Abstract
Background. Both intraoperative hypotension and hypertension have been reported to increase the occurrence of acute kidney injury (AKI). However, the impact of the intraoperative pulse pressure (PP) on the latter complications remains relatively unknown.
Objectives. To explore whether high intraoperative PP values are associated with postoperative AKI.
Materials and methods. The data for this study come from a prospective cohort study in which patients who underwent abdominal surgery between October 1, 2018 and July 15, 2019 in university hospital in Katowice, Poland were included in the analysis. Pre- and intraoperative data, including blood pressure measurements, were acquired from medical charts. Several PP thresholds were applied: >50, >55, >60, >65, >70, >75, >80, >85, and >90 mm Hg. Additionally, by analyzing the maximal PP during the procedures, the cutoff point for the occurrence of outcomes was estimated. Postoperative AKI was considered as the outcome of the study. Univariable and multivariable analyses were performed to assess PP relationship with AKI.
Results. Four hundred and ninety-four patients were included in the analysis. The AKI was present in 32 (6.5%) cases. The receiver operating characteristic (ROC) curve analysis estimated a cutoff point of >84 mm Hg of maximal PP to be associated with the outcome. The PP values above 80 mm Hg and onward were successfully included in the multivariable statistical models. A model in which PP > 90 mm Hg (odds ratio (OR) = 4.03; 95% confidence interval (95% CI): [1.53; 10.62]) was included, had the best predicting value in predicting hypoperfusion injury (area under the receiver operating characteristics (AUROC) = 0.88). Apart from PP, intraoperative hypotension, presence of chronic arterial hypertension, chronic kidney disease, and procedure duration were independently associated with AKI.
Conclusions. High intraoperative PP may be associated with the occurrence of postoperative AKI. However, the effect of high PP should be confirmed in other noncardiac populations to prove the generalizability of our results.
Key words: acute kidney injury, hemodynamic monitoring, general surgery, pulse pressure
Background
Hypoperfusion-related organ injury is a fairly frequent perioperative complication.1, 2, 3, 4 Intraoperative hypotension (IOH) has been linked with postoperative myocardial injury (MI), acute kidney injury (AKI) and stroke.1, 2, 3 Perioperative Quality Initiative (POQI) consensus statement on intraoperative blood pressure underlines that mean arterial pressure (MAP) below 60–70 mm Hg and systolic blood pressure (SBP) below 100 mm Hg are associated with hypoperfusion-related organ injury and death.4 However, hypertensive events during surgery may also worsen the prognosis, as intraoperative episodes of SBP above 160 mm Hg have been correlated with the risk of myocardial injury and infarction.4 Lastly, diastolic blood pressure (DBP) below 50 mm Hg is also reported to be harmful.5
Although ambulatory pulse pressure (PP) is considered one of the best predictors of cardiovascular risk, it has been poorly investigated in the perioperative period.6 The association between high preoperative PP values and the relationship with postoperative complications (mainly myocardial infarction, AKI and stroke) has been explored mostly in cardiosurgical patient populations. The POQI has called for further research on the matter in noncardiac surgery.7
Objectives
In an exploratory fashion, we sought to verify whether elevated intraoperative PP values are associated with postoperative AKI in the abdominal surgery population.
