Abstract
Sepsis is defined as an infection that causes the immune system to attack the body, subsequently leading to death. Some findings suggest that there is a high level of correlation between tumor necrosis factor (TNF) activity and susceptibility to sepsis. We used MEDLINE, Scopus and Web of Science databases to conduct an automated search covering the years 2000–2019. The Meta-analysis of Observational Studies in Epidemiology (MOOSE) criteria and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used for the meta-analysis. The selected studies were evaluated based on their focus on the TNF-α –308 A/G polymorphism, sepsis and sepsis mortality. Based on this inclusion criterion, 24 papers out of 782 were chosen for the meta-analysis. The meta-analysis was performed using Review Manager. The comparison of TNF1 and TNF2 among the patients was calculated in the 2 groups and the odds ratio (OR) was used to construct the forest plots. The meta-analysis of the OR in Asian and Caucasian populations does not prove the influence of TNF variant on sepsis risk.
Key words: sepsis, tumor necrosis factor, 308A/G polymorphism, single nucleotide polymorphisms
Introduction
Sepsis is a serious burden to healthcare worldwide. It develops mostly in elderly patients, preterm infants or low-birth-weight infants.1, 2 An exigent factor is the association of a vital genetic component to both the risk of developing sepsis and the subsequent outcome regarding survival. Various studies have shown a connection between widespread variations in human DNA, genetic polymorphism and sepsis-related mortality.3 Several biomarkers show elevated levels in sepsis conditions, such as TLR4 (toll-like receptor 4) single nucleotide polymorphisms (SNPs), rs4986790, and rs4986791, but not the SERPINE1 (serpin peptidase inhibitor, clade E (nexin, plasminogen activator inhibitor type 1), member 1) rs1799768 polymorphism.4, 5, 6, 7, 8
Tumor necrosis factor alpha (TNF-α) plays a vital role in many serious conditions, such as diabetes, cancer, etc.9, 10 The studies conducted in the past have produced mixed results on the role of TNF-α in a weakened health condition. One study reported TNF-α as a risk factor in the North Indian and Japanese populations, as well as Chinese and Turkish children. Studies in Germany and Hungary revealed a negative correlation between preterm infants and low-birth-weight infants.8, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21
Several papers examining the relationship between TNF-α and sepsis risk and outcome have been published. However, various conflicting reports have emerged, and it is not easy to replicate initial studies in many cases.22, 23 We speculated that a synthesis of the results of these studies would be more understandable, as it would provide more accurate estimates of the clinical effects of the TNF-α –308 A/G polymorphism, since many studies had insufficient power to suggest that TNF2 was correlated with the development of sepsis or sepsis-related mortality.24, 25, 26
Objectives
This study aimed to investigate whether having a variant TNF-α –308 genotype (TNF2 or non-G/G) is linked to a higher risk of sepsis or sepsis-related mortality.
Materials and methods
The meta-analysis was carried out according to the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines. We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) normative recommendations with the registration No. XMU # SM/IRB/2020/1021.
Data sources and searches
The keywords used were “TNF-α –308 A/G”, “tumor necrosis factor”, “sepsis”, “septic shock”, and “sepsis risk”. The search was carried out in MEDLINE, Scopus, and Web of Science databases.
Study selection
Articles from the years 2000–2019 were considered. A total of 782 articles were retrieved, out of which only 45 full-text articles were found to be eligible. Out of those, 24 were used for the purpose of the meta-analysis after their quality had been assessed.
Figure 1 shows the process of the selection of studies.
Exclusion and inclusion criteria
The selected studies were evaluated using 2 parameters: 1) focus on TNF-α –308 A/G polymorphism; and 2) focus on sepsis and sepsis risk.
To improve the readability, we used the term “sepsis” when referring to health disorders such as sepsis, severe sepsis, septic shock, septicemia, or infection-related systemic inflammatory response syndrome.
