Abstract
Background. Hepatitis B virus (HBV) infection is one of the most common infections, affecting 248 million people worldwide. Hepatitis B virus can progress to cirrhosis, liver failure and hepatocellular carcinoma (HCC).
Objectives. To analyze the clinical characteristics and survival time of HCC occurrence in patients with HBV infection after virus turning negative.
Materials and methods. The Kaplan–Meier and log rank survival analysis were performed to compare the overall survival (OS) of the patients with HCC in different groups.
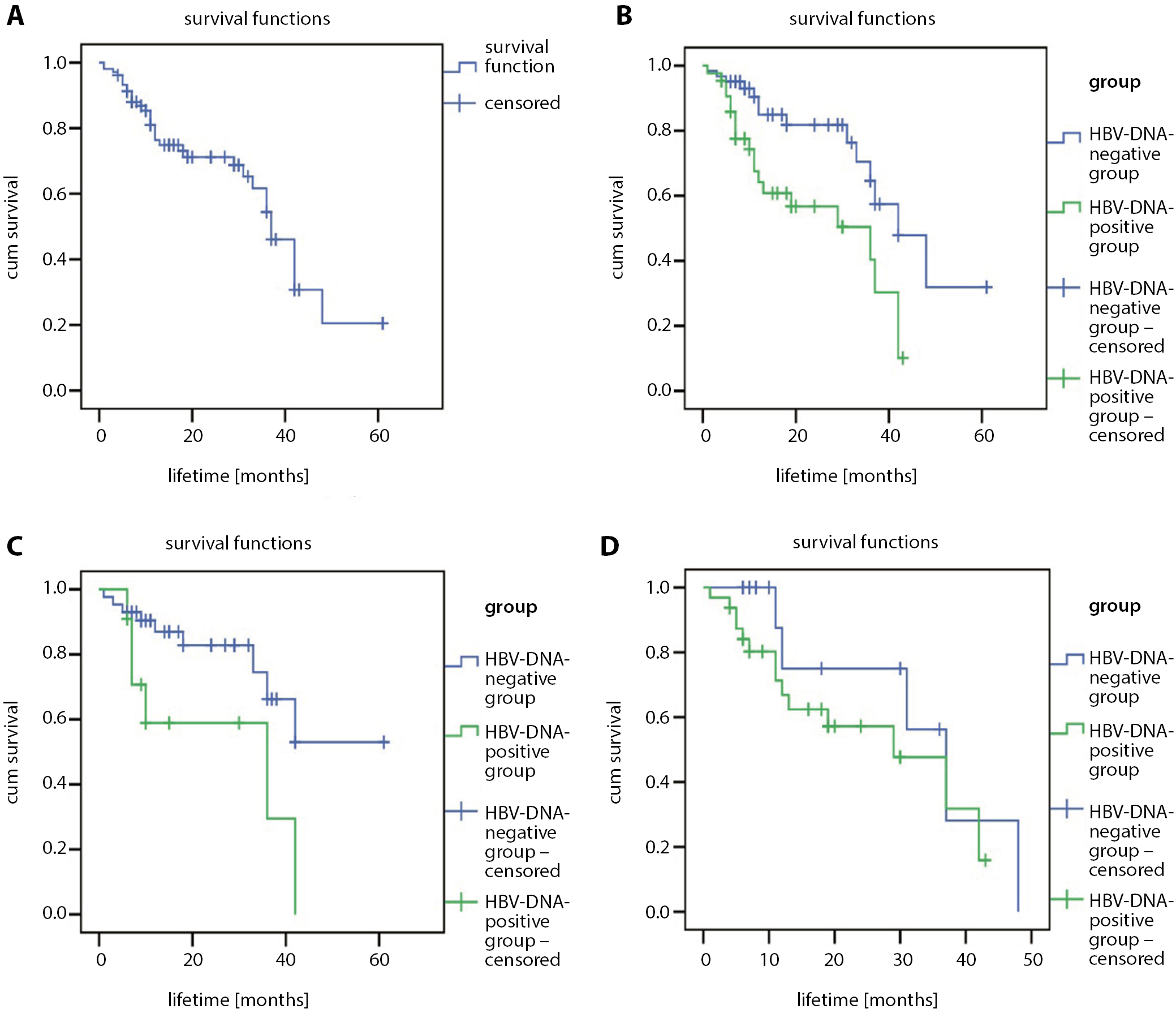
Results. The 1-, 3- and 5-year OS rates of the 104 investigated patients were 76.4%, 54.4% and 20.5%, respectively. The median survival time was 37 months. The median survival time of HBV-DNA-negative group was longer than that of the HBV-DNA-positive group (negative compared to positive: 42 compared to 36, p = 0.003). The 5-year OS rate of patients receiving antiviral therapy before HCC diagnosis in the HBV-DNA-negative group was higher than that in the HBV-DNA-positive group (negative compared to positive: 53.0% compared to 0%, p = 0.022). There was no significant difference in the 5-year OS rate in patients who did not receive antiviral therapy before HCC diagnosis between HBV-DNA-negative and HBV-DNA-positive groups (p = 0.195).
