Abstract
Background. Pulse pressure (PP) is a pulsatile component of blood pressure (BP), strongly correlated with arterial stiffness (AS) and impacting prognosis. Disproportionally increased PP values in individuals with autosomal dominant polycystic kidney disease (ADPKD) should be expected, given the multifactorial cardiovascular involvement in the natural course of this disease.
Objectives. To investigate ambulatory PP in a group of ADPKD patients, and to examine the impact of age, sex, kidney function, hypertension, circadian rhythm, and antihypertensive drugs (AH) on studied parameters.
Materials and methods. A total of 130 ADPKD patients (median age 41 years, 35% men) who underwent 24-hour BP measurement with portable oscillometer Spacelabs 90217, were included in the study and their recordings were retrospectively analyzed. Demographic data and the medical history including antihypertensive treatment were collected, ADPKD was diagnosed based on the criteria by Pei et al., and estimated glomerular filtration rate (eGFR) was calculated according to the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation.
Results. Pulse pressure in the whole group was 46 (IQR: 42–53) mm Hg and it was significantly higher in men than in women and during the day compared to nighttime. There was a negative correlation of PP with eGFR and a positive correlation with age. Pulse pressure was not different in ADPKD patients with or without a diagnosis of hypertension.
Conclusions. Ambulatory PP is not substantially increased in ADPKD patients across different stages of CKD. It follows a regular pattern of being increased with age, male sex, daytime, and decreasing eGFR, but not with the diagnosis of hypertension.
Key words: hypertension, ADPKD, pulse pressure
Background
Pulse pressure (PP) is defined as an arithmetic difference between systolic blood pressure (SBP) and diastolic blood pressure (DBP).1 However, being easily accessible and extremely simple to obtain, it seems not appreciated highly enough in the clinical settings, given that PP surpasses many other indices, including mean arterial pressure (MAP), as a predictor of cardiovascular complications. Pulse pressure is also an established risk factor for unfavorable cardiovascular and kidney outcomes in patients with chronic kidney disease (CKD).2, 3, 4
Arterial stiffness (AS) is a consequence of arteriosclerosis, loss of elastin, collagen replacement, calcification, thickening of the artery wall, and impaired endothelium-dependent vasodilatation.5 It leads to high PP with disturbed perfusion flow, causing hypertrophy and/or hyperplasia of smooth muscles in the arterial wall, and end organ damage.6
Interestingly, patients with autosomal dominant polycystic kidney disease (ADPKD), the most common genetic cause of kidney failure, present with all the abovementioned contributors to increased PP and AS. Progressive renal dysfunction in ADPKD is caused by an enlargement of cysts, and it often leads to a dependence on renal replacement therapy (RRT).7 Endothelium damage occurs in the early stages of the disease.8 Also, abnormal function of polycystins, products of genes directly involved in the pathogenesis of ADPKD, is responsible for vascular damage.9, 10
Objectives
The aim of the study was to investigate ambulatory PP in a group of ADPKD patients and to examine the impact of age, sex, kidney function, hypertension, circadian rhythm, and antihypertensive drugs (AH) on studied parameters.
Materials and methods
Patients older than 18, recruited from a single outpatient center, with an established diagnosis of ADPKD based on clinical criteria by Pei et al., were referred to ambulatory blood pressure monitoring (ABPM) between 2014 and 2018.11 Renal function was assessed by estimating creatinine clearance using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation. Hypertension was diagnosed based on prior medical history or the use of AH agents. The exclusion criteria included diabetes mellitus and other comorbidities influencing the vascular phenotype.
For ambulatory 24-hour BP monitoring, portable oscillometer was used – Spacelabs 90217 (Spacelabs Healthcare Company, Issaquah, USA). Monitors were programmed to obtain readings at 15-minute intervals during daytime (6:00–22:00) and 30-minute intervals during nighttime (22:00–6:00). The arm cuff was positioned on the nondominant upper limb. Pulse pressure was calculated as SBP–DBP. Mean arterial pressure was calculated as DBP plus 1/3 of PP.
The collection and analysis of blinded data were approved by Ethical Committee of the Medical University of Gdansk, Poland (approval No. NKBBN 429/2019).
Statistical analyses
For statistical analysis, we used STATISTICA v. 10 (StatSoft Inc., Tulsa, USA). The normality of distribution was tested using the Shapiro–Wilk test. Data did not follow a normal distribution and were expressed as medians and interquartile range (IQR). We compared groups with the Mann–Whitney U test or Kruskal–Wallis analysis of variance (ANOVA) by ranks, as appropriate. Correlations were expressed as Spearman’s coefficients. Differences and associations were considered significant for p < 0.05.
Results
Data of 24-hour ABPM from 130 ADPKD patients were collected. The median age of the study group was 41 (33–51) years and there were 46 men (35%). Mean estimated glomerular filtration rate (eGFR) in the whole group was 79 (54–90) mL/min/1.73 m2. Median SBP was 126 (120–133) mm Hg and median DBP was 74 (75–84) mm Hg. Characteristics of the group stratified according to the CKD stage are displayed in Table 1. A majority (68%) of patients were in G1 or G2 CKD stage.
