Abstract
Background. Dextran sodium sulfate (DSS)-triggered ulcerative colitis (UC) model in animals provides a valuable platform to preclinically evaluate the outcome of drug candidates for UC. Dandelion root extracts (DRE) have a therapeutic effect on UC. However, the protective mechanism of DRE against UC remains unknown.
Objectives. To discover the targeting pathway involved in DRE-induced protection against UC.
Materials and methods. The UC model was developed in C57BL/6 mice by oral administration of DSS. Following DSS exposure, sulfasalazine (SASP), low dose of DRE (DRE-L), moderate dose of DRE (DRE-M), high dose of DRE (DRE-H), and DRE-H plus mitogen-activated protein kinases (MAPK) agonist (DRE-H+MA) were administered to the mice. Colon Mucosal Damage Index (CMDI) and histopathological analysis were used to evaluate the colonic mucosal damage. The cytokine levels were detected using commercial enzyme-linked immunosorbent assay (ELISA) kits. The MAPK pathway activation was determined with western blotting.
Results. We found that DRE-H attenuated DSS-triggered colonic mucosal damage. The DSS-induced inflammatory responses and oxidative stress in the bloodstream and colon tissues were dramatically inhibited by DRE-H administration. Also, this plant impaired DSS-provoked phosphorylation levels of extracellular signal-regulated kinases (ERK), c-Jun N-terminal kinases (JNK), p38 mitogen-activated protein kinases (p38), p65, and IκB. More importantly, MAPK agonist, BIM-23A760, removed the protective effect of DRE-H on the bloodstream and colon tissues.
Conclusions. The DRE-H is capable of relieving DSS-induced UC, and its mechanism links to the MAPK pathways.
Key words: mouse, MAPK, ulcerative colitis, dextran sodium sulfate, dandelion root extracts
Background
Ulcerative colitis (UC) is an inflammatory bowel disease (IBD) featured with recurrent attacks, poor outcome, prolonged illness, and high potential of colorectal carcinogenesis.1 The annual incidence of UC has been largely increasing in China, and the current clinical drugs, including anti-inflammatory and immunosuppressive compounds, have several adverse side effects such as osteoporosis, neurotoxicity and gastrointestinal intolerance2; in consequence, more and more studies attempt to exploit new or alternative approaches to UC.3 Dextran sodium sulfate (DSS), one of the sulfated polysaccharides, is a standard reagent used to create an experimental UC model in animals, as most pathological symptoms during DSS application are similar to those of UC patients.4 Therefore, the DSS-triggered UC animal model provides a valuable platform to either develop new drug targets by constant progress in understanding the etiology and pathogenesis of UC, or pre-clinically evaluate the outcomes of drug candidates for UC.
It has long been known that dandelion (Taraxacum, Asteraceae family), a traditional Chinese herbal medicine, together with other herbs, can treat various ailments.5 Recent attempts have led to discoveries of anticancer, anti-inflammatory and antioxidative functions of this plant.6, 7, 8 By stimulating multiple death signaling pathways, the dandelion root extracts increase cell apoptosis of colorectal and pancreatic cancer and leukemia.9, 10, 11 Inhibiting PI3K/AKT pathway is a mechanism for dandelion root extracts to ameliorate lipopolysaccharide (LPS)-triggered inflammation,12 and reducing lipid peroxidation contributes to the antioxidative activity of dandelion root extracts on the alcohol-induced liver.13 Although the protective effect of this plant on UC has been revealed,14 the exact mechanism is unknown. Such a broad function of dandelion root extract might be due to its active chemical constituents, including sesquiterpenes, various triterpenes, phytosterols, and phenolic compounds.15 Among the phenolic compounds, hydroxycinnamic acid derivatives (chlorogenic, caffeic, 4-coumaric, 3-coumaric, ferulic acids) are mainly reported, whereas flavonoids and hydroxybenzoic acid derivatives have a relatively low level.16, 17, 18 These chemical constituents likely act individually, additively or in synergy, in order to modulate different tissue-specific functions.19 Still, the physiological functions of dandelion and its clinical utilization clues the ability of cells to respond to its bioactive components through fundamental and widespread intracellular mechanisms.
Mitogen-activated protein kinases (MAPK) harbor 3 well-known subfamily members, extracellular signal-regulated kinases (ERK), c-Jun N-terminal kinases (JNK) and p38 mitogen-activated protein kinases (p38), that perform autophosphorylation or phosphorylate their substrates to activate or deactivate downstream molecules.20 These kinases are ubiquitous and conserved in eukaryotes, and they are essential modulators for cell pathology, cellular physiology and various diseases.21 Recent evidence proves that MAPK signaling pathways show a crucial correlation with the pathogenesis of UC, due to the abundant release of cytokines and inflammatory mediators by their activation.22, 23, 24 Given the clues that MAPK pathways can be utilized by dandelion or its extracts to ameliorate the inflammation in vitro and bacterial disease in animals,25, 26, 27 we proposed that dandelion root extracts may relieve experimental UC by regulating MAPK signaling pathways.
Objectives
In this study, we hypothesized that MAPK signaling pathways might participate in the protection against UC by dosing dandelion root extracts in experimental UC. Therefore, this study aimed to discover the interrelationship between dandelion root extracts and MAPK pathways in the DSS-triggered experimental mouse model.
