Abstract
Degenerative disease of the spine (DDS) is one of the most common pathological conditions in humans. The clinical presentation of DDS is highly variable, ranging from mild pain to severe neurological symptoms. When more severe clinical symptoms are present, it is necessary to use imaging methods, such as magnetic resonance imaging (MRI), to confirm the diagnosis and establish the extent of the disease in order to determine proper treatment. There are several MRI changes which, based on clinicoradiological studies, are believed to be potential sources of pain and other clinical symptoms in DDS, including compression of the nerve root or spinal cord by disc herniations or osteophytes, recent (“active”) disc herniation, Modic type 1 degenerative changes of the vertebral bodies, degenerative changes of the vertebral endplates (erosive intervertebral osteochondrosis), marked degenerative changes of the facet joints and ligamenta flava, degenerative spinal canal stenosis, degenerative spondylolisthesis, and Baastrup’s disease. The authors analyzed the relationship of the MRI findings mentioned above with clinical symptoms of DDS, as well as the differentiation between DDS and nondegenerative diseases, which can manifest with similar clinical signs. The role of contrast-enhanced MRI and advanced MR techniques (e.g., high field MRI, functional MRI and MR spectroscopy) was also discussed. To establish an appropriate treatment for DDS, it is important to emphasize in the MRI report specific changes, which might be the cause of the pain and other clinical signs, as well as to rule out nondegenerative lesions, especially neoplasms, infections and rheumatoid disorders.
Key words: degenerative disease of the spine, magnetic resonance imaging, back pain, neurological claudication, myelopathy
Introduction
Degenerative disease of the spine (DDS) is one of the most common pathological conditions in humans. The clinical presentation of DDS is highly variable, ranging from mild pain to severe neurological symptoms.1, 2 In benign cases, the diagnosis of DDS is based on the clinical signs and physical examination. When more severe clinical symptoms are present, it is necessary to use imaging methods to confirm the diagnosis and establish the extent of the disease, which is necessary to determine the proper treatment.3
Plain X-rays of the spine are usually used as an initial imaging method in DDS; however, their value is limited to the bone changes (e.g., vertebral body osteophytes) and narrowing of the intervertebral spaces, which are indirect signs of degenerative disc disease (DDD). The imaging modality of choice in patients with more severe DDS is magnetic resonance imaging (MRI).4 It is believed that MRI can help define the anatomic basis of the pain and autonomic nervous system syndrome in patients with disc herniations.5 Computed tomography (CT) is a complementary method to better assess bone lesions. The other imaging methods (nuclear medicine modalities, myelography and discography) are rarely used due to their low availability or invasiveness.6
The difficulty of interpreting MRI studies in DDS is associated with the multi-level and multistructural patterns seen on MRIs. Degenerative disease of the spine is commonly located at multiple levels, and multiple structures are involved at each level.7, 8 The so-called discosomatic (discovertebral) unit consists of 5 elements: intervertebral disc, vertebral bodies, facet joints, ligamenta flava, and longitudinal ligaments. Each of these structures can be affected by the degenerative process.9 Moreover, any particular part of the discosomatic unit may show various MRI findings. For instance, DDD can present as a black disc disease (dehydration), disc calcifications, gas in the disc (vacuum phenomenon), disc bulging, and disc herniation.10, 11, 12 Vertebral body degeneration involves Modic type 1, 2 and 3 changes, osteophytes, and erosive osteochondrosis (degeneration of vertebral endplates).7, 8, 13, 14 There are also specific MRI patterns like degenerative spinal stenosis (changes in all elements of the discosomatic unit), degenerative spondylolisthesis and Baastrup’s disease (degeneration of the spinous processes).3, 4, 9, 15, 16, 17
The most important clinical symptoms of DDS are6: 1) persistent back pain; 2) radicular (pain) symptoms; 3) neurological deficits (limb paresis, myelopathy); 4) sensory impairment; and 5) neurogenic claudication. The crucial problem is how to correlate the pain and other clinical symptoms with particular MRI findings, i.e., how to establish which of the multiple MRI changes are responsible for the clinical signs.
