Abstract
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic has caused high morbidity and mortality and has been a source of substantial challenges for healthcare systems globally. Despite a full recovery, a significant proportion of patients demonstrate a broad spectrum of cardiovascular, pulmonary and neurological symptoms that are believed to be caused by long-term tissue damage and pathological inflammation, which play a vital role in disease development. Microvascular dysfunction also causes significant health problems. This review aimed to critically appraise the current data on the long-term cardiovascular sequelae of coronavirus disease 2019 (COVID-19), with a primary focus on cardiovascular symptoms such as chest pain, fatigue, palpitations, and breathlessness, and more significant disease entities including myocarditis, pericarditis and postural tachycardia syndrome. Potential risk factors identified in recent studies that contribute towards the development of long COVID are also included alongside a summary of recent advances in diagnostics and putative treatment options.
Key words: COVID-19, inflammation, microcirculation, SARS-CoV-2
Introduction
The term “long COVID” was first used by a patient as a hashtag on Twitter shortly after the pandemic spread in May 2020,1 and quickly gained popularity within the scientific community. Even though long coronavirus disease (COVID), also known as post-coronavirus disease 2019 (COVID-19) or chronic COVID syndrome, affects many patients each year, it is a poorly understood illness, and its definition is not entirely clear. This is because the syndrome includes several hundred complications with completely different degrees of severity that impact functioning with varying duration. The disorder also affects psychophysical performance and nearly every organ within the human body. The World Health Organization (WHO) defines long COVID as a condition that “occurs in individuals with a history of probable or confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, usually 3 months from the onset of COVID-19 with symptoms that last for at least 2 months and cannot be explained by an alternative diagnosis”.2 However, researchers often use the revised criteria, which makes it difficult to determine the frequency at which it occurs. In a recently published meta-analysis, the global prevalence of long COVID was estimated at 0.43 and was higher for hospitalized than non-hospitalized patients. Researchers also described heterogeneity of the results of the included studies (from 0.09 to 0.81).3
Objectives
Since long COVID is a new disease entity with a short observation period, this review aimed to present possible pathophysiological mechanisms, risk factors, cardiovascular manifestations, diagnostics, and treatments using the limited literature available. A complete understanding of this disease is crucial to providing adequate evidence-based patient care.
