Abstract
Background. Dextran sulfate sodium (DSS)-induced murine colitis is the most commonly used model for the analysis of the pathogenesis of inflammatory bowel disease (IBD) and for the assessment of the efficacy of putative therapeutics. It has been suggested that mice should be given 2.5–10% DSS for 3–7 days to establish the model.
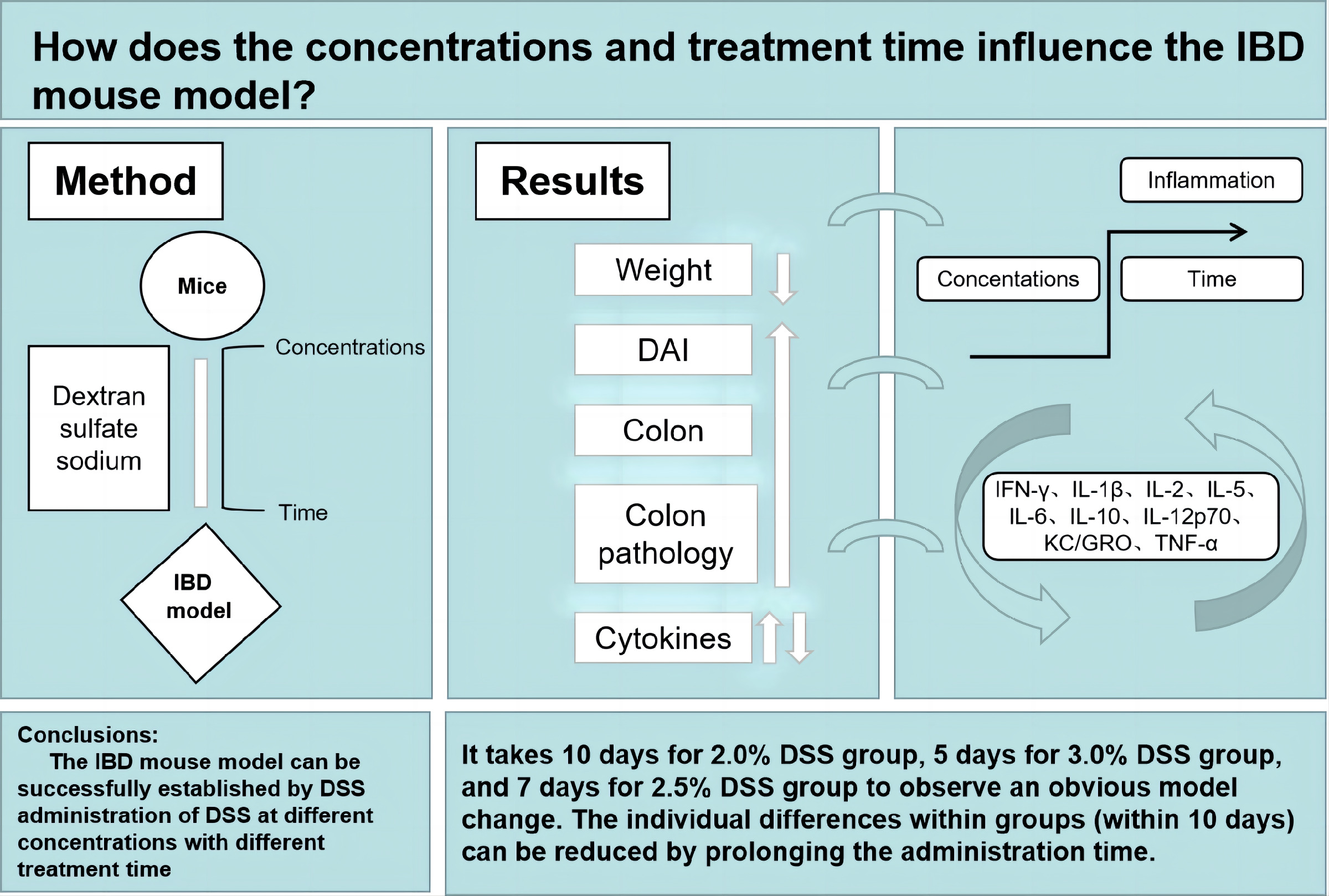
Objectives. To compare the IBD model in C57BL/6J mice given free access to water containing DSS at concentrations of 2.0%, 2.5% or 3.0% for 5, 7 or 10 days.