MRI findings which may be responsible for the pain and other clinical symptoms in DDS
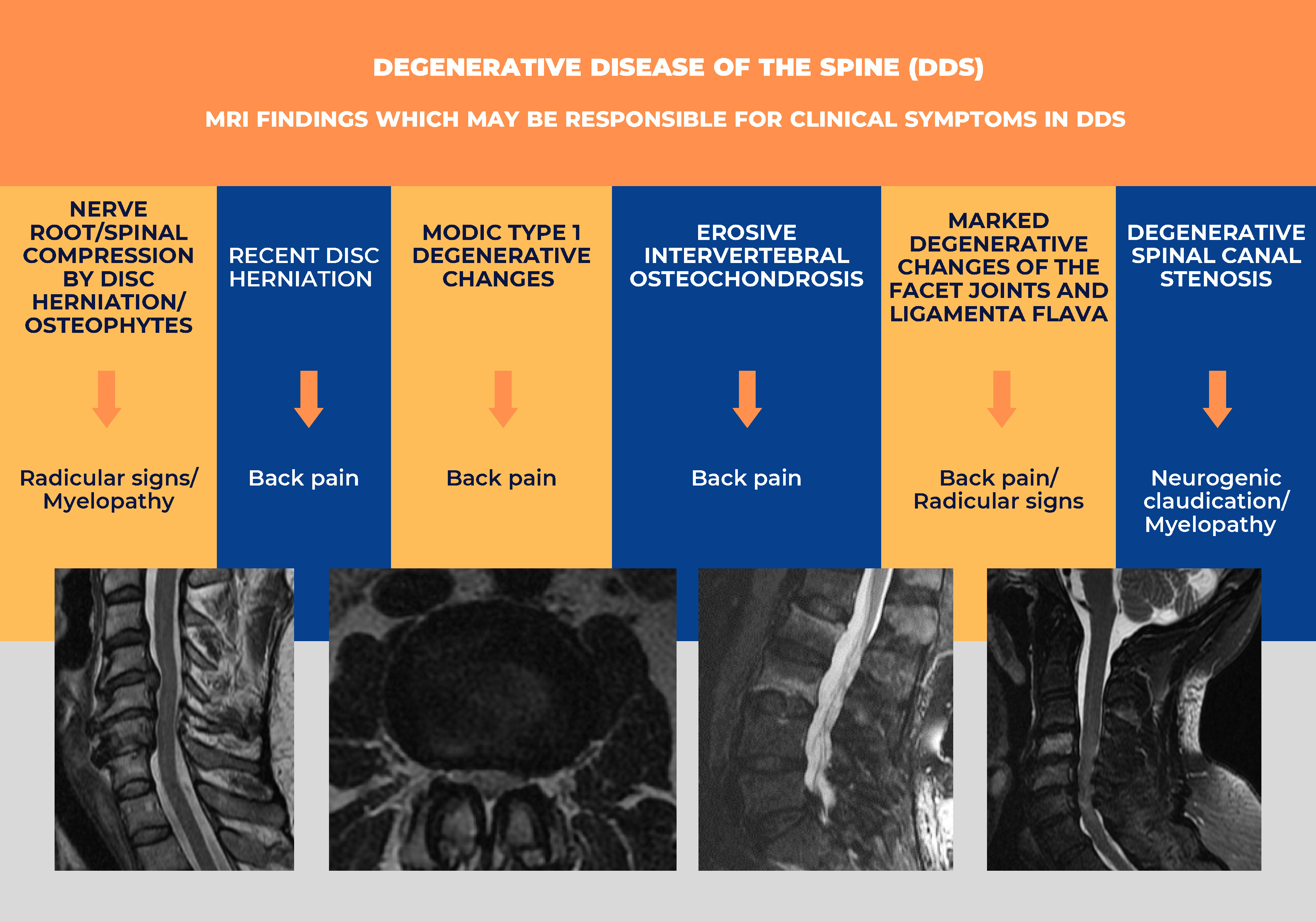
There are several MRI changes which, based on clinicoradiological studies, are believed to be a potential source of pain and other clinical symptoms in DDS3, 4, 7, 9, 16, 17: 1) compression of the nerve root or spinal cord by disc herniations or osteophytes; 2) recent (“active”) disc herniation; 3) Modic type 1 degenerative changes of the vertebral bodies; 4) degenerative changes of the vertebral endplates (erosive intervertebral osteochondrosis); 5) marked degenerative changes of the facet joints and ligamenta flava; 6) degenerative spinal canal stenosis; 7) degenerative spondylolisthesis; and 8) Baastrup’s disease.
