Abstract
Background. Puerarin demonstrates a protective effect in many cardiovascular diseases. However, the role of puerarin in acute myocardial infarction (AMI)-induced injury and the exact molecular mechanisms are not fully understood.
Objectives. To investigate whether puerarin pretreatment improves cardiac function and to study the mechanism of action of puerarin.
Materials and methods. Thirty rats were grouped into sham group, AMI group and AMI+puerarin (PUE) group at random (n = 10 per group). Except for the sham group, a model of AMI was established via left anterior descending artery ligation. The PUE group received puerarin 120 mg/(kg × day) for 7 days before the operation. Echocardiography was used for evaluation of cardiac function in rats and TUNEL staining for measuring myocardial apoptosis. The expression levels of p-PI3K, t-Akt, p-Akt, Bax, Bcl-2, and cleaved caspase-3 proteins were measured with western blot.
Results. Compared to the sham group, the AMI group demonstrated poor cardiac function and decreased p-PI3K, p-Akt and Bcl-2 proteins levels, while Bax, cleaved caspase-3, and myocardial apoptosis levels increased. Compared with the AMI group, the PUE group showed significant improvement in cardiac function and increased protein expression of p-PI3K, p-Akt and Bcl-2, while Bax and cleaved caspase-3 levels decreased and myocardial apoptosis was attenuated.
Conclusions. Puerarin pretreatment in AMI can effectively improve cardiac function by inhibiting myocardial apoptosis. The molecular mechanism of this protective effect may be mediated by activating the PI3K/Akt pathway in cardiomyocytes.
Key words: puerarin, cardiac function, PI3K/Akt signaling pathway, acute myocardial infarction, myocardial apoptosis
Background
As a myocardial perfusion obstructing disease, acute myocardial infarction (AMI) remains the leading cause of morbidity and mortality worldwide.1 Myocardial apoptosis is a form of cellular death in cardiomyocytes, which leads to the loss of cardiomyocytes and participates in myocardial injury during myocardial infarction (MI).2 At present, many articles have shown that myocardial apoptosis is the chief cell injury in AMI and plays an important role in reducing cardiac function, while the inhibition of myocardial apoptosis is beneficial in preventing and treating myocardial injury caused by ischemia and hypoxia.3, 4, 5 Nevertheless, the molecular mechanisms of cardiomyocyte apoptosis in AMI are complex and diverse. A previous study found that AMI-mediated myocardial apoptosis via inhibition of PI3K/Akt signaling is one of the main causes of progressive cardiac dysfunction.6 Therefore, it is reasonable to propose that intervention measures to reduce myocardial apoptosis by activating this signaling pathway can protect the heart from AMI-related dysfunction and improve heart function.
Puerarin is the major isoflavone extracted from Radix puerariae, which can dilate blood vessels, improve microcirculation, increase blood flow, reduce blood pressure, and prevent coronary artery disease.7 Studies have found that puerarin has a positive preventive and therapeutic effect on diabetes, hypertension, arteriosclerosis, and cerebral ischemia.8 However, effects of puerarin on cardiomyocyte apoptosis and cardiac function in AMI rat models and its exact molecular mechanisms are not clear, and no relevant research currently exists. Thus, facilitating the realization of the relationship between puerarin and myocardial apoptosis will be helpful for the prevention and therapy of AMI.
Objectives
Therefore, we investigated the effects of puerarin pretreatment on cardiomyocyte apoptosis and the PI3K/Akt pathway in an AMI rat model to elucidate the underlying protective mechanism of puerarin.
Materials and methods
Animal preparation
Animal experiments were conducted based on the guidelines for the Care and Use of Experimental Animals and were approved by the Animal Use Ethics Committee of Guangxi Medical University, Nanning, China. Our study was conducted in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Thirty Sprague Dawley (SD) rats (male, weight 250–300 g) were purchased from the experimental animal center of Guangxi Medical University. The rats were kept in a room with temperature of 23 ±2°C and humidity of 50–60%, and sufficient food and water were guaranteed for a 12 h/12 h light-dark cycle.