Materials and methods
Preparation of dandelion root extracts
Premier Herbal Inc. (Toronto, Canada; lot No. 318121) provided dandelion roots for the present study. The root extract was isolated according to the published protocol.19 Briefly, the appropriate dandelion roots were extracted by soaking in 95% ethanol at 1:10 w/v (plant material to solvent ratio) under uninterrupted stirring for 3 days, changing the solvent every 24 h. The extract was filtered, and the solvent was removed using a rotary evaporator (Vacufage plus; Eppendorf, Hamburg, Germany). The dry extract of dandelion root was frozen at −80°C for long-term storage and redissolved by the same solvent in 100 mg/mL before use. According to the published analysis on reversed-phase high-performance liquid chromatography,19 the main chemical constituents of dandelion root extract were hydroxybenzoic acid derivatives and hydroxycinnamic acids (gallic acid, chlorogenic acid, caffeic acid, vanillic acid, syringic acid, p-coumaric acid, and ferulic acid). For the animal experiment, 100 mg/mL of dandelion roots extracts were finally diluted in sterile distilled water to the indicated concentrations for the gavage.
Animals and experimental design
Male and female (half-and-half) specific pathogen-free (SPF) C57BL/6 mice, 6−8 weeks of age, weighing 18 ±3 g, from the same litters, were commercially obtained from Animal Center of Hainan Medical University and housed in an environmentally controlled breeding room (temperature: 20 ±2°C, humidity: 60 ±5%, 12 h/12 h dark/light cycles) in cages, and fed with sterile water and standard laboratory rodent diets. They were maintained under internationally accepted principles for laboratory animal use. The animal protocol complied with the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines and was approved by the Institutional Animal Care and Use Committee of Hainan Medical University, China (approval No. DLMU2020012341).
