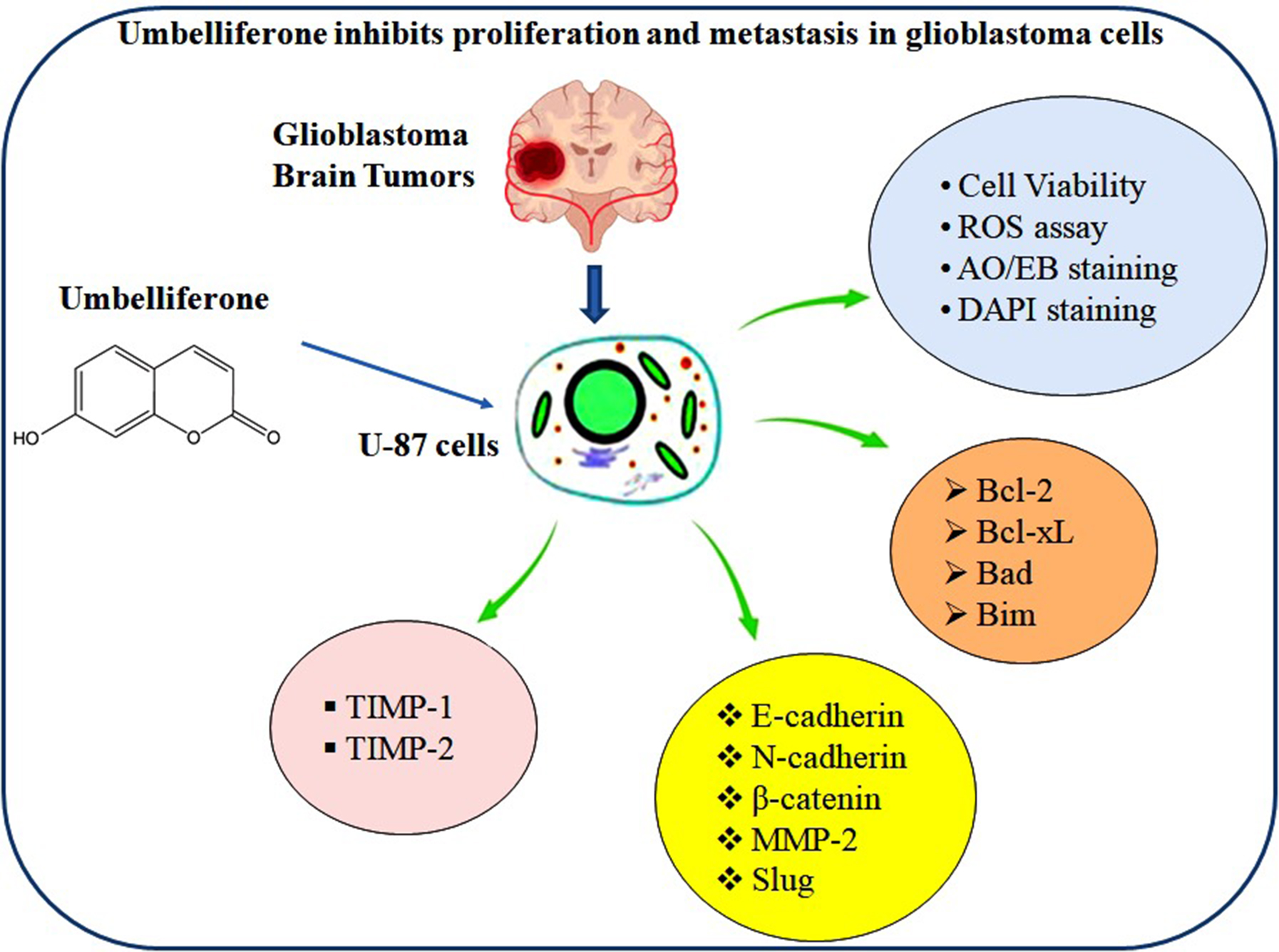
Objectives. Umbelliferone (UMB) is found extensively in many plants and shows numerous pharmacological actions against inflammation, degenerative diseases and cancers. However, its anticancer effects on GBM cells have not yet been explored.