Compression of the nerve root or spinal cord by disc herniations or osteophytes
This is a very common radiological finding in DDS. The anterior surface of nerve roots and the spinal cord are located very close to the posterior aspect of the intervertebral discs and vertebral bodies. Therefore, even a small disc herniation or osteophyte could compress the adjacent nerve root or spinal cord, especially in patients with degenerative spinal canal stenosis in whom the intraspinal space is already compromised.7, 10, 12, 18
On the other hand, many small herniations or osteophytes do not produce clinical symptoms until they compress the epidural space or dural sac. In patients who present clinically with radicular symptoms or myelopathy, one should check MR images for direct compression of the nerve root or spinal cord. The best visualization of nerve root compression is provided by axial T2-weighted images, while spinal cord compression can be appreciated in sagittal and axial planes (Figure 1).6, 9, 19
Prolonged compression of the spinal cord can lead to secondary changes in the spinal cord, which during earlier phases is compatible with edema and ischemia, while later phases correlate with myelomalacia and gliosis. The longer the compression lasts, the more severe are the myelopathic symptoms. The changes mentioned above can be seen on MR T2-weighted images as hyperintense foci within the spinal cord.10 However, in many patients with myelopathic symptoms, no changes in the spinal cord are visible on MRI even if it is compressed. The reason for this is the limited spatial resolution of MR images. The sensitivity of MRI in detecting spinal cord lesions can be increased by the use of diffusion tensor imaging (DTI), which provides the quantitative assessment of the spinal cord impairment by calculating fractional anisotropy (FA) and other DTI parameters.20, 21, 22 Another promising technique is functional MRI (fMRI). Some studies demonstrated a positive correlation between functional connectivity and volume of activations in blood-oxygen-level-dependent (BOLD) signals on fMRI and postsurgical outcomes in patients with degenerative cervical myelopathy.21 Finally, the use of high-field MRI systems (3 or 7 Tesla units) might improve sensitivity in detecting spinal cord changes in patients with degenerative myelopathy.23
A possible cause for the discrepancy between the clinical symptoms of radiculopathy or myelopathy and MRI signs is the supine position of the patient during MRI examination, while in many cases, the compression occurs only in the standing or sitting position. One of the solutions is the use of vertical MRI units, in which the patient stands or sits, and dynamic scanning with flexion or extension is performed.24 However, such MRI units are rarely available; hence, an alternative known as axial-loaded imaging has been developed. This is based on special devices which provide a calculated compression of the patient’s feet and head to imitate natural loading in the standing position.25 This can help to visualize the compression of the nerve roots or spinal cord, which is not visible on plain MRIs.
Recent (“active”) disc herniation
Although disc degeneration is generally associated with low signal intensity on T2-weighted images due to decreased water content (dehydration), foci of high intensity (high intensity zones (HIZ)) can be seen in the posterior parts of bulging or herniated discs.26 The significance of HIZ is controversial. It is believed by some authors that HIZ in the posterior part of degenerated or bulging discs represents a tear in the annulus fibrosus that can lead to disc herniation.27 However, it has not been fully confirmed by radiopathological studies. On the other hand, the increased signal in bulging and especially herniated disks means an increased water content that could be caused by edema and inflammation in or around the bulging/herniated disc and, thus, could be the source of pain.26, 27, 28
This hypothesis can be confirmed by the common occurrence of HIZ in patients with marked back pain and recent disc herniations, even without compression of nerve roots, while in patients with older disc herniations, which usually do not have HIZ, the clinical symptoms are associated with radicular or spinal cord compression (Figure 2).10, 27, 28
Additional evidence of an inflammatory process in patients with recent disc herniations who present with back pain is the contrast enhancement around the herniations, which can be seen in rare cases when gadolinium contrast medium is used in patients with DDS.29 As in other places, the inflammatory reaction commonly results in pain.