Conclusions. Among HBV-infected patients, a significant proportion of virus-negative patients develop liver cancer and require long-term continuous monitoring. A long-term effective antiviral therapy can improve the survival rate of patients with liver cancer. This study revealed important clinical characteristics of HCC patients and provided useful information for their clinical management and monitoring.
Key words: HBV-DNA, hepatocellular carcinoma (HCC), hepatitis B virus (HBV), survival rate
Background
Hepatitis B virus (HBV) infection is one of the most common infections, affecting 248 million people worldwide.1 Hepatitis B virus causes chronic infection in patients and can lead to cirrhosis, hepatocellular carcinoma (HCC) and other serious liver diseases.2 Primary liver cancer is the 5th most frequent cancer and the 2nd main reason of cancer death worldwide.3, 4 The main pathological types of primary liver cancer include HCC, intrahepatic cholangiocarcinoma (ICC) and hepatocellular carcinoma combined with cholangiocarcinoma (CHCC-CC).5 Hepatocellular carcinoma accounts for more than 90% of primary liver cancer cases, and in China, more than 90% of patients suffering from primary liver cancer die from HCC each year, which is associated with a rising rate of annual HBV infection.6, 7 In addition, genetic conditions leading to chronic liver disease, such as hemochromatosis and alpha-1 antitrypsin deficiency, also increase the risk of HCC.8 Hepatocellular carcinoma occurs mainly in the context of cirrhosis, hepatitis B or C virus infection or nonalcoholic steatohepatitis.9 Hepatocellular carcinoma is the most rapidly rising cause of cancer-related deaths in developed countries and is anticipated to increase further.9, 10 Chronic HBV or hepatitis C virus (HCV) infection are the most common causes of HCC,9, 10, 11 accounting for 78.5% of HCC-related deaths worldwide in 2013.3 It is a global public health threat that causes considerable liver-related morbidity and mortality.12
So far, serum HBV-DNA level has become a major marker for monitoring virus replication and evaluating the efficacy of antiviral therapy in patients with chronic HBV infection.13 In clinical practice, HBV replication can be assessed by viral DNA load.14 A study has proven that the excessive degree of HBV-DNA is an independent threat component for the occurrence of HCC.15 Another study showed that when HBV-DNA stages were less than 2000 IU/mL, the chance of HCC remained, with an annual incidence of 0.06%.16 However, it is still unknown how the clinical features and survival rates of HCC in HBV-infected individuals differ from those in HBV-negative patients. The prognosis of patients with liver cancer has been very poor, with a 5-year survival rate of less than 20%.17 However, there is little data on the survival trends in HCC patients for whom the HBV was tested negative.
Objectives
The aim of this study was to analyze the traits and survival time of HCC occurrence in patients with HBV infection after HBV turning negative.
Materials and methods
Ethical approval
All patients signed informed consent. The study protocol was in line with the ethics guidelines of the Declaration of Helsinki and approved by the local ethics committee of the First Hospital of Shanxi Medical University (approval No. K028).
Patients
All patients with HCC caused by HBV infection diagnosed at the Department of Infectious Diseases of the First Hospital of Shanxi Medical University from June 2014 to June 2017 were included. During diagnosis of HCC, according to the titer of HBV-DNA, the patients were divided into 2 groups: HBV-DNA-negative group and HBV-DNA-positive group.