After 3 days of acclimatization, mice were administered 2% DSS (w/v) (36–50 kDa; MP Biomedicals, Illkirch-Graffenstaden, France) in their drinking water to induce acute UC. Mice were randomly separated into 7 groups (n = 10 per group): 1) control group (mice received regular drinking water, CON group); 2) DSS-induced UC model group (mice received 2% w/v DSS in drinking water for 10 days, UC group); 3) DSS + sulfasalazine (SASP) group (100 mg/kg/day for 10 days); 4) DSS + low dose of dandelion root extracts (DRE; 0.5 mg per mouse) group (mice received 2% w/v DSS in drinking water for 10 days, together with the administration by gavage of 1 mg per mouse of DRE twice a day, DRE-L group); 5) DSS + middle dose of DRE group (1 mg per mouse, DRE-M group); 6) DSS + high dose of DRE group (2 mg per mouse, DRE-H group); and 7) DSS+DRE-H+MAPK agonist group (DRE-H+MA group). Sulfasalazine is a certain drug used to treat UC clinically.28 The MAPK agonist, BIM-23A760,29 was provided by IPSEN Bioscience (Cambridge, USA)30 and injected into the tail vein (4 μg per mouse) every other day. The volume of gavage and tail vein injection was 400 μL and 100 μL, respectively. Mice were observed for 10 days and then sacrificed by CO2 inhalation.31 Serum and colon samples were collected for the subsequent analysis.
Colon Mucosal Damage Index
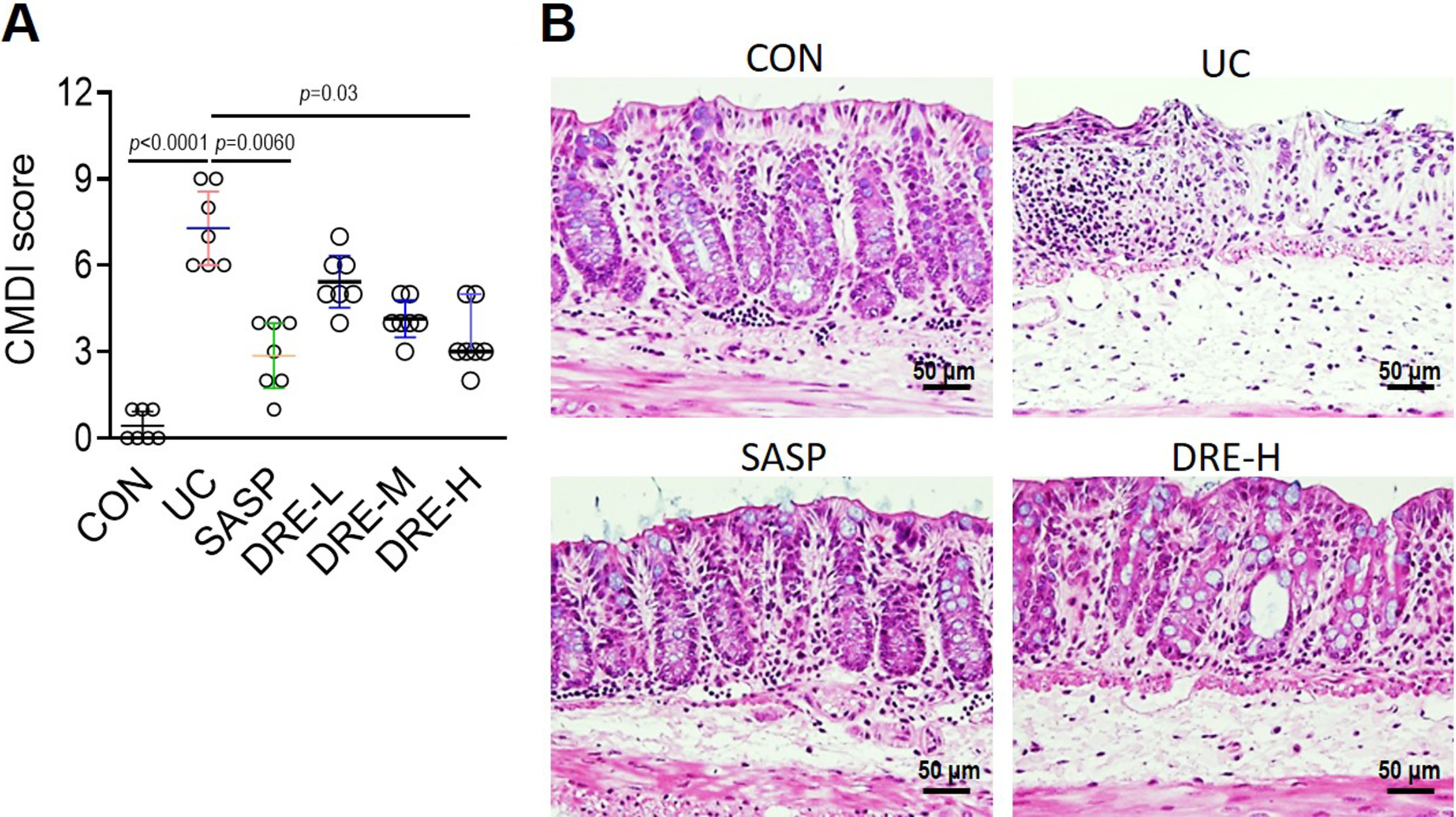
Colon Mucosal Damage Index (CMDI) was evaluated according to the following criteria32: (1) ulcer and inflammation: 0 points – normal; 1 point – focal congestion, no ulcer; 2 points – ulcer without congestion or thickening of the intestinal wall; 3 points – one place with inflammatory ulcer; 4 points – ulcers and inflammatory sites in 2 places; 5 points – the central part of the damage along the colon extension ≥ 1 cm; 6–10 points – the injury along the length of the colon extended ≥ 2 cm; for each increase of 1 cm of damage, the score increased by 1 point; (2) the presence of adhesion: 0 points – normal; 1 point – slight adhesion; 2 points – the main adhesion.