Materials and methods. This research intended to assess the antitumor efficacy of UMB and the molecular mechanism of cell–cell adhesion proteins in human U-87 GBM cells. The cytotoxicity assay, intracellular reactive oxygen species (ROS), cell adhesion proteins, and cell apoptosis actions of UMB were assessed using 3-[4, 5-dimethylthiazol-2-yl]-2, 5-diphenyl tetrazolium bromide (MTT), dichlorodihydrofluorescein diacetate (DCFH-DA), 4′,6-diamidino-2-phenylindole (DAPI), acridine orange/ethidium bromide (AO/EB), and western blot.
Results. The findings revealed that UMB reduced the proliferation of GBM cells and cell adhesion proteins, while augmenting apoptosis through the elevation of cellular ROS. Bcl-2 family protein levels of Bcl-2 and Bcl-XL were mitigated; conversely, the pro-apoptotic proteins Bad and Bim were elevated upon treatment with UMB in a quantity-dependent way. Furthermore, UMB-treated GBM cells suppressed N-cadherin, β-catenin, Slug, and matrix metalloproteinase 2 (MMP-2) expression, whereas they showed enhanced TIMP protein and E-cadherin levels.
Conclusions. Our findings suggest that UMB can prevent proliferation and metastasis and stimulate apoptosis in GBM cells.
Key words: glioblastoma, proliferation, apoptosis, umbelliferone, cell adhesion proteins
Background
Gliomas are the predominant kind of malignancy in the central nervous system and are accountable for a significant number of human deaths.1 Gliomas comprise 24.7% of all primary brain tumors and 74.6% of all malignancies, according to the American Brain Tumor Association (ABTA).2 Glioma patients have an extremely low survival rate; with current treatments, the average survival duration for low-grade gliomas is less than 60 months, and for advanced phases, it is fewer than 15 months.3 As a recurrent brain tumor in adults, glioblastoma multiforme (GBM) is an extremely aggressive malignancy owing to its highly invasive and vascularizing nature.4 Currently, available standard therapy for GBM is surgical resection with subsequent radiotherapy and temozolomide chemotherapy.5 Because of the level of distortion and chemoresistance, GBM certainly relapses despite recent advancements in these treatments.6 Thus, it is urgently required to advance innovative therapeutic approaches for the management and improvement of GBM survival rates.
Reactive metabolic byproducts, such as reactive oxygen species (ROS), greatly influence both harmful and positive actions. Reactive oxygen species in cells act as secondary messengers in signaling cascades that are a threat to usual physiological actions, including development and differentiation.7, 8 Conversely, hypergeneration of ROS can impair biomolecules,9 which leads to cell integrity harm followed by cell pathology. Recently, ROS have been shown to promote tumorigenesis, angiogenesis and metastasis.10 However, extreme accumulation of ROS has been recognized to bring about apoptotic cell death.11 Metastasis is the primary cause of cancer-associated mortality,12 along with catastrophic organ dysfunction following the establishment and uncontrolled progression of exogenous cancer cells surrounded by normal tissue. Adhesion and cell–cell interaction are crucial tools for effective tissue function and homeostasis by describing compartmentalization and polarity in cells.13 Among the numerous adhesion molecules, the key group is the cadherins superfamily, which are transmembrane proteins that are essential in the development of adherens junctions.14 Matrix metalloproteinases (MMPs) are endopeptidases in a zinc-dependent family that are responsible for destroying constituents of the extracellular matrix (ECM),15 tumor cell incursion, angiogenesis, and the subduing of antitumor immune surveillance.16 There are natural endogenous secreted proteins comprising tissue inhibitors of metalloproteinases (TIMPs) that constrain the actions of MMPs.17 Hence, TIMPs control cellular functions, including proliferation, movement and survival, by regulating ECM deprivation through communication with MMPs.18
Natural herbal constituents have been shown to be vital sources of medicines and models for drug design. The well-known 7-hydroxycoumarin umbelliferone (UMB) is extensively found in numerous popular plants from the Umbelliferae family comprising garden angelica, coriander, carrot, etc. It displays numerous pharmacological actions against microbial infections, inflammation, degenerative diseases, and cancers.19 It employs anticancer activities against colon cancer,20 laryngeal cancer21 and liver cancer.22
Objectives
This report was intended to explore the anticancer efficacy of UMB in terms of cytotoxicity, apoptosis and metastasis in the most commonly studied GBM cells.