Studies were omitted if 1) the triggers of systemic inflammatory reaction syndrome (SIRS) or multiple organ dysfunction syndrome (MODS) were specifically unrelated to infection (e.g., following heart surgery); 2) the reseached causes of sepsis were nonbacterial infections (such as parasites, fungi and viruses); or 3) there were duplicate reports. The research architecture or language used had no impact on which studies were included in the meta-analysis.
The titles and abstracts of publications found using the search strategy were reviewed by 1 reviewer (HN). Any journal that was considered to be significant; full text was downloaded. The reviewers were not blinded to study authors or outcomes. In the end, both reviewers agreed on which findings should be included in the meta-analysis. A kappa (κ) statistic was used to assess the level of agreement. A kappa (κ) value of 0.87 indicated a high level of agreement between the reviewers, and hence the selection of included studies was agreed upon (Table 1).
Extraction of data
and methodological approach
The primary outcomes were the development of sepsis and mortality among patients with sepsis. Two researchers (FL and HM) used a structured data collection form to retrieve the data.
The included studies compared G/G with G/A, G/A with A/A, and hence the meta-analysis compared the studies taking into consideration G/G with G/A or A/A allele combinations. The G/A or A/A allele combinations are referred to as TNF2 and G/G as TNF1. The meta-analysis also considered the ethnicity of the study population, as it is believed to have a confounding role in the influence of TNF on sepsis risk.
Patients with related symptoms or illnesses and being at risk for sepsis served as the reference population for the sepsis study. Only people with sepsis were included in the probability of mortality study. Control subjects from the general population, such as healthy blood donors or healthy volunteers, were not included in either study.
We conducted stratified analyses by variables that we believed could confound the primary analysis, including 1) ethnicity (Asian compared to Caucasian); 2) sepsis classification (sepsis, severe sepsis, and septic shock); 3) age (adults compared to pediatric patients); and 4) immune status (immune-competent compared to immune-compromised patients). In the stratified study, only results of at least 2 trials per group were considered; therefore, age, immune condition, and 2 elements relevant to methodologic accuracy (primer sequence and sepsis definition) were excluded.
Statistical methods
The meta-analysis was performed using Review Manager v. 5 software (The Cochrane Collaboration, Copenhagen, Denmark). The comparison of TNF1 and TNF2 among patients was calculated in the 2 groups. Meta-analyses were done using a random effects model (Mantel–Haenszel method) and heterogeneity was evaluated using I2 statistic. The odds ratio (OR) and 95% confidence intervals (95% CIs) were used to construct forest plots. The value of p < 0.05 was considered statistically significant. The publication bias was assessed using a funnel plot in which the standard error of the log of the OR was plotted against the log of the OR.
Results
The flow diagram of trial recognition and collection is presented in Figure 1. A total of 782 titles and abstracts were reviewed, with 24 meeting the requirements for inclusion in the final meta-analysis. The following 21 articles were excluded: 14 articles with no study on sepsis susceptibility or result, 3 articles with inadequate evidence on TNF-α –308 A/G polymorphism status and 4 articles with a secondary release.
Table 1 shows the demographic data of the patients examined in the 24 included studies. A total of 4764 patients were analyzed in 24 studies: 15 papers concerned white population, 8 Asian population and 1, a mixed population. Sepsis was classified as sepsis (in 8 studies), extreme sepsis (in 5 studies), septicemia (in 1 study), septic shock (in 7 studies), or 2 of the above forms of sepsis, defined as combined patient groups (in 3 studies). Fifteen researchers examined sepsis susceptibility, 5 examined sepsis death and 4 examined each of them.
In 15 studies, TNF2 (G/A or A/A) was compared to TNF1 (G/G) in 8 studies, and allele A frequency in 1 sample, the TNF 308 genotype was identified as G/G, G/A, or A/A.29 The data from the 15 studies that reported on G/G, G/A, or A/A were translated to TNF2 (G/A or A/A) or TNF1 (G/G), yielding a total of 23 studies that could be used to assess the effect of TNF2 (Table 2, Table 3). The data from the 15 studies that reported on G/G, G/A or A/A were translated to TNF2 (G/A or A/A) or TNF1 (G/G), yielding a total of 23 studies that could be used to assess TNF2 effect (Table 2, Table 3).