Histology analysis
The colon samples were collected and fixed in 4% paraformaldehyde solution overnight, and then embedded in paraffin and sliced into 4-μm sections. The paraffin sections were stained with hematoxylin for 5 min, followed by phosphate-buffered saline (PBS) washing and dying in eosin solution for 30 s.33 The histopathological changes were observed using a light microscope (BX50; Olympus Corp., Tokyo, Japan).
Enzyme-linked immunosorbent assay
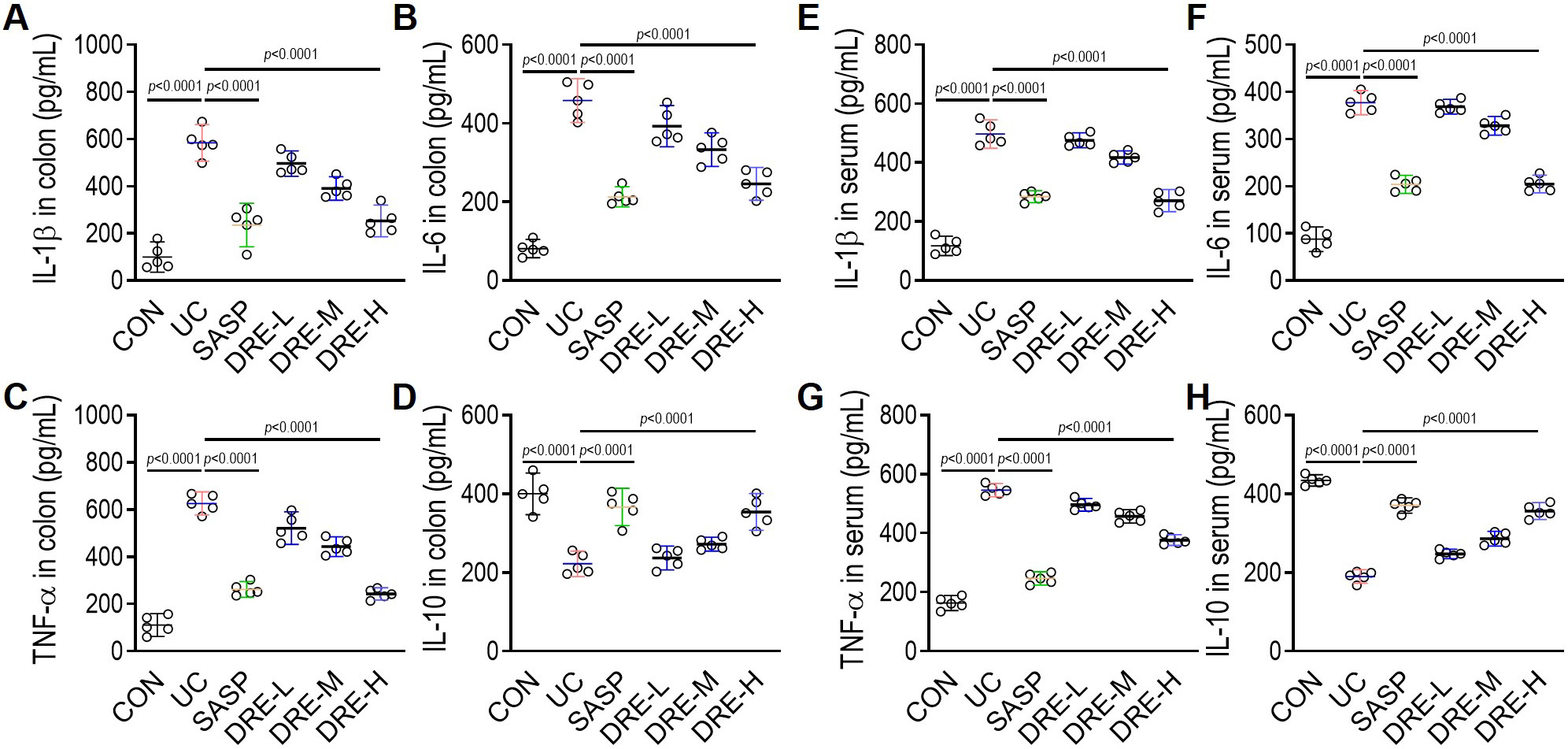
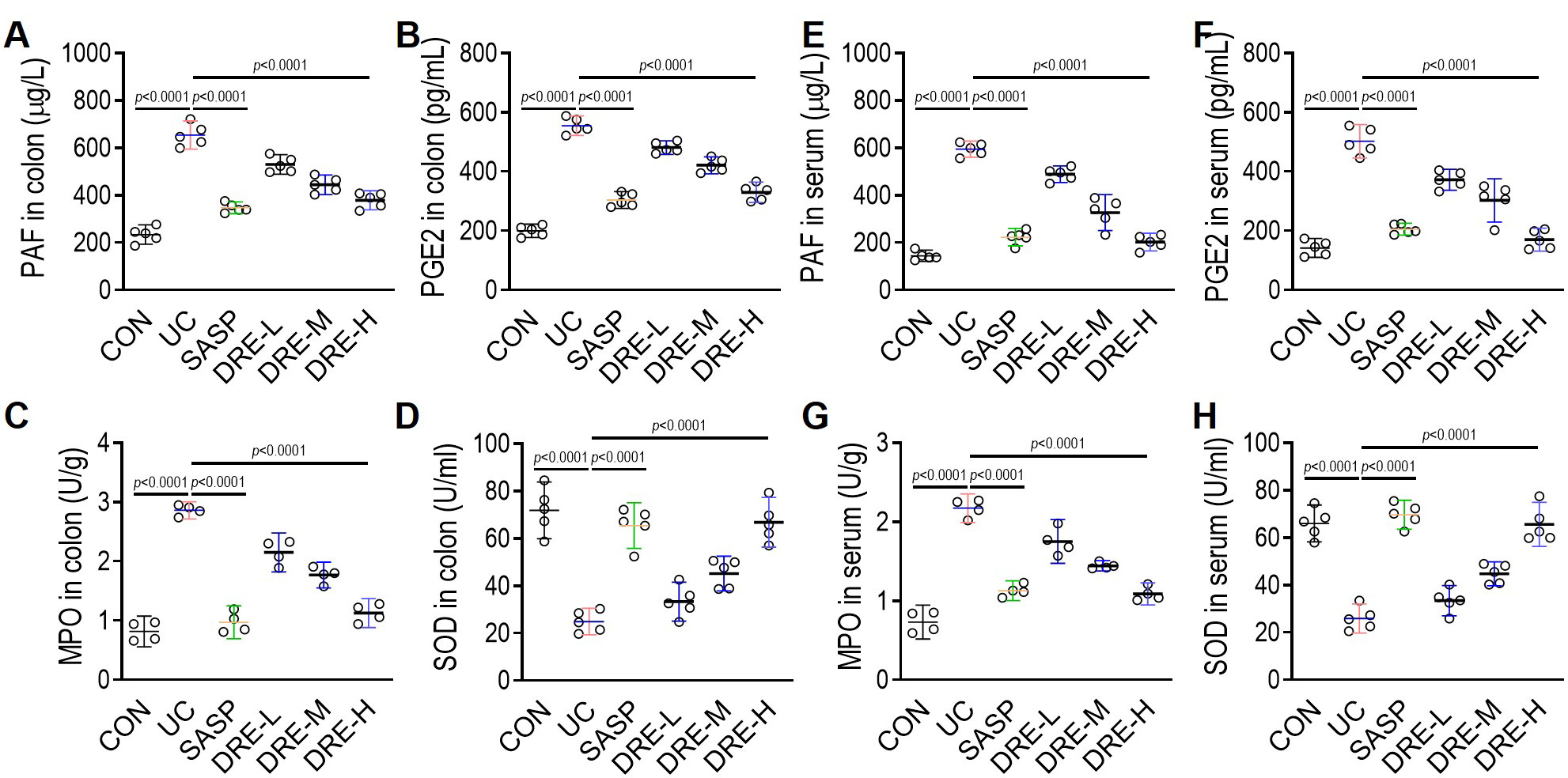
The levels of mouse interleukin (IL)-1β (Nanjing Senbeijia Biological Technology, Nanjing, China), IL-6 (Nanjing Senbeijia Biological Technology), tumor necrosis factor alpha (TNF-α) (Nanjing Senbeijia Biological Technology), IL-10 (Nanjing Senbeijia Biological Technology), platelet-activating factor (PAF; cat No. MBS2700185; MyBioSource, San Diego, USA), prostaglandin E2 (PGE2, MyBioSource), myeloperoxidase (MPO; cat. No. EMMPO; Invitrogen, Waltham, USA), and superoxide dismutase (SOD; cat. No. EIASODC; Thermo Fisher Scientific, Waltham, USA) were assessed in the colon homogenates and serum samples, using the abovementioned commercially available kits according to the manufacturer’s specifications.34
Western blotting
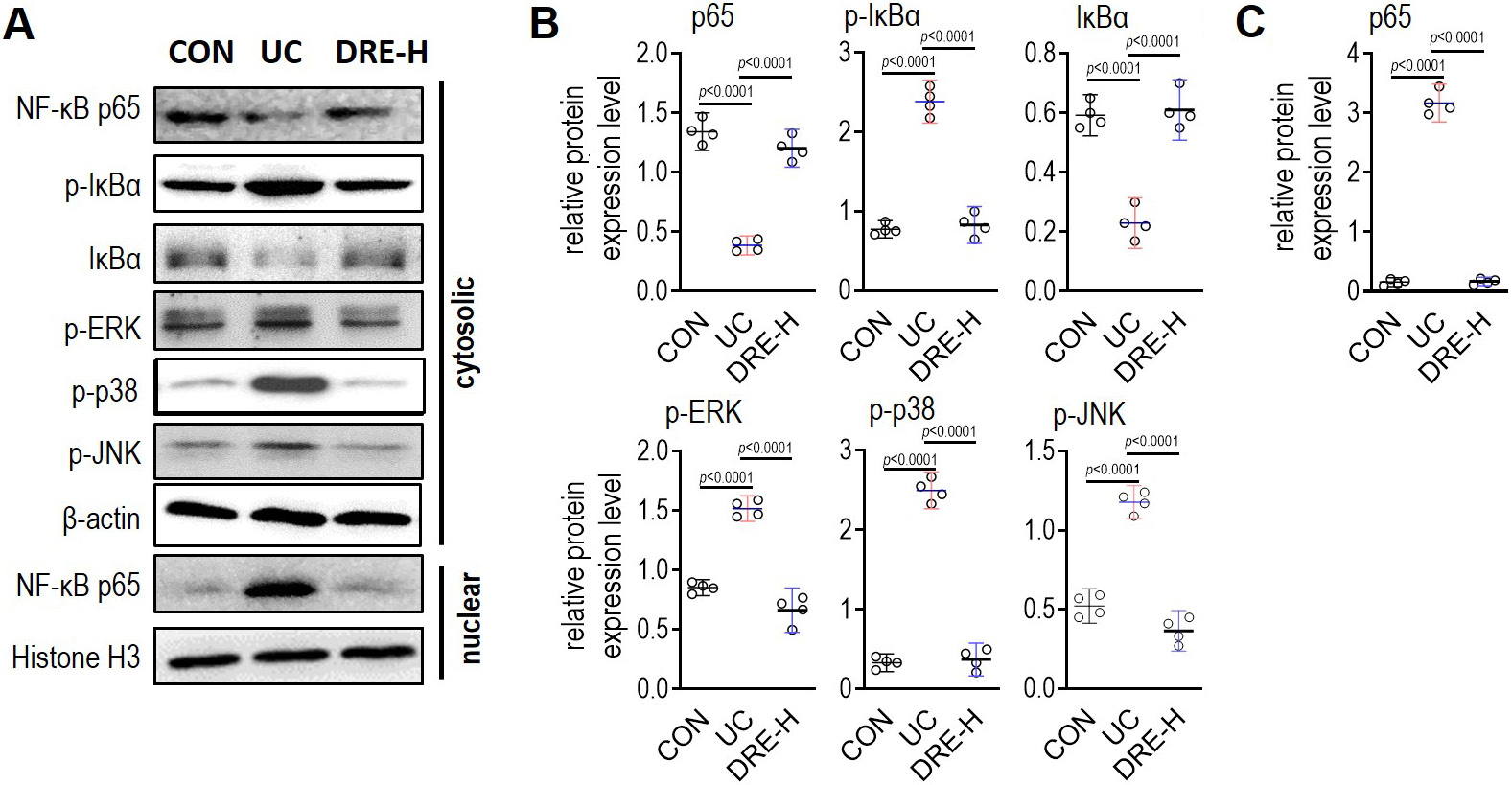
For the immunodetection, 50 µg of total protein extracts from cytosol or nucleus in Laemmli sample buffer (Bio-Rad, Hercules, USA) were resolved on 10% or 15% sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and then transferred to nitrocellulose membranes for western blotting. The