Injection of gadolinium contrast medium can be useful in patients after herniated disc surgery presenting with failed back surgery syndrome (FBSS) to differentiate between recurrent disc herniation and other causes of persistent pain after surgery.30, 31 The other complementary MRI techniques in FBSS include fMRI and MR neurography.31
Further research based on fMRI, T1, T2, and T2* mapping, as well as MR spectroscopy, may provide new information concerning the relationship between pain and degenerative disc changes.32, 33
Modic type 1 degenerative changes of the vertebral bodies
Modic type 1 degenerative changes are believed to represent inflammatory reactions of the vertebral bodies during the course of DDS. They are located in the direct vicinity of the vertebral endplates, usually on both sides of the degenerated intervertebral disc. Their signal is hyperintense on T2-weighted/Fat-Sat T2-weighted MR images and hypointense on T1-weighted images. This pattern is compatible with the increased water content in Modic type 1 lesions, which are caused by edema and inflammation, and can result in pain.7, 8, 11, 13 Moreover, if, for any reason, gadolinium contrast medium is used, an enhancement of the Modic type I areas might be observed, which is further evidence of their inflammatory background (Figure 3).34
This inflammatory nature is the reason for the term “aseptic spondylodiscitis”, which is sometimes used to describe Modic type 1 changes. Actually, in some cases, Modic type 1 changes must be differentiated from true infectious spondylodiscitis. Apart from clinical and laboratory markers of infection like fever or increased C-reactive protein (CRP) and white blood cell (WBC) levels, the MRI pattern in infectious spondylodiscitis is different. First of all, in Modic type 1 changes, the adjacent intervertebral disk has a low signal on T2-weighted images due to degenerative dehydration, while in infectious spondylodiscitis, the signal of the disc is increased due to infectious infiltration. Besides, the vertebral endplates in Modic 1 changes are intact, while in infectious spondylodiscitis, they are destroyed. Finally, the infectious infiltration often extends to the paravertebral and extradural spaces, which does not occur in Modic type 1 degenerative changes.4, 35 The pattern of contrast enhancement after administration of gadolinium is a useful clue; in DDS, the enhancement is limited to the regions of the vertebral endplates and facet joints, while in spondylodiscitis, it may also involve extradural and paraspinal areas.36, 37
Degenerative changes of the vertebral endplates (erosive intervertebral osteochondrosis)
The vertebral endplates consist of cartilage and cortical bone from the vertebral body surfaces adjacent to the intervertebral disc.38 The endplates are highly vulnerable to the degenerative process as they are, on the one hand, loaded by body weight and, on the other hand, provide the blood supply to the intervertebral disc. Degeneration of the endplates, especially of their cartilaginous component, is considered a source of pain.9, 34, 38
On MR images, the degeneration of the endplates appears as an irregular outline in their cortical bone, which can be seen in all sequences.6, 9 This pattern can also mimic infectious spondylodiscitis. However, in erosive intervertebral osteochondrosis, the black line consistent with the endplate cortical bone is always intact, even if its outline is very irregular (Figure 4), while in infectious spondylodiscitis, this black line is blurred. The other crucial difference is the signal of the adjacent intervertebral disc, which is low on T2-weighted images in erosive intervertebral osteochondrosis, due to infectious infiltration.35 The use of contrast media may also help in the differential diagnosis, as the pattern of enhancement in spondylodiscitis is different from that in vertebral degeneration.36, 37
Marked degenerative changes of the facet joints and ligamenta flava
Facet joints are commonly affected in DDS. As they bear significant stress and weight, they are highly vulnerable to degeneration.39 Besides, as the facets are richly innervated, their degeneration can result in back pain or sciatica.40
The radiological signs of facet degeneration seen on MRI or CT include deformation and osteophytes of the articular processes as well as narrowing of the joint spaces. (Figure 56, 9, 41 In severe degeneration, T2-weighted fat-suppressed MRI sequence could reveal the hyperintense signal of the articular process while post-contrast T1-weighted fat-suppressed sequences can show enhancement.34, 36, 37 These findings are compatible with edema and an inflammatory reaction, and thus could be a source of pain. The rare manifestation of a synovial cyst can protrude into the spinal canal.5, 6, 9
Ligamenta flava are located in the posterolateral parts of the spinal canal adjacent to the facet joints. Degeneration of the ligamenta flava causes thickening, which compromises the posterior-lateral parts of the spinal canal and compresses the posterior-lateral aspects of the dural sac.6 In active degeneration with an inflammatory component, contrast enhancement of the ligamenta flava can be observed.36, 37
The degeneration of the facet joints and ligamenta flava usually occur together. Apart from pain caused by degenerated facets, they also contribute to spinal stenosis and the clinical symptoms of spinal stenosis (see below).
Degenerative spinal canal stenosis
Narrowing of the spinal canal (stenosis) may be caused by degeneration of any structure of the discosomatic unit, e.g., disk herniation, osteophytes and thickening of the ligamenta flava. However, the term degenerative spinal canal stenosis usually refers to the degeneration of all or most of the structures of the discosomatic unit.6, 9 The typical pattern of spinal canal stenosis consists of: 1) intervertebral disc bulging; 2) osteophytes of the posterior edges of the vertebral bodies; 3) deformation, osteophytes and (rarely) synovial cysts of the facet joints; and 4) thickening of the ligamenta flava.