Inclusion and exclusion criteria
All patients met the following inclusion criteria: HBV infection and diagnosis in accordance to the Chronic Hepatitis B Prevention and Control Guidelines (2015 Edition).18 Hepatocellular carcinoma was diagnosed in accordance to the Guidelines for Diagnosis and Treatment of Primary Liver Cancer in China (2017).19 Exclusion criteria were as follows: patients coinfected with different viruses, including HCV, hepatitis D virus (HDV) or human immunodeficiency virus (HIV), and patients with different liver diseases, including alcohol-related or non-alcohol-related liver disease, autoimmune liver disease, drug-induced liver diseases, and other factors.
Follow-up
Until June 30, 2017, all hospitals followed up the patients using both passive and active approaches, with active follow-up by telephone every 3 months. In addition, hospital staff linked patient records to local population-based cancer registry data, which provided the information on their survival. The survival time of the patients was calculated starting from the date of HCC diagnosis to the last follow-up date, and the survival time of the patients who had already died was calculated from the date of HCC diagnosis to the patient’s death.
Data collection
Patients were directly questioned and the past medical records were collected and thoroughly analyzed for the diagnosis of HCC, including the following: 1) general information: gender, age, history of diseases, personal history, family history, etc.; 2) laboratory examination: HBV-DNA, hepatitis B serologic test, platelet (PLT) count test, serum alanine aminotransferase (ALT), serum albumin (ALB), total bilirubin (TBIL), alpha-fetoprotein (AFP), etc.; 3) other data: use of antiviral drugs, survival rate after HCC diagnosis, etc.
Statistical analyses
Statistical evaluation was carried out with the use of SPSS (v. 26.0; IBM Corp., Armonk, USA) and Stata (v. 12.0; StataCorp LLC, College Station, USA) software. The 1-, 3- and 5-year overall survival (OS) rates and median survival time were analyzed using the Kaplan–Meier method. The differences between the survival curves were analyzed using the log rank test. Two-sided values of p < 0.05 were considered statistically significant.
Results
Baseline data of liver cancer
patients investigated
A total of 104 individuals were recruited in this study, including 61 patients in HBV-DNA-negative group and 43 patients in HBV-DNA-positive group. The age of patients in the HBV-DNA-negative group, which consisted of 48 (78.69%) males and 13 (21.31%) females, was between 50 and 63 years, with a median of 62 years. According to the data, 18 (29.51%) patients were smokers, 8 (13.11%) patients had diabetes and there were 15 (24.59%) patients with HBV family history. Also, 47 (77.05%) patients were diagnosed with HCC complicated with cirrhosis; 43 (70.49%) patients had received antiviral therapy before HCC diagnosis; 35 (57.38%) patients had normal body mass index (BMI) (18.5–23.0 kg/m2). As for the HBV-DNA-positive group, including 31 (72.09%) males and 12 (27.91%) females, the patients ranged in age from 43 to 62 years, with a median of 52 years. In this group, 13 (30.23%) patients were smokers, 4 (9.30%) patients had diabetes and there were 14 (32.56%) individuals with HBV family history. Also, 40 (93.02%) patients were complicated with cirrhosis, 11 (25.58%) patients had received antiviral therapy before HCC diagnosis and 25 (58.14%) individuals had normal BMI (18.5–23.0 kg/m2). Compared with HBV-DNA-positive group, patients in the HBV-DNA-negative group had lower proportion of liver cirrhosis (77.05% compared to 93.02%, p = 0.034) and higher proportion of receiving antiviral therapy (70.49% compared to 25.58%, p < 0.001) (Table 1).