membranes were first stained with Ponceau S to confirm the transfer efficacy. After blocking with 5% skim milk dissolved in Tris-buffered saline (TBS) containing 0.05% Tween-20 (TBST) (VWR, Lutterworth, UK) for 2 h at room temperature, membranes were incubated with anti-phospho-ERK, anti-phospho-p38, anti-phospho-JNK, anti-β actin, anti-Histone H3, anti-phospho-IκBα, anti-IκBα, anti-phospho-p65-S536, or anti-p65 (ABclonal, Woburn, USA) at appropriate dilutions, followed by goat anti-rabbit secondary antibody conjugated with horseradish peroxidase (HRP). Positive band intensities were detected using a gel documentation system (Fujifilm LAS-3000 Imager, Tokyo, Japan).35
Statistical analyses
Nonparametric Kruskal–Wallis test with Dunn’s multiple comparison test were only used in the data shown in Figure 1A, since it did not fit normal distribution (Gaussian).36 One-way analysis of variance (ANOVA) with Tukey’s multiple comparison test or Welch’s ANOVA test with Dunnett’s T3 multiple comparisons test were used in the remaining data from Figure 2, Figure 3, Figure 4, Figure 5, Figure 6. The Shapiro–Wilk and Kolmogorov–Smirnov tests were performed for normal distribution. The Shapiro–Wilk test is performed based on regression and correlation, and Kolmogorov–Smirnov test is the empirical distribution function test.37 They analyze the normality of data in a different way. Therefore, the data with a sample size greater than 4 was considered to have a normal distribution only if it passed both tests, while a normal distribution of data with a sample size less than 5 can only be evaluated by the Shapiro–Wilk test. The Bartlett’s test was used for evaluating the equal variances.38 All data analyses were performed using GraphPad Prism software v. 8.4.0 (GraphPad Software, San Diego, USA). A value of p < 0.05 was considered statistically significant.