Materials and methods
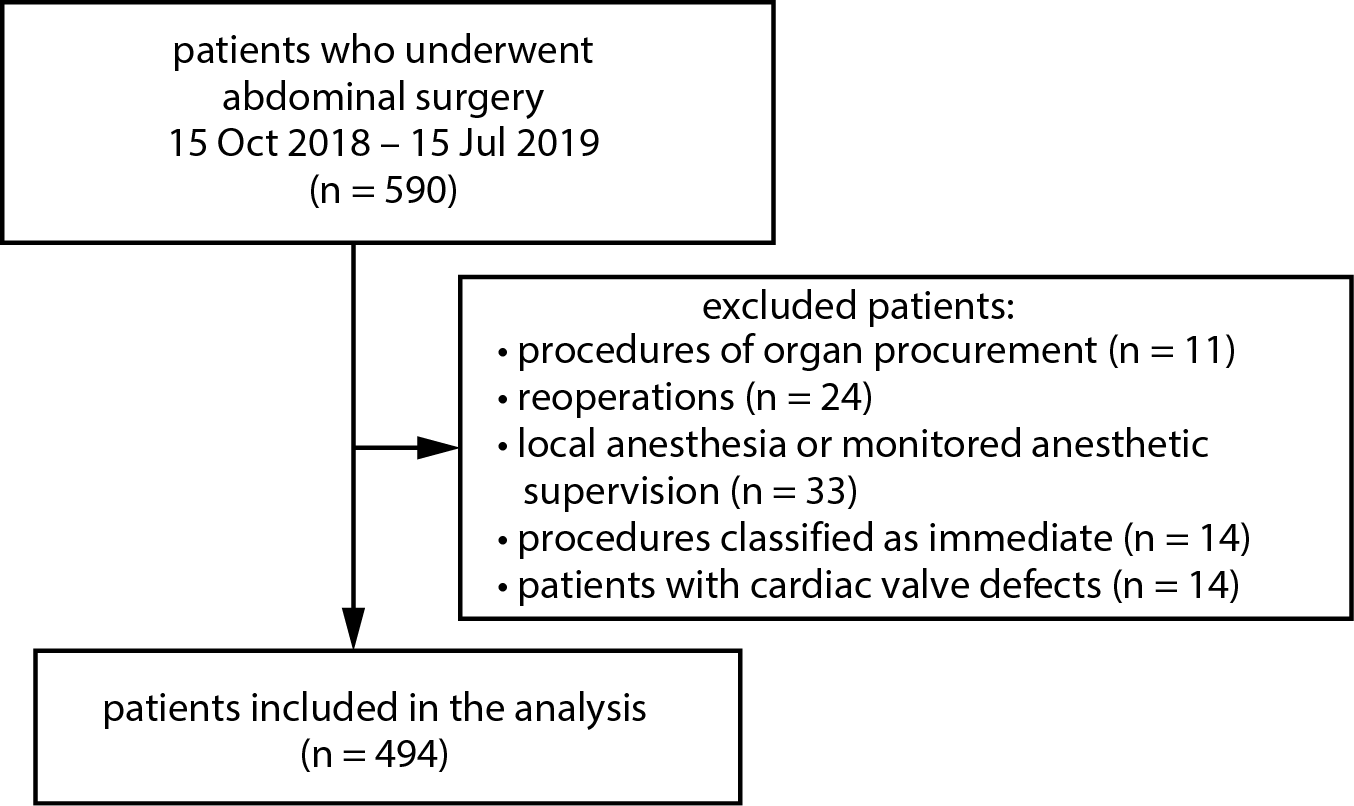
The data used in this study come from a prospective cohort study previously published by our team.8 We screened 576 consecutive patients who underwent abdominal surgery between October 1, 2018 and July 15, 2019,
in a University Hospital in Katowice, Poland. Procedures of organ procurement (n = 11), reoperations (n = 24), procedures performed in local anesthesia or monitored anesthesia supervision (n = 33), procedures classified as immediate according to the National Confidential Enquiry into Patient Outcome and Death (NCEPOD) Classification of Intervention9 (n = 14), and patients with proven cardiac valve defects (n = 14) were excluded from the study (Figure 1). Demographic and medical data were recorded, including sex, age, weight, height, and comorbidities and their pharmacological treatment, according to the International Classification of Diseases (ICD-10) criteria.10 Body mass index (BMI) and Charlson comorbidity index (CCI) were subsequently calculated. Type and duration of anesthesia, as well as type, duration and urgency of surgery were recorded. Perioperative risk was assessed based on an individual patient’s risk, according to the American Society of Anesthesiologists (ASA) physical status (PS) classification,11 and procedural risk, according to the European Society of Cardiology and European Society of Anaesthesiology recommendations.12 Primary arterial hypertension was diagnosed based on medical records.
