Abstract
Background. Ischemia/reperfusion (I/R) can cause damage to distant organs. Rutin is known to have antioxidant and anti-inflammatory properties, and inhibits cytokine and polymorphonuclear leukocyte (PMNL) infiltration. It may prevent the development of reperfusion injury.
Objectives. This study aimed to examine the role of PMNLs in distant organ (lung) injury after a liver I/R procedure, and to evaluate the protective effects of rutin in rats using biochemical and immunohistochemical methods.
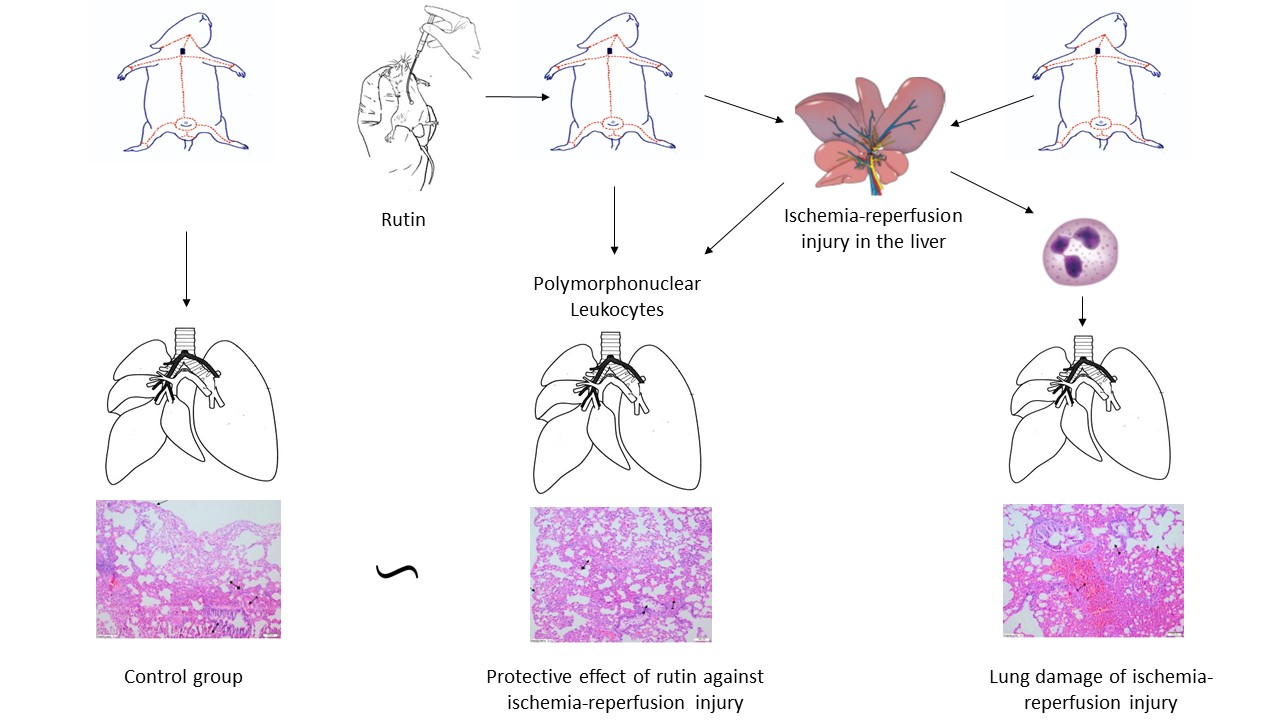
Materials and methods. In this study, 18 Wistar albino male rats (255–275 g) were used. Experimental animals were divided into 3 groups: a liver I/R (LIR) group, a 50 mg/kg rutin+liver I/R (RLIR) group and a sham operation (SG) control group. Experimental results obtained from the RLIR group were compared with the LIR and SG groups.
Results. Blood malondialdehyde (MDA) levels in the RLIR and SG groups were significantly lower compared to the LIR group (p < 0.001). Blood myeloperoxidase (MPO) activity in the RLIR and SG groups was significantly lower compared to the LIR group (p < 0.001). Total glutathione (tGSH) levels in the RLIR and SG groups were significantly higher compared to the LIR group (p < 0.001).
Conclusions. Rutin can be used to prevent distant organ (lung) damage due to liver I/R. However, more extensive studies are needed on this issue.
Key words: rutin, distant organ damage (lung), ischemia/reperfusion
Background
Ischemia is defined as a condition in which blood flow and the oxygen supply to tissues are reduced or completely obstructed due to various reasons. Reperfusion is the process in which blood flow is restored to the ischemic tissue.1 Tissue damage caused during the ischemic period increases with reperfusion.
Although ischemia/reperfusion (I/R) injuries have been examined in many tissues and organs, the pathophysiology of I/R injuries has not been fully elucidated. Studies have argued that I/R injuries are complex pathological processes which start with deoxygenation of the tissue, followed by oxidative stress, and result in an inflammatory response involving the activation of polymorphonuclear leukocytes (PMNLs) and the excessive formation of free radicals, which are responsible for the I/R injury. The reperfusion process causes these free radicals formed in the ischemic area to enter the systemic circulation. Activated PMNs are not only present in the primary tissue where the I/R event occurred, but are also transported to other tissues by the circulation, and cause damage to other organs (remote tissues). Activated PMNLs cause increased pulmonary capillary permeability, leading to the passage of PMNLs into the tissue.2
In a study by Yu et al., it was stated that an I/R procedure applied to the liver can cause severe inflammatory damage not only in the liver but also in the lung.3 This information suggests that local I/R injuries may cause systemic damage to distant organs. Moreover, vascular permeability due to activated PMNLs and oxidative stress are the major components in distant organ injury. In the present study, the impact of rutin (3, 3′, 4′, 5, 7-pentahydroxyflavone-3-rhamnoglucoside) on distant organ (lung) damage due to liver I/R was investigated.