With regard to results obtained from hepatitis B serologic test, the difference in hepatitis B e-antigen (HBeAg)/ hepatitis B e-antibody (HBeAb) between the HBV-DNA-negative group and the HBV-DNA-positive group was statistically significant (p = 0.021). Moreover, the proportion of HBeAg (+)/HBeAb (–) was lower (16.39% compared to 34.88%), and the proportion of HBeAg (+)/HBeAb (–) was higher (31.15% compared to 11.63%) in the HBV-DNA-negative group than that in the HBV-DNA-positive group, while the proportion of HBeAg (+)/HBeAb (–) was almost the same (52.46% compared to 53.49%) in these 2 groups. The other measurements also showed that the proportion of patients with normal ALT level (<40 U/L) in the HBV-DNA-negative group was higher than that in the HBV-DNA-positive group (55.74% compared to 27.91%, p = 0.017). There was no statistically significant distinction between 2 groups regarding TBIL (p = 0.181) and ALB (p = 0.065) levels. In PLT comparison, the proportion of people with normal PLT level (100–300 × 109/L) in the HBV-DNA-negative group was lower than that in the HBV-DNA-positive group (49.18% compared to 60.47%). However, the difference between the 2 groups regarding AFP was not statistically significant (p = 0.128). The details of the analysis are presented in Table 1.
Survival and univariate analysis
At the end of the follow-up period, there were 35 deaths due to liver cancer, 1 patient survived for more than 5 years and the median survival time was 37 months (95% confidence interval (95% CI): [32.13; 41.87]). The 1-, 3- and 5-year survival rates after the HCC diagnosis were 76.4%, 54.4% and 20.5%, respectively (Figure 1A). In the HBV-DNA-negative group, there were 15 liver cancer deaths, median survival time was 42 months (95% CI: [31.19; 52.81]), and 1-, 3-, and 5-year survival rates after the HCC diagnosis were 84.9%, 64.6% and 15.9%, respectively. However, in the HBV-DNA-positive group, the median survival time was 36 months (95% CI: [11.68; 60.32]) and the 1-, 3- and 5-year survival rates after the HCC diagnosis were 64.2%, 40.3% and 0%, respectively. The survival rate of HBV-DNA-negative group was higher than that of HBV-DNA-positive group, and the difference was statistically significant (χ2 = 8.864; p = 0.003; Figure 1B). A further analysis of the patients who acquired antiviral remedy before the HCC diagnosis confirmed that 1-, 3- and 5-year survival rates were 87.0%, 66.3% and 53.0% in the