Grouping and modeling
Thirty SD rats were randomly divided into 3 groups: sham operation group, AMI group and AMI+puerarin (PUE) group, with 10 rats in each group. With reference to the method described by Zhang et al., in the PUE group, 120 mg/(kg × day) puerarin was injected intraperitoneally for 7 days before the AMI model was established.9 This dose has been shown to improve cardiac function in rats with AMI. The AMI model was established by ligating the left anterior descending (LAD) coronary artery following the method described by Curaj et al.10 In brief, 1% pentobarbital sodium (40 mg/kg) was intraperitoneally injected into rats for anesthesia. Endotracheal intubation was then performed and breathing was assisted by a ventilator, followed by left thoracotomy at the 3rd and 4th ribs to expose the heart. Next, a 5-0 polypropylene monofilament suture was placed around the LAD, which was 1–2 mm away from the left auricular tip. After ligation, pale staining of the left ventricular anterior wall and apex, transient weakening of heartbeat, and ST-segment elevation of related ECG leads were seen, which verified a successful performance of LAD occlusion. The chest layers were closed, and the rats were put on a thermostatic blanket until they woke up from anesthesia. For control, the rats in the sham group underwent a similar thoracotomy without coronary artery ligation.
Echocardiography
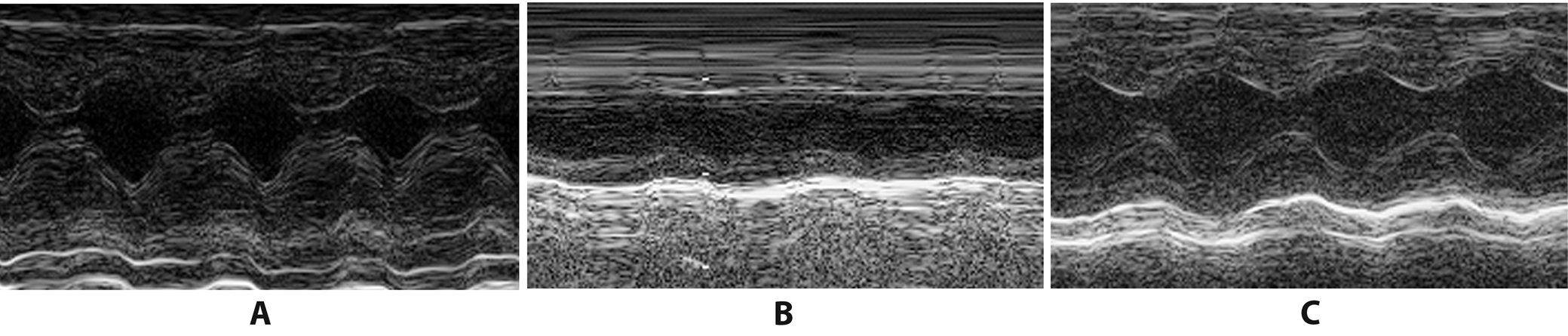
Three days after the operation, we evaluated cardiac function in the 3 groups of rats. The transthoracic echocardiographic study was conducted by an ultrasound physician blind to this research. The left ventricular ejection fraction (LVEF), left ventricular fractional shortening (LVFS), left ventricular end-systolic diameter (LVESD), and left ventricular end-diastolic dimension (LVEDD) were measured using a Hewlett-Packard Sonos 7500 ultrasonic instrument with a 12.0 MHz probe (Philips Technologies, Andover, USA). The measurements were averaged over 3 cardiac cycles.
Sample collection and processing
After echocardiography, 2 mL 10% KCl was injected via the tail vein into rats to arrest the heart in diastole and to enable immediate harvesting. Rat death was determined by respiration and cardiac arrest, loss of nerve reflexes, pupil dilation, and muscle relaxation. Then, the ventricles were infused with cold saline until the rinsing solution was not stained. The atria and large vessels were separated, followed by separation of the left and right ventricles (including the ventricular septum). Next, parallel to the atrioventricular sulcus, the left ventricle was laterally divided from the midpoint of the long axis into apical and basal parts. The apical part was immediately frozen in liquid nitrogen and then transferred to −80°C storage for subsequent western blot detection. Furthermore, the basal part was fixed in 4% paraformaldehyde for 24 h and cut into 4-μm slices after paraffin embedding for terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining.