Rutin is a P1 vitamin flavonoid. It is known to have antioxidant and anti-inflammatory properties, and inhibits cytokine and PMNL infiltration.4, 5 Rutin has the potential to bind free oxygen radicals and convert them into more stable non-reactive species, as well as end the free radical chain reaction.6 Rutin exerts a cleaning effect by inhibiting 1,1-diphenyl-2-picrylhydrazyl (DPPH) radicals.7 The lipid peroxidation inhibition and superoxide radical-clearing potential of rutin depends on the dose of rutin used.6 In the inflammation zone, pro-inflammatory cytokines such as tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β) and interleukin-6 (IL-6) are secreted from pulmonary cells and alveolar macrophages. The secretion of TNF-α, IL-1β, IL-6, and monocyte chemotactic protein (MCP) encourages the production of more effective pro-inflammatory cytokines and chemokines such as macrophage inflammatory protein (MIP), keratinocyte-induced chemokine, cytokine-induced neutrophil chemoattractant (CINC), and macrophage inflammatory protein-2 (MIP-2), which mediates the recruitment of PMNLs, macrophages and lymphocytes. Studies have shown rutin to suppress TNF-α and IL-1β production, which are known as pro-inflammatory mediators.9 In umbilical vein endothelial cells (HUVECs) during the acute period induced lipopolysaccharides (LPS), mice with acute liver damage intoxicated with carbon tetrachloride (CCI4), and renal inflammation, rutin was found to reduce TNF-α formation.10, 11, 12 Rutin was shown to reduce leukocyte infiltration of the lung by reducing the expression of pro-inflammatory cytokines. In addition, it has been demonstrated that rutin protects vascular barrier integrity and reduces hyperpermeability.11 Based on this information, it was hypothesized that rutin may help prevent the development of pulmonary damage in liver I/R procedures.
Objectives
Ischemia/reperfusion causes distant organ damage. Rutin may prevent the development of reperfusion injury. This study aims to examine the role of PMNL in distant organ (lung) injury in liver I/R procedures and the protective effect of rutin in rats using biochemical and immunohistochemical methods.
Material and methods
Animals and chemicals
This experimental study included a total of 18 male albino Wistar rats weighing 255–275 g. The experimental animals were supplied by the Medical Experimental Application and Research Center of Atatürk University (Erzurum, Turkey). The rats were housed in batches and provided with food and water ad libitum, at a temperature of 22°C with 12-hour light/dark cycle until the experiments. The study was conducted at the Atatürk University Experimental Studies and Research Center. Thiopental sodium was supplied by the IE Ulagay (Istanbul, Turkey), and rutin was obtained from Solgar (Leonia, USA).
The animals were divided into 3 groups: liver I/R (LIR) group, 50 mg/kg rutin plus liver I/R (RLIR) group and sham operation control (SG) group, with 6 rats in each.
Ethical standards
This study was approved by the Ethics Committee at the University of Erzincan (approval No. 28.06.2018/148). The experimental procedure was approved by the Erzurum Atatürk University Animal Research Committee. The animal experiments were performed in accordance with the International Guidelines for the Use and Care of Laboratory Animals.
Surgical and pharmacological procedures
All surgical procedures were carried out under general anesthesia using an intraperitoneal injection of 25 mg/kg of thiopental sodium at appropriate intervals, with xylazine sniffing. Rutin was administered at a dose of 50 mg/kg to the rats in the RLIR group by oral lavage 1 h before anesthesia induction, whereas rats in the SG and LIR groups were administered the same amount of distilled water. Following thiopental sodium injection, the animals were kept anesthetized for an appropriate period to perform the surgery. The appropriate anesthesia period for surgical intervention was understood as the period required to immobilize the animals in supine position. After immobilization was confirmed, the anterior abdomen was accessed through a 3.5–4 cm vertical incision and a laparotomy was carried out in all rats.
Rats in the SG group were not subjected to further procedures after the abdominal cavity was closed. Rats in the RLIR and LIR groups were subjected to total hepatic ischemia by clamping the bile duct, portal vein and hepatic artery for 1 h, and then reperfused for 6 h.
At the end of the surgical procedure, all rats were sacrificed using high-dose anesthesia and their lung tissue was removed. Biochemical and histopathological examinations were carried out on the removed lung tissue. Experimental results obtained from the RLIR group were compared to the LIR and SG groups.
Sample preparation
Blood samples collected from the animals to be placed in separation gel were taken in drainage serum tubes. All collected samples were left to incubate at room temperature for 15 min and centrifuged for 15 min at 1500 × g for separation. All samples were kept at −80°C until analysis. Before dissection, all tissue samples were rinsed with a phosphate-buffered saline (PBS) solution, and the lung tissues were then homogenized in a 50 mM ice-cold phosphate buffer at a pH of 7.4, which was suitable for measuring the variables. The obtained homogenates were then subjected to centrifugation at 5000 rpm for 20 min at 4°C. The supernatant portion was separated for analysis of the protein concentrations of myeloperoxidase (MPO), malondialdehyde (MDA) and total intracellular glutathione (tGSH). The protein supernatant levels were determined utilizing the technique described by Bradford.13 The results were expressed in grams after being divided by the concentration of protein.5
MDA, MPO and tGSH analyses
Malondialdehyde levels were determined using the technique described by Ohkawa et al. Briefly, the absorbance of the pink complex formed as a result of the reaction between MDA and thiobarbituric acid (TBA) at a high temperature (95°C) was measured spectrophotometrically at a wavelength of 532 nm.14 The MPO-mediated H2O2 oxidation reactions containing a 4-aminoantipyrine/phenol solution was used as a substrate to determine MPO enzyme activity.15 The tGSH analysis in serum and tissue was performed according to the method defined by Sedlak and Lindsay. DTNB (5,5’-dithiobis [2-nitrobenzoic acid]) disulfite is chromogenic in the medium, and DTNB is reduced easily by sulfhydryl groups. The yellow color observed after the reduction of DTNB was read spectrophotometrically at a wavelength of 412 nm.16
Histopathology
All tissues were fixed in 10% formaldehyde, dried using ethanol, and then embedded in paraffin blocks. The tissues were cut into 4 mm-thick sections, deparaffinized with xylene, and rehydrated with alcohol and water. Hematoxylin and eosin (H&E) were used to stain the tissue samples, which were then analyzed under a light microscope (Olympus pX53; Olympus Corp., Tokyo, Japan) equipped with a digital camera system (Olympus UTVO. 5XG-3; Olympus Corp.) by the same pathologist, who was blinded to the procedure.