Materials and methods
Chemicals
Umbelliferone, Dulbecco’s modified Eagle’s medium (DMEM), foetal bovine serum (FBS), antibiotics, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), 2′−7′-dichlorodihydrofluorescein diacetate (DCFH-DA), 6-diamidino-2-phenylindole (DAPI), sodium dodecyl sulphate (SDS), dimethyl sulphoxide (DMSO), and phosphate-buffered saline (PBS) were acquired from Gibco (Waltham, USA). The primary and secondary antibodies for Western blot analysis were purchased from Beyotime Biotechnology (Beijing, China). Analytical-grade biochemicals and solvents were used.
Cell culture
Human U-87 GBM cells were acquired from Shanghai Aiyan Biotechnology Co., Ltd (Shanghai, China) and cultured in DMEM medium, which contained 10% FBS, streptomycin (100 μg/mL) and penicillin (100 U/mL) in a 5% CO2 atmosphere with below 95% humidity at 37°C.
Cell proliferation assay
Human glioma cell viability was evaluated using the MTT test.23 Briefly, U-87 GBM cells were sowed into 96-well plates (1×105 cells/well) and cultured at 37°C in a 5% CO2 wet incubator. Once they were incubated overnight, the medium was separated, washed with PBS in the cells and incubated through different concentrations of UMB (5–60 μM/mL) for 1 day. Then, a 10 μL solution of MTT was supplemented to the treated cells and sustained for another 4 h. Subsequently, crystals of formazan were dissolved by treating with DMSO (150 μL). The optical density (OD) was determined at 490 nm using a multifunctional plate reader (BD Biosciences, Franklin Lakes, USA). Cell proliferation was measured as a percentage of viability against control GBM cells (100%). The IC50 value was determined using the method shown below:
cell viability inhibition (%) = (control OD – test OD) × 100.
Determination of intracellular ROS
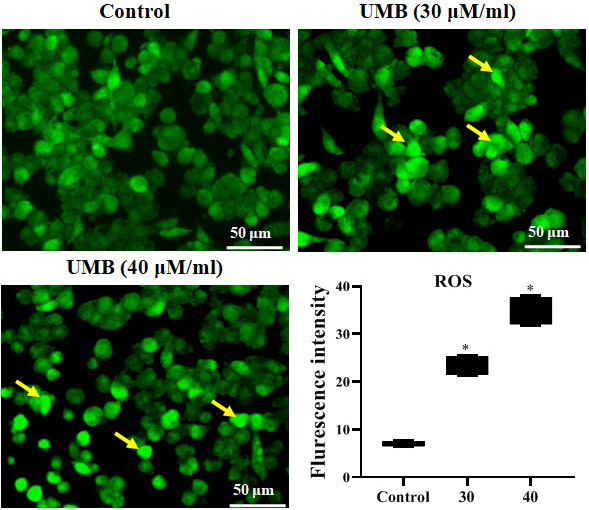
Human glioma cells were sowed in 6-well plates each and kept for 1 day; then, different concentrations of UMB (30 or 40 μM/mL) were added. Next, the control and treated cells were stained with 10 μM of DCFH-DA and preserved for 30 min at 37°C. The stained cells were washed with ice-cold PBS twice to remove any excess dye. The excitation and emission fluorescence were measured using a PR 4100 microplate reader, which is a next-generation 8-channel absorbance microplate reader (Bio-Rad Laboratories India Pvt. Ltd., Gurgaon, India), as described previously.24
Evaluation of apoptosis using dual staining with AO/EB
The apoptotic morphology of the human U87 cells after exposure to UMB was evaluated using acridine orange/ethidium bromide (AO/EB) staining.25 Glioblastoma multiforme cells were supplemented with different concentrations of UMB (30 or 40 µM/mL) and kept for 1 day. The UMB-supplemented and control cells were treated with an AO/EB (100 µg/mL each dye) mixture. All groups were kept in the dark at room temperature for 20 min, ensuring unattached dye was separated using PBS and detected using a fluorescence microscope (model BX51; Olympus Corp., Tokyo, Japan).