Results
Dandelion root extracts ameliorate
DSS-triggered colonic mucosal injury
Oral administration of DSS is a common approach to induce UC in mice.39 As expected, the UC was successfully established in C57BL/6 mice. It was indicated by the high CMDI score and the mucosal epithelial cell degeneration and necrosis, as well as an increased number of infiltrating mucosal leukocytes in histopathological staining of the colon sections (Figure 1A,B). Sulfasalazine, a therapeutic drug for UC,40 and various doses of DRE were used to treat UC mice. The SASP group and the DRE-H group had significantly lower CMDI score than the UC group, while the low or middle dose of DRE (DRE-L or DRE-M) groups showed only slight but no statistically significant reduction of CMDI score compared to the UC group (Figure 1A). Therefore, the DRE-H group was selected for the following experiments. Consistently, the mild epithelial cell degeneration and necrosis, as well as a small number of inflammatory cells were observed in colonic sections derived from SASP and DRE-H groups (Figure 1B).
Dandelion root extracts weaken
DSS-triggered inflammatory cytokines
The effects of DRE on the DSS-induced colonic mucosal inflammation were evaluated by assessing several inflammatory markers in mouse serum and intestinal tissues. As shown in Figure 2A–H, the proinflammatory cytokines, TNF-α, IL-6, and IL-1β, were markedly enhanced, and the anti-inflammatory cytokine, IL-10, was significantly reduced in serum and colon tissues of the UC group. More importantly, all of the abnormal changes of these inflammatory indicators induced by DSS were reversed, mainly by the oral administration of SASP or DRE-H, suggesting the anti-inflammatory action of DRE in UC mice.
Dandelion root extracts improve
DSS-triggered oxidative injury in the colon
Several factors (PAF, PGE2, MPO, and SOD) related to colonic oxidative injury were further assessed to determine the antioxidative effect of DRE in UC mice. The DSS substantially triggered oxidative injury, as demonstrated by the upregulation of PAF, PGE2 and MPO levels, and the downregulation of the activity of SOD in either colonic tissues or serum samples (Figure 3A–H). Interestingly, SASP and DRE-H dramatically ameliorated oxidative stress in UC mice by increasing SOD activity and decreasing PAF, PGE2 and MPO levels. In addition, the antioxidative activity between SASP and DRE-H groups is similar.