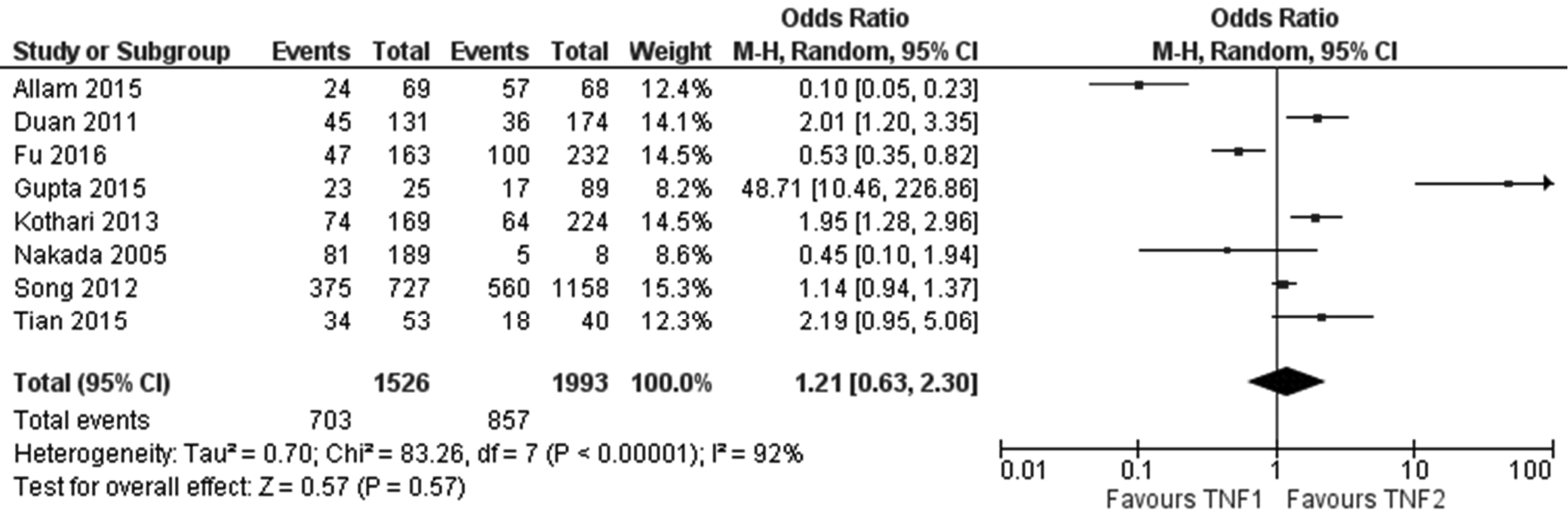
Figure 2 shows the forest plot of the role of TNF-α during sepsis among Asian population (n = 8 studies). There was no significant effect of TNF phenotype on risk of sepsis (OR 1.21, 95% CI: [0.63; 2.30], p = 0.57, I2 = 92%). The heterogeneity value I2 is high (>80%) indicating inconsistency between the included studies.