Materials and methods
The literature search used the PubMed, medRxiv and Google Scholar databases, references from relevant articles, and internet sources, including WHO reports and the web pages of the British Medical Journal (BMJ) and Office for National Statistics (Cardiff, UK). Search terms included “post-COVID-19”, “post-COVID syndrome”, “long COVID”, “chronic COVID syndrome”, “post-acute COVID-19 syndrome”, “myocarditis”, “pericarditis”, “arrhythmia”, and “post-COVID-19 tachycardia syndrome”. No filters were applied for the starting dates, with the last literature search performed on March 22, 2023. The authors screened the titles and abstracts to identify relevant papers.
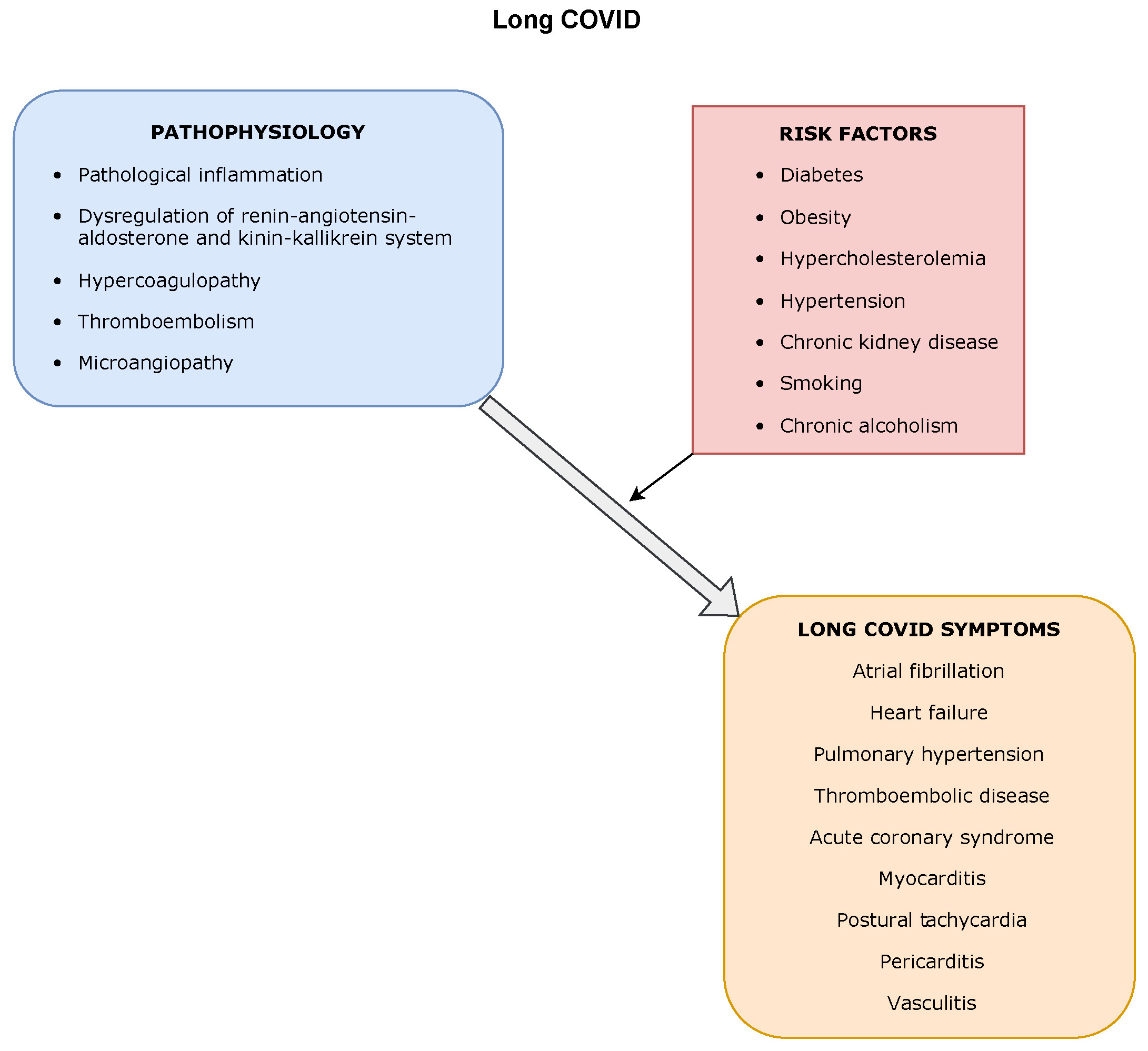
Risk factors
The currently available data show that the most prominent risk factors for long COVID are acute COVID-19 with admission to the intensive care unit (ICU) and/or ventilator support, female sex, and more than 5 symptoms during the 1st week of illness.4, 5, 6 There was no difference in morbidity between patients treated with oxygen alone, invasive ventilation or continuous positive airway pressure.5, 7 The risk of developing long COVID increases when the patient smokes tobacco or has underlying comorbidities such as diabetes, hypertension, obesity, cardiovascular diseases (CVDs), chronic alcoholism, and chronic kidney disease (CKD).8, 9, 10 However, many patients with light/mild acute symptoms who were not hospitalized also develop long COVID symptoms.5, 11, 12 Some studies show that a history of depression, anxiety or antidepressant use can influence the likelihood of developing long COVID.4, 13, 14 Reports also suggest that older age groups are at risk of developing long COVID and that children may experience symptoms similar to adults.15 Evidence is also available indicating that the virus variant affects the risk of developing long COVID, with a higher risk associated with Delta than with Omicron BA.1 or Omicron BA.2 variants. Studies on the effects of vaccines on long COVID risk are inconclusive, with some researchers demonstrating a significant risk reduction in vaccinated patients; the recent data do not support these findings and claim that there is only a 15% risk reduction. Nonetheless, even though vaccination does not eliminate the risk, it reduces it to some extent.16, 17
Putative pathophysiology
The mechanisms of long COVID are likely multifactorial, and there seems to be a continuity of pathogenesis of acute illness. The etiology may include pathological inflammation leading to long-term tissue damage, dysregulation of the renin–angiotensin–aldosterone system (RAAS) and the kinin–kallikrein system (KKS), a virus-induced cytokine and bradykinin storm, dysregulated immune responses, endothelialitis, and mast cell activation syndrome (MCAS).