Determination of cardiomyocyte apoptosis
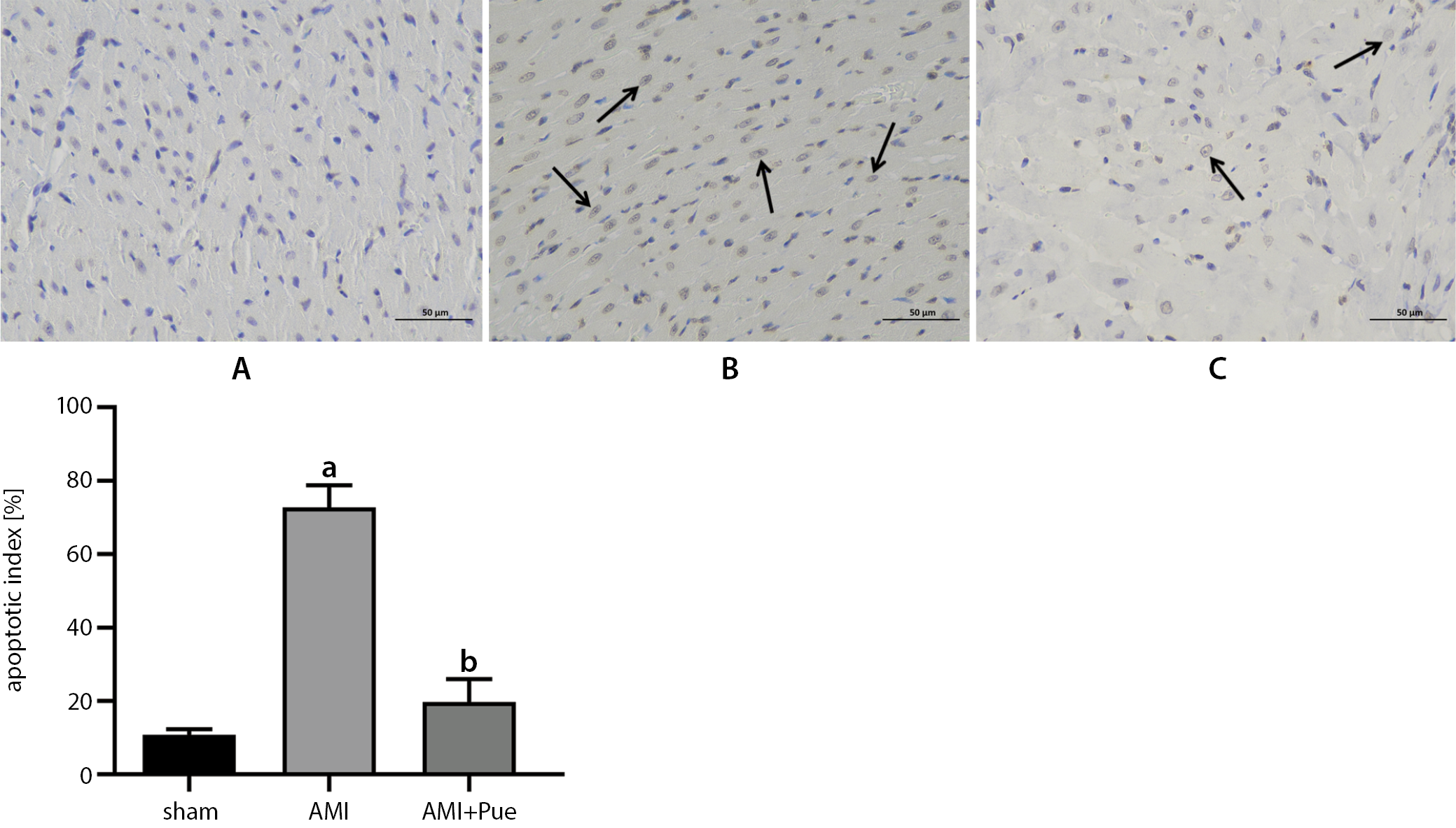
Cardiomyocyte apoptosis was detected using the TUNEL apoptosis detection kit (Roche, Indianapolis, USA) manufacturer instructions. As observed by light microscopy, the normal nucleus was light blue, while the apoptotic nucleus was yellowish brown (TUNEL-positive). In each slice specimen, 20 non-overlapping regions (magnified ×400) were randomly selected to calculate the total number of cardiomyocytes and the number of apoptotic cardiomyocytes in the infarcted and marginal regions. The apoptotic index (AI) was calculated as:
Western blot detection
Total myocardial proteins were extracted from the apex portion with protein lysis buffer, and the bicinchoninic acid (BCA) assay was used to determine the protein concentration. Next, equivalent protein samples were separated using SDS-PAGE assay and then electrically transferred to polyvinylidene fluoride (PVDF) membrane (Merck Millipore, Burlington, USA). The membrane was blocked with 5% bovine serum albumin (BSA) or skim milk for 1 h at room temperature and then incubated overnight at 4°C with primary antibodies against p-PI3K, p-Akt, t-Akt, Bax, cleaved caspase-3, Bcl-2, or GAPDH. The cleaved caspase-3, p-Akt and t-Akt antibodies were acquired from Cell Signaling Technology (Beverly, USA), while p-PI3K, Bax, Bcl-2, and GAPDH were obtained from Abcam (Cambridge, USA). Then, the membrane was washed 5 times with tris-buffered saline with Tween 20 (TBST) and incubated at room temperature for 1 h with secondary antibody conjugated with horseradish peroxidase (HRP; Abcam). Finally, the signals on the membrane were detected using the chemiluminescence detection equipment (Pierce, Holmdel, USA). The protein bands were evaluated and quantified using the ImageJ software (National Institutes of Health, Bethesda, USA).
