Abstract
Background. Postpartum hemorrhage (PPH) is a serious condition that can lead to several complications. Many different factors precipitate PPH.
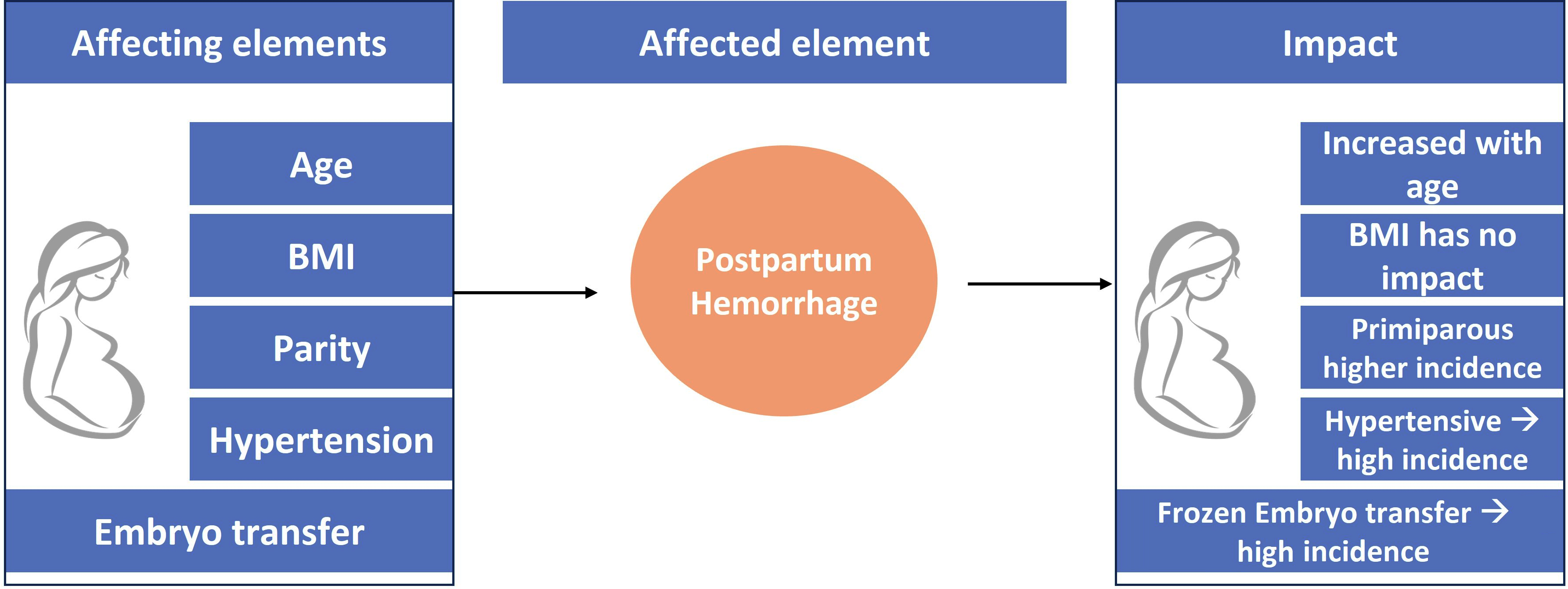
Objectives. To assess the role and impact of different factors such as body mass index (BMI), age, hypertension, parity, and embryo transfer on PPH.
Materials and methods. Thirty-one studies have been included in the current meta-analysis. The outcomes of these studies were analyzed using a random-effects model and it was used to calculate the mean difference (MD) with 95% confidence interval (95% CI) in order to quantify the impact of different risk factors on PPH.
Results. Regarding the maternal age, older subjects (≥35 years) showed a significantly higher incidence of PPH (MD = 1.15, 95% CI: 1.03–1.27, p = 0.01). On the other hand, BMI had no impact on the incidence of PPH (MD = 0.76, 95% CI: 0.26–2.24, p = 0.62). At the same time, primiparous subjects, those with hypertension and those in whom frozen–thawed embryo transfer (FTET) was performed showed a significantly higher PPH incidence (MD = 1.27, 95% CI: 1.02–1.56, p = 0.03; MD = 1.51, 95% CI: 1.42–1.61, p < 0.001; and MD = 1.43, 95% CI: 1.11–1.85, p = 0.006, respectively).