Pathological inflammation and dysregulated immune responses
The SARS-CoV-2 persistence in the body is a possible consequence of deoxyribonucleic acid (DNA) integration of the viral genome18, 19 and its presence in immunologically privileged sites.20 These mechanisms contribute to the phenomenon in which patients who recovered from COVID-19 continuously obtain positive polymerase chain reaction (PCR) results.18, 19 Viral ribonucleic acid (RNA) and antigens may prompt some level of immune activation leading to the development of long COVID,4 with the process of presenting antigens to autoreactive dysfunctional T-cells by antigen-presenting cells making long COVID pathophysiology similar to autoimmunity.4, 21 Furthermore, some studies have shown that B-cells produce antibodies against neutrophils, interferons, cell nuclei, cyclic citrullinated peptides, and connective tissues, indicative of autoimmunity.4, 22
Hyperinflammatory syndrome, which can contribute to long COVID, results from dysregulated host innate immune responses and decreased absolute lymphocyte count.23 Endothelialitis, which takes part in cardiovascular system dysfunction and its symptomatology, is caused by viral elements within endothelial cells and inflammatory cell infiltration of the myocardium.20, 24
Dysfunction of the renin–angiotensin–aldosterone system and the kinin–kallikrein system
Adherence of the viral S protein to angiotensin-converting enzyme 2 (ACE2) is a primary route for SARS-CoV-2 cellular entry. Locations such as the intestines, kidneys and heart have high expression of ACE2, while its expression is moderate in blood vessels. There are studies showing the downregulation of ACE225, 26 and its disintegrin and metalloproteinase domain-containing protein 17 (ADAMS17)-mediated shedding from the cell surface27 caused by SARS-CoV-2, which leads to RAAS dysregulation and plays a significant role in long COVID development. Under normal physiological conditions, RAAS consists of the angiotensin II (Ang II) type 1 receptors (AT1R) (via which signaling components can intensify cardiac fibrosis and hypertrophy, vasoconstriction, endothelial dysfunction, and vascular inflammation) and Ang II type 2 receptors (AT2R) (which promote vasodilatation, cardioprotection and decreased platelet aggregation).25 Due to ACE2 downregulation and cytokine-induced hyperinflammation, elevated Ang II/AT1R axis activity contributes to myocardial injury, microvascular thrombosis and dysfunction of numerous organs, as shown in Figure 1. In addition, ACE2 shedding initiates a cytokine storm by aggravating the response of tumor necrosis factor alpha (TNF-α) and interleukin (IL)-6R.27, 28 Some have theorized that MCAS and the resultant cytokine storm are associated with long COVID.8, 29
The KKS exerts biological effects through bradykinin-1 (B1) and bradykinin-2 (B2) receptor activation. While B2 receptors are present in all tissues, B1 receptor expression increases during cellular stress and inflammation. Elevated expression of ACE contributes to the downregulation of the KKS as well as suppresses its cardioprotective activity, and hyperinflammation causes moderate stimulation of B1 receptors, leading to a cytokine storm. A bradykinin storm causes myocardial and endothelial dysfunction due to fibrotic accumulations, and may be a consequence of ACE2 reduction and downregulation of des-Arg9-bradykinin (DABK) degradation.25, 30
Mast cell activation syndrome
Mast cell activation syndrome is an inflammatory disease caused by mast cells mistakenly releasing excessive amounts of inflammatory cytokines. Some have theorized that a connection exists between prolonged COVID-19 symptomatology and hyperactive mast cells in patients with underlying primary MCAS. Due to the expression of ACE2, mast cells become an easy target for SARS-CoV-2. Nevertheless, there is a lack of experimental studies confirming this hypothesis.31, 32
Microvascular dysfunction
Microcirculation analysis, related to the development and improvement of research techniques that allow for microvascular assessment, is continuously gaining more attention. Great hopes are placed on functional diagnosis in coronary microcirculation, as it would allow for the detection of changes in the small arteries of the heart and help determine whether coronary microcirculation dysfunction causes symptoms. Microcirculatory responses to hypoxia can be a prognostic factor for COVID-19, with hypoxia-inducible factor (HIF) playing a critical role in the response. Its actions induce protective molecules that are vital to maintaining a well-functioning response to hypoxia.33