HBV-DNA-negative group and 58.9%, 29.5% and 0% in the HBV-DNA-positive group, respectively. There was a substantial distinction in the survival rate between the 2 groups (χ2 = 5.285; p = 0.022; Figure 1C). For patients with no antiviral therapy before the HCC diagnosis in the HBV-DNA-negative and the HBV-DNA-positive groups, 1-, 3- and 5-year survival rates were 75.0%, 56.3% and 0%, and 66.9%, 31.8% and 0%, respectively. There was no large difference in survival rates (χ2 = 1.679; p = 0.195), but the survival rate of the HBV-DNA-negative group was relatively high (Figure 1D).
Relationship between antiviral therapy and survival time
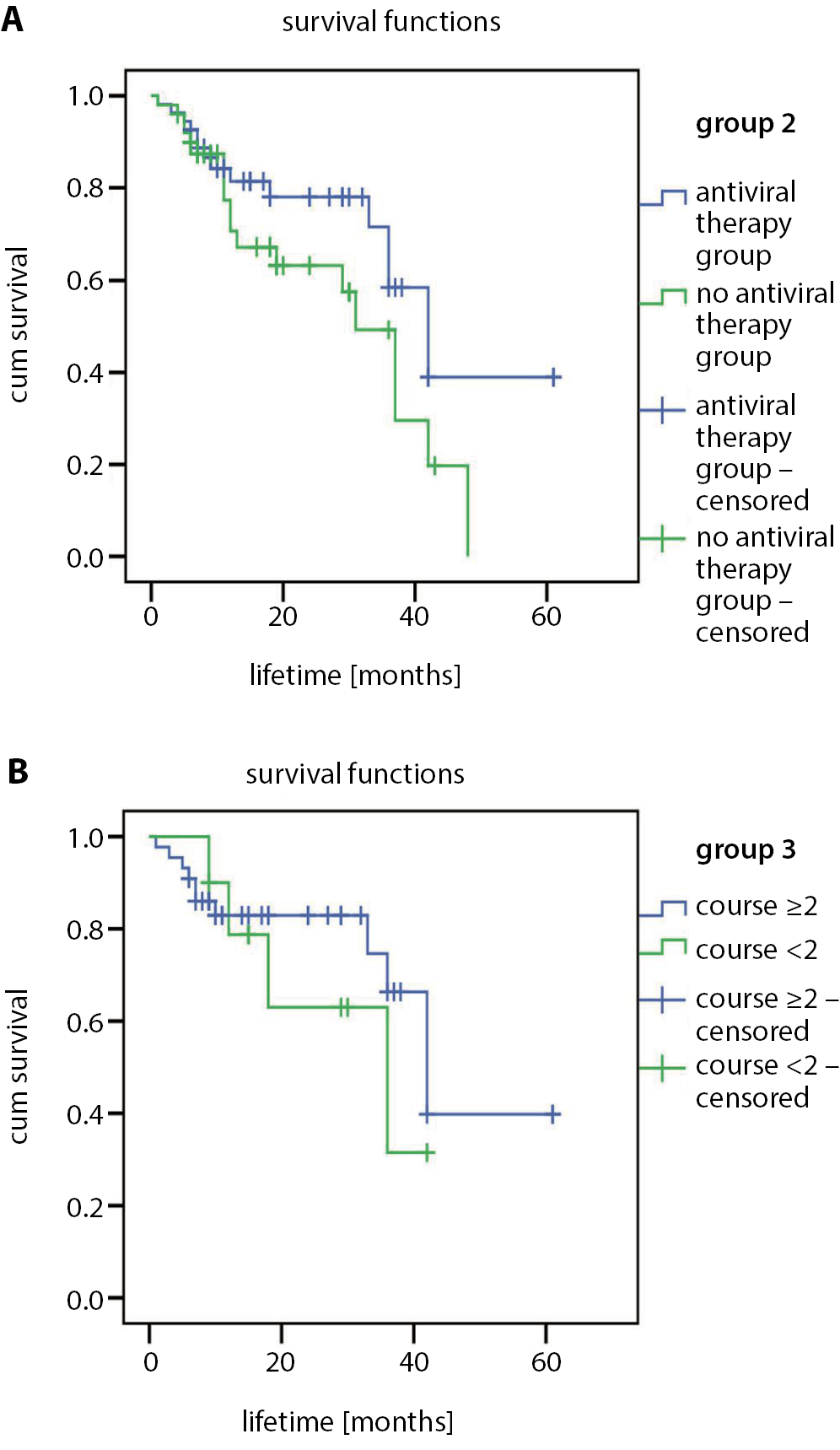
The median survival time of patients receiving antiviral therapy before the HCC diagnosis was 42 (95% CI: [33.93; 50.07]) months, and that of patients without antiviral therapy was 31 (95% CI: [24.74; 37.26]) months. The 1-, 3- and 5-year survival rates for both groups were 81.4%, 58.5% and 19.5%, and 70.6%, 29.6% and 0%, respectively. There was no significant difference between the survival rates of the 2 groups (χ2 = 2.885; p = 0.089) (Figure 2A). In addition, there was no large difference between the survival rate of patients treated with sustained antiviral for more than 2 years and those treated with antiviral therapy for less than 2 years (χ2 = 0.454; p = 0.500) (Figure 2B). However, patients who underwent antiviral treatment for more than 2 years before the diagnosis of HCC experienced a higher 3- and 5-year survival rates than patients without the treatment (66.3% and 39.8% compared to 31.5% and 0%, respectively).
Discussion
In recent years, with the emergence of nucleotides and their analogues, the clinical prognosis of HBV-associated end-stage liver disease has significantly improved. However, some patients still develop HCC after viral negative transformation, suggesting that the inhibition of viral replication and liver inflammation alone cannot eliminate HCC. This study collected and compared the characteristics of HBV-related HCC patients at diagnosis and after turning positive or negative, in order to provide the scientific evidence for the occurrence and clinical regularity of HCC after HBV infection. Up to now, the therapy of HCC has involved mainly surgical treatments, antiviral therapy and a few other methods. Patients with HBV-associated HCC have significantly improved survival rates when the combination therapy model is employed.