Statistical analysis
In the present study, statistical analysis was conducted using SPSS software v. 17 (IBM Corp., Armonk, USA). The data was expressed as mean ± standard deviation (SD), and the number of repetitions for each group of data was at minimum n = 3. The Student’s t-test was used for comparisons of 2 groups, and one-way analysis of variance (ANOVA) was used for comparing 3 groups. A p-value of <0.05 was considered statistically significant. GraphPad Prism software v. 8.0 (GraphPad Software, San Diego, USA) was used for statistical mapping.
Results
Puerarin ameliorated cardiac function following AMI
Table 1 and Figure 1 present the results of the echocardiography. Briefly, systolic function decreased in the AMI group compared to the sham group, which manifested as decreased LVFS and LVEF and increased LVESD and LVEDD (p < 0.05). However, compared with the AMI group, in the PUE group, the cardiac function index significantly improved after AMI; this was reflected in the increase of LVFS and LVEF and the decrease of LVESD and LVEDD. These results suggest that puerarin pretreatment could improve cardiac function in AMI rats.
Effects of puerarin on cardiomyocytes apoptosis after AMI
Cardiomyocyte apoptosis was detected usingTUNEL staining (Figure 2). A small amount of myocardial apoptosis was observed in the sham group. However, apoptosis was notably higher in the AMI group than in the sham group (p < 0.05), and most of the apoptotic cells were concentrated in and around the infarcted area. Meaningfully, compared with the AMI group, the number of apoptotic cardiomyocytes were reduced significantly in the PUE group (p < 0.05). The rates of myocardial apoptosis in the sham, AMI, and PUE groups were 10.33 ±1.35%, 72.70 ±6.10% and 19.77 ±6.21%, respectively. Overall, these data indicate that puerarin pretreatment could reduce the apoptosis rate.
Effects of puerarin on cleaved caspase-3, Bcl-2 and Bax protein expression