The SBP and DBP were measured on a nondominant arm using an automated noninvasive oscillometric BP monitoring device (Dräger Infinity Gamma XL; Dräger, Lübeck, Germany) with a cuff of appropriate size, depending on a patient’s arm circumference, and recorded in 5-minute intervals during anesthesia, from the first preinduction measurement until the last measurement during recovery from anesthesia in the operating theater. The MAP values were automatically calculated. Pulse pressure was calculated as the difference between SBP and DBP. The need for norepinephrine (NE) use and its doses, together with intraoperative fluid balance, were analyzed.
Taking into consideration other studies on clinical consequences of abnormal PP, and the fact that PP revolves usually around values of 40 mm Hg, we distinguished following absolute PP thresholds: >50, >55, >60, >65, >70, >75, >80, >85, and >90 mm Hg.5, 13, 14, 15 Additionally, by analyzing the maximal PP during the procedure, the best cutoff point associated with the occurrence of AKI was estimated. We used maximal PP (presented as the median of all maximal PPs recorded among all the patients) and not an average or a median, due to a number of reasons. Firstly, the average value is much more confounded by extreme values of the distribution of numbers. Secondly, choosing maximal PP over average or median PP is better suited for finding a cutoff point for PP that is associated with postoperative AKI. In this study, we explored the role of high PP values; therefore, we naturally sought higher values and tried to find thresholds that would be easily identifiable by a clinician. In contrast, the average or the median value reflects rather a global trend in values and does not bring any specific information.
Moreover, we analyzed the occurrence of high systolic (defined as SBP > 160 mm Hg16), low diastolic (defined as DBP < 50 mm Hg17) and low mean arterial pressure (defined as MAP < 60 mm Hg18). We excluded pre-induction measurements in order to assess only those BP values that occurred during anesthesia.
In the postoperative period, the incidents of AKI were recorded and defined as a serum creatinine increase ≥0.3 mg/dL within 48 h or an increase in serum creatinine by ≥1.5 times baseline, which is known or presumed to have occurred within the prior 7 days.19 This outcome was considered as the endpoint. In addition, incidents of AKI were classified as stages based on Kidney Disease Improving Global Outcomes (KDIGO) guidelines.20
STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) statement was applied for appropriate reporting.21
Statistical analysis was performed using MedCalc statistical software v. 18.1 (MedCalc Software Ltd., Ostend, Belgium). Continuous variables were expressed as median and interquartile range (IQR). Qualitative variables were expressed as absolute values and/or percentages. Between-group differences for quantitative variables were assessed using the Mann–Whitney U test. Their distribution was verified with the Shapiro–Wilk test. The χ2 tests were applied for qualitative variables. The correlation was assessed using Spearman’s rank correlation coefficient. The receiver operating characteristic (ROC) curve analysis was implemented to assess the relationship between AKI and maximal PP. In order to control the potential confounding factors, we used multivariable logistic regression with all variables that achieved p-value <0.1 in the univariable analysis. The Hosmer–Lemeshow test was performed to assess the goodness-of-fit of multivariable logistic regressions. If applicable, odds ratios (ORs) and area under the receiver operating characteristics (AUROC) with 95% confidence intervals (95% CIs) were calculated. All tests were two-tailed. A value of p < 0.05 was considered statistically significant.
Results
A total number of patients included in the analysis was 494, out of which 239 (46%) were male. The median age of participants was 65 years (IQR 46–68). Older age, higher ASA-PS class and higher CCI were found to be significant preoperative risk factors for the occurrence of AKI. Detailed preoperative population characteristics are presented in Table 1, whereas intraoperative population characteristics are presented in Table 2. The primary outcome (AKI) was diagnosed in 32 (6.7%) patients. According to KDIGO criteria, 24 patients (75%) suffered from stage 1 AKI, 5 patients (15.6%) from stage 2 AKI and 3 patients (9.4%) from stage 3 AKI.19 Pre-induction PP was not associated with the outcome (Table 1).
In patients who developed AKI, PP more negatively correlated with DBP than in patients without AKI (Table 3).
