Abstract
Background. Behçet’s disease (BD) is a chronic inflammatory vasculitis affecting multiple organs. Uveitis is frequently seen in patients with BD, especially in Turkish population.
Objectives. To investigate vascular endothelial growth factor (VEGF) gene polymorphisms along with the levels of VEGF and VEGF receptors in patients with Behçet’s uveitis (BU).
Materials and methods. Fifty-five BD-associated uveitis patients and 30 age- and sex-matched controls were included in this case-control study. The genotypes of the single nucleotide poymorphisms (SNPs): rs2010963 (+405G), rs3025039 (+936T) and rs699947 (−2598A) of the VEGF-A gene were determined using real-time polymerase chain reaction (RT-PCR) and serum levels of VEGF and VEGF receptors were measured using enzyme-linked immunosorbent assay (ELISA).
Results. No associations of the VEGF gene polymorphisms were observed in BD uveitis patients, but arthritis was present in 53.3% of patients not possessing CT genotype in C3025039→T polymorphism (p = 0.024). Although there were no statistically significant differences in serum VEGF-A, VEGF-C and soluble vascular endothelial growth factor receptor-3 (sVEGFR-3) levels (p < 0.05), serum vascular endothelial growth factor receptor-1 (VEGFR-1) and sVEGFR-3 levels were significantly higher in the BD group (p < 0.001 and p = 0.001, respectively). In addition, VEGF-C/soluble vascular endothelial growth factor receptor-2 (sVEGFR-2) ratio was significantly higher (p < 0.001), while VEGF-A/VEGFR-1 and VEGF-C/sVEGFR-3 ratios were significantly lower (p < 0.001 and p = 0.033, respectively) in BD patients compared to controls. Also, VEGF-C/sVEGFR-3 (p = 0.024, r = 0.37) and VEGF-C/sVEGFR-2 (p = 0.020, r = 0.38) ratios were positively correlated with disease duration.
Conclusions. The significant changes in sVEGFR-3 levels and VEGF-C/sVEGFR-3 ratio has shown that lymphangiogenesis processes might take place in the pathogenesis of BD uveitis, and these parameters can be important indicators of evaluation of BD patients with uveitis together with disease duration.
Key words: vascular endothelial growth factor, VEGF gene polymorphism, Behçet’s disease, Behçet’s uveitis
Background
Behçet’s disease (BD) was described by Turkish dermatologist Dr. Hulusi Behçet as a disease with triple symptoms: oral aphthae, genital ulcers and uveitis with hypopyon.1 Behçet’s disease is a systemic immuno-inflammatory vasculitis occurring in young adults that is characterized by endothelial cell dysfunction. The most prominent feature of BD is systemic, dermal and ocular vasculitis with lymphocytic infiltration of vessels with different diameters. Ocular involvement can be the first finding in most BD patients from 2 to 4 years after disease onset, and it is one of the most important causes of morbidity as it may result in blindness.2, 3 It is also more frequent and more severe in Turkish and Japanese BD patients. In a study conducted in Turkey, eye involvement in form of uveitis/retinal vasculitis was reported in 29.1% of patients, while early age of onset and male gender were poor prognostic factors for ocular findings.4, 5
Although many studies have been published since BD was first described, its etiopathogenesis is not fully defined yet. The disease tendency appears to be related to HLA-B gene polymorphisms.6, 7, 8, 9, 10 Human major histocompatibility complex class I chain-related gene A was also shown to be related to the disease pathogenesis.8, 11 However, the exact mechanisms about the genes related to the disease pathogenesis are still obscure.