Statistical analyses
Statistical analysis was performed using IBM SPSS Statistics for Windows v. 20 (IBM Corp., Armonk, USA). The results for continuous variables were reported as mean ± standard deviation (M ±SD) for normally distributed variables and as median (interquartile range (IQR), Q1–Q3) for non-normally distributed data. The Shapiro–Wilk test was used to test for normal distribution of continuous variables. One-way analysis of variance (ANOVA) was used to compare continuous variables between groups when the variables were normally distributed. For non-normally distributed variables, the Kruskal–Wallis test was performed. Before ANOVA, the homogeneity of the variances was tested using Levene’s test. After ANOVA, a Tukey’s honestly significant difference (HSD) test was performed as a post hoc test when the homogeneity assumption was met. Otherwise, a Games–Howell test was used as the post hoc test. Dunn’s test was performed as a post hoc test after the Kruskal–Wallis test. A p-value <0.05 was considered statistically significant.
Results
Biochemical results
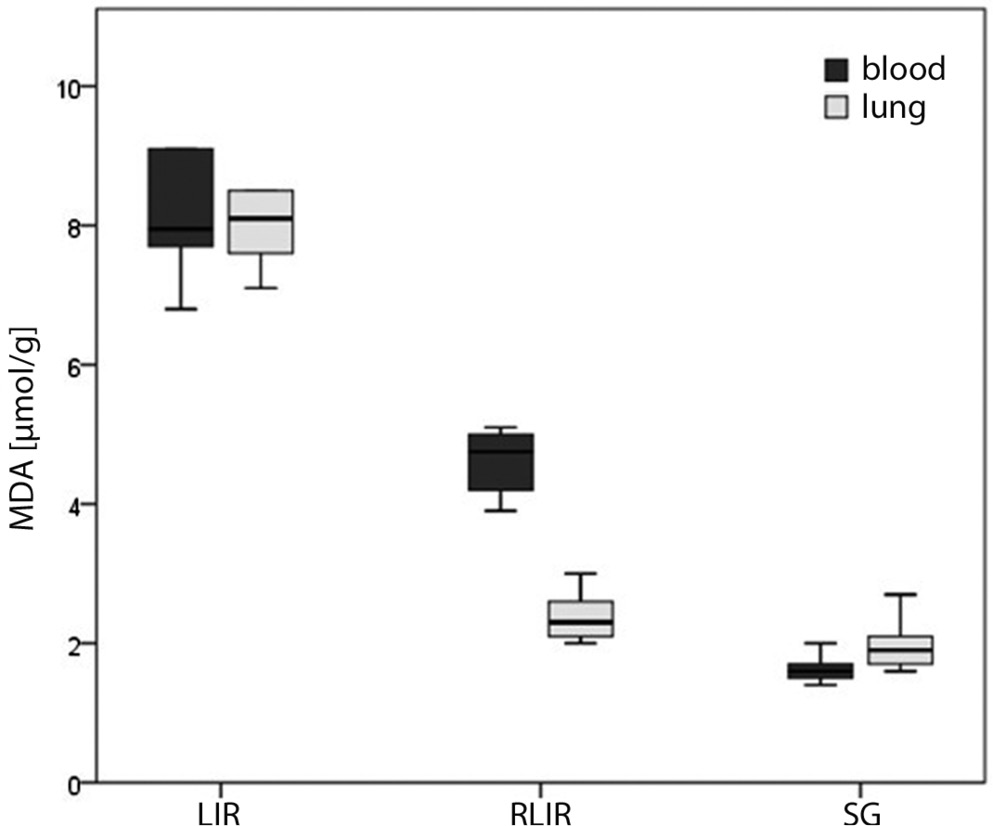
As shown in Table 1 and Figure 1, blood MDA levels in the LIR, RLIR and SG groups were 8.1 ±0.9 µmol/g protein, 4.6 ±0.5 µmol/g protein and 1.6 ±0.2 µmol/g protein, respectively. Lung MDA levels were 7.9 ±0.5 µmol/g protein, 2.4 ±0.4 µmol/g protein and 2.0 ±0.4 µmol/g protein, respectively. The total MDA levels in blood and lung in the RLIR and SG groups were significantly lower compared to the LIR group (p < 0.001, ANOVA, F = 180.5; p < 0.001, ANOVA, F = 339.3, respectively).