Conclusions. The weight of the subjects is not a risk factor for PPH. However, a higher incidence of the disease can be observed in older, hypertensive and primiparous subjects, as well as those in whom FTET performed.
Key words: BMI, postpartum hemorrhage, maternal, pregnancy, age
Introduction
Postpartum hemorrhage (PPH) is a medical emergency, regardless of the type of delivery.1 Postpartum hemorrhage is defined as blood loss after delivery greater than 500 mL and it occurs in 18% of births. About 3% of all vaginal births are accompanied by severe PPH,2 and this condition is a leading cause of maternal mortality. Severe PPH is defined as blood loss of more than 1000 mL.1
Each year, about 14 million of people lose their lives due to PPH.3 About half of all postpartum deaths occur during the first 24 h, and the majority (66%) happen within the first week. It is difficult to recognize the signs of a medical emergency during labor and the first 24 h after giving birth, which contributes to maternal mortality.4 The global prevalence of PPH is estimated at 6% and 10%.5 According to the previous systematic review of the epidemiology of PPH,6 the overall prevalence of PPH is between 6.09 and 10.55% (7.23% when objectively examined and 5.40% when evaluated subjectively). The evaluation of prevalence according to study design revealed a 5.95% prevalence in observational studies and a 13.94% prevalence in randomized controlled studies.7
Postpartum hemorrhage-related deaths are more prevalent in places with limited access to medical care. The consequences of PPH may be exacerbated in such settings by factors such as malnutrition, malaria and anemia. In places with suitable infrastructure and medical care, PPH-related deaths are quite uncommon.8 To reduce the occurrence of PPH, midwives should assess the expectant mother’s risk factors during each prenatal appointment.9 Pregnant women should be included in the care plan design, and high-quality medical care should be made available to them.7 Midwives play an integral role in assessing risks, implementing safeguards and obtaining medical attention.10
According to the World Health Organization (WHO), postpartum care should cater to the individual requirements of both mothers and newborns, to avoid potential health problems and provide prompt medical attention in the event of any.11 Over 2/3 of women with PPH have no identifiable risk factors.12 No risk factors for PPH have been discovered, and the risk of PPH associated with high multiparity and many pregnancies has grown, according to the World Health Organization’s Recommendations for the Prevention and Treatment of PPH.3 A history of PPH, being nulliparous, being multiparous (going into labor more than 5 times), having coagulopathy (congenital or acquired, use of drugs such as aspirin or heparin), abnormal placental attachment, being older than 30, anemia, excessive stretching of the uterus (multiple pregnancies, polyhydramnios), and fetal macrosomia are all risk factors for PPH in the antepartum period.1, 13, 14 The meta-analysis approach has been used to analyze some of the maternal characteristics in the antepartum period that either increase or decrease the risk of PPH, in order to identify risk factors that are directly associated with PPH.
Over 5 million babies have been born thanks to in vitro fertilization (IVF) as a therapy method of infertility.15 The use of frozen–thawed embryos has become commonplace ever since the first successful live birth was reported using a cryopreserved embryo transfer in 1984.16, 17, 18 Improved perinatal outcomes of frozen–thawed embryo transfer (FTET) due to the fast refining of embryo cryopreservation procedures have made FTET a competitive option for fresh embryo transfer (FET).19, 20 In light of the mounting evidence showing that FET is superior to FET, “freeze-all” cycles are replacing FETs in clinical practice.15, 21 The current data that support embryo cryopreservation are based on low-quality randomized controlled trials and individual observational studies,8, 9 which lack the power to demonstrate statistically significant differences in some perinatal outcomes when low background risk of complications exists. There is still some debate as to whether FTET provides better clinical outcomes. Quantifying the risks of complications and bad birth outcomes calls for a meta-analysis.
Objectives
The study aims to assess the role and impact of different factors such as body mass index (BMI), age, hypertension, parity, and embryo transfer on the incidence of PPH.