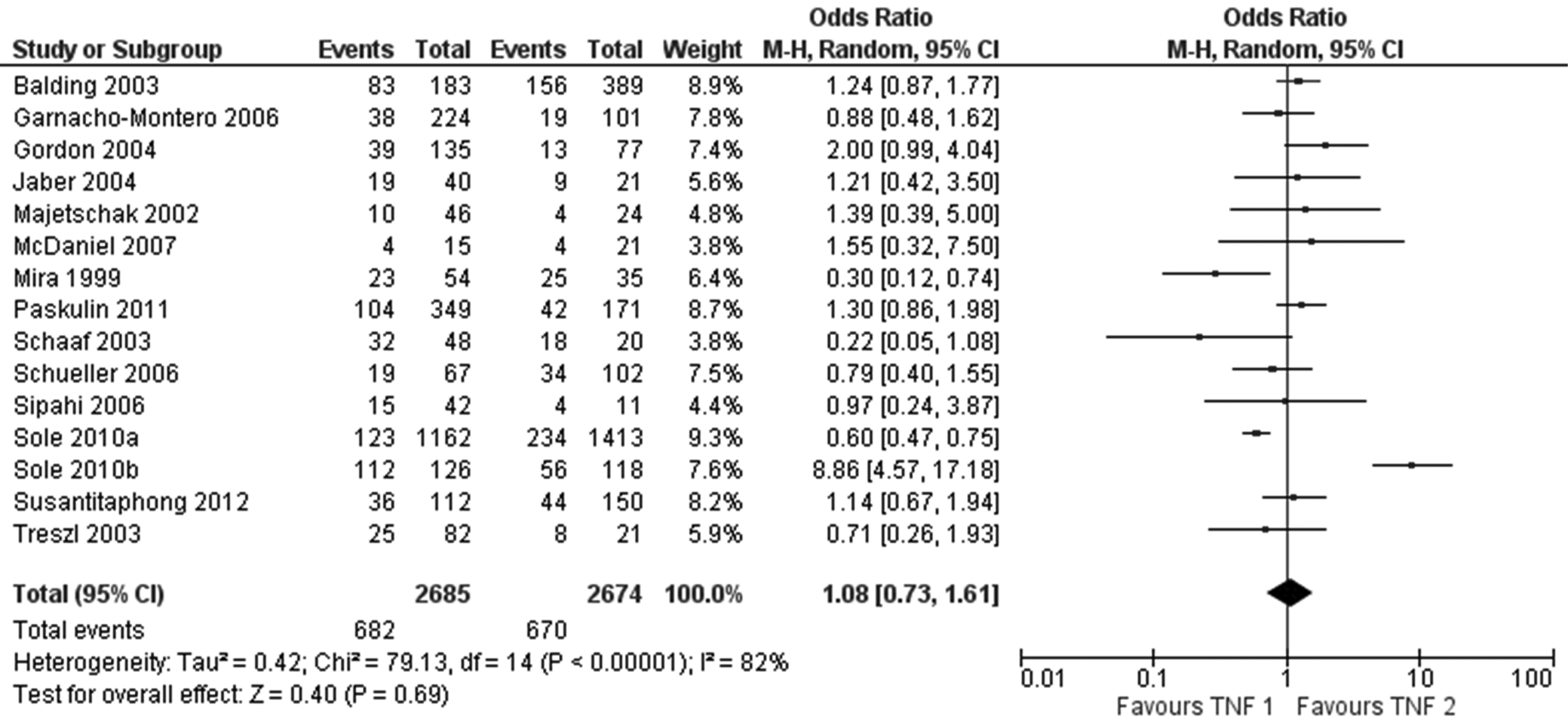
Figure 3 shows the forest plot between the roles of TNF-α during sepsis among Caucasian population (n = 15 studies). There was no significant effect of the TNF phenotype on risk of sepsis (OR 1.08, 95% CI [0.73; 1.61], p = 0.69, I2 = 82%). A high I2 heterogeneity value (>80%) indicates the inconsistency between the studies.