Coronavirus disease 2019 disturbs blood flow in the microcirculation, which results in a high mortality rate. The pathomechanism is complex and consists of many elements acting together to significantly dysregulate homeostasis. The RAAS system is mainly responsible for maintaining optimal blood pressure, though overactivation of this endocrine system causes proliferation and inflammation.34 The ACE2 is a central part of the RAAS system, and its imbalance leads to hypertension, obesity, inflammation, thrombosis, and insulin resistance. Its reduced activity is due to the fact that the virus uses the receptor to enter the cells,35 and as ACE2 levels drop, the Ang II/AT1R axis becomes overactive. The release of complement factors and the endothelial expression of tissue factor (TF) and plasminogen activator inhibitor-1 (PAI-1) increase, leading to a hypercoagulable state.36 Moreover, higher expression of ACE2 was demonstrated in pericytes of patients with underlying heart failure, meaning they may have a greater risk of progressing to severe disease during COVID-19 infection.37
Systemic inflammation, as a result of immune responses to COVID-19 infection, causes dysregulation of the entire immune system, leading to a cytokine storm activating T-cells and macrophages.35 Another important aspect of the pathophysiology of microvascular dysfunction is oxidative stress, which leads to a type 2 myocardial infarction due to a supply and demand mismatch. Another effect of oxidative stress is the activation of the sympathetic nervous system, with subsequent catecholamine release resulting in vasoconstriction and increased cardiomyocyte oxygen demand.34
Increased blood viscosity and decreased red blood cell deformability is an important risk factor that occurs due to low estrogen levels (one of the causes of higher male mortality), obesity, vitamin D deficiency, diabetes, and hypertension.38 Furthermore, a crucial part of microcirculation disorders is the damaged endothelium leading to an imbalance between endothelial nitric oxide synthase (eNOS) and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2 (NOX-2). Production of nitric oxide (NO) has a vasodilating effect and is dependent on eNOS. Low concentrations of NO cause hypertension, thrombosis and vascular inflammation. In contrast, NOX-2 activity increases the number of free radicals that further damage the endothelium.34, 35, 36, 39 It was also found that perivascular cells (myocytes, pericytes and adipocytes) damaged by coronavirus cause the compression of the coronary microcirculation and can release toxic agents.34 In summary, disturbances in the microcirculation occur during SARS-CoV-2 infection, with the virus damaging the endothelium, causing its remodeling and loss of function. Microcirculation disorders may persist after the infection and be the basis for the development of many long COVID disorders. The summary of the processes leading to microvascular dysfuntion is presented in Figure 2.
Cardiovascular manifestations
The association between SARS-CoV-2 infection and an increased risk of late cardiovascular problems was observed early in the pandemic. A recent study on a large group showed that people who had contracted COVID-19 were more likely to develop incident CVDs, including cerebrovascular disorders, dysrhythmias, inflammatory heart disease, ischemic heart disease, heart failure, thromboembolic disease, and other cardiac disorders, within 12 months of illness. The increased risk was independent of other factors and confirmed previously conducted research.
Cardiovascular long COVID most often manifests as long-lasting symptoms, of which the most persistent include palpitations, fatigue, chest pain, and back pain. The complications can also occur in patients who seem to have fully recovered from infection and whose symptoms were relatively mild. Thus, there is a need to develop diagnostic and therapeutic strategies for post-infection patients.40, 41, 42
Myocarditis
Myocarditis is an inflammation of the heart muscle, most commonly being a consequence of viral infection. The pathogenesis of COVID-related myocarditis is unknown, yet the main hypotheses assume the involvement of molecular mimicry mechanisms and the effect of viruses on ACE2 regulation.43 Even though there is evidence of SARS-CoV-2 genomes in endomyocardial biopsies, it is suspected that myocarditis is caused in most patients by the hyperinflammatory response triggered by the virus.44, 45 Moreover, heart muscle injury was assessed using biochemical and imaging tests soon after the infection and was present in most patients. Due to the destruction of myocytes, this condition deteriorates cardiac function, causing heart failure, cardiomyopathy and sudden cardiac death. There have been multiple studies on myocarditis after COVID-19 infection, with Puntmann et al. showing cardiac involvement in 78% of patients and ongoing myocardial inflammation in 60% after a median time of 71 days.46 Cardiac involvement was also present in later studies showing that cardiac magnetic resonance (CMR) is useful for patient monitoring.47 Further research established that myocardial injury, assessed using troponin levels during infection, can be predictive of later CMR abnormalities.48