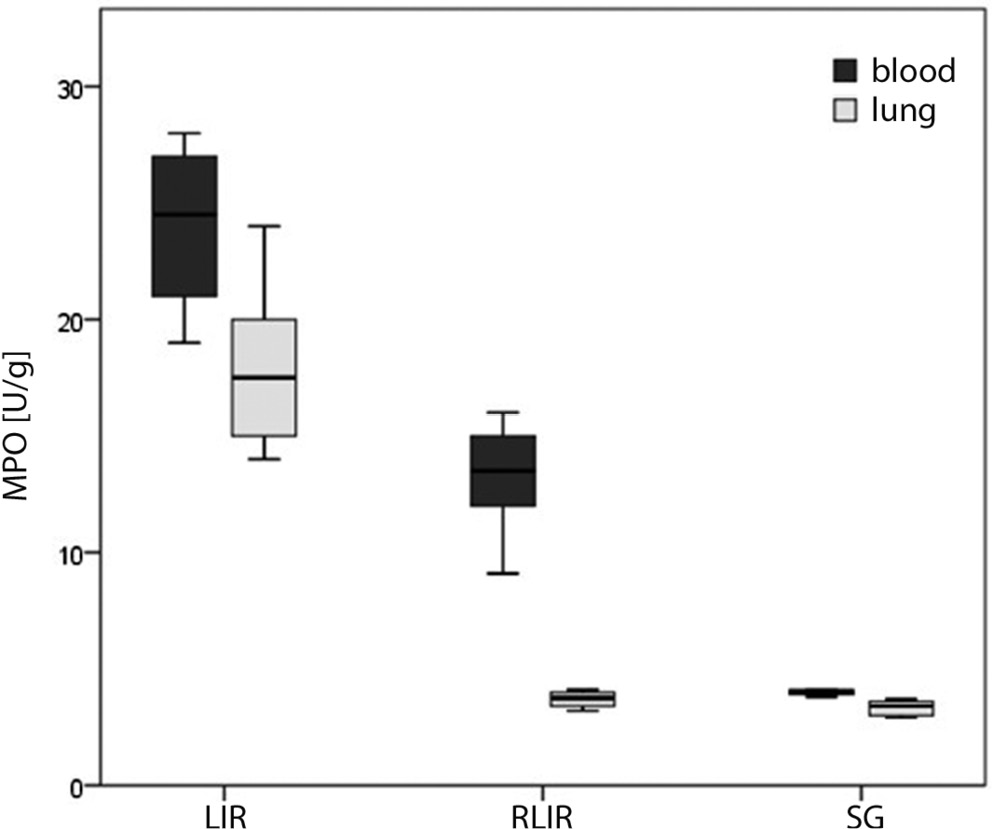
As shown in Table 1 and Figure 2, blood MPO levels in the LIR, RLIR and SG groups were 24.0 ±3.6 µmol/g protein, 13.2 ±2.5 µmol/g protein and 3.9 ±0.3 µmol/g protein, respectively. Lung MPO levels were 18.0 ±3.7 µmol/g protein, 3.7 ±0.4 µmol/g protein and 3.3 ±0.3 µmol/g protein, respectively. Significantly lower total MPO levels were observed in the RLIR and SG groups compared to the LIR group (p < 0.001, ANOVA, F = 96.7; p < 0.001, ANOVA, F = 88.5, respectively).
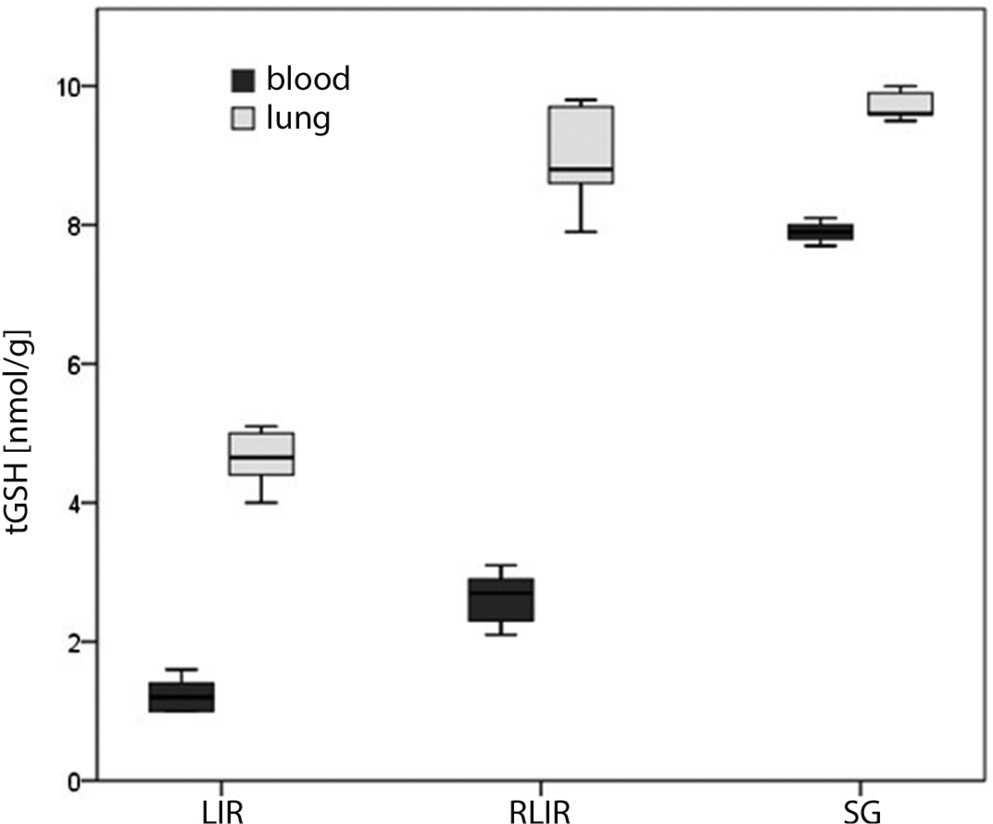
As shown in Table 1 and Figure 3, tGSH levels in the LIR, RLIR and SG groups were 1.2 ±0.2 nmol/g, 2.6 ±0.4 nmol/g and 8.1 ±0.4 nmol/g in the blood, and 4.6 ±0.4 nmol/g, 8.9 ±0.7 nmol/g and 9.5 ±0.5 nmol/g in the lung, respectively. The total tGSH levels in the RLIR and SG groups were significantly higher compared to the LIR group (p < 0.001, Kruskal–Wallis test, p < 0.001, ANOVA, F = 139.2, respectively).