Angiogenesis is a multistage process controlled by several pro-angiogenic and anti-angiogenic factors. Vascular endothelial growth factor (VEGF) is a potent endothelium-specific cytokine which modulates vasculogenesis and angiogenesis by acting as a major mitogen for endothelial cells. The VEGF plays an important role in the regulation of angiogenesis, wound healing, inflammation, and tumor progression.12 Neutrophils, macrophages and vascular endothelial cells, which are involved in BD pathogenesis, primarily produce VEGF. The main stimulant in VEGF production in systemic and retinal endothelial cells is inflammation induced by pro-inflammatory cytokines. Since VEGF is a potent mitogen for dermal and ocular microvascular endothelial cells, VEGF expression in vascular beds of BD patients may be important in the clinical course of the disease. In our previous study, we indicated that elevated serum VEGF and sVEGFR-1, and – more importantly – VEGF/sVEGFR-1 ratio could play an important role in the development of thrombosis in BD.13 Thus, it has been suggested that plasma VEGF levels in BD may be affected by the course of the disease and may vary depending on the eye involvement or disease activity. There are also studies showing that VEGF is elevated especially in the active stage of BD and high VEGF levels can be associated with loss of vision as an additional risk factor.14, 15
The VEGF gene is highly polymorphic and more than 30 different single nucleotide polymorphisms (SNPs) have been previously reported. These polymorphisms are important due to their effects on VEGF production and their relationship with BD. Studies have shown that both +405G/C (rs2010963) and +936C/T (rs3025039) SNPs reduce VEGF production.14, 15, 16, 17, 18 However, Salvarani et al. reported that VEGF (rs2010963) and VEGF-2549 18bp insertion/deletion (I/D) polymorphisms were associated with BD susceptibility.19 In addition, Nam et al. found that in the Korean population, the VEGF (rs2010963) polymorphism was associated with the development of ocular inflammation in BD.20
Objectives
In this study, we aimed to evaluate: (1) the serum levels of VEGF-A, VEGF-C, vascular endothelial growth factor receptor-1 (VEGFR-1), soluble vascular endothelial growth factor receptor-2 (sVEGFR-2) and soluble vascular endothelial growth factor receptor-3 (sVEGFR-3), and correlation of their levels with the severity of BD; (2) the influence of the presence of polymorphisms (rs2010963, rs3025039 and rs699947) on the regulatory region of the VEGF gene, and to emphasize the importance of the detected polymorphisms in terms of disease development and severity in BD patients with ocular involvement in the Turkish population.
Materials and methods
The study protocol was approved by the Institutional Local Ethics Committee of Ankara Numune Training and Research Hospital, Ankara, Turkey (approval No. E-17-1612). The research protocol complies with the Declaration of Helsinki and written informed consent was obtained from all participants.
The present study is a cross-sectional study carried out in 55 Behçet’s uveitis (BU) patients and 30 age/sex-matched controls admitted to Rheumatology and Eye Clinics of Gulhane and Numune Training and Research Hospital, Ankara, Turkey.
Criteria from the International Study Group for BD were used to evaluate BD patients.21 The disease activity was assessed with the Turkish version of BD Current Activity Form (BDCAF).22 The BDCAF score was calculated by adding up the scores on each item, and ranged from 0 to 12. Behçet’s uveitis patients were defined as the ones with edema, non-perfusion, neovascularization, and atrophy of the retina and/or optic disc.23
Overnight fasting blood samples were obtained from the antecubital vein and collected in blood tubes containing clot activator for serum separation. Sera were obtained by centrifugation at 3000 g × 10 min. For single nucleotide polymorphism (SNP) analysis, whole blood samples were collected into ethylenediamine tetraacetic acid (EDTA)-containing tubes.
Detection of serum levels
of the study parameters
Serum VEGF-A(E-EL-H0111), VEGF-C(YLA0798HU), VEGFR-1(E-EL-H1087), sVEGFR-2(YLA1212HU), and sVEGFR-3(YLA1379HU) levels were measured using quantitative enzyme-linked immunosorbent assay (ELISA) kits (Elabscience, Wuhan, China for VEGF-A and VEGFR-1; Shanghai BiotechCo., Shanghai, China for VEGF-C, sVEGFR-2 and sVEGFR-3). The measurements were carried out using ELISA plate reader Bio-Tek Synergy HT (Biotek Instruments Inc., Winooski, USA). Intra- and inter-assay coefficients of variation (CVs) were 4.69% and 4.2%, respectively, with a sensitivity of 18.75 pg/mL for VEGF-A, <8%, <10% and 10.58 pg/mL, respectively, for VEGF-C, 5%, 4% and 75 pg/mL, respectively, for VEGFR-1, <8%, <10% and 22 pg/mL, respectively, for sVEGFR-2, and <8%, <10% and 114 pg/mL, respectively, for sVEGFR-3.