The impact of the SARS-CoV-2 virus on the cardiac muscle of professional athletes has yet to be established. The studies show discrepant results, though all available research was performed in small patient groups.49, 50, 51 Vaccination seems to be the only effective method for preventing COVID-19 and its complications. However, post-vaccination myopericarditis should not be overlooked.52, 53, 54 Recommendations for CMR imaging criteria of active myocarditis provided by the Journal of the American College of Cardiology scientific expert panel consists of an increase in at least 1 T1-based method, including T1 mapping, and 1 T2-based method, including T2 mapping.55 This method of imaging, in addition to clinical manifestations of myocarditis, is currently the gold standard non-invasive diagnostic modality.56
Pericarditis
Pericarditis is common in acute SARS-CoV-2 infection but can also occur after recovery. There are minimal data concerning its epidemiology, though it seems to be a common manifestation of long COVID57 and is thought to be associated with the ongoing inflammation that results from the persistence of viral nucleic acid. In most cases, it is diagnosed by exclusion and 2 of the following symptoms: pericardial effusion, chest pain, electrographic changes, and pericardial friction. Isolated, small pericardial effusion is much more common than pericarditis.31, 46, 58 One study demonstrated that 30.9% of patients had myocarditis and/or pericarditis 10 weeks after COVID-19 pneumonia. Therefore, some refer to both as myopericarditis (pericarditis with associated myocarditis on CMR without left ventricular wall motion abnormalities) or perimyocarditis (when left ventricular wall motion abnormalities are present), depending on the dominating clinical image.59 A study by Dini et al. reported a high prevalence (22%) of pericarditis in patients previously affected by COVID-19 after at least 12 weeks from the end of the infection.57
Long COVID tachycardia syndrome and other arrhythmias
Tachycardia is thought to be a common feature following acute SARS-CoV-2 infection. Persistent, symptomatic tachycardia constitutes a syndrome called long COVID tachycardia syndrome, which presents clinically as postural orthostatic tachycardia syndrome (POTS), inappropriate sinus tachycardia or sinus tachycardia, with 54% of patients reporting persistent symptoms and an increased resting heart rate. Holter monitoring and other devices measuring the heart rhythm may allow for diagnosis and monitoring of progress in an outpatient setting. Moreover, tachycardia can be a valuable quantitative indicator of long COVID severity and reflects not only autonomic dysfunction and possible damage to the heart but also the general condition of the patient.31, 60, 61, 62
The first studies comparing the available treatments for long COVID tachycardia syndrome have already appeared, with one showing that ivabradine, which acts on the sinus node, was more effective than carvedilol.63 Knowledge of the prevalence of POTS and inappropriate sinus tachycardia in patients suffering from long COVID is limited, as most studies retrospective or have small sample sizes.64 Based on the available data, the proportion of COVID-19 survivors developing POTS and inappropriate sinus tachycardia is estimated at 2–22% and 4–20%, respectively.65, 66, 67
In addition to sinus tachycardia, long COVID may include other arrhythmias such as atrial fibrillation (AF), ventricular tachyarrhythmias (VT), bradyarrhythmias, and conduction defects. It is suspected that multiple mechanisms may lead to this complication, though the primary pathophysiology is driven by the inflammatory response. Cytokines, especially TNF-α, IL-1 and IL-6, may cause myocardial damage or systemic effects that indirectly influence the heart muscle.68 A slightly different mechanism has been described for VT, in which patients who limited their physical activity due to infection had a higher incidence of VT.69 Ingul et al. found cardiac arrhythmias of unknown clinical importance 3 months after hospitalization due to COVID-19 in 27% of participants, premature ventricular contractions in 18% and non-sustained ventricular tachycardia in 5%.70
Another problem faced by patients is arterial pressure dysregulation. In one study, the hazard ratios for high blood pressure persisted for up to 6 months after infection.71 A summary of the studies on cardiovascular consequences is presented in Table 1.