Materials and methods
Study design
This systematic review and meta-analysis of clinical trials was included in the epidemiological declaration and had a set study protocol. For data collection and analysis, several of databases were searched.
Data pooling
Prospective and retrospective cohort studies focusing on the assessment of the impact of several risk factors on the incidence of PPH were chosen for this analysis. Human-related studies were included, regardless of language. There were no restrictions regarding the sample size. We did not include reviews, editorials or letters to the editor because they do not report a measure of association. The study procedure is depicted in Figure 1.
Eligibility and inclusion
Analysis of the impact of different maternal characteristics and embryo transfer on the incidence of PPH was used to construct a summary.
The sensitivity analysis comprised only papers reporting the role of age, BMI, hypertension, parity, and embryo transfer on PPH. Several methods of analysis were employed to demonstrate the impact of different factors on PPH.
The inclusion criteria were as follows:
1. Retrospective, prospective or cohort studies;
2. The target population consisted of individuals with certain maternal characteristics related to age, BMI, parity, hypertension, and embryo transfer;
3. The intervention regimen of the included studies was based on comparing the incidence of PPH among subjects with different levels/scores for each analyzed outcome.
The exclusion criteria were as follows:
1. Studies that failed to identify the incidence rate (event number or percent) of PPH;
2. Review articles, letters, books, and book chapters;
3. Studies not focusing on the comparison between different groups regarding the outcomes of the study.
Identification
According to the PICOS principle, a protocol of search strategy was developed32 and defined as follows: P (population) – female subjects with a previous history of pregnancy; I (intervention/exposure) – PPH incidence; C (comparison) – age (<35 years and ≥35 years), BMI (<25 kg/m2 and ≥25 kg/m2), blood pressure (hypertensive and nonhypertensive women), parity (primiparity and multiparity), and embryo transfer method (FTET and FET); O (outcome) – number or percent of occurrence for each parameter in different conditions; S (study design) – cohort studies.33
Using the keywords and associated phrases listed in Table 1, we conducted a complete search of the PubMed, OVID, Cochrane Library, Embase, and Google Scholar databases concerning studies published between January 2000 and August 2022. The titles and abstracts of all the publications that had been collated into reference managing software have been reviewed. The 2 authors (CZ and CW) indentified suitable studies.
Screening
According to the following criteria, data were compiled to include: study- and subject-related features in a standard format, the surname of the first author, the period of the study, the year of publication, the country of the study, the design of the study, the population type recruited in the study, the total number of subjects, qualitative and quantitative evaluation method, demographic data, clinical and treatment characteristics, information source, outcome evaluation, and statistical analysis results.34 Each study was assessed for bias, and the methodological quality of the chosen studies was evaluated by 2 abovementioned authors in a blinded fashion.
The Newcastle–Ottawa Scale (NOS), a quality and bias assessment tool developed specifically for observational research, was used to evaluate the quality and bias of the study. The NOS examines the sample, the comparability of cases and controls, and the exposure in observational studies, and the resuls is expressed by assigning values between 0 and 9. Studies with a rating of 7–9 are of the highest quality and have the lowest risk of bias compared to those with a rating of 4. Studies with a rating of 4–6 are considered to be of moderate quality. Each study underwent a methodological evaluation by the 2 abovementioned authors (CW and CZ).
Statistical analyses
In the current meta-analysis, the mean difference (MD) with a 95% confidence interval (95% CI) was calculated using a random-effects model. Since using the fixed-effects model requires a high similarity between included studies and low heterogeneity (I2) level, all groups were analyzed using the random-effects model due to high heterogeneity in some groups and inconsistent methodology in other groups. The I2 index (determined using Reviewer Manager v. 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark) and expressed in the form of forest plots), in a form of a numeric value ranging from 0% to 100%, was calculated. Values ranging from 0% to 25%, 50% and 75% indicated no, low, moderate, and high heterogeneity, respectively. As previously stated, the subcategory analysis was performed by stratifying the initial evaluation into result categories. Publication bias was investigated quantitatively using the Begg’s test and publication bias was considered present if p > 0.05.37 To obtain the p-values, a two-tailed test was used. The statistical analysis and graphs were presented with Reviewer Manager v. 5.3 and jamovi software v. 2.3 (https://www.jamovi.org/) using the dichotomous model.