In this study, 83.65% of individuals were diagnosed with cirrhosis complicated with HCC, which was consistent with the data from the previous research stating that cirrhosis is a high risk factor for HEB complicated with HCC.20, 21 This study discovered that the percentage of patients with cirrhosis was considerably different between HBV-DNA-positive group and HBV-DNA-negative group, and the percentage of patients with cirrhosis was lower in the HBV-DNA-negative group. Therefore, some HBV-DNA-negative patients with chronic HEB will develop HCC even if it does not progress to liver cirrhosis. In this regard, we hypothesized that some HBV-DNA-negative patients had received a long-term antiviral therapy before progressing to cirrhosis, and that antiviral treatment can improve or reverse liver fibrosis and cirrhosis. A study has shown that entecavir (ETV) could significantly improve or reverse hepatic fibrosis or cirrhosis in HBeAg-negative or HBeAg-positive individuals, and the Ishak fibrosis scores improved by 88%.22 Lok have collected and analyzed the results of nucleotide analogues (NAs) that were observed for more than 10 years.23 They found that 1 year of NAs treatment reduced liver inflammation in 50~70% of chronic hepatitis B (CHB) patients. After 3–5 years of long-term treatment, fibrosis and cirrhosis could be resolved in most patients.
Antiviral remedy is the key to deal with chronic HBV infection. Effective antiviral remedy can lengthen the survival time of patients with liver cancer. Long-term research on lamivudine (LAM) and adefovir (ADV) has proven that antiviral remedy reduces the occurrence of HCC in patients with chronic HBV infection.24 A multicenter study in Taiwan additionally confirmed that 4-year ETV treatment appreciably reduced the danger of liver cancer, cirrhosis events and mortality in patients with hepatic B-associated cirrhosis, with a 60% reduction of the threat of liver cancer as compared to the non-antiviral group.25 A retrospective study in 632 patients with hepatitis-associated liver cancer found that the 2-year survival rate of patients with hepatitis-associated liver cancer before and after antiviral remedy was significantly greater than that of patients without antiviral therapy (χ2 = 33.792; p = 0.000, χ2 = 33.179; p = 0.000).26 Therefore, antiviral remedy before and after HCC diagnosis is beneficial. In our study, 43 HBV-DNA-negative and 11 HBV-DNA-positive patients received effective antiviral therapy. The remaining 32 individuals who did not obtain antiviral cure developed liver cancer. This was constant with the study by Choi et al. stating that not receiving effective antiviral treatment is an independent cause of HCC in patients with hepatic B cirrhosis.27 In addition, there was no significant difference in 1-, 3- and 5-year survival rates between patients who were administered antiviral remedy before diagnosis and those who did not (χ2 = 2.885; p = 0.089). This may be associated with the older age of patients in the antiviral remedy group at the time of HCC diagnosis, different treatment measures taken after HCC diagnosis and the application of antiviral drugs.
Different researchers have different views on the relationship between HBeAg and HCC. In an 8-year prospective follow-up study of 18,154 HBeAg-positive patients in Taiwan, these patients were found to have a relatively high risk of liver cancer, which was 6 to 7 times higher than in the HBeAg-negative patients.28 Zhou et al. determined that there was no significant difference between the incidence of HCC and serum E-antigen positivity in patients with chronic HBV infection.29 Another study reported an even higher risk of HCC in HBeAg-negative patients.30 In our study, HBeAg-negative patients accounted for a large proportion of the whole study population. Compared with the HBV-DNA-positive group, HBeAg+/HBeAb− ratio was lower and HBeAg−/HBeAb− ratio was higher in the HBV-DNA-negative group. We speculated that this was related to a long-term antiviral remedy in the HBV-DNA-negative group. Some patients were negative for HBeAg after treatment, but no serological changes of HBeAg were observed. This does not rule out the possibility that some HBeAg-negative patients with chronic HBV are tested negative right now, but were HBV-DNA-positive before undergoing antiviral treatment, mainly due to mutations in anterior C and BCP regions of the virus strain during the immune clearance period.31 After a long-term antiviral therapy, the expression of the mutated HBeAg was highly reduced and HBV-DNA became negative. Whether it is necessary to further define the HBeAg and HBeAb of patients before antiviral treatment needs to be further examined.