Dandelion root extracts impact
the MAPK signaling pathways
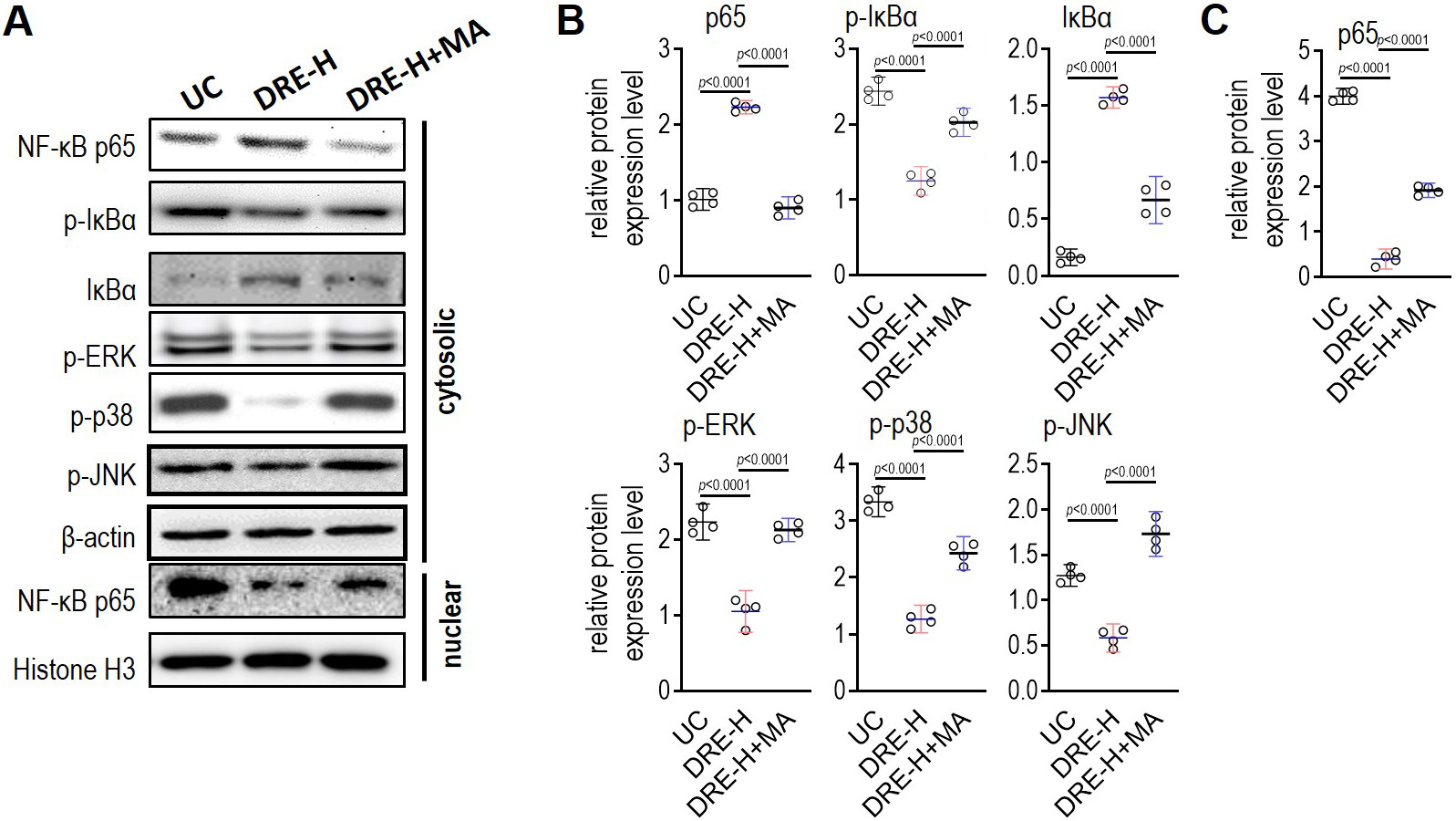
First, it was investigated whether the action of DRE against UC is related to MAPK signaling pathways. As shown in Figure 4, the phosphorylation level of ERK, JNK or p38 in colon tissues was strongly induced by the oral administration of DSS, and the application of DRE-H entirely blocked the DSS-induced high phosphorylation of the 3 indicators. Nuclear factor kappa B (NF-κB) is downstream of MAPK signaling pathways. The phosphorylation levels of NF-κB p65 and IκBα, 2 indicators of NF-κB activation, were obviously enhanced by the DSS treatment, but the abundances of IκBα and p65 in the cytoplasm were significantly decreased by the DSS exposure (Figure 4A,B). The production of NF-κB p65 in nuclei was enhanced by the DSS treatment, suggesting the robust translocation of p65 into nuclei (Figure 4A,C). Notably, the administration of DRE-H could completely reverse all the abovementioned DSS-triggered alterations (Figure 4), revealing that DRE-H likely blocks the activation of NF-κB through the inhibition of phosphorylation levels of ERK, JNK and p38.
Dandelion root extracts target MAPK to improve DSS-triggered UC
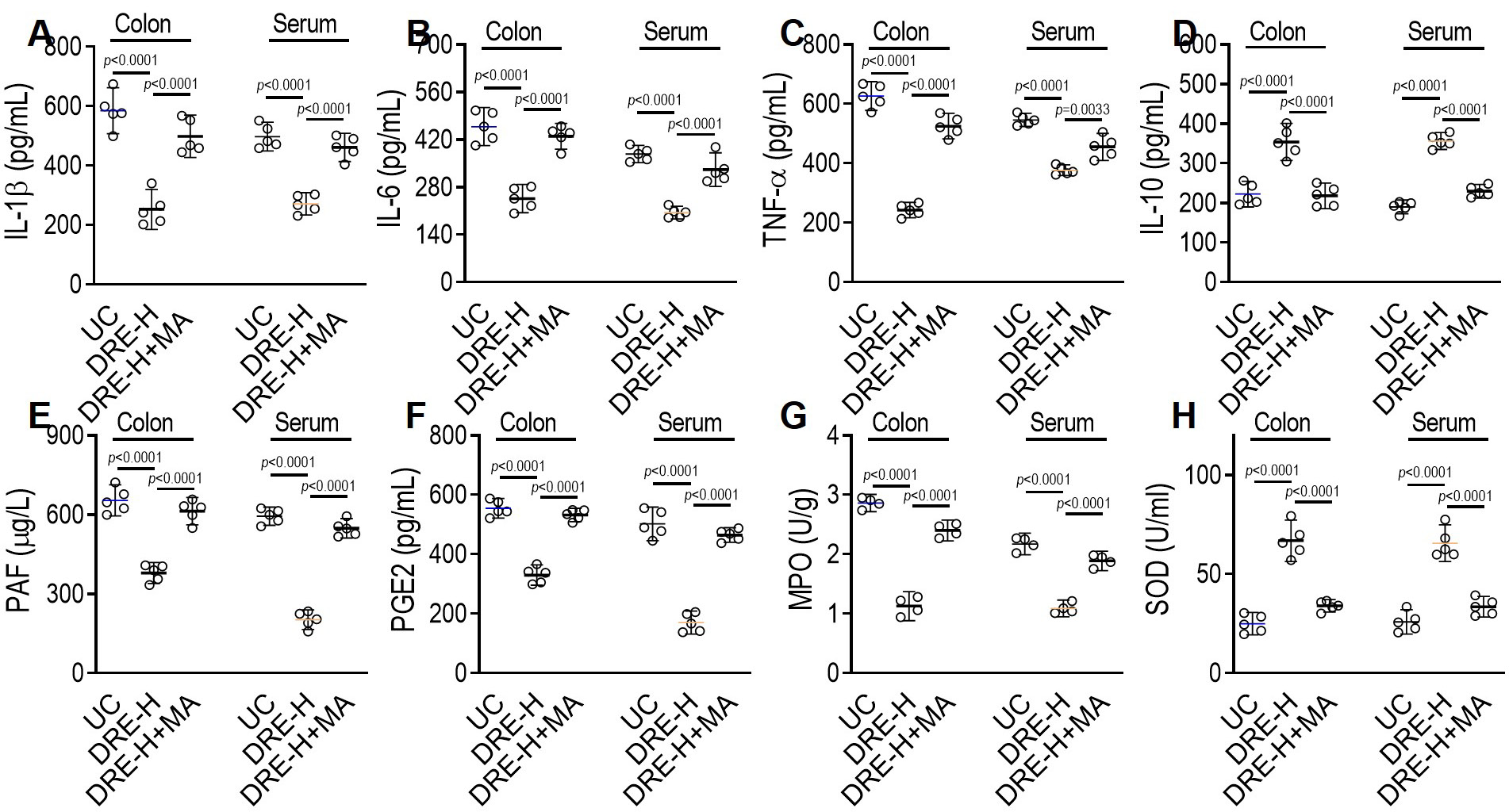
To investigate further whether the therapeutic effect of DRE against UC is achieved by suppressing MAPK signaling pathways, the MAPK agonist, BIM-23A760, was applied in C57BL/6 mice during DRE-H administration. Interestingly, BIM-23A760 removed anti-inflammatory and antioxidative actions of DRE-H by reinducing IL-1β, IL-6, TNF-α, PAF, PGE2, and MPO, and resuppressing the levels of IL-10 and SOD in serum and tissue samples (Figure 5A–H). At the same time, the inhibitory effect of DRE-H on DSS-triggered activities of ERK, JNK and p38, and their downstream transcriptional factor NF-κB was omitted by this agonist (Figure 6A–C), which seems to prove that the re-activation of MAPK pathways could block the therapeutic effect of DRE-H against UC. In other words, the inhibition of MAPK signaling pathways could be a mechanistic explanation for DRE to relieve UC in mice.