Materials and methods. Female mice (9 weeks old) were given access to drinking water containing DSS (2.0%, 2.5% or 3.0%) for 5–10 days. Body weight and colon length were then measured. Signs of edema, epithelial layer disruption, inflammatory cell infiltration, and cytokine induction, and severe colitis-related clinical signs were observed and analyzed.
Results. Weight of the mice decreased and disease activity index (DAI) score immediately increased in all 3 groups. The colon of mice in the 3.0% DSS group was shortened after 5 days, and the colon of mice in the 2.0% and 2.5% DSS groups was shortened after 7 days. A significantly increased intestinal injury score was observed on day 5 in the 3.0% DSS group, on day 7 in the 2.5% DSS group and on day 10 in the 2.0% DSS group. Cytokines were found to be elevated in all 3 groups after 5 days of DSS exposure, with higher DSS concentrations and longer administration times found to be associated with more serious inflammation of the intestinal tract. After 10 days of DSS administration, all mice in the 3.0% DSS group died.
Conclusions. It took 10 days for the 2.0% DSS group, 5 days for the 3.0% DSS group and 7 days for the 2.5% DSS group to develop obvious observable changes related to the induction of the IBD model. The individual differences within groups (within 10 days) could be reduced by prolonging the administration time. Excessive DSS concentration and longer DSS administration time (exceeding 7 days) may increase mortality of the mice.
Key words: concentration, inflammation, inflammatory bowel disease, dextran sulfate sodium, mouse model
Background
Inflammatory bowel disease (IBD) is a lifelong condition occurring in both adolescence and adulthood. It involves intestinal inflammation caused by impaired epithelium and can lead to aberrant gut microbiota and immune cell infiltration.1, 2 The clinical symptoms of IBD encompass abdominal pain, diarrhea, mucus, pus, bloody stool, and weight loss. In the long term, the development of IBD increases the risk of disability and cancer. Although the etiology of IBD is still elusive, research has shown that genetic, environmental and gut microbial factors are potential contributors to its development.3, 4, 5
Over the past decades, a variety of IBD models, such as spontaneous colitis models, adoptive transfer models, genetically modified models, and inducible colitis models, have been successfully developed.6 Animals, including mice, rats, dogs, and rabbits, have been used to establish these models.7 The C57BL/6J inbred mouse strain is commonly employed in dextran sulfate sodium (DSS)-induced IBD models and is found to be more suitable for chronic colitis induction than the BALB/c mouse strain.8
Among various animals and methods used for modeling IBD, establishing a C57BL/6J mouse model using DSS is relatively simple and therefore widely used. Perše and Cerar reported that adding 2.0% or 5.0% DSS to drinking water for 4–9 days was sufficient to induce acute colitis.6 However, reports on model differences after DSS administration within this concentration range are scarce. Hence, DSS was used in this study at 3 different concentrations (2.0%, 2.5% and 3.0%) to establish a C57BL/6J mouse model of IBD and to observe changes in symptoms and cytokine levels.
Objectives
This study aimed to compare models of IBD in C57BL/6J mice given free access to drinking water containing DSS at concentrations of 2.0%, 2.5% or 3.0% for 5, 7 or 10 days.
Materials and methods
Ethics statement
All experimental procedures involving animals were approved by the Laboratory Animal Care (approval No. SCXK-2018-0003) and Institutional Animal Care Use Committee (IACUC) at Shanghai University of Traditional Chinese Medicine (approval No. PZSHUTCM210416003).
Animals
The 9-week-old C57BL/6J wide-type (WT) female mice (weight >20 g) were purchased (Shanghai Lingchang Biology Science and Technology Co. Ltd., Shanghai, China) and housed in the animal facility of Longhua Hospital, Shanghai University of Traditional Chinese Medicine (Shanghai, China). All experiments were performed according to the local, institutional and federal regulations for vertebrate animal research. Mice were reared in a normal environment (temperature 22 ±2°C, humidity 40–60%), with a 12-hour light/dark cycle, and fed ad libitum for 1 week. The animals (n = 75) were divided into a 2.0% DSS group, 2.5% DSS group, 3.0% DSS group, and a control group (n = 21 mice in each DSS group and n = 12 in the control group). To induce colitis, 10-week-old female mice were administered DSS of different concentrations via drinking water, and their body weight, activities and condition (such as diarrhea and bloody stools) were observed and recorded. After 5 days of induction, 3 mice from each IBD group and 4 mice from the control group were euthanized. After 7 and 10 days of induction, 6 mice from each IBD group and 4 mice from the control group were euthanized. Decisions on the experimental protocol were reached through simple random sampling.