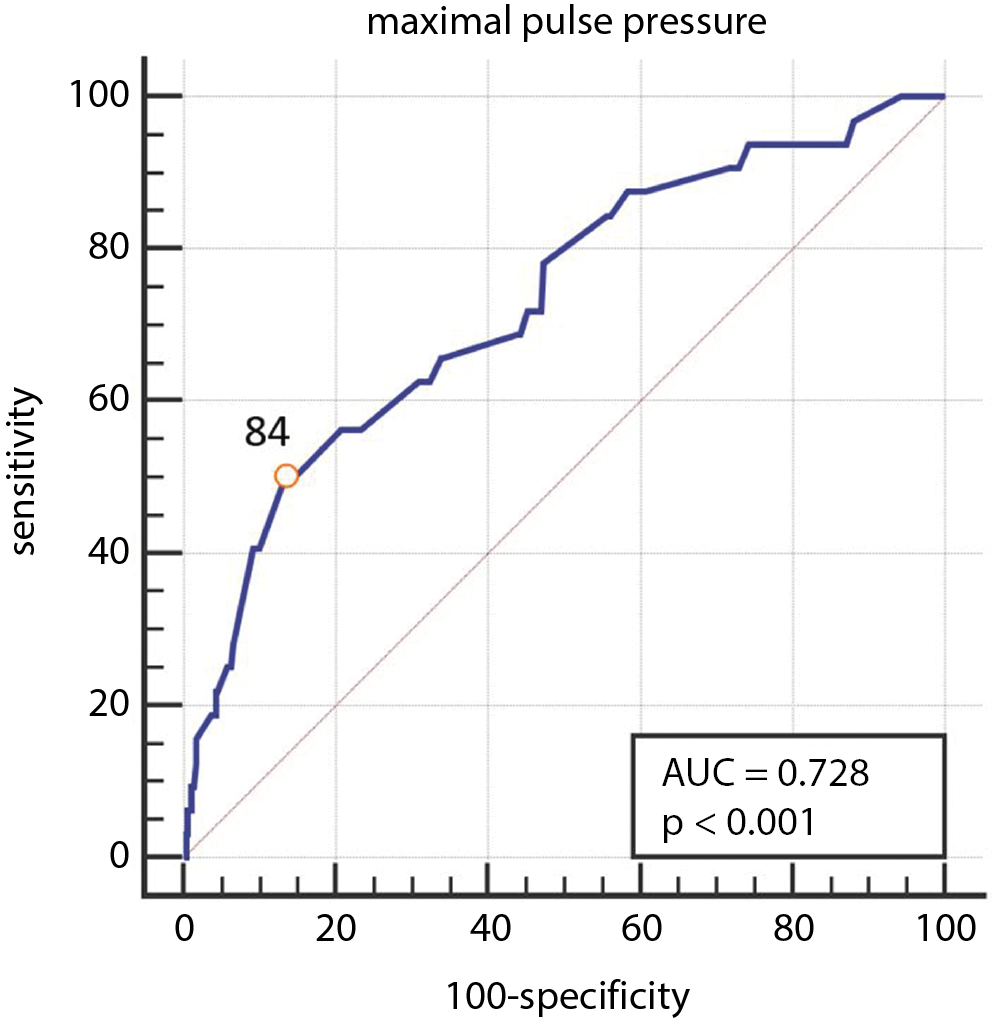
Maximal PP registered over the course of the procedure was associated with the outcome (AUROC = 0.728; p < 0.001), with a cutoff point >84 mm Hg (Figure 2).