Apoptosis assessed using DAPI staining
Human GBM cells were sowed at 1×105 cells per plate and added to UMB (30 or 40 µM/mL) and then fixed with 4% paraformaldehyde for 10 min at 37°C. These preserved GBM cells underwent DAPI staining to evaluate the nucleus changes related to apoptotic cell death using a method described previously.26 Then, all samples were fixed on a glass slip and examined through a BX51 fluorescence microscope (Olympus Corp.).
Western blotting analysis
Human GBM cells were supplemented with 30 or 40 µM/mL of UMB and cultivated for 1 day. The cell lysates were set with lysis buffer in ice-cold conditions, ensuring protease inhibitors, and western blot analysis was performed. Concisely, protein measurement was achieved using a Protein BCA Assay Kit (Pierce Chemical Co., Rockford, USA). After these were quantified, they were electrophoretically dispersed and moved into a polyvinylidene fluoride (PVDF) film. At that point, the film was blocked with a probe preserved at room temperature for 1 h, treated with primary antibodies in a 1:1,000 dilution (Bcl-XL, Bcl-2, Bim, Bad, N-cadherin, E-cadherin, β-catenin, MMP-2, Slug, TIMP-1, and TIMP-2), and set aside overnight at 4°C. Then, horseradish peroxidase (HRP)-conjugated secondary antibodies were added. The protein bands were successively stained and then visualized. The protein band quantification was determined using densitometry with ImageJ software (National Institutes of Health (NIH), Bethesda, USA) and normalized to GAPDH expression.
Statistical analyses
The statistical analysis of the data from each group was conducted using GraphPad Prism v. 8.0.2 (GraphPad Software, San Diego, USA) and IBM SPSS v. 25.0 (IBM Corp., Armonk, USA). Measurement data were presented as medians (min–max). As the sample size was too small to verify normal data distribution, the differences between the groups were analyzed using the nonparametric Kruskal–Wallis test with Dunn’s post hoc test. Subsequently, significant differences among multiple groups were examined using the Kruskal–Wallis test, and Dunn’s post hoc test was employed for multiple comparisons. A statistically significant data divergence was considered when p < 0.05. All tests in this study were bilateral.
Results
Table 1, Table 2, Table 3 show the results of comparing variables among groups.
Antiproliferative and cytotoxic effects of UMB on glioma cells
Human U-87 GBM cell viability was assessed using the MTT test with different concentrations (5–60 μM/mL) of UMB. The data revealed that UMB reduced viability through its cytotoxic and antiproliferative actions on GBM cells in a dose-dependent way. Umbelliferone treatment at a concentration of less than 10 μM did not expressly alter the antiproliferation and cytotoxicity results for GBM cells. Conversely, higher concentrations of UMB (30 and 40 μM/mL) substantially reduced (p < 0.05) the viability of both U-87 cells compared to an untreated control. Using MTT assay, we detected the IC50 values of the GBM cells. Recognized based on the figure of the inhibitory concentration data of 30 and 40 μM/mL, UMB was taken for further trials (Table 1, Figure 1).
Impact of UMB on intracellular ROS accumulation in human GBM cells
The formation of intracellular ROS is related to numerous stimuli, and it can trigger cell death and cell cycle arrest. Intracellular ROS levels were elevated in U-87 cells after being supplemented with 30 or 40 µM/mL UMB in an amount-dependent way, in contrast to the control. To clearly distinguish the accumulation of ROS, the DCFH-DA-labeled cells were examined under a BX51 fluorescence microscope (Olympus Corp.). The intensity of ROS was greatly augmented with 40 µM/mL of UMB (Table 2, Table 3, Figure 2).