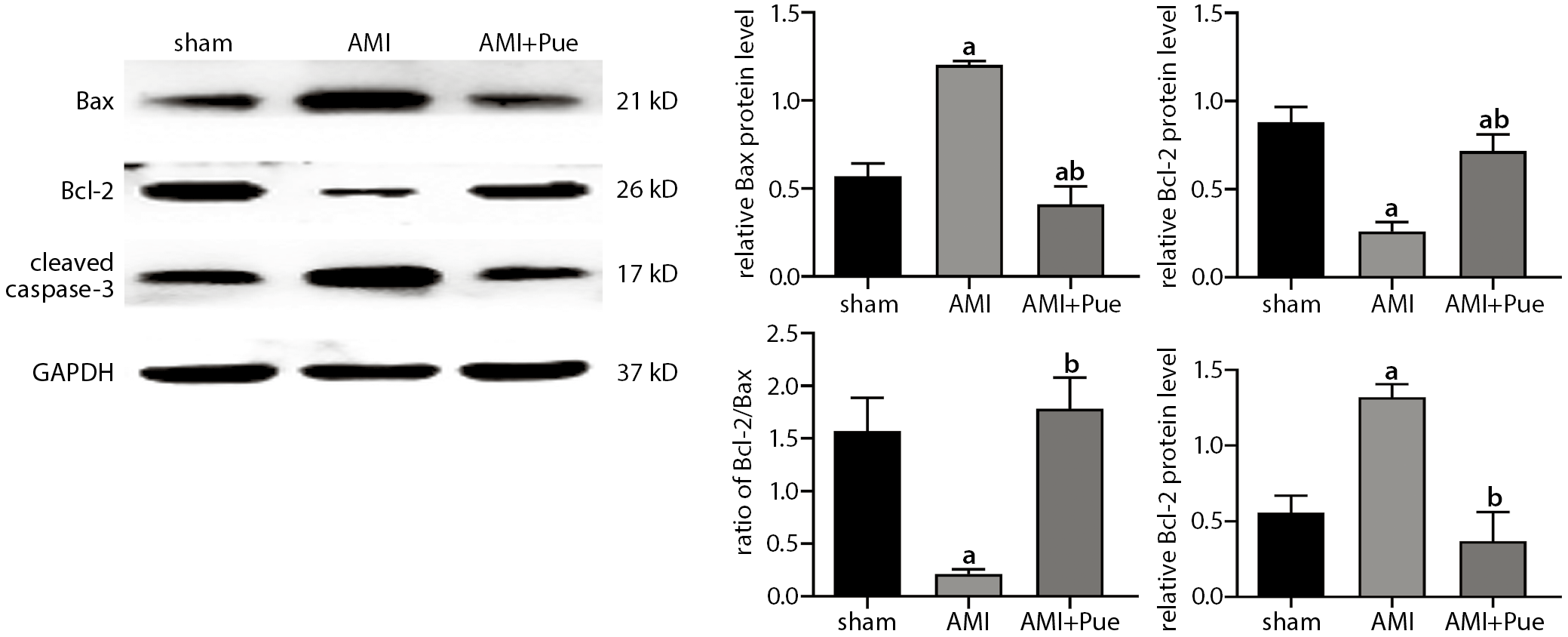
Cleaved caspase-3, Bcl-2 and Bax protein expression was detected to confirm myocardial apoptosis after AMI. Compared to the sham group, the cleaved caspase-3 and Bax protein expression increased in the AMI group, while Bcl-2 protein levels decreased distinctly and caused a significant decrease in the Bcl-2/Bax ratio (p < 0.05) (Figure 3). After pretreatment with puerarin, Bcl-2 protein expression increased, while cleaved caspase-3 and Bax decreased, resulting in a significant increase in the Bcl-2/Bax ratio. This indicates that puerarin has an anti-apoptotic effect on myocardial injury caused by AMI.
Effect of puerarin on protein expression in the PI3K/Akt signaling pathway
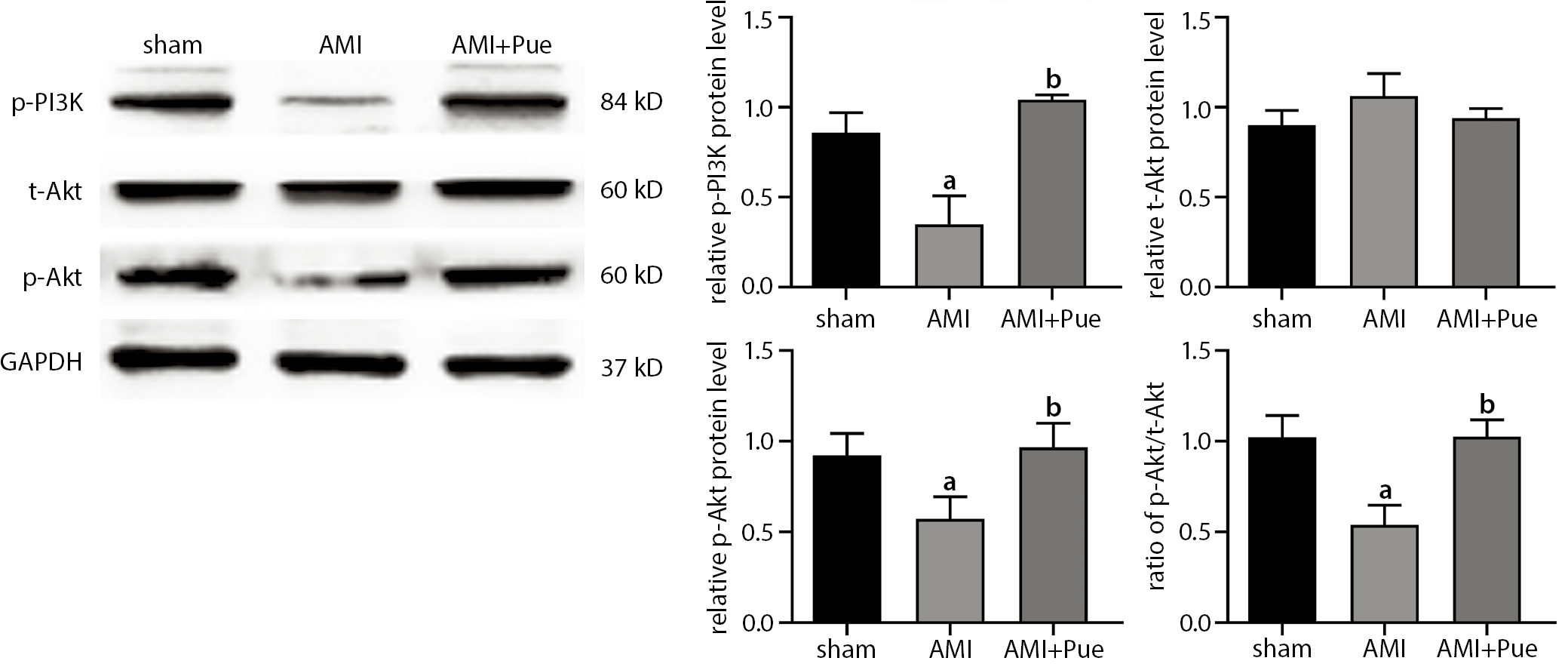
Notably, there was no difference in total Akt expression among the 3 groups (Figure 4). Myocardial level of p-Akt and p-PI3K in the AMI group and PUE group was lower compared to the sham group (p < 0.05). Nevertheless, after puerarin pretreatment, myocardial level of p-PI3K as well as p-Akt was increased significantly (p < 0.05), which suggests that puerarin improved rat cardiac function after AMI, possibly by activating PI3K/Akt pathway.
Discussion
The data provided in this work demonstrate the following 3 points. First, myocardial apoptosis plays a considerable role in myocardial injury after AMI, which may be caused by inhibition of the PI3K/Akt signaling pathway. Second, puerarin pretreatment can significantly reduce myocardial apoptosis and improve myocardial dysfunction induced by AMI. Third, the potential protective mechanism of puerarin may be activation of the PI3K/Akt signaling pathway, thus reducing myocardial apoptosis.