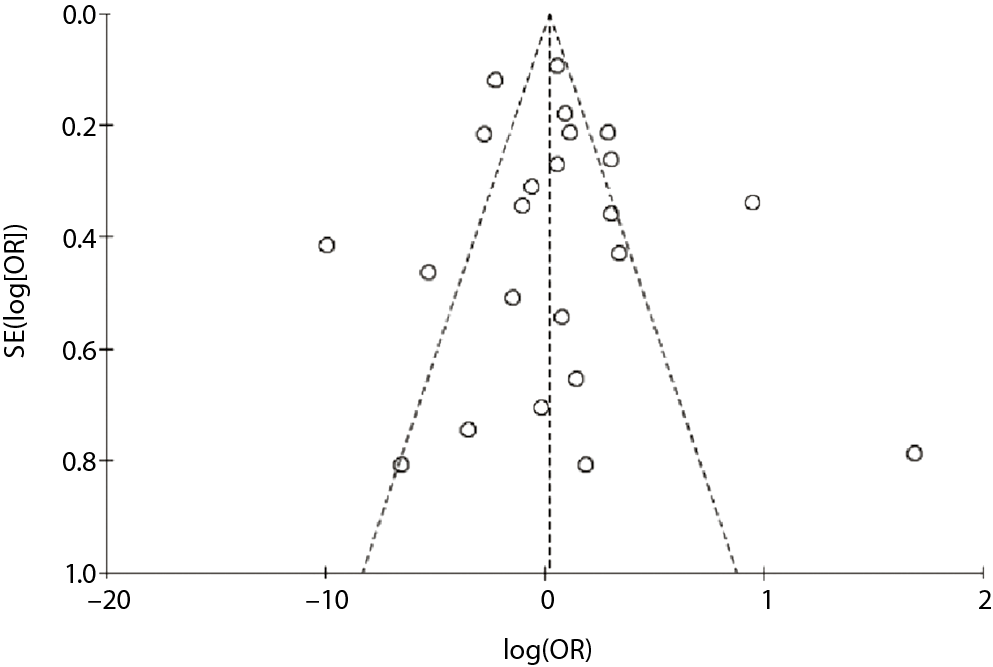
Figure 4 is the funnel plot for the assessment of publication bias. The funnel plot was asymmetrical, indicating the possibility of publication bias.
However, based on the OR results of the meta-analysis, there was a higher risk of sepsis in Asian population than in Caucasian population, and there was no significant effect of TNF phenotype on sepsis risk in either population.
Discussion
Tumor necrosis factor alpha has been identified as an important pro-inflammatory cytokine in diseases like psoriatic arthritis, rheumatoid arthritis, ulcerative colitis, Crohn’s disease and other autoimmune diseases, as well an in healthy people. It is located on the p arm of chromosome 6 within the major histocompatibility complex, where genetic alterations in the TNF-α locus are known to be involved directly in high TNF-α production.47 Many polymorphisms inside the TNF-α promoter positioned at −1031 (T→C), −863 (C→A), −857 (C→A), −851 (C→T), −419 (G→C), −376 (G→A), −308 (G→A), −238 (G→A), −162 (G→A), and −49 (G→A) have been identified, but nucleotide position −308 directly affects TNF-α production. A SNP within the promoter of the gene for TNF-α results in 2 allelic forms, TNF1 with guanine as common allele and TNF2 with guanine substituted by adenosine.48 The polymorphism in TNF-α –308 A/G has been associated with several pathologies like parasitic, bacterial and viral infections; autoimmune diseases like systemic lupus erythematosus, rheumatoid arthritis and ankylosing spondylitis; acute rejections for transplants57, 58, 59, 60; cancers10 and coronary artery disease.52 It has been observed in the past that TNF-α has been involved in sepsis-induced immune depression through increased apoptosis.49 The TNF2 allele has been linked to sepsis, but the evidence is ambigous. The present meta-analysis aimed to analyze the role of TNFα –308 genotype (TNF1 or TNF2) in a higher risk of sepsis.