Umbelliferone-induced apoptosis of human GBM cells revealed using dual staining with AO/EB
Apoptotic alterations in U-87 cells were visualized using dual staining with AO/EB. The GBM control cells exhibited evenly stained viable green cells (Table 2, Table 3, Figure 3A). The UMB-treated GBM cells presented higher apoptosis variations compared to the controls in a dose-dependent mode. Umbelliferone-treated (30 or 40 μM/mL) U-87 cells revealed apoptotic cells, chromatin condensation and membrane blebbing in what seemed like light greenish-yellow dots. Supplementation with UMB at a dose of 40 μM/mL exposed late apoptotic alterations in GBM cells, which presented as an orange-red color.
Umbelliferone-triggered apoptosis of human GBM cells displayed using DAPI staining
Human U-87 GBM cells stained with DAPI revealed typical viable cells with normal nuclei (Table 2, Table 3, Figure 3B). Treatment of the glioma cells with UMB stimulated apoptosis that intensified the nuclear morphology and fragmentation of the nuclear bodies compared to the control cells. When UMB (30 or 40 μM/mL) was added to the glioma cells, they exhibited chromatin reduction, membrane blebbing, destruction of the nuclear envelope, and cellular collapse. These effects underlined that UMB-triggered apoptosis occurs in a quantity-dependent way.
Studies on the effects of UMB on Bcl-2 family protein expression
In human GBM cells supplemented with UMB (30 or 40 μM/mL), pro-apoptotic Bad and Bim levels were elevated, whereas anti-apoptotic Bcl-2 and Bcl-XL mitigated their protein levels. These results established the apoptotic action of UMB in a quantity-dependent way (Table 2, Table 3, Figure 4).
Influence of UMB on metastatic protein expression
For U-87 human GBM cells supplemented with UMB (30 or 40 μM/mL), E-cadherin protein expression was elevated, whereas N-cadherin, β-catenin, MMP-2, and Slug showed attenuated protein expression. These results confirmed the apoptotic action of UMB in a concentration-dependent way (Table 2, Table 3, Figure 5).
Protein expression analysis of TIMP-1 and TIMP-2
TIMP-1 and TIMP-2 expressions were downregulated in untreated glioma cells. The GBM cells supplemented with 30 or 40 μM/mL UMB upregulated TIMP-1 and TIMP-2 protein expression in a quantity-dependent way (Table 2, Table 3, Figure 6).
Discussion
Malignant carcinomas are a growing problem in the medical community due to delayed diagnosis, elevated metastasis, augmented resistance, and the massive complexity of currently existing medications. The GBM is the most common glioma, with the highest grade of malignancy among brain tumors, the least-favorable prognosis and the greatest likelihood of metastasis and recurrence.1, 3 Temozolomide is the most beneficial medication accepted for GBM treatment, but its drawback is drug resistance, which frequently happens in medical use, eventually causing treatment failure.5 At present, several treatments are available for GBM; however, their effectiveness is partly owing to their broad ramifications. As such, researchers are mainly focused on the development of innovative medications with reduced complications. Over 50% of cancer medicines are produced from natural ingredients.
Umbelliferone, also known as 7-hydroxycoumarin, is an extensive natural component of the coumarin family. It has been documented to have analgesic, anti-inflammatory, antinociceptive, and bronchodilatory actions.27, 28 Umbelliferone has been demonstrated to display anticancer and immunomodulatory actions by constraining tumor progression of sarcoma 180 in mice models.29 Kielbus et al.21 stated that UMB repressed laryngeal cancer cell proliferation and migration. They reported that UMB mitigated the proliferation and migration of laryngeal RK33 malignant cells in a quantity-dependent way.21 Umbelliferone has also been tested for anticancer activities against 7,12-dimethylbenz(a)anthracene-prompted mammary carcinomas in rats.30 To the best of our knowledge, the current study is the first report to exhibit the cytotoxic, apoptotic and antimetastatic prompting actions of UMB in human U-87 GBM cells in vitro. The MTT assay showed that treatment with UMB mitigated cell proliferation in a dose-dependent way in glioma cells, which indicates that UMB might be an active antitumor candidate, as shown in this research.