Histopathology
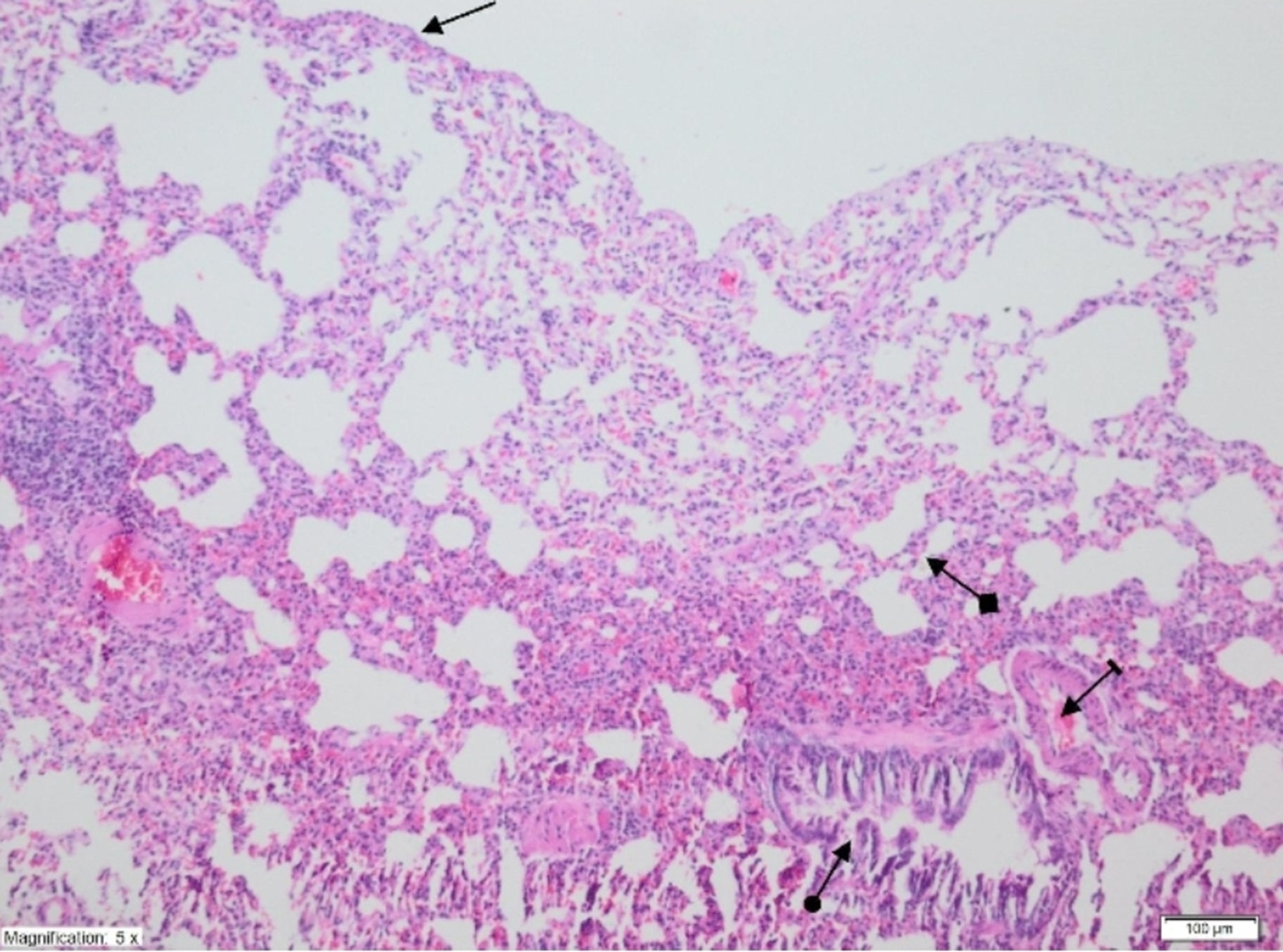
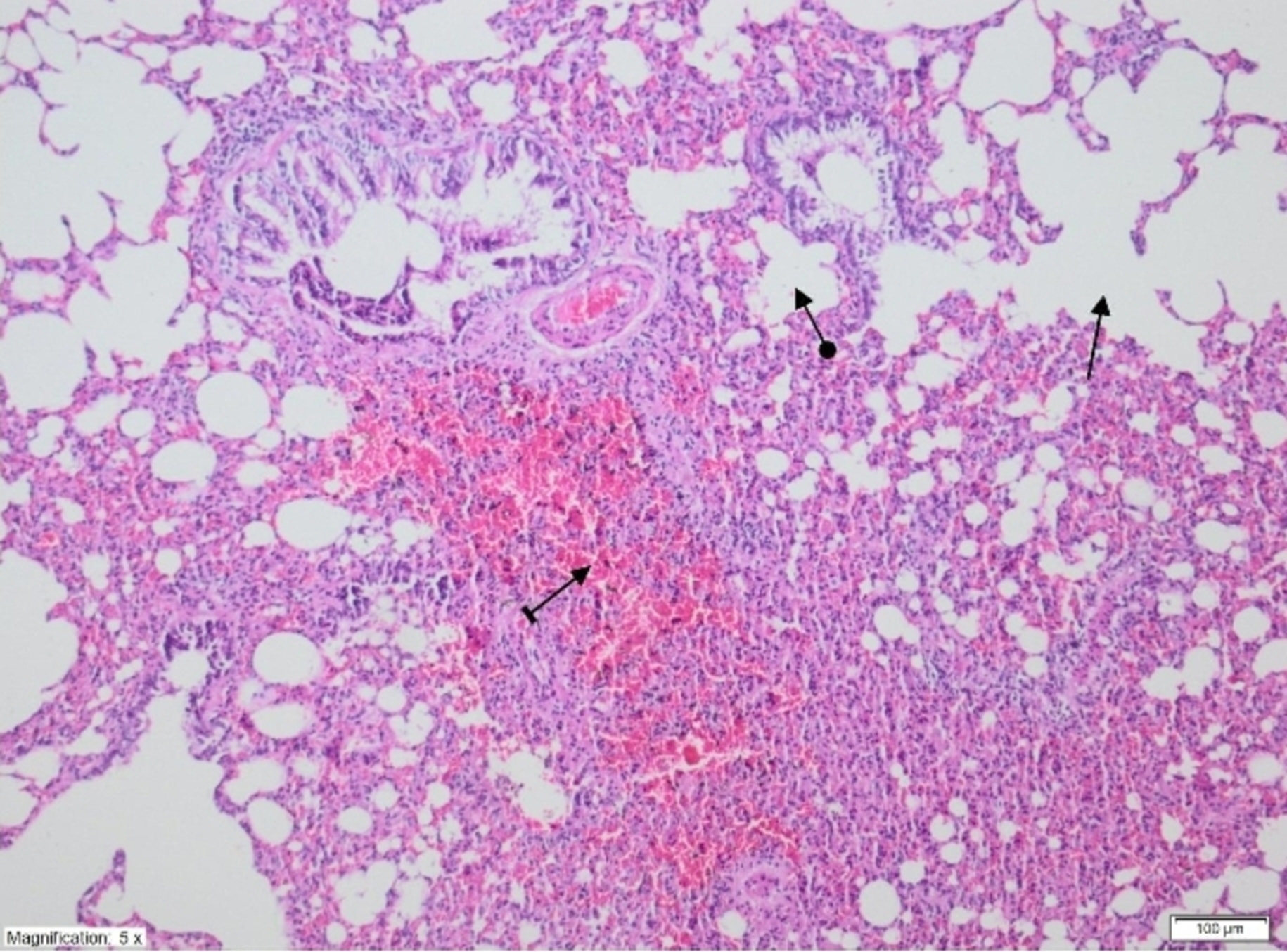
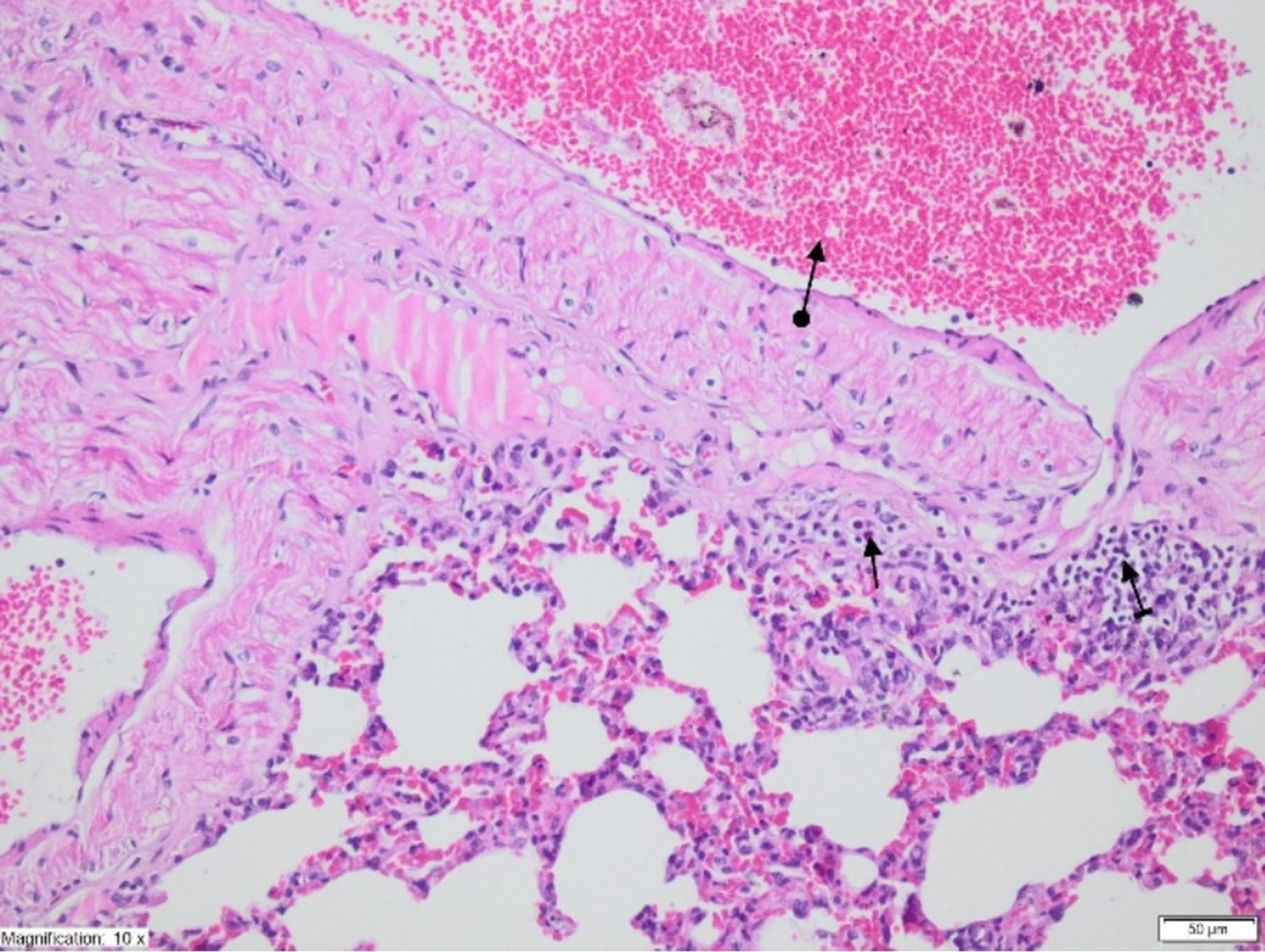
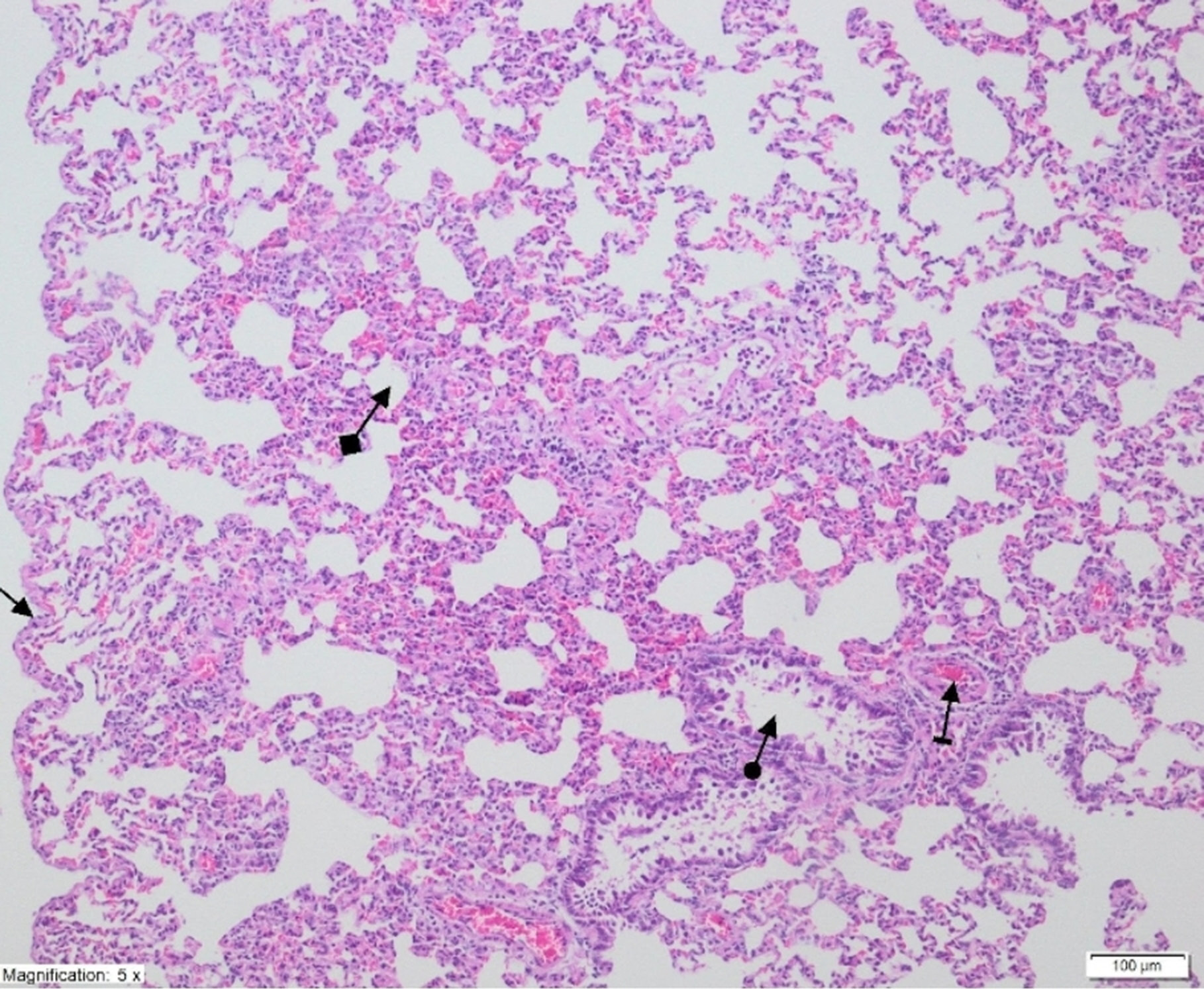
On histopathological examination, the alveolar vein, bronchiole structures and pleural mesothelium were identified as normal in pulmonary tissues in the SG group (Figure 4). However, significant areas of alveolar degeneration, edema and hemorrhage were observed in the pulmonary tissues of the LIR group (Figure 5). Congestion, PMNL and lymphocytic infiltration were observed in the lung tissues in the LIR group (Figure 6). In the lung tissue of the RLIR group, the pleural mesothelium, and alveolar, vascular and bronchial structures were identified as normal, which was similar to the observations in the SG group (Figure 7).
Discussion
Reperfusion is an important surgical treatment for reversing ischemic damage. It has been reported that a considerable amount of free oxygen radicals are formed during re-oxygenation of ischemic tissue. After ischemia, local and systemic damage is initiated by reperfusion via free oxygen radicals and inflammatory mediators. The reperfusion process causes the free radicals formed in the ischemic area to enter the systemic