Diagnostics
Long COVID diagnosis is currently based on reported symptoms and a history of confirmed SARS-CoV-2 infection. Many studies suggest that diagnostic tests should be performed to assess long COVID cardiovascular manifestations. The American College of Cardiology expert panel recommends performing basic cardiac tests (electrocardiography, cardiac troponin levels and echocardiography) as a minimum in patients at risk of developing CVD after COVID-19, which includes those with a moderate to severe disease course, CVD history, or risk factors such as age, obesity, hypertension, diabetes, and other comorbidities. The current state of knowledge leaves the physician with broad discretion. A better understanding of the epidemiology and pathomechanisms could contribute to creating more universal management guidelines.72, 73
Laboratory tests
One study showed that patients presenting long COVID symptoms had increased N-terminal pro B-type natriuretic peptide (NT-proBNP) and NO levels, while troponin remained within normal limits. There was no statistically significant difference in other oxidative stress markers between people with and without symptoms.74 Some studies revealed no difference in systemic inflammatory biomarkers, such as C-reactive protein (CRP), procalcitonin and IL-6, between patients who recovered from COVID-19 with and without persistent symptoms.4, 14, 75, 76, 77, 78 However, another study demonstrated a connection between these biomarkers and radiological abnormalities of the heart at 2-to 3-month follow-up of discharged patients.4, 79 Patients with long COVID had elevated levels of endothelin-1, which indicates endothelial hypoperfusion.80 Furthermore, some symptomatic patients continued to display increased D-dimer levels 2 months after hospital discharge following acute SARS-CoV-2 infection.81
Imaging and others
Numerous articles have evaluated the role of electrocardiogram (ECG), magnetic resonance imaging (MRI) and echocardiography in screening patients with cardiac manifestations of long COVID.55 One study showed that patients presenting long COVID symptoms had sinus tachycardia in resting ECG or atrial tachycardia in 24-hour Holter monitoring.74 Other indicators of myocardial damage on ECG are ST-segment and T wave changes.82 Noninvasive examination methods, such as computed tomography (CT), MRI and echocardiography, can help rule out coronary artery disease (CAD), myopericarditis or congestive heart failure, and assess left/right ventricle function and regional wall motion.83, 84, 85, 86 In transthoracic echocardiography of 102 long COVID patients, there were 4 patterns of abnormalities, including impaired left ventricular function (LVF) in 35 subjects, increased estimated systolic pulmonary artery pressure in 51, diastolic dysfunction with normal LVF in 66, and pericardial effusion/thickening in 23.85 The summary of diagnostic methods helpful in identifying long COVID is shown in Figure 3.
Treatment
Since long COVID is a relatively new condition, pilot studies are primarily available. As such, all administered drugs should be used with the utmost care. Initially, it was assumed that therapy with drugs that inhibit RAAS could cause increased cellular ACE2 expression, which may facilitate virus entry and exacerbate the infection. However, several studies found that these drugs had no negative effect on mortality and should not be discontinued.87, 88, 89 Moreover, some studies indicate their positive impact in the context of SARS-CoV-2 infection, which is related to their antioxidant and anti-inflammatory effects.90 Sulodexide is a promising drug for endothelial disorders, and a small study showed that it can reduce long COVID symptoms, such as chest pain, palpitations and fatigue, after a 21-day follow-up.91 In addition, the hypothetical positive effects of anticoagulants on long COVID are being increasingly discussed. However, no scientifically robust evidence exists to recommend their use in this population at this stage.92, 93
Guidelines for implementing rehabilitation into long COVID treatment plans, as well as inclusion and exclusion criteria to adapt it to patients’ individual needs are under development. Nonetheless, most patients will benefit from light aerobic and breathing exercises.4
Limitations
The primary limitation that may affect the conclusions of this study is that COVID-19 and the associated long COVID are new disease entities. For this reason, the short observation period limits the results and conclusions drawn. Larger studies are required in the future to determine the precise influence of long COVID on health, particularly for putative treatments based on small pilot studies. No relevant recommendations exist, and treatment is based on hypotheses and clinical practices.
Conclusions
The COVID-19 outbreak has proven to be a substantial challenge for healthcare systems. Although the media interest focused on acute disease, the long-term effects of the epidemic will continue to affect citizens’ health for a long time. Unfortunately, the literature and research on long COVID are still scarce, making it challenging to plan appropriate patient care and treatment. In our article, we focused on the cardiovascular aspects of long COVID and devoted a large part to its pathogenesis because, in the absence of research and standards for diagnosis and treatment, it is crucial to primarily understand the mechanisms in order to implement an optimal therapeutic strategy. Patients with risk factors such as diabetes, hypertension, obesity, CVDs, and CKD require special attention and should be closely monitored.
Myocarditis, pericarditis and tachycardia syndrome are the main cardiovascular manifestations of long COVID. Research is ongoing in the fields of vasculitis, CAD and thromboembolism to better understand their role in long COVID. Continuously developing imaging techniques and laboratory tests allow for the detection of patients who may suffer from long COVID. Treatment is mainly based on symptoms and includes both pharmacological and non-pharmacological approaches. Hopefully, precise therapeutic methods will emerge as research on the pathophysiology and diagnostic processes continues, resulting in better clinical outcomes.