It has been stated that stable levels of ROS are vital for preserving typical physiological roles.7, 8 The generation of intracellular ROS accompanies oxidative stress, which ultimately leads to the process of apoptosis. Additional ROS production in a biological system is harmful to proteins and DNA, which may eventually cause cell death. When there are upsurges in ROS generation, morphological variations occur and cause late apoptotic modifications, as evidenced by the dual staining of AO/EB in the current research. Next, to confirm that UMB triggered apoptosis, DAPI staining was performed. The fluorescent dye DAPI attaches to the A-T-rich DNA section. U-87-stained cells were examined under a fluorescent microscope, and the images presented fragmented DNA after treatment with UMB. Our findings revealed that elevated ROS generation occurred following UMB administration at different concentrations (30 or 40 μM/mL), in contrast to the control GBM cells, in a quantity-dependent manner. This finding also confirmed the ROS-stimulating action of UMB on U-87 cells.
To clarify the outcomes, we assessed the level of apoptotic protein expression following supplementation of glioma cells with UMB. The proportion of pro-apoptotic and anti-apoptotic proteins is vital during cell destiny determination. Our findings revealed that UMB attenuated the level of anti-apoptotic (Bcl-XL and Bcl-2) proteins and elevated the level of pro-apoptotic (Bad and Bim) proteins in a dose-dependent way. Anti-apoptotic and pro-apoptotic proteins also offer the creation of discrete channels for the permeabilization of the mitochondrial membrane.31 Finally, we showed that the mediator molecules for cell death by UMB are Bim and Bad, which are involved in the crucial mechanisms of cell apoptosis in the mitochondrial pathway.
The current study revealed that UMB-treated glioma cells enhanced E-cadherin and TIMPs while mitigating the protein levels of N-cadherin, β-catenin, MMP-2, and Slug, compared to untreated GBM control cells. Numerous researchers have assessed the arrays of classical cadherin expression in GBM. The expression of N-cadherin in GBM has been examined in different patient cohorts, with nearly 60–80% of cases.32 E-cadherin levels have been further showed as rare or lacking in gliomas.33 Slug protein is a well-considered transcriptional repressor, which can directly attach to and curb the E-cadherin promoter and other cell–cell adhesion within epithelial cells.34 The cytoplasmic domain of cadherin also contributes to cell–cell adhesion by alleviating the cadherin/catenin complex and binding this complex to the actin cytoskeleton at the membrane. It controls an extensive variety of typical physiological developments, comprising embryo enlargement, cell propagation, differentiation, gene expression, and apoptosis.35 Preceding reports have established that the TIMP gene family members are differentially expressed in numerous tumors.36, 37 As MMPs are greatly associated with the prognosis of multiple cancers, TIMPs, at least theoretically, may affect diagnosis in a few malignancy cases as precise inhibitors. In brain tumors, including GBM, several reports have explored the disparity in the expression of TIMPs.38 In the current study, UMB enhanced the protein expression levels of TIMP-1 and TIMP-2 in GBM cells in a dosage-dependent way.
Limitations
As a limitation, microRNA interference level molecular studies were not performed.
Conclusions
This report showed that UMB subdues the proliferation of human U-87 GBM cells through the stimulation of ROS-mediated apoptosis and its cytotoxic effects. Additionally, it could also suppress metastasis of the GBM cells by modulating the cadherin/β-catenin complex-mediated cell–cell adhesion in human glioblastoma cells. These outcomes suggest that UMB may prove to be a key component of antitumor agents or antitumor lead molecules. However, further research endeavors involving the precise molecular mechanisms of its antimetastatic effects are urgently required.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.12804976. The package includes the following files:
Supplementary Fig. 1. Results of Kruskal–Wallis test as presented in Figure 1.
Supplementary Fig. 2. Results of Kruskal–Wallis test as presented in Figure 2.
Supplementary Fig. 3. Results of Kruskal–Wallis test as presented in Figure 3.
Supplementary Fig. 4. Results of Kruskal–Wallis test as presented in Figure 4.
Supplementary Fig. 5. Results of Kruskal–Wallis test as presented in Figure 5.
Supplementary Fig. 6. Results of Kruskal–Wallis test as presented in Figure 6.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.

















.jpg)
.jpg)
.jpg)
.jpg)
.jpg)