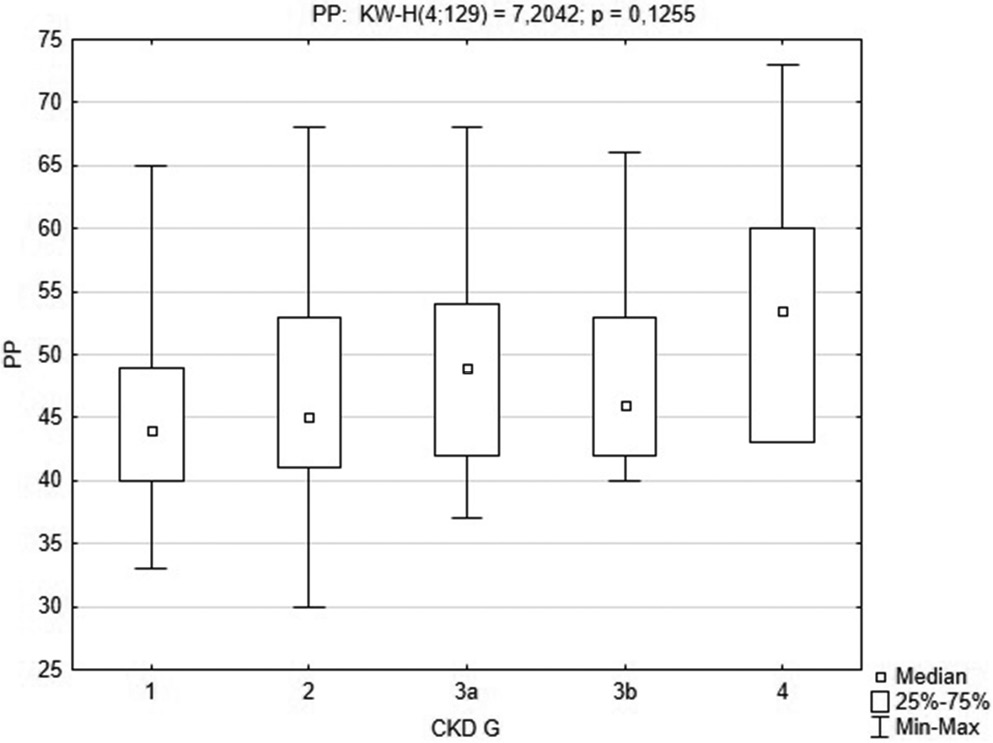
Pulse pressure in the whole group was 46 (42–53) mm Hg, and it was significantly higher in men than in women (49 (44–55) mm Hg compared to 44 (41–51) mm Hg, respectively, Z = −2.77; pMann–Whitney = 0.005). Also, there was a significant diurnal difference in PP: 47 (42–54) mm Hg during the day compared to 45 (41–52) mm Hg at night, Z = 3.60; p = 0.001. Figure 1 displays a comparison of PP according the CKD grade Kruskal–Wallis H test ((KW-H) (4; 129) = 7.2042; p = 0.126 (we excluded CKD G5 patients from this analysis as there was only 1 such patient in this group)). We found a weak correlation (Spearman’s test) between PP and age (r = 0.210; p < 0.05).
Pulse pressure was not different in patients with or without a diagnosis of hypertension.
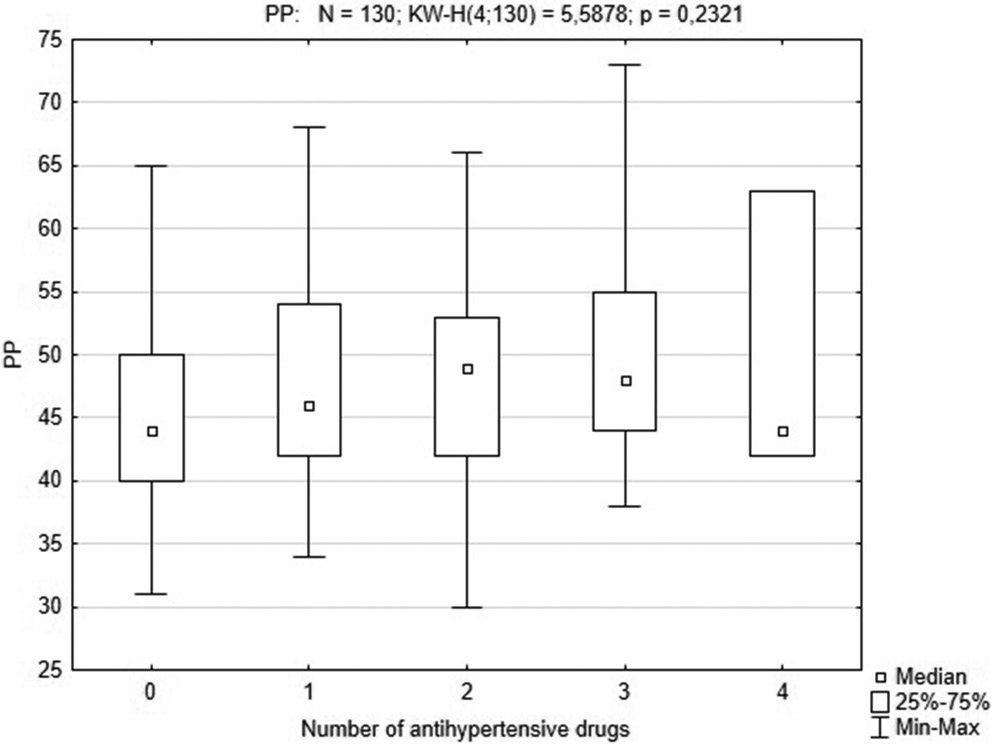
Eighty-seven patients were treated with AH. We analyzed the impact of the number and class of AH on the PP value (Table 2). Pulse pressure was not different between patients non-treated or treated with a different number of AH (1–4 drugs, Figure 2). Pulse pressure was higher in a group of patients that received blockers of α1-adrenergic receptors (AB) (54 (51–61) mm Hg in non-treated patients compared to 46 (42–53) mm Hg in treated ones; pMann–Whitney = 0.038). The AB treatment was not significantly associated with a different MAP (97 mm Hg compared to 95 mm Hg; Z = −0.94; pMann–Whitney = 0.344) for treated and non-treated patients, respectively.