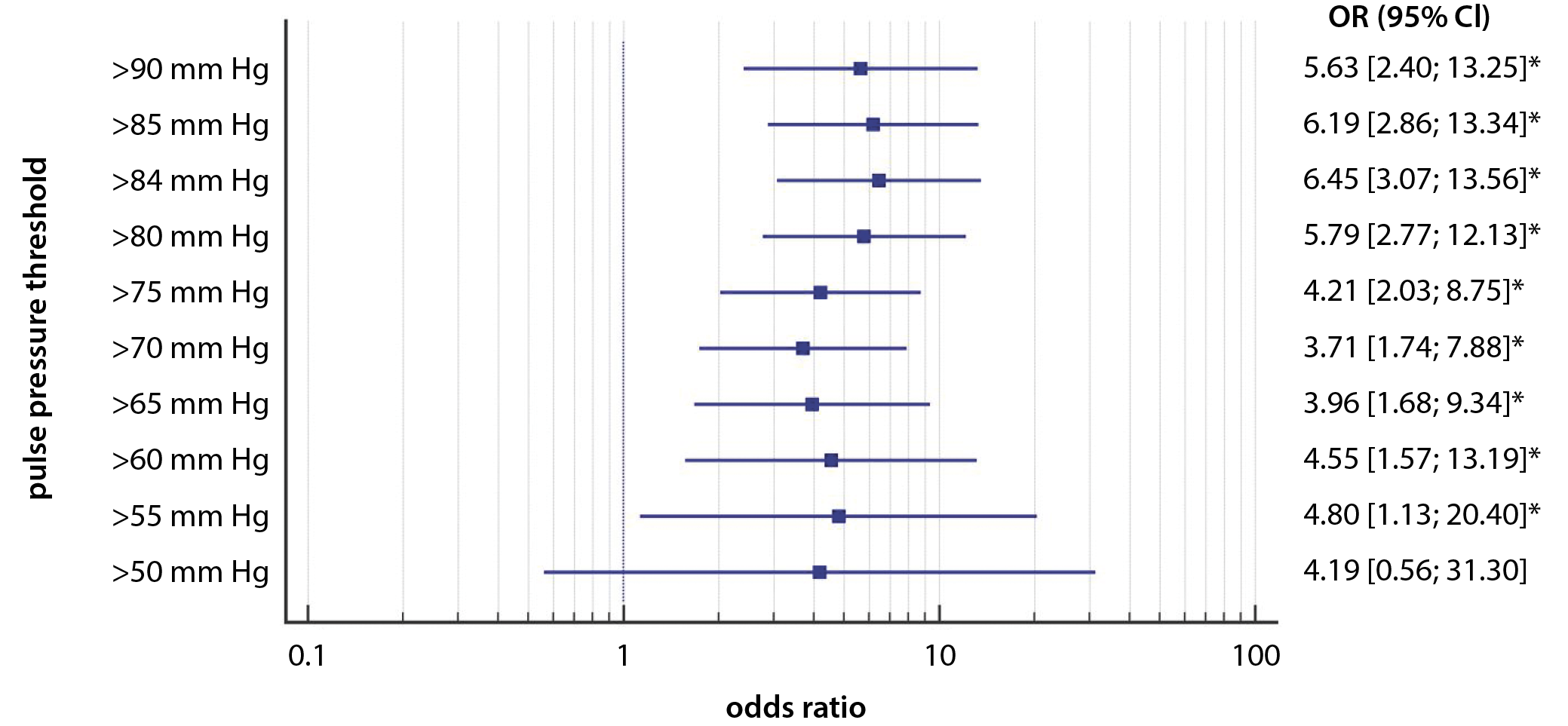
In univariable analyses, all PP thresholds, except for >50 mm Hg, were statistically significant predictors of AKI (Figure 3). In multivariable logistic regressions, PP values >80, >84, >85, and >90 mm Hg were included in the final statistical models. It was discovered that PP > 90 mm Hg predicted AKI with the highest accuracy, even after the adjustment for various confounding factors, including intraoperative hypertension (Table 4). Low DBP (<50 mm Hg) and high SBP (>160 mm Hg) were not significant in the multivariable models.
Discussion
The main finding of our exploratory study is that increasing intraoperative values of PP were associated with the occurrence of postoperative AKI. This association persisted after adjusting for confounding factors (most importantly: high SBP and low DBP). We found a cutoff point of >84 mm Hg of maximal PP to be associated with AKI. In regards to the predetermined thresholds, PP above 80 mm Hg and onward was linked to AKI. Pulse pressure above 90 mm Hg, out of all PP thresholds applied, appeared to be the best predictor of postoperative AKI.