Dextran sulfate sodium administration
The molecular weight of DSS (MP Biomedicals Inc., Santa Ana, USA) ranged from 36 kDa to 50 kDa.2 Dextran sulfate sodium solutions (2.0%, 2.5% and 3%) were prepared using sterile drinking water and 100 mL of DSS was given to 5–6 mice per cage for 2 days. Before replenishing the DSS solutions, the remaining liquid was discarded.
Experimental design
After 1 week of acclimation, the 10-week-old mice were randomized into 4 groups and given access to distilled water containing 2.0%, 2.5% or 3.0% DSS, or distilled water for 10 days. Their body weight, excrement and fecal bleeding were monitored daily. Three or 6 mice from different groups were euthanized using pentobarbital sodium (0.1–0.2 mL, 0.1%) via intraperitoneal injection on the 5th, 7th and 10th day. Serum samples were collected for subsequent testing, the entire large intestine was harvested and the colon length was measured (Figure 1).
Weight and disease activity index
Body weight was recorded and a small fecal sample was collected from each mouse every day. Three major physical endpoints, including weight loss, diarrhea and rectal bleeding, were assessed using the disease activity index (DAI) scoring system9 (Table 1). Fecal samples were examined for consistency and tested for blood using the Fecal Occult Blood Complete test kit (BASO Diagnostics Inc., Zhuhai, China).
Histopathological evaluation
of colitis
Mid and distal colon samples were fixed in 4% buffered formalin, dehydrated through a gradient of ethanol, clarified with xylene, embedded in paraffin, then cut into 4-μm-thick sections and stained with hematoxylin and eosin (H&E). Colonic histopathological evaluation was performed and recorded by 2 blinded investigators. The tissue sections were assessed using grading standards described by Morris et al. (Table 2) and Scheiffele et al. (Table 3).10, 11
Meso scale discovery
Meso Scale Discovery (MSD) was carried out using commercially available kits (K15048D and K0081254; Lab Ex., Shanghai, China). Mouse serum samples were thawed on ice, diluted twice, centrifuged (3000 g at 4°C for 5 min), and left at room temperature for 20 min. Then, 1000 μL diluent was added and samples were left to sit for 20 min before being mixed thoroughly. Next, the antibody diluent (3 mL with 60 μL of each test antibody), a washing solution (0.05% phosphate-buffered saline (PBS) and Tween-20) and a plate reading solution were prepared. The washing solution (150 μL/well) was used to wash each well 3 times. Then, 50 μL of sample and detection antibody were added to each well, and the plate was sealed with sealing film and shaken at room temperature for 2 h. The washing process was repeated 3 times. Next, detection antibody (25 μL) was added to each well, the plate was sealed with sealing film and shaken at room temperature for 2 h. Once again, the washing process was repeated 3 times. Finally, 150 μL of plate reading solution was added to each well, and plates were read using the Meso QuickPlex SQ 120 (Meso Scale Discovery, Rockville, USA).
Statistical analyses
The IBM SPSS v. 22.0 (IBM Corp., Armonk, USA) and GraphPad Prism 9 (GraphPad Software, San Diego, USA) software were used for all analyses. The normality of data was initially assessed using the Shapiro–Wilk method. Normally distributed data are presented as mean and standard deviation (M ±SD), and non-normal data are presented as median (Me) and quartiles (Q25, Q75). The Kruskal–Wallis H test was used to analyze data that was not normally distributed, as well as normally distributed data without homogeneity of variance. For data that had normal distribution and homogeneity of variance, the comparisons between the 2 groups were made using the Student’s unpaired t-test. The multi-group comparisons and trends were analyzed with repeated measures analysis of variance (rm-ANOVA). The value of p < 0.05 represented a statistically significant difference.
Results
Body weight
Weight loss is a sufficient, objective and economical indicator of DSS-induced colitis in mice.12 Loss of >20% of initial body weight is a physiological indicator of animal stress and imminent demise.13 After 3 days of ad libitum access to DSS-spiked water, the weight of the 3 treatment groups decreased. Weight loss in the 2.0% and 2.5% groups did not differ; however, significantly different weight loss was found between the DSS groups and the control group. Furthermore, weight loss decreased at a faster rate in the 3.0% DSS group. After 10 days, the DSS-spiked water was removed and replaced with distilled water. However, mice in the 3.0% DSS group continued to lose weight and eventually died. On the 7th day after DSS removal, body weight in the 2.0% and 2.5% groups had gradually increased and returned to pre-DSS weight (Figure 2).