In this study, rats developed left ventricular systolic dysfunction and cardiomyocyte apoptosis increased in them after AMI modeling. These results, like previous studies, indicate that cardiomyocyte apoptosis plays an important role in the irreversible injury of cardiomyocytes caused by AMI. However, after puerarin pretreatment, cardiac function was improved and cardiomyocyte apoptosis was reduced in AMI rats. These results, confirmed in regard to several different aspects, validate the conclusion that puerarin improves cardiac function by inhibiting cardiomyocyte apoptosis induced by AMI. In addition, when exploring the molecular mechanism of action of puerarin, it was found to act at least partially through the activation of the PI3K/Akt signaling pathway. At present, although reperfusion therapy reduces the mortality of AMI, it also increases the risk of heart failure caused by the loss of cardiomyocytes in secondary injury. Therefore, in combination with reperfusion therapy, exploring and developing multiple adjuvant drugs to reduce cardiomyocyte loss will provide important support for the prevention and treatment of MI. To some extent, our research provides new evidence that puerarin exerts an inhibitory effect on myocardial apoptosis induced by AMI through PI3K/Akt pathway. However, in order to translate this into clinical application, there are still many problems that need to be further studied and resolved.
Although percutaneous coronary intervention can significantly improve patient survival, it is undeniable that AMI survivors still face a high risk of death within a few years of an AMI incident.11 Data showed that the three-year all-cause mortality of AMI survivors tends to be greater than 50%.12 As an important form of cell death after injury, apoptosis plays a very important and key role in AMI-induced myocardial injury, which occurs early in AMI.3, 13 Importantly, several studies demonstrated that anti-apoptotic therapies could decrease myocardial apoptosis and significantly ameliorate cardiac dysfunction caused by AMI.1, 14, 15 Therefore, a therapeutic strategy to limit cardiomyocyte loss by inhibiting apoptosis may be a feasible method to prevent and treat AMI.