Stenosis can be increased by degenerative spondylolisthesis (see below). It usually involves multiple levels, most often in the lower lumbar and lower cervical segments of the spine (Figure 5, Figure 6).4
Degenerative spinal canal stenosis can be classified as central spinal canal stenosis (narrowing of the central part of the spinal canal) and lateral spinal canal stenosis (narrowing of the lateral recesses of the spinal canal and the neural foramina). The latter could be subdivided into lateral recess stenosis and foraminal stenosis.5, 6, 9
The typical clinical manifestation of central lumbar spinal canal stenosis is neurological claudication, which means pain, tingling or cramping in the lower back, legs, hips, and buttocks that can be accompanied by weakness in the legs. These symptoms are enhanced during standing and walking, and relieved when leaning down and sitting.42 On the other hand, cervical and thoracic central spinal canal stenosis causes spinal cord compression and thus presents clinically as myelopathy (pain, numbness and weakness in the neck, back, as well as in upper and/or lower extremities, motor impairment, etc.).4, 10
Lateral spinal canal stenosis (both lateral recess and foraminal) is associated with compression of the radicles or spinal nerves; therefore, its main clinical manifestation is radicular pain.9 In fact, most cases of central and lateral stenosis are combined and result in combined clinical symptoms.
Magnetic resonance imaging provides a very good evaluation of degenerative spinal canal stenosis, especially in axial (central and lateral recess stenosis) and lateral sagittal planes (foraminal stenosis). Computed tomography can be used complementarily for better assessment of the boney elements contributing to stenosis. A detailed analysis of the imaging studies can identify the levels or structures responsible for clinical symptoms.4, 6, 9, 19
Degenerative spondylolisthesis
Degenerative spondylolisthesis develops due to chronic facet joint and intervertebral degeneration, as well as ligamentous laxity, which finally results in vertebral instability and slipping (subluxation) of the upper vertebral body in relation to the inferior one. Typically, the upper vertebral body moves anteriorly to the adjacent lower vertebral body; however, it can also move posteriorly, which is called retrolisthesis.16
The instability caused by spondylolisthesis can cause pain apart from that associated with facet joint degeneration. This can be confirmed by Modic type 1 changes, which are often seen in the vertebral bodies at the level of the spondylolisthesis. Besides, degenerative spondylolisthesis contributes to spinal canal stenosis and can present with clinical symptoms of stenosis.4, 9
In young patients, degenerative spondylolisthesis needs to be differentiated from spondylolisthesis caused by an interarticular pars defects of the posterior vertebral arch (spondylolysis), which can be detected using CT and MRI (including post-contrast MRI).36, 37, 43
Baastrup’s disease
Baastrup’s disease (kissing spines syndrome) occurs when the adjacent spinous processes (usually in the lumbar region) are close enough to touch, which results in degenerative changes. It could be a part of generalized DDS or caused by hyperlordosis of the lumbar spine. Patients experience pain in the midline that worsens with extension and is relieved during flexion of the lumbar spine.17
In T2 fat-suppressed MRI sequences, the affected spinous processes demonstrate a high signal, which is consistent with edema and an inflammatory reaction, thus explaining the pain. On CT, the space between the involved spinous processes is narrowed, and their adjacent surfaces have irregular outlines and osteosclerotic areas.4, 17
Differentiation of DDS
with nondegenerative diseases
An additional problem with clinicoradiological correlations in DDS is that it is sometimes mimicked by nondegenerative diseases. Although DDS is extremely common, other diseases can present with similar clinical symptoms. One example is infectious spondylodiscitis, which can have similar clinical and MRI appearances to Modic type 1 vertebral body changes and especially erosive osteochondrosis (see above).35 Back pain can be caused by neoplastic disease, especially metastases. In doubtful cases, MRI and CT can be supplemented with a bone scan or PET/CT. Finally, the source of the pain could be spondyloarthropathies (e.g., ankylosing spondylitis); therefore, it is useful to evaluate the sacroiliac joints which are commonly affected in spondyloarthropathies that could be easily diagnosed on MRIs of the sacroiliac joints.44
Conclusions
Magnetic resonance imaging reports in patients with DDS should emphasize all changes which might cause pain and other clinical signs (e.g., compression of the nerve root, Modic 1 changes, degeneration of the endplates, facet joint degeneration, and spinal canal stenosis). It is necessary to rule out nondegenerative lesions, especially neoplasms and infections, as well as possible causes of clinical symptoms from adjacent structures, e.g., sacroiliac joints, and post-contrast imaging is helpful in the differential diagnosis. Advanced MR techniques, such as high-field MRI, fMRI, T1, T2, and T2* mapping, MR spectroscopy, or MR neurography, may contribute to a better understanding of the relationship between clinical symptoms and radiological findings in DDS.