To our knowledge, this is the first study investigating the role of intraoperative PP in abdominal surgery in such a complex manner. It is known that increased ambulatory PP is strongly associated with cardiovascular events, not only in the general population but also in cardiac surgery setting, irrespective of the presence of chronic arterial hypertension.6, 22, 23 Pulse pressure stands as a proxy for general vascular health and reflects cardiovascular risk better than isolated measurements of either SBP or DBP.24 Generally, a value of PP is determined by stroke volume, left ventricle contractility and arterial compliance. Interestingly, pre-induction PP values (a reflection of baseline PP) alone were not significantly related to the outcome. In studies by Abbott et al. and Mitrev et al., it was found that increasing values of ambulatory and pre-induction PP were significantly related to the increased occurrence of postoperative MI and AKI.5, 25 It must be remembered, however, that those studies were performed among cardiac surgery patients with pre-existing cardiac morbidities, and the effect of preoperative PP might have been more significant than in the noncardiac setting. The fact that in our cohort pre-induction PP was not associated with AKI, gave us more space to explore the impact of intraoperative values. Nevertheless, intraoperative PP positively correlated with the pre-induction values. The negative correlation between PP and DBP was especially interesting, since it was 2 times stronger in patients with the compromised outcome. Lowered DBP is known to decrease coronary perfusion and could be associated with the development of hypoperfusion-induced organ injury.16, 26, 27 However, after taking into account low DBP (<50 mm Hg) in multivariate analyses, PP thresholds remained significant and low DBP was not included in the models.
We discovered that patients who experienced AKI exhibited higher values of PP, and the ORs varied, depending on the threshold applied. Contrary to our hypothesis, Ahuja et al., in a large cohort of 23,000 patients, found that PP below 35 mm Hg was linked to postoperative MI and AKI.17 Indeed, in our cohort, the AKI group experienced lower minimal PP compared to the non-AKI group (median 25 mm Hg compared to 30 mm Hg). Low PP is thought to predict cardiovascular events in patients with impaired cardiac function: decreased contractility of left ventricle causes SBP to achieve lower values and negatively impact the value of PP. It must be remembered that Ahuja et al. explored only the lowest values of PP and called for further research regarding high intraoperative PP.17
High PP could influence systemic circulation in numerous ways. First, kidneys have a high resting blood flow. With the increase of PP, perfusion of this organ becomes more pulsatile and it is thought to damage endothelium and smooth muscle and induce shear stress, which can cause plaque to rupture and form thrombosis.28, 29, 30 Additionally, high PP can decrease flow-mediated vasodilation.31 What is also worth mentioning is that increased PP causes aortic lumen to decrease, which results in ventricular-aortic decoupling, characterized by cardiac output that is too great to be accommodated by aortic lumen (leading to the impaired cardiac output with preserved systolic function).5, 32
Limitations
The abovementioned findings should be analyzed with caution due to possible confounding factors. First, the vast majority of patients had their BP measured with the oscillometric method. Due to an imperfect algorithmic method of distinguishing SBP and DBP, such patients have a higher risk of discrepancy between the registered and real BP. The discrepancy, especially in SBP, is more often expressed in patients with stiffer arteries and higher PPs.33 In a study by Kayrak et al., oscillometric measurements led to the underestimation of PP in a group of patients with isolated systolic hypertension (but not in subjects with mixed hypertension).34 Secondly, the true association between high intraoperative PP and AKI is, to a certain extent, determined by the preoperative PP values. Despite the fact that pre-induction PP was not significantly related to the outcome in our analysis, it is possible that intraoperative PP is only a reflection of the overall cardiovascular condition and does not impair organ perfusion in a short-term period (such as the duration of surgical procedure). Thirdly, pre-induction BP value was defined as baseline MAP. It is possible that such measurement does not represent the true baseline, as it could be influenced by stress or premedication. Additionally, the BP measurements were recorded in 5-minute intervals, and therefore, a risk of underrecognition of PP changes exists. Finally, our analysis was restricted to a limited population of abdominal patients, which reduces the generalizability of our results into all noncardiac surgery settings.
Conclusions
High intraoperative PP may be associated with AKI in patients undergoing abdominal surgery. However, the effect of high PP should be confirmed in other noncardiac populations to prove the generalizability of our results.