Selection of gene regions and SNP analysis
The reference sequences of the VEGF gene to be examined were downloaded from the National Biotechnology Information Center database of the National Library of Medicine within the American National Institutes of Health (NIH; Bethesda, USA). The primer pairs were specifically designed for the respective region of the VEGF-A gene for each SNP from the gene promoter region as rs2010963, rs3025039 and rs699947. The DNA concentration and purity were measured using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Munich, Germany). Genotyping of the relevant SNPs were performed using Taqman probes (Sigma-Aldrich, St. Louis, USA). The polymerase chain reaction (PCR) was performed for each sample as follows: 10 ng of genomic DNA, mixture of 10 µL of Probe Master Kit (04707494001; Sigma-Aldrich), 1 µL Reagent mix (TIB MolBiol, Berlin, Germany) and 4 µL diH2O were mixed to carry out polymerase chain reaction described below.
The amplification of DNA regions was carried out with LightCycler 480 (Roche Diagnostics, Basel, Switzerland) real-time PCR (RT-PCR) device. An initial denaturation of 1 min at 95°C was followed by 45 amplification cycles with 5 s at 95°C, 15 s at 60°C and 15 s at 65°C.
Statistical analyses
Statistical analyses were performed using IBM SPSS Statistics for Windows v. 21.0 (IBM Corp., Armonk, USA).
Categorical variables were presented as numbers or percentages and compared using the χ2 or Fisher’s exact test, as appropriate. Numerical variables were tested for normality using histogram and Kolmogorov–Smirnov test. Normally distributed variables were presented as mean (standard deviation (SD)) and variables which were not normally distributed were presented as median (interquartile range (IQR)). To compare numerical variables, the Mann–Whitney U test was used when the variables were not normally distributed, and Student’s t-test was used for normally distributed variables. The correlations between VEGF-A, VEGF-C, VEGFR-1, sVEGFR-2, sVEGFR-3, VEGF-A/VEGFR-1, VEGF-A/sVEGFR-2, VEGF-C/sVEGFR-2, VEGF-C/sVEGFR-3, and patient features such as age, age at diagnosis, and disease duration were analyzed using the Spearman’s correlation test. An increase in one parameter as the other parameter decreased was considered as negative correlation, and an increase in one parameter as the other parameter increased was considered as positive correlation. Genotype distribution and allele frequencies of patients with the BD group and control group were analyzed using the χ2 test. Odds ratios with 95% confidence intervals (95% CIs) were used to describe the risk of uveitis. The level of statistical significance was set at p < 0.05.
Results
There was no statistically significant difference between the 2 groups with respect to age and gender. Seventy-three percent (40) of BD patients and 70% (21) of controls were male. The mean age of the BD patients was 37.8 ±9.1 years while that of controls was 39.2 ±11 years.
Clinical features and disease manifestations presented by BD patients are summarized in Table 1. Serum levels of VEGF-A, VEGF-C, VEGFR-1, sVEGFR-2 and sVEGFR-3, and parameters calculated using factor receptor ratio among patients and control groups are presented in Table 2. According to these data, there were no statistically significant differences in mean serum VEGF-A and VEGF-C levels between the 2 groups. On the other hand, serum VEGFR-1 and sVEGFR-3 levels were statistically significantly higher in BD patients compared to controls, while there was no such difference in sVEGFR-2 levels. In addition, VEGF-C/sVEGFR-2 ratio was significantly higher (p < 0.001), while VEGF-A/VEGFR-1 and VEGF-C/sVEGFR-3 ratios were significantly lower (p < 0.001 and p = 0.033, respectively) in BD patients compared to controls.
When groups of patients with certain clinical manifestations were compared, VEGF-C/sVEGFR-2 ratios were significantly lower while VEGF-A/sVEGFR-2 ratios were above but close to the borderline statistical significance in patients with pathergy when compared to patients without this symptom (125 compared to 151, p = 0.048; 20.5 compared to 53.2, p = 0.053, respectively). The VEGF-A levels and VEGF-A/VEGFR-2 ratios were also lower in patients with arthritis; however, no statistically significant difference was obtained (p = 0.080 and p = 0.086, respectively). Also, considering the relationship between polymorphisms and clinical findings of BD, no arthritis was observed in patients with CT genotype, while arthritis was only present in 53.3% of patients without CT genotype in C3025039→T polymorphism (p = 0.024).