Watanabe et al. found that the PLT level was related to the improvement in patients with HBV-associated HCC, and low PLT level was an independent indicator of the possible occurrence of HCC.32 A study has shown that the peripheral blood PLT count of HBV-associated HCC patients is different and dynamic.33 The peripheral blood PLT count of patients with early HCC was considerably lower than that of patients with advanced HCC. Some scholars believe that HCC can synthesize PLT biotin, which can cause paraneoplastic PLT hyperplasia. The larger the tumor size, the more often the synthesis of bopoietin and the higher the peripheral blood PLT count.34 In this study, HBV-DNA-negative patients had lower PLT levels than HBV-DNA-positive ones. This may be associated with the greater number of HBV-DNA-negative patients receiving antiviral therapy and regular follow-up. Most patients in the HBV-DNA-positive group were diagnosed with advanced HCC, resulting in PLT accompanied by paracancerous hyperplasia.
In our study, the serum ALT level was decreased in the HBV-DNA-negative group, which may be associated with the higher percentage of antiviral treatment in the HBV-DNA-negative group. Antiviral remedy can enhance liver inflammation. Consistent with the results obtained by Furman et al., serum ALT levels were lower in HCC patients with negative HBV-DNA during medium and long-term NAs treatment than in HCC patients with positive HBV-DNA.35
With the global implementation of infant HBV vaccination programs, the incidence of HBV virus infection in birth cohorts has gradually declined. However, due to the long incubation period of HBV infection, an immense percentage of patients with chronic HBV infection develop HCC or even die. In 2009, Nguyen et al. found that the prognosis of HBV-related HCC was very poor, the median survival time was less than 16 months, and the 1-, 3- and 5-year survival rates were 36–67%, 10–56% and 15–26%, respectively.36 In this study, the median survival time of HCC patients was 37 months, and the 1-, 3- and 5-year survival rates were 76.4%, 54.4% and 20.5%, respectively.
The HBV-DNA stages (negative or positive) are also associated with the prognosis of HBV-associated HCC patients. One study has proven that the 2-year survival rate of HBV-DNA-negative group is higher than that of HBV-DNA-positive group (p = 0.007).37 Stratified evaluation confirmed that HBV-DNA was associated with survival rate, and the difference between the 500 IU/mL HBV-DNA group and the 105 IU/mL HBV-DNA group was statistically significant (p = 0.009), suggesting that the higher the baseline HBV-DNA level, the lower the survival rate and the worse the prognosis after the HCC diagnosis. In this study, there was a significant distinction in the survival rate between HBV-DNA-negative and HBV-DNA-positive group (χ2 = 8.846; p = 0.003). Similarly to previous studies, patients in the HBV-DNA-negative group survived longer. The difference between the 2 groups may be related to HBV reactivation (PHR) after liver cancer treatment. One of the risk factors for the recurrence of liver cancer is HBV activation.26 The occurrence of PHR may be related to surgical resection and interventional therapy of HCC. Lao et al. found a PHR risk after hepatectomy or transarterial chemoembolization (TACE).38 Preoperative HBV-DNA level, hepatic blood flow, degree of cirrhosis, and other factors are related to PHR, even if the preoperative HBV-DNA titer is less than 500 copies/mL, there is still a risk of postoperative virus infection. Lai and Yuen also conducted a similar study.24 The HBV-associated HCC patients with negative HBV-DNA were at risk of hepatitis B reactivation after TACE, and PHR could further damage liver function and affect the prognosis of a patient. A further analysis of the individuals treated with antiviral therapy before HCC showed that the HBV-DNA-negative group had longer survival time and higher survival rate than the HBV-DNA-positive group, which was related to the dual effect of antiviral remedy and the negative HBV-DNA.
Limitations
The sample size of our study was relatively small, and further multicenter studies with large sample size are needed to confirm the outcomes of the study.
Conclusions
In summary, HBV-infected patients are at hazard of developing HCC even after turning virus-negative. When diagnosed with HCC, most patients have received long-term and effective antiviral therapy, thus decreasing the degree of cirrhosis, which indicates that antiviral therapy is one of the effective means for the remedy of liver cancer. At the same time, continuous monitoring of the disease is necessary to achieve early detection, diagnosis and therapy of HCC.