Disease activity index scores
Disease activity index scores between DSS groups were not significantly different, although significant differences were observed between the 3 DSS groups and the control group. Scores increased most rapidly in the 3.0% DSS group (Figure 3).
Colon length
A dose-dependent decrease in colon length was found in mice exposed to DSS when compared to the control. However, when the colon shortened to approx. 6 cm, there were no differences found among the 3 groups. Shortened colon length was observed in all 3 treatment groups after 5 days of exposure to DSS. After 7 days of DSS exposure, the change of colon length of mice in each group became shorter (Figure 4, Figure 5) (Table 4, Table 5). This suggests that 2.5% DSS induces a stable model of IBD.
Pathological and histological score
Significantly increased pathological and histological scores were found in the 3.0% DSS group on day 5. Compared with the control group, the scores for the 2.5% DSS group were significantly higher on the 7th day. On the 10th day, scores had increased in all 3 DSS groups, though there was no difference between them (Figure 6) (Table 6, Table 7, Table 8, Table 9).
Cytokines
Ten serological markers, including interferon-γ (IFN-γ), interleukin (IL)-1β, IL-2, IL-4, IL-5, IL-6, IL-10, IL-12p70, platelet-derived growth factor-inducible protein KC/growth-regulated oneogene (KC/GRO), and tumor necrosis factor alpha (TNF-α), were determined with MSD (Figure 7). The IL-4 decreased in the treatment groups to below the detection threshold of MSD, yet it could be detected in the control group (0.15 ±0.13 pg/mL). The IFN-γ and IL-5 levels increased as the concentration and exposure time to DSS increased in each treatment group. However, there was no statistical difference between the mice exposed to DSS and the control group, which indicated that the activity of IFN-γ and IL-5 had little significance on the model. Similarly, the levels of the anti-inflammatory marker IL-10 were higher in the treatment groups compared to the control group, though the increase was not significant, which suggests that this cytokine was not central to the development of the IBD model. Furthermore, IL-1β, IL-12p70, KC/GRO, and TNF-α were all significantly elevated after 5 days of exposure to DSS, though there was no difference between the groups after 7 days. Nonetheless, the levels of IL-6 significantly increased, which indicates that it is a useful biomarker of the IBD model.
Mortality
Six mice remained in each of the DSS groups after the 10th day of the experimental protocol, except for the 3% DSS group, in which 1 mouse died on the 9th day. After the DSS-spiked water was removed and the mice were given access to distilled water, all mice in the 3% DSS group died. There was no mortality in the other 2 DSS groups.
Discussion
This study showed differences in a mouse model of IBD induced using 2.0%, 2.5% and 3.0% DSS added to drinking water. After 10 days of exposure to DSS-spiked drinking water, animals from 3 concentration groups displayed an IBD model phenotype. These model-related changes occurred fastest in the mice exposed to 3% DSS, though the differences between the 3% and 2.5% fed mice were subtle before the 7th day. Furthermore, 10 days of exposure to 3% DSS led to a >20% decrease in body weight and an increased mortality rate.
Serological testing revealed that the cytokine IL-6 is a good indicator for the development of the IBD model phenotype. The IL-6 is produced by T cells, B cells, monocytes, and polymorphonuclear leukocytes (PMNs), and plays a role in both pro-inflammatory and anti-inflammatory immune responses.14 Most inflammatory markers were upregulated after 5 days of DSS exposure, though the fact that there were significant differences among the treatment groups indicated that the IBD model had yet to be established. However, the differences in serological markers were similar between the DSS groups after 7 days, and the IBD model was established in the 3.0% group. After a further 3 days of DSS exposure, the model had developed also in the 2.0% and 2.5% groups.