Puerarin is the main monomer component in Radix puerariae, used in traditional Chinese medicine (Kudzu root), which has anti-oxidative, anti-inflammatory and anti-apoptotic effects.16, 17 As mentioned previously, puerarin can dilate blood vessels, improve coronary circulation, increase blood flow, and reduce blood pressure and heart rate.7, 8 Because of its rich sources and high relative safety, as well as multiple mechanisms of action, puerarin possesses broad application potential in cardiovascular disease.18 In addition to these benefits, previous studies have demonstrated that puerarin alleviates cadmium-induced hepatocyte injury by inhibiting apoptosis and restoring autophagy.19 Similarly, in lead-induced rat kidney injury models, puerarin reduces renal apoptosis by regulating the PI3K/Akt/eNOS pathway.17 Moreover, puerarin protects the brain from ischemic injury by inhibiting apoptosis of astrocytes, which may be connected with activation of the PI3K/Akt and MAPK/ERK pathways.20 Deng et al. demonstrated that puerarin can inhibit the expression of tissue factor induced through oxidative low-density lipoprotein by inhibiting the activation of NF-κB and ERK1/2 and activating the PI3K/Akt/eNOS pathway.21 In the present work, the results indicate that puerarin distinctly reduced myocardial apoptosis in an AMI rat model, thereby alleviating myocardial injury induced by AMI.
As myocardium is a terminally differentiated tissue, damaged cardiomyocytes cannot be regenerated and repaired. Therefore, protecting the activity of cardiomyocytes against ischemic diseases is essential. Myocardial apoptosis is widespread during AMI, leading to the loss of a large number of cardiomyocytes and severe cardiac dysfunction. In this situation, the activation of anti-apoptotic pathways, as well as the expression of specific anti-apoptotic proteins, are particularly important for the protection of cardiac function.6 Interestingly, PI3K/Akt is not only an important signaling pathway that mediates apoptosis, growth and survival, but also a common pathway for a variety of cardiovascular drugs to achieve myocardial protection.6, 22, 23, 24
In cells, a vital function of activated PI3K is to inhibit programmed cell death, and Akt – as a direct downstream target mediating PI3K-dependent cell-survival response – is also closely related to anti-apoptosis.25 The PI3K/Akt pathway can regulate the levels of apoptosis-related proteins, such as caspase-3, Bax and Bcl-2. Bcl-2 is a significant and indispensable anti-apoptotic protein, while Bax is an important pro-apoptotic protein, so the Bcl-2/Bax ratio is a crucial factor in determining the apoptosis threshold.26 Moreover, the aspartic acid-specific protease family, caspases, are also considered to be a key factor in inducing apoptosis.27 Caspase-3 is a well-known family member that catalyzes apoptosis, and its activation is the most critical and important executor of apoptosis.28, 29 One of the mechanisms through which the PI3K/Akt signaling pathway reduces apoptosis is via phosphorylated PI3K activating Akt, which then phosphorylates Bad, thus freeing Bcl-2 to inhibit Bax.30 In this study, the decrease in Bcl-2 and p-Akt after AMI injury means decreased anti-apoptotic activity in the heart. However, puerarin pretreatment upregulated the expression of p-Akt and p-PI3K, as well as Bcl-2. The increased expression of phosphorylated proteins in the PI3K/Akt pathway means that this pathway was activated. Therefore, the protective effect of puerarin on myocardial injury may be closely related to the PI3K/Akt signaling pathway.
Limitations
The limitations of this study need to be discussed. First, puerarin was administered through intraperitoneal injection, while the most common and appropriate route of drug application clinically is oral. We will explore the effect of oral administration and the best therapeutic dose of puerarin in the future. Second, in this study, there is a lack of strong evidence proving a direct causal relationship between the expression of PI3K and Akt and cleaved caspase-3, Bax and Bcl-2. Third, PI3K inhibitors were not used in this study to confirm that puerarin indeed exerts cardioprotective effects by activating the PI3K/Akt signaling pathway. In future research, we will strive to improve these shortcomings.
Conclusions
The PI3K/Akt pathway is inhibited after AMI and exerts a key effect on myocardial apoptosis induced by AMI. Pretreatment with puerarin can distinctly activate the PI3K/Akt signaling pathway, inhibit cardiomyocyte apoptosis induced by AMI and ameliorate cardiac function.