circulation. They then induce direct damage or indirectly exert a stimulating effect on the activation of neutrophils and the production of cytokines. Most of these mediators lead to the migration of neutrophils into tissues by neutrophil–endothelial interactions, and the migrating neutrophils degranulate and damage the tissue. Reperfusion of ischemic tissue leads to a disruption in the endothelium-dependent dilatation of arterioles, formation of leucocyte plugs in capillaries, and increased fluid filtration. This in turn causes leakage of plasma proteins out of post-capillary venules and thus disrupts microvascular functions. Restoring blood flow can lead to the development of multisystem organ dysfunction and mortality. This damage is observed especially in myocardial, renal and pulmonary tissues. Animal studies have correlated the degree of reperfusion injury with the degree of PMNL activation.17
The PMNLs release various enzymes, such as MPO and free oxygen radicals, which in turn exacerbates the damage.18 In a recent study by Kumbasar et al., MPO activity was reported to increase in ovarian tissues undergoing I/R.19 There are numerous studies showing that MDA levels increase in the damaged tissue formed as a result of LIR.20, 21 Under normal physiological conditions, the oxidant–antioxidant balance is maintained in favor of antioxidants. Disruption of this balance leads to tissue damage. This condition is called oxidative stress, and oxidant–antioxidant balance is used to determine whether tissue damage will occur. It has been shown in various I/R injury models that oxidant levels increase, antioxidant levels decrease, and the balance between oxidants and antioxidants changes in favor of oxidants. This demonstrates that enzymatic and non-enzymatic antioxidant mechanisms are continuously working in a controlled manner against oxidant mechanisms in tissues. Total glutathione is one of the critical non-enzymatic endogenous antioxidants that play a role in cellular defense against oxidative damage. It directly reacts with hydrogen peroxide (H2O2), hydroxyl (OH−), superoxide (O2−), and alkoxy (RO•) radicals in living tissues to protect the cell from the damaging effects of free radicals.22 High tGSH levels are an important marker of cellular function and viability. Decreased tGSH levels indicate a poor intracellular defense system and are regarded as a damage marker.23 Oxidation parameters increase during the reperfusion period after ischemia.