Results regarding colon length could be an important indicator for the development of the IBD mouse model, as colon length decreased in line with increased cytokine levels. Generally, IBD is related to dysregulated mucosal immune responses and barrier function of the intestinal epithelium.1 Indeed, interleukins are typically used to explore the etiology of IBD and observe the therapeutic effect of drugs. Also, IFN-γ, IL-1β, IL-12p70, and TNF-α are thought to promote inflammation and injury,15, 16 while IL-4, IL-5 and IL-10 are related to the inhibition of inflammation.17, 18 Secreted by T cells, IL-2 is a cytokine that can regulate CD4, CD8, natural killer (NK), and B cells, and increased IL-2 is used as a marker to signal the development of colonic intestinal inflammation.19 At the same time, IL-5 is related to type 2 immune responses. It can act on eosinophils and basophils during the immune-mediated mechanisms in IBD,20 and has been found in patients after intestinal surgery.12 The chemokine CXC motif chemokine ligand 1 (CXCL1) participates in the migration of inflammatory cells to the sites of injury, and its levels have been shown to be elevated in Crohn’s disease (CD) patients compared to healthy controls. Indeed, the correlation between CXCL1 and disease activity is even stronger than it is for C-reactive protein (CRP).21 In summary, a higher DSS concentration was associated with increased cytokine levels, yet no significant differences were found among the treatment groups as the time of exposure to DSS was prolonged.
Dextran sulfate sodium is widely used to establish IBD models. Many factors, including the molecular weight of DSS, age, sex, the strain of mice, and the administration method, are conventionally considered to exert influence on the model of IBD. Furthermore, microorganisms, especially intestinal flora, have been shown to have an impact on the model. Some microorganisms are thought to be favorable for modeling, and the onset of IBD is understood to result from a combination of microorganisms and innate intestinal immunity.12 A previous study showed that CD was driven by Th1/Th17 cells (through the production of IFN-γ and IL-12) and ulcerative colitis (UC) by Th2 cells (through the production of IL-4, IL-5, IL-10, and IL-13). Dextran sulfate sodium-induced colitis, though not a typical Th1-like model, more closely matches UC in humans.22 Indeed, the induction of DSS is more likely to cause colorectal damage in the distal regions, which is similar to the intestinal changes found in human UC patients in clinical practice.
In this study, factors that may interfere with the modeling of IBD were selected to create a protocol that would allow for the exploration of the effects of DSS concentration and time. Therefore, all of the investigated factors should be taken into consideration when using the study as a modeling reference resource. Based on the cytokine expression profile of the model, researchers could modify the DSS concentration and exposure time to match their research goals.
Limitations
The limitations of the study include the small number of animals, and the use of a single mouse strain and of female mice only. The addition of more animals, varied strains and the use of both sexes in future studies would provide more insight and more reliable results. In addition, further research should include mice that have been allowed to recover naturally from DSS-induced intestinal inflammation. Such mice would allow researchers to evaluate the efficacy of drugs for IBD.
Conclusions
The present study investigated the impact of a narrow range of DSS concentrations on the IBD modeling in mice. This provided an accurate and safe modeling concentration, as well as a suitable time period over which to expose the mice to DSS. Exposure to drinking water spiked with 2.5% DSS for 10 days seems to be a compromise that resulted in a higher success rate and lower intragroup differences. After removing DSS, the mice recovered slowly through self-regulation. However, a continuous use of DSS at a high concentration led to a high mortality rate. The periodic use of such DSS doses could be used in the development of the chronic mouse model of inflammatory bowel disease.
The physiological and cytokine profile changes that were observed in the DSS-induced IBD model are useful markers for exploring the etiology of IBD and for observing the effects of putative therapeutics. Furthermore, the results of this study can provide a useful resource for researchers attempting to establish an IBD mouse model that can match their own research needs.
A mouse model of IBD was successfully established using DSS at different concentrations and with different exposure times. It took 10 days for the 2.0% DSS group, 5 days for the 3.0% DSS group and 7 days for the 2.5% DSS group to develop observable changes indicative of IBD. Furthermore, intragroup differences (within 10 days) could be reduced by prolonging the exposure time. However, excessive DSS concentration and longer exposure time (exceeding 7 days) led to increased mortality.
Supplementary files
The supplementary files are available at https://doi.org/10.
5281/zenodo.7305141. The package contains the following files:
Supplementary File 1. The original data of the study, including the weight change record, DAI score change record, colon length, colon pathological score, and measurement data of cytokines.
Supplementary File 2. The data of the statistical analyses of this study, including the initial statistical analysis (normal distribution, homogeneity of variance, statistical test) and the repeated statistical test during the revision of the text.
Supplementary Table 1. Test methods and test values of each observation index in the study.