Results
After a review of 1526 relevant articles, a total of 31 studies published between January 2000 and August 2022 were included in the meta-analysis because they fit the inclusion criteria.22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52 Table 2 summarizes the findings of these investigations.
Age
Ten studies (the study by Butwock et al. was presented twice in the same analysis, one for American subjects and the other for Swedish subjects) including 3,168,287 subjects reported data stratified according to age (subjects ≥35 years compared subjects <35 years; Figure 2). Older subjects had a higher incidence of PPH compared with subjects younger than 35 years (MD = 1.15, 95% CI: 1.03–1.27, p = 0.01, I2 = 92%).
BMI
Nine studies including 791,597 subjects reported data stratified according to BMI (BMI ≥ 25 kg/m2 compared to BMI < 25 kg/m2; Figure 3). The weight of included subjects had no impact on the incidence rate of PPH, as both groups showed nonsignificant rates (MD = 0.76, 95% CI: 0.26–2.24, p = 0.62, I2 = 100%).
Parity
Eight studies including 1,120,995 subjects reported data stratified according to parity (primiparous compared to multiparous subjects; Figure 4). Multiparous subjects were associated with a lower incidence of PPH compared to primiparous subjects (MD = 1.27, 95% CI: 1.02–1.56, p = 0.03, I2 = 39%).
Hypertension
Six studies including 365,354 subjects reported data stratified according to the presence of hypertension during pregnancy (hypertensive compared to nonhypertensive subjects; Figure 5). Hypertensive subjects had a higher PPH incidence compared with nonhypertensive pregnant subjects (MD = 1.51, 95% CI: 1.42–1.61, p < 0.00001, I2 = 0%).
Embryo transfer
Three studies including 8610 subjects analyzed the impact of embryo transfer method (FTET compared to FET; Figure 6). Frozen–thawed embryo transfer showed a significantly higher incidence of PPH compared with FET (MD = 1.43, 95% CI: 1.11–1.85, p = 0.006, I2 = 48%).
It was not possible to assess the impact of individual characteristics such as ethnicity on the comparison results because no data on these variables had been analyzed in the study. In addition, publication bias was assessed using the Begg’s test and it showed a nonsignificant bias for all included groups with a value of p > 0.05. The obtained p-values for age, BMI, parity, hypertension, and embryo transfer method analysis were p = 0.65, p = 0.61, p = 0.4, p = 0.47, and p = 0.99, respectively.
The risk of bias assessment was evaluated using NOS (Table 2). Twenty-one studies had a score between 7 and 9, which reflect a low risk of bias and high methodological quality. Ten studies showed a moderate risk of bias, achieving a score ranging from 4 to 6 points.
Discussion
A total of 5,454,843 subjects from different countries examined in 31 cohort studies were included in the current meta-analysis.
Every successful PPH treatment must begin with a prompt diagnosis. Learning about the potential causes of PPH is crucial. According to the National Institute for Health and Care Excellence (NICE), women with PPH risk factors should give birth in a unit equipped to handle emergencies.53 This meta-analysis found that a BMI of 25 kg/m2 or higher has no impact on the PPH incidence rate. Regarding maternal age, older subjects (≥35 years) showed a significantly higher incidence of PPH (MD = 1.15, 95% CI: 1.03–1.27, p = 0.01). On the other hand, BMI had no impact on the incidence of PPH (MD = 0.76, 95% CI: 0.26–2.24, p = 0.62). At the same time, primiparous subjects, those with hypertension, and those with FTET showed a significantly higher PPH incidence (MD = 1.27, 95% CI: 1.02–1.56, p = 0.03; MD = 1.51, 95% CI: 1.42–1.61, p < 0.001; and MD = 1.43, 95% CI: 1.11–1.85, p = 0.006, respectively).