Free oxygen radicals are also known as reperfusion mediators. Tok et al. demonstrated that I/R treatment results in decreased tGSH levels and cause serious oxidative damage to rat kidneys; such damage can be reduced with the use of antioxidants.24 Demiryilmaz et al. reported that tGSH levels decreased and the levels of MDA increased in ovarian tissue treated with I/R.25 The lungs are a large reservoir of neutrophils, and a significant proportion of I/R injuries occur in the lungs.26 Fantini et al. emphasized that lung injuries due to reperfusion are characterized by non-hydrostatic pulmonary edema resulting in progressive hypoxemia, pulmonary hypertension, decreased lung compliance, and adult respiratory distress syndrome (ARDS).27 In addition, humoral mediators produced during reperfusion cause end-organ damage in regions far away from the focus of the I/R, and the lungs are the target organs.27
Ischemia/reperfusion injury to the lung can be observed on light microscopy examination of histopathological preparations stained with H&E.28, 29 The accumulation of neutrophils in capillary beds, interstitial edema, thickening of the alveolar septum, and the presence of protein-containing exudates in alveolar air cavities are common. Quantitative injury assessment is possible by examining 10 or 20 different sites under high magnification and counting the PMNLs in slides prepared in the same way.30
Various substances, such as superoxide dismutase, allopurinol, catalase, mannitol, vitamin C, pravastatin, alpha-tocopherol, L-carnitine, and pentoxifylline, have been used as treatments. These medications have been shown to be effective in eliminating the negative effects of free oxygen radicals after I/R. These antioxidant substances have shown a protective effect against distant tissue organ damage after I/R, either by decreasing pulmonary microvascular permeability and preventing neutrophil accumulation or by activating the antioxidant system. Dietary antioxidants such as flavonoids found in vegetables, fruit and beverages are known to protect the human body against a range of diseases such as cancer, diabetes, osteoporosis, and cardiovascular and neurodegenerative disorders.31 Flavonoids in vegetables are used more often due to their better compatibility with the human body and fewer side effects.32
Rutin, also known as vitamin P, is a flavonol abundantly found in many foods including apples, vegetables, tea, and wine. The antioxidative, antibacterial, anti-inflammatory, antitumor, anti-ulcer, antidiarrheal, antimutagenic, immunomodulating, and vasodilatory characteristics of rutin have been shown.33
In one study, rutin was compared to ascorbic acid (Vc) and butylated hydroxytoluene (BHT), which are commonly used in the pharmaceutical industry. Inhibition of DPPH, which measures rutin antioxidant activity, was found to be higher than that of Vc but similar to that of BHT. The reducing capacity of rutin and the hydroxyl radical (OH) sweeping effect has been found to be similar to that of BHT. The effect of rutin inhibiting lipid peroxidation was greater than that of Vc.6 Therefore, it is recommended to use antioxidants in combination.34
In addition, it has been shown that rutin alleviates I/R injuries that develop in organs such as the heart, brain, liver, kidney, and testes.35, 36, 37, 38, 39 In their study, Adefegha et al. found that rutin has a protective effect against reactive oxygen-mediated oxidative damage due to its anti-inflammatory activity, as seen in rat models of acute inflammation.40 Studies on LPS-induced acute lung injury (ALI) have shown rutin treatment to not only prevent histopathological changes in lung tissue, and also inhibit the transit of polymorphonuclear granulocytes into bronchoalveolar lavage fluid. The LPS-mediated inflammatory reactions, including increased lipid peroxidation and secretion of pro-inflammatory cytokines, are prevented by rutin treatment in a dose-dependent manner.5 It has been reported that the decreased activities of antioxidant enzymes such as superoxide dismutase (SOD), glutathione peroxidase (GPx), catalase, and heme oxygenase-1 (HMOX1) in LPS-induced injuries can be reversed by rutin.5
Our experimental findings show that the I/R procedure caused significant oxidative damage to liver tissue. It was observed that I/R induced in the liver also caused I/R injuries in distant organs such as the lungs, resulting in hemorrhage, edema, and PMNL infiltration into the lung parenchyma. In our study, the difference in blood and lung MDA levels in the LIR, RLIR and SG groups was statistically significant (p < 0.001). The levels of MDA were significantly lower in the lungs compared to the blood in the RLIR group (p < 0.001). In the LIR group, MPO activity was higher compared to the RLIR and SG groups (p < 0.001). Our experimental results and literature review show that the antioxidant properties of rutin prevent I/R-induced oxidative damage to the lungs caused by PMNL migration in distant organs after I/R injuries to the liver. In our study, tGSH, an important antioxidant in the body, decreased in blood and lungs in the LIR group, increased in the RLIR group and had the highest value in the SG group (p < 0.001). Rutin administration significantly prevents the increase of oxidation parameters such as MDA and MPO, and the decrease in tGSH level in rats with distant organ (lung) injury as a result of liver I/R injury (p < 0.001).
The results of this study showed that immunohistochemical findings were consistent with our biochemical findings.
Limitations
Many substances have been implicated in the pathogenesis of I/R distant organ damage. However, these items could not be included in our study. In addition, many substances are thought to be useful in I/R injuries. Therefore, there is a need for comprehensive studies including items other than rutin. It is also important to examine the molecular histopathology of the tissues.
Conclusions
Ischemia/reperfusion injuries are complex pathological processes that start with the deoxygenation of tissue, continue with change the oxidant/antioxidant balance in favor of oxidants, and develop with an inflammatory response. In I/R injuries, MDA and MPO levels increase and tGSH levels decrease. The administration of 50 mg/kg of rutin significantly prevented I/R injuries. It was found that rutin also showed antioxidant activity in addition to its known anti-inflammatory activity. The results of this study suggest the possible utilization of rutin in clinical practice to prevent I/R injuries.