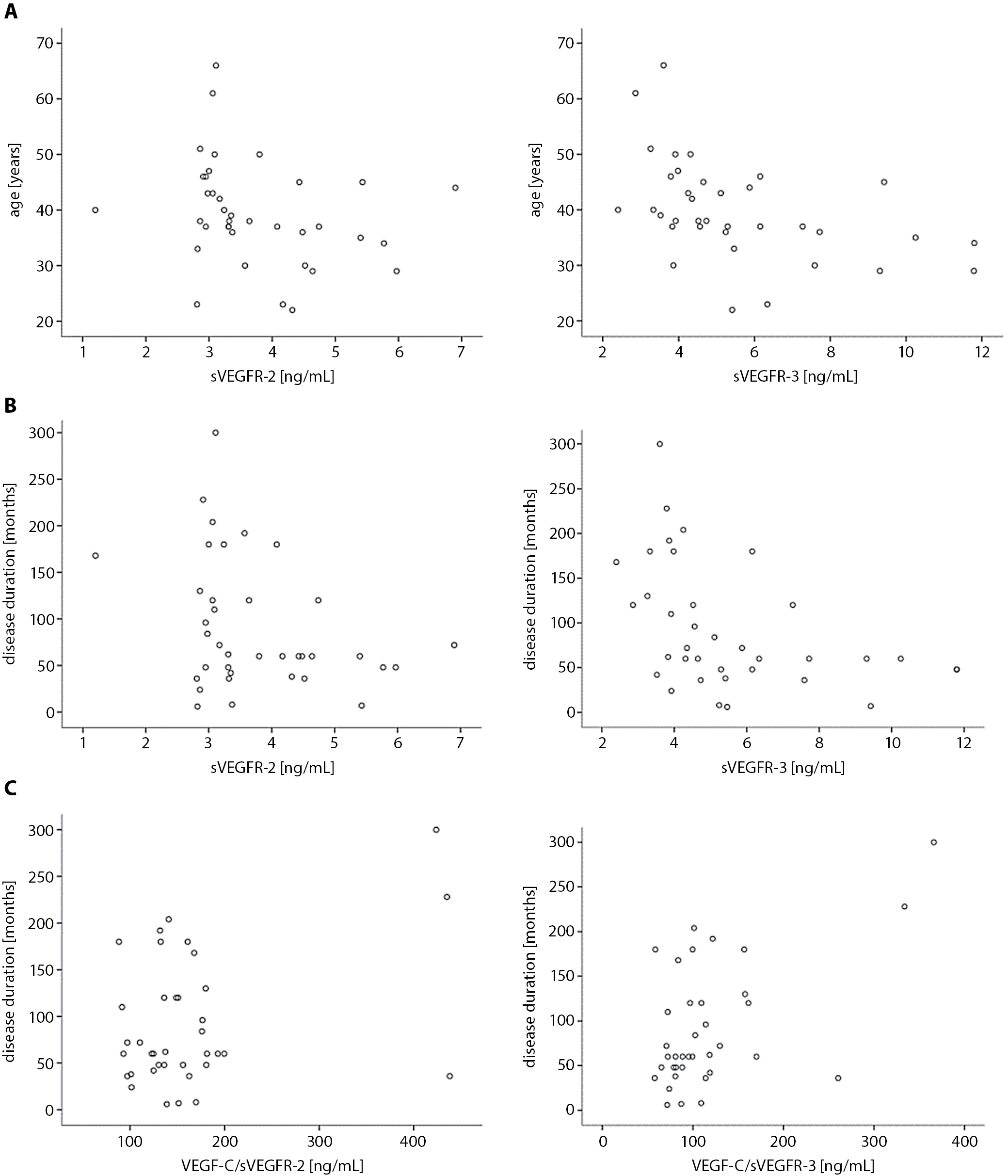
There was a moderately negative correlation between serum sVEGFR-2 and sVEGFR-3 levels and age (p = 0.032, r = −0.35 and p = 0.003, r = −0.47, respectively) and disease duration (p = 0.048, r = −0.33 and p = 0.006, r = −0.44, respectively). In addition, VEGF-C/sVEGFR-3 (p = 0.024, r = 0.37) and VEGF-C/sVEGFR-2 (p = 0.020, r = 0.38) were positively correlated with disease duration (Table 3 and Figure 1).
Genotype and allele frequencies of patient and control groups were calculated for detected SNPs. The genotype and allele frequencies of the 3 selected polymorphisms, reported to regulate the production of VEGF, were not significantly different between BD and controls. The presence of any genotype or allele did not show any increased disease risk (Table 4).