According to the Society of Obstetricians and Gynaecologists of Canada, women older than 35 are more likely to have adverse birth outcomes, such as miscarriage, complications during pregnancy, premature delivery, or to bear more than 2 babies in 1 delivery (i.e., twins or more).54 Maternal age ≥40 years increases the risk of PPH, according to both the NICE53 and the Royal College of Obstetricians and Gynecologists (RCOG) clinical guidelines55 for the prevention and management of PPH. Pregnant women aged ≥35 years had a higher risk of adverse pregnancy and delivery outcome.56 After conducting a meta-analysis, Walker et al. found that inducing labor did not increase the incidence of cesarean section for women aged ≥35 years.57 In their meta-analysis, Wood et al. found that inducing labor had no effect on cesarean section rates among women aged ≥45 years in a subgroup analysis.58 The current meta-analysis indicated that older age is related to adverse events such as PPH, which is consistent with the recommendations of RCOG.55 The studies included in the meta-analysis covered study populations different countries. Maternal age is a significant risk factor for PPH in many nations because of the low quality of care provided to mothers aged ≥35 years throughout pregnancy, delivery and the postpartum period.
According to the WHO, having a BMI ≥ 25 kg/m2 is associated with an increased chance of comorbidities, and the risk of complications increases with increasing obesity. According to NICE guidelines, an increased risk was associated with a BMI ≥ 35 kg/m2.53 According to RCOG (2011), having a BMI ≥ 35 kg/m2 is associated with an increased risk of PPH. Obese pregnant women have a higher risk of perinatal problems and interventionist delivery, according to research by Bogaerts et al.59 The current meta-analysis found no significant difference between the ideal body weight and obese subjects regarding the PPH rate. In addition, the current study included obese subjects with a BMI ≥ 25 kg/m2, and the studies that showed a significantly lower impact of obesity stated that a BMI ≥ 35 kg/m2 is related to such adverse events. Hence, we cannot make a general conclusion indicating that obesity is related to PPH.
Post-term birth is more common in first-time mothers, as indicated by the American College of Obstetricians and Gynecologists in their clinical management guide for obstetricians/gynecologists (OB/GYNs).60 According to a comprehensive review and meta-analysis by McDonald et al.,61 parity increased the risk of premature delivery in IVF and twin pregnancies; however, this association was not statistically significant. A comprehensive review conducted by Wang et al. found that primiparity is a risk factor in perineal lacerations.62 According to the results of the present research, first-time mothers are at a higher risk of developing PPH.
According to research published in 2013, hypertension rates increase in pregnancy, and preeclampsia rates are 4 times greater than the mean in 5% of pregnant women worldwide. The risk of PPH is increased by prenatal hypertension, preeclampsia and eclampsia, according to the RCOG clinical guidelines for the prevention and management of PPH.55 In this way, the current results are consistent with the clinical standards. Postpartum hemorrhage is a potential complication of maternal hypertension.
Postpartum hemorrhage was more common in the FTET group, as shown in the current study. Whether or not the embryo cryopreservation process had a negative impact on the increased risk ratios of these events is unknown. The risk of hypertension problems was found to be greater in a FTET compared to FET in observational cohort research comparing outcomes in sibling pregnancies in women.63 Somewhat inconclusive results came from a short study conducted in China that compared FET with vitrified and slow-frozen embryo transfer.64
Limitations
This study may have been skewed by the exclusion of many trials from the meta-analysis. However, our meta-analysis excluded studies that did not meet the inclusion criteria. In addition, some of the included studies have not evaluated the impact of race on the represented outcomes. Thus, it was not possible to assess the impact of ethnicity on the results. Some of the included studies have moderate methodology quality, as evaluated using the NOS score. Variables such as nutritional status were not considered in the included studies, which may have skewed the results. Study results could be biased if there are unpublished articles and uncollected data.
Conclusions
The weight of the subjects is not a risk factor for PPH. However, a higher incidence of the disease can be observed in older (≥35 years), hypertensive and primiparous subjects, as well as those in whom FTET was performed. Hence, controlling the blood pressure and using the optimum embryo transfer method could result in beneficial outcomes regarding PPH. Nevertheless, future clinical multicenter studies are needed to reach a more sensible conclusion.