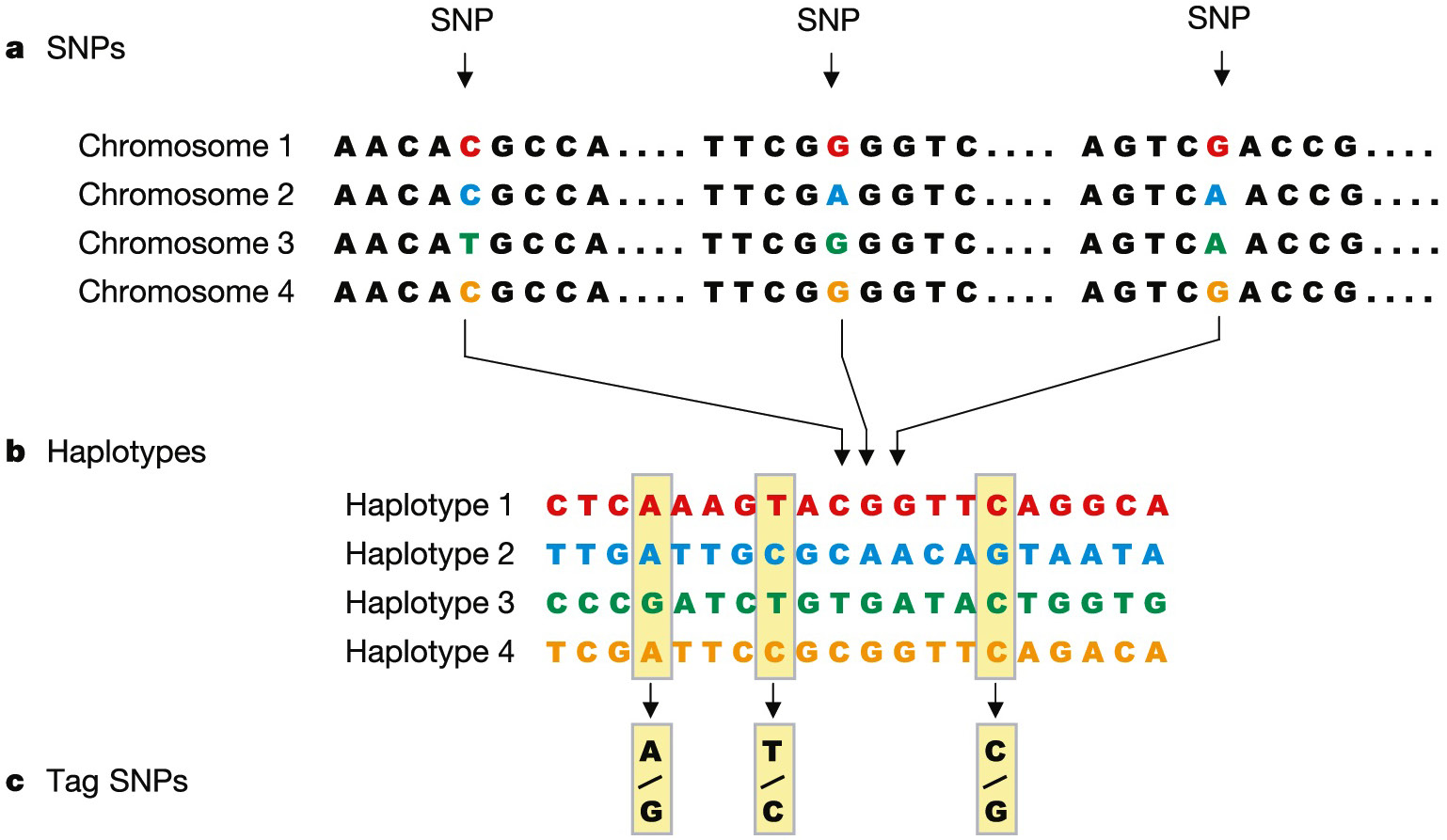
The Human Genome Project (HGP) focused on an individual’s DNA sequence. The following stage was designed to compare DNA sequences from various populations. The HapMap is a collection of human genetic diversity. It was completed in 2005 and uses SNPs to discover huge blocks of DNA sequence known as haplotypes that are inherited together (Figure 5). Researchers examine haplotypes in persons with and without certain diseases to elucidate information from the analyzed data. The haplotypes shared by persons with the disease are then analyzed in depth to search for genes that are linked to a particular condition. Scientists have already used the data in order to find a gene linked to age-related macular degeneration, a condition that causes blindness in elderly patients.9 The International HapMap Consortium is predicted to play a significant role in finding many more disease-related genes in the future.51
In a small number of DNA samples, the HGP,49 the SNP Consortium50 and the International HapMap Project51 found over 10 million common DNA variations, mostly SNPs. Genome-wide association studies have effectively found hundreds of unique genomic loci that have an impact on human diseases, due to the knowledge on SNPs and associated linkage disequilibrium patterns.4
Nonetheless, our understanding of human genetic variation in terms of variant type, frequency and population diversification is inadequate. Only common DNA variations (minor allele frequency (MAF) 5%) have been thoroughly investigated, despite the fact that low MAF variations are likely to account for a significant portion of genetic risk for common diseases. Systematic investigations of other forms of variations, particularly copy number variation, have just lately begun to shape our understanding of their frequency spectra, population distributions and linkage disequilibrium patterns.