In the evaluation of treatment patterns of patients, it was observed that only 25 patients were receiving colchicine alone, while the rest of them were receiving combined therapy including any combination of 2 medication from the following: cortisone, colchicine and azathioprine. Twelve patients out of the rest were receiving colchium + cortisone treatment, while 18 were receiving colchium + azathioprine treatment.
Discussion
The most prominent feature of BD is systemic vasculitis and endothelial dysfunction, while the most prominent feature of uveitis is the occurring ophthalmologic complication.24 A considerable evidence suggests that VEGF is also upregulated by pro-inflammatory cytokines, which participate in the pathogenesis of chronic ocular inflammation in BD.25 It is also known that the VEGF gene is highly polymorphic.26, 27 Although there are many studies evaluating the relationship between different complications and clinical presentations of BD patients and VEGF levels, the studies investigating the polymorphisms playing a role in the pathogenesis of BD are rare.19, 20 Considering vasculitis as the pathological lesion underlying most of the clinical findings and incidence of uveitis in Turkish BD patients, we performed the first study in Turkish BD population, aiming to investigate the levels of different VEGFs and VEGFRs, as well as the presence of any genotype or allele related to VEGF.
This is also the first study investigating serum angiogenesis, lymphangiogenesis mediators (VEGF-A, VEGF-C) and corresponding receptor levels (VEGFR-1, sVEGFR-2, sVEGFR-3) simultaneously. Considering uveitis and retinal vasculitis as ocular manifestations in patients with BD, the increased synthesis of VEGF in the retinal cells is crucial in the development of ocular neovascularization.28 From this point of view, several studies revealing increased levels of VEGF in patients with BD in active stage of ocular disease have been performed.13, 14, 15, 16, 29 In these studies, there was a critical difference but no significant correlation between VEGF serum levels of BU patients and normal control.30, 31 On the other hand, in a recent study aiming to investigate VEGF levels in the aqueous humor of patients with BU and Fuchs’ uveitis syndrome, Simsek et al. observed a significant difference only between Fuchs’ uveitis syndrome and control subjects.32 Unlike the findings of Yalçındağ et al. and Ozdamar et al., but compatible with Simsek et al., mean serum VEGF-A levels were not statistically different in our study. Also, VEGF-C levels were not significantly different in BD patients compared to controls in our study.30, 31, 32 This might be due to the relatively small number of patients in the study group. In addition, considering the fact that the vascular disorder is not the main pathology in most patients with uveitis, and since endothelial dysfunction is a complex, multistep mechanism, VEGF levels may not change directly in the BD group like ours, where only BU comes to the fore. Also, uveitis is rarely known to be progressed together with neovascularization. Considering the overproduction of VEGF, even in the absence of neovascularization, it has been suggested that VEGF plays a pro-inflammatory role rather than an angiogenic role in the pathogenesis of uveitis. Moreover, the increase in VEGF levels may also be due to pro-inflammatory cytokines released from the area of inflammation.32, 33
In current literature, there is lack of studies evaluating VEGF-C levels in both uveitis caused by BD and/or other diseases/pathologies. This situation is mainly due to the unique lymphatic circulation of the eye. Although there is no known lymphatic outflow from the eye, recent studies have shown that corneal limbus, ciliary body, lacrimal gland, orbital meninges and extraocular muscles possibly contain lymphatic vessels, and that the choroid might have a lymphatic-like system after the novel lymphatic endothelial markers (e.g., LYVE-1, podoplanin) and lymphangiogenic factors (e.g., VEGF-C) have been proposed to be found in the choroid.34 The presence of lymphatic-like features may be related to clinical implications in inflammatory eye diseases like uveitis.35 Based on this, we evaluated the levels of VEGF-C and its soluble receptors, sVEGFR-2 and sVEGFR-3, in BU patients.35 In our study, we found a significant difference in VEGF-C and sVEGFR-2 levels, while there was only a significant but slight difference in sVEGFR-3 levels between patients and control subjects. Contrary to our findings, Nakao et al. found a significant increase in VEGF-C but not in VEGFR-2 or VEGFR-3 in a study evaluating choroidal neovascularization from uveitis.34, 36 Another report demonstrates both VEGF-C and sVEGFR-3 levels as below the detection limit in vitreous samples in a study group including uveitis patients.37 On the other hand, various studies aimed to assess the relationship between other inflammatory pathologies of the eye and lymphangiogenesis. However, the complete agreement between the results of these