Discussion
There seems to be a consensus that DSS acts as a toxic factor, damaging gut epithelial cells and destroying the integrity of the mucosal barrier, thereby disseminating gut bacteria or other microbes into the lamina propria.41 This dissemination provokes immune and concomitant inflammatory responses by recruiting neutrophils and other leukocytes to the injured sites and accumulating several leukocyte-expressed proinflammatory cytokines, including IL-1β, IL-6 and TNF-α; the latter aggravate the disease further.42 Our data consistently showed that leukocytes and their secreted cytokines were accumulated, and that MPO, the principal constituent of neutrophil cytoplasmic granules, as well as PAF, the stimulator for leukocyte adhesion, were enhanced in DSS-exposed colon tissues. In the current study, the application of DRE-H reduced the accumulation of neutrophils and other leukocytes, as demonstrated by the reduction of DSS-triggered leukocytes infiltration, proinflammatory cytokines, MPO activity, and PAF level. Moreover, the DSS-triggered increase of proinflammatory cytokines, MPO and PAF in the bloodstream were also improved by the oral administration of DRE-H. Besides its effect on proinflammatory cytokines, DRE-H also supported the anti-inflammatory cytokine, IL-10. The IL-10 is deficient in patients with UC and has been proposed as a potent anti-inflammatory therapy in this disease.43 The mechanisms responsible for its anti-inflammatory action have been recently explored, and this anti-inflammatory action is dependent on the promotion of mitophagy that clears dysfunctional mitochondria featured by low membrane potential and a high level of reactive oxygen species (ROS).44 Without the effective IL-10 signaling, damaged mitochondria are accumulated and result in dysregulated activation of the inflammasome and the production of IL-1β. Thus, all of the abovementioned evidence supports the protective effect of DRE-H on UC, which is in line with the previous findings.14
The NF-κB is an inflammation-associated signal essential for the host defense to different infections and intracellular changes of reduction-oxidation state.45, 46 Without any stimulations, NF-κB forms an inactive and stable complex with inhibitory protein IκB in the cytoplasm. Once the cells get activated by various stimuli, the inhibitory protein IκB is phosphorylated and then dissociated from NF-κB. After the translocation into the nucleus and a subsequent attachment to the κB binding sites, NF-κB triggers the expression of several downstream genes, including inflammatory mediators.47, 48 In this study, DSS treatment largely elicited the phosphorylation of NF-κB and IκB, while DRE-H diminished their phosphorylation, indicating the anti-inflammatory molecular mechanism dependent on NF-κB for DRE. Three prominent MAPK pathway members, ERK, JNK and p38, can arouse the action of NF-κB.49 Interestingly, we found that these 3 kinases were all provoked in DSS-injured tissues and had a similar expression pattern as NF-κB, suggesting that their activation likely modulated the activation of NF-κB in circulation and colon tissues from DSS-treated animals. Given the strong ability of DRE-H to weaken the phosphorylation levels of ERK, JNK and p38, it is therefore reasonable to hypothesize that the mechanism of action of this plant seems to require the involvement of MAPK signaling pathways to inhibit NF-κB activation and ensuing inflammatory events. The MAPK agonist BIM-23A760 was simultaneously injected into animals during DRE application to examine this proposal. After the agonist usage, the effect of DRE-H on protecting animals against UC disappeared, as demonstrated by the measurement of the level of inflammatory cytokines, PAF, MPO, PGE2, and SOD, and the phosphorylated levels of NF-κB, IκB, ERK, JNK, and p38. These data support the mechanism through which DRE can inhibit UC in DSS-administrated rats by blocking MAPK signaling pathways.
Limitations
Firstly, this study only clarified the action and involved the mechanism of total extracts of dandelion root in DSS-induced UC model. The potential bioactive compounds (hydroxybenzoic acid derivatives and hydroxycinnamic acids) need to be further exaxmined. Secondly, there might be other signaling pathways involved in the protection of DRE, which should be also investigated in future studies. Lastly, our study only validated the effect of DRE on DSS-induced UC. The function of this plant on experimental UC triggered by other drugs, including 2,4,6-trinitrobenzenesulfonic acid (TNBS), Oxazolone and acetic acid, remains unknown.
Conclusions
The present study shows that DRE-H treatment protects mice against DSS-induced UC. This protective effect is attributed to its anti-inflammatory and antioxidative roles by inhibiting MAPK pathways and its downstream target – NF-κB. These findings do not only further support the potential application of dandelion for UC but also provide a mechanistic explanation for the protective function of dandelion against UC.