According to Elahi et al.,52 TNF polymorphisms are found in a region of polymorphic variation and they are in linkage disequilibrium with the human leukocyte antigen (HLA) genes and with each other. Due to the differences in the distribution of HLA alleles, there can be variation in TNF polymorphisms and various discrepancies can be associated with different geographical locations.
The current study does not demonstrate the influence of TNF variants on sepsis in Asian and Caucasian populations. However, the number of studies in Asian population is small (n = 8), thus limiting an inference to be made in general. The heterogeneity values are high (>80%), suggesting a considerable inconsistency between the studies. Potential sources of heterogeneity are age, sample size, genotyping method, and type of sepsis.
The TNF2 allele frequencies have been identified for various ethnic groups, with Asian population (TNF2 prevalence ranging from 1.7%51 to 5.1%54) having a lower TNF2 allele frequency than Caucasian population (TNF2 prevalence ranging from 21.7%55 to 23.0%56). To see whether the connection between TNF2 and sepsis varied by ethnicity, we conducted a stratified study. Our findings indicate that the connection between TNF2 and sepsis is not significant in the Asian community compared to other ethnic groups. Given the secondary nature of the study, this finding should be regarded as hypothesis-generating.
The authors has also researched whether the impact of TNF2 varied, based on how sepsis was defined (sepsis, extreme sepsis or septic shock) in our stratified studies. The fact that many different health conditions such as pneumonia, bacteremia, sepsis, septicemia, acute sepsis, or septic shock have been analyzed, has made it challenging to interpret past findings. The authors were able to conduct systematic review because this meta-analysis used published consensus criteria for sepsis55 and the rest of the articles in our meta-analysis used these same definitions.
We discovered that the way sepsis was defined had little effect on the connection between TNF2 and sepsis susceptibility. Because of the limited number of trials in each subgroup, determining whether sepsis classification influenced the relationship between TNF2 and sepsis mortality is more complicated. Therefore, future research into this topic is critical. However, the present meta-analysis helped to overcome the limitations of the individual studies and thoroughly examined TNF-α role in sepsis.
One of the strengths of the study is that it was focused solely on TNF-α –308 A/G, which helped in conducting a thorough search and analysis of the impact of this single polymorphism. Other genetic variants, on the other hand, are very likely to affect sepsis risk. As a result, future experiments should examine the effects of other polymorphisms, and eventually, many polymorphisms would have to be considered.
In addition, the current meta-analysis reveals the possibility of obtaining a more stratified study by considering similar studies in each stratum. A meta-regression could also be carried out to examine the hidden factors affecting the phenomenon.
Limitations
The study has a few limitations, such as limited sample size in most of the analyzed studies, that affects the statistical power. There is a high heterogeneity among the different ethnic backgrounds. Lastly, there is a strong need for more studies to determine the clarification on genetic roles and cytokine production during sepsis.
Conclusions
The relationship between the TNF–308 G/A polymorphism and sepsis was quantitatively synthesized in this meta-analysis. It does not suggest an association between the G/A allele combination of TNF-α and sepsis risk in the Asian and Caucasian populations. We believe that integrating the effects of correlation research will help us better understand the impact of polymorphisms on disease outcomes.