studies and the detailed role of the VEGF-C/VEGFR-3 or VEGF-C/VEGFR-2 pathway has not been shown yet.38 In our study, we found a significant difference in VEGF-C and sVEGFR-2 levels, while there was only a significant but slight difference in sVEGFR-3 levels between BD patients and control subjects. As a result, there was only a barely significant difference in the VEGF-C/sVEGFR-3 ratio between the BD group and control group. What is more, there was no change in VEGF-C levels in the BD group, while sVEGFR-3 levels increased significantly. Thus, it is of critical importance to evaluate VEGF-C values together with VEGF-C/sVEGFR-3 ratio, as sVEGFR-3 is known to function purely in lymphangiogenesis to counterbalance the VEGF-C increase. Since sVEGFR-3 levels were significantly higher in the BD group in our study, lower VEGF-C/sVEGFR-3 ratios were thought to be individual indicators of the limitation of lymphangiogenesis. On the other hand, sVEGFR-2 plays a key role in both angiogenesis and lymphangiogenesis. That is why we evaluated the changes in both VEGF-A/sVEGFR-2 and VEGF-C/sVEGFR-2 ratio, and had the opportunity to co-evaluate angiogenesis and lymphangiogenesis in the BD group. The obtained data have shown that, considering the receptors, sVEGFR-2 was the only parameter that did not show any significant change. Although there was no change in any of VEGF-A, VEGF-C and sVEGFR-2 levels, there was a significant decrease in VEGF-C/sVEGFR-2 levels. This shows that even though factor levels do not increase in BD patients, the angiogenesis process was only regulated by VEGF-A/sVEGFR-1 ratio, while lymphophiogenesis have been regulated by decrease in both VEGF-C/sVEGFR-2 and VEGF-C/sVEGFR-3 ratios. Besides the slightly significant decrease in VEGF-C/sVEGFR-2 ratio, the limited increase in sVEGFR-3 and borderline significant decrease in VEGF-C/sVEGFR-3 ratio in the BD group can be accepted as a trigger of limited lymphangiogenesis. Apart from this, the significant increase in VEGFR-1 levels and significant decrease in VEGF-A/VEGFR-1 ratio in favor of angiogenesis suppression in the BD group. It also shows the importance of evaluation of sVEGFR-2 levels in conjunction with VEGFR-1 and sVEGFR-3 levels in assessing angiogenesis and lymphangiogenesis. Our data have also shown that the relevant receptors do not always change their levels in the same direction. In clinical situations that do not emphasize significant changes in VEGF levels like our BD group, the evaluation of the growth factors together with their relevant receptors will provide a better understanding of clinical presentation and development of complications. Furthermore, the changes in opposite directions in levels of different receptors reacting with the same factor show that these receptors can be affected by different determinants. Although this study mainly aims to evaluate the process of angiogenesis and lymphangiogenesis in patients with BU by evaluating VEGFs and their receptor levels, the results have also revealed the importance of determinative factors in altering the level of VEGF receptors in disease progression. Inflammation is one of these major determinants. When the importance of inflammation in progression of vascular pathologies is considered, our study has proven that the inflammation can be a key factor in the alteration of receptor levels. In this manner, the present study can influence and guide the future studies. Furthermore, we suggest that anti-VEGF therapies, which are used quite widely in our era, should not focus solely on the changes in VEGF levels, but also on the receptor levels and related factor/receptor ratios in order to estimate the counterbalancing rate of the relevant factor by its receptors.
In the study by Kamoun et al., serum VEGF levels did not correlate with the age of BD patients or with the disease duration. Although our findings are compatible with the mentioned study in terms of receptor levels and ratios, we observed a negative correlation between serum sVEGFR-2 and sVEGFR-3 levels and age and disease duration, as well as a positive correlation between VEGF-C/sVEGFR-3 and VEGF-C/sVEGFR-2 ratios with disease duration.16 The fact that sVEGFR-2 and sVEGFR-3 levels show negative correlation with the age and disease duration may suggest that this relationship is related not only to the disease process. However, the positive correlation between VEGF-C/sVEGFR-3 and VEGF-C/sVEGFR-2 ratios only with disease duration is a particularly important and novel finding. Considering the significant decreases in VEGF-C/sVEGFR-2 ratio and VEGF-C/sVEGFR-3 ratio in the patient group may cause a dilemma. Significant correlation of both ratios in the same direction is sufficient to associate these parameters directly with the disease duration. In addition, as a parameter most significantly correlated with disease duration, VEGF-C/sVEGFR-3 ratio demonstrates that the evaluation of the lymphangiogenesis process is important, especially in the follow-up of the disease period in patients with BU.