Discussion
High PP is strongly correlated with stiffened arteries which cannot dampen pulsatile flow and may cause the destruction of the arterial walls, microvascular remodeling and microinfarcts, particularly in high-flow organs like the brain and kidneys, sensitive to PP.12 Furthermore, AS increases left ventricular pulsatile work, wall stress and myocardial oxygen consumption, and causes left ventricular hypertrophy (LVH).13 The LVH preceding hypertension has been described in early ADPKD and could be explained by several rationales.14 Polycystin 1 and polycystin 2 are present in endothelial cells, vascular smooth muscle cells, cardiomyocytes, and fibroblasts. Insufficient polycystin dosage leads to the reduced availability of nitric oxide synthase in the vascular endothelium and results in insufficient vasodilatation.9 This is observed also in people with ADPKD without hypertension or renal decline.15 Vasoconstriction in ADPKD is also supported by a high level of endothelin I.8 In this way, the balance between vasodilatation and vasoconstriction is impaired and causes vascular remodeling. Interestingly, also the mechanism of bone formation and vascular calcification may be at play in ADPKD and influence AS across different stages of ADPKD.16
Given the results of the HALT Progression of Polycystic Kidney Disease (HALT-PKD) study,17 one might expect disproportionally increased PP values in ADPKD individuals. However, it seems not to be the case. The median value of 24-hour PP in a group of 130 patients with ADPKD was 46 (42–53) mm Hg. Although the exact target PP value has not been established, it is known that individuals in the highest tenth of the 24-hour PP distribution had a significantly higher-than-average cardiovascular risk in multivariable-adjusted models.18 We stratified PP in our group according to age and sex, as PP is known to be higher in men and to increase with age. Both statements held true in our cohort, although the rate of PP rise accelerates after attaining 50 years of age, and our group was relatively young (median: 41 years). In a recently published study analyzing the results from the International Database on Ambulatory Blood Pressure in Relation to Cardiovascular Outcome (IDACO) that comprises over 11,000 participants, the mean PP was higher (49.7 ±10.0 mm Hg).19 Also, median PP in adults >50 years of age from our study (50 mm Hg) fits into the low-middle PP category (41.17–51.19 mm Hg) from IDACO.19 This may come as a surprise, given the aforementioned facts imposing a substantial risk of AS and vascular resistance on ADPKD patients. On the other hand, this finding goes in line with the excellent prognosis and low mortality rate in RRT patients with ADPKD, as compared to other etiologies.
Patients treated with AB had higher 24-hour PP values, although we found no difference in MAP between AB receivers and non-receivers. This fact can be explained by the effects of sympathetic vascular tone on arterial pressure and its involvement in the wave reflection and modulation of the aortic PP. However, the most probable explanation of this finding is older age and lower eGFR in patients treated with AB. The best practice remains using renin–angiotensin–aldosterone (RAA) blockade as the therapy of the first choice in different stages of ADPKD, as reflected in both arms of the HALT-PKD study.17, 20
Limitations
The first limitation of the study is a cross-sectional design. We were able to infer about associations but not of any causative relations between analyzed groups of AH and PP. Second, only a single ABPM measurement was performed and we might have missed some fluctuations in PP with time. Finally, we did not have a control group of individuals without ADPKD for comparisons. Adjusting for age, hypertension and stage of CKD would be very difficult in such a group. Third, we report only a single-center experience. However, due to a reliable protocol of measurement and verified equipment, we believe that our results are easily replicable in other centers.
Conclusions
Ambulatory PP is not substantially increased in ADPKD patients across different stages of CKD. It follows a regular pattern of being increased with age, male sex, daytime, and decreasing eGFR, but not with the diagnosis of hypertension.