Since vasculitis is the generally accepted pathology in BD and the change in VEGF levels is revealed in several studies, it is assumed that BD may occur more frequently in the carriers of the VEGF gene polymorphism. There is a limited number of studies evaluating different SNPs of the VEGF which have only been reported in Italian, Korean and Tunisian BD patients.16, 19, 20 Based on these studies, we found it appropriate to evaluate 3 different SNPs that we considered to be common and important (rs2010963 (+405 G), rs3025039 (+936 T) and rs699947 (−2598 A)). In Italian BD patients, −634 C/G, +936 C/T polymorphisms and an 18 bp I/D genotype at −2549 allele of the VEGF promoter region were selected by Salvarani et al., and they showed that the carriers of the −634 C and −2549 I alleles were susceptible to develop BD.19 In another study performed by Kamoun et al., although there was no association between VEGF polymorphisms and the susceptibility to BD, a positive association between VEGF 18bp I/D polymorphism and ocular involvement was found, and lower frequency of the 18 bp I/I genotype was observed in patients with ocular inflammation among patients with severe BD.16 Moreover, the distribution of VEGF serum level among the genotypes showed that VEGF level was significantly higher in severe BD patients for VEGF −634 GG and 18 bp D/D genotypes. On the other hand, Nam et al. reported that there was no association between VEGF gene polymorphisms and BD (−2578 C/A, −1154 G/A, −634 C/G, and −936 C/T alleles were evaluated); however, −634 C/C genotype polymorphism, which had been associated with a lower production of VEGF, showed a slight tendency to be protective against the ocular inflammation in BD.20 Our data were in accordance with those obtained in the Korean population; however, they contradict the reports in the Italian and Tunisian BD populations. The differences between these reports may be explained by the heterogeneity of the genetic background of different populations studied and the disequilibrium between polymorphisms within regions. In the present study, there was no association between the 3 VEGF gene polymorphisms and BU in Turkish patients. Moreover, when VEGF levels were considered in our study, it was not surprising that we obtained similar clinical presentations with the relevant SNPs.
Different treatment strategies were applied to the patients included in the study. Drugs of choice were cortisone, colchicine and azothioprine. Only 25 patients were receiving colchicine alone, while the rest of them were receiving combined therapy including any two of these drugs. Colchicine is known to inhibit the chemotactic activity of neutrophils and its activity in vasculitis is likely associated with the downregulation of cell surface adhesion molecules and decreased neutrophil adhesion and migration. Corticosteroids have an inhibitory effect on VEGF gene expression and circulating VEGF levels in a dose-dependent manner.39, 40 To our knowledge, there are no studies showing the direct effect of azathioprine treatment on VEGF. When the antiangiogenic effects of colchicine and cortisone are considered, VEGF levels are expected to be lowered in patients receiving the treatment. On the other hand, it was noticed that the statistically significant difference between patients with uveitis and controls was observed in VEGFR-1 levels rather than VEGF-A levels. Considering all the information, it is hardly likely for the treatment patterns to create a significant difference directly on VEGF levels.
Limitations
The present study has some limitations. First, our study had a relatively small sample size. Second, half-lives of circulating factors or receptors were not evaluated in the study since only immediate levels of these parameters were assessed. Third, since this was a cross-sectional study, some of our patients were already taking different types of mostly anti-inflammatory medications for the treatment of BD symptoms; therefore, we could not totally neglect the effects of these medications on VEGF levels or inflammatory markers. Fourth, although so many SNPs associated with VEGF have been reported to date, we were only able to evaluate 3 of them that were supposedly more related to our patient population.
Conclusions
In conclusion, in contrast to the results of the Italian and Tunisian population, no associations with the VEGF gene polymorphisms were observed in Turkish BU patients. The differences in genetic background of different populations and composition of clinical presentation, as well as complications of patient groups in which SNP analysis is performed seem to be especially prominent. This study highlights the need for a further investigation of the role of VEGF polymorphisms and VEGF serum levels in BD susceptibility and its clinical manifestations. Thus, because lymphatic system contributes to the pathogenesis of immune diseases, lymphatic-targeted drug design may provide new agents for the treatment of uveitis.