Abstract
Background. Physical activity undertaken in the treatment process additionally increases the oxygen demand of the working muscles. It seems interesting to see whether a delivery of an enriched respiratory mixture can have an impact on lower acidification of working muscles and oxygenation of tissues.
Objectives. To assess tissue saturation and the level of acidification at rest and during exercise while breathing atmospheric air or an oxygen-enriched mixture.
Materials and methods. Tissue saturation and lactate concentration at rest and during exercise were assessed in 18 females with an average body mass index (BMI) of 42 kg/m2. The study regimen was as follows: day 1 – cardiopulmonary exercise testing (CPET) – determination of the threshold load; day 2 – 20 min of physical effort on a cycloergometer (threshold load, breathing atmospheric air); day 3 – 20 min of physical effort on a cycloergometer (threshold load, breathing mixture enriched with oxygen). Saturation measurements were performed in 3 places on the patient’s body by measuring absorbance via near-infrared spectroscopy (NIRS).
Results. A significant decrease in heart rate (HR) at rest was found when using the oxygen-enriched air mixture (Z = 2.1339, p = 0.0328 (effect size (ES) = 0.478)). During the exercise, a significant decrease in saturation was shown only for the midpoint of the quadriceps muscles (Z = 2.1572, p = 0.309 (ES = 0.600)). Medium effect sizes were shown by the difference in resting and exercising lactate concentration change between the experimental models studied (Z = 2.5041, p = 0.0122 (ES = 0.707)). In the experimental models studied, different medium effect sizes were demonstrated in the resting and exercising lactate concentration change.
Conclusions. Oxygen-enriched air mixture contributes to reducing hypoxia in working muscles of obese people. Oxygen supplementation can result in higher physical fitness levels. The implementation of oxygen-enriched air mixture is a promising therapeutic strategy for obese patients who exhibit high lactate concentrations after exercise.
Key words: obesity, tissue saturation, near-infrared spectroscopy (NIRS)
Background
Obesity as a chronic disease
Obesity is currently considered an increasingly severe social and health-related issue. World Health Organization (WHO) qualifies obesity as a chronic disease along with circulatory diseases, diabetes, cancer, and respiratory diseases. Treating excess weight and obesity remains a major challenge, faced by both doctors and patients. The primary treatment of obesity consists of exercise and diet. Pharmacological therapy or surgical interventions are recommended in cases where insufficient weight loss was achieved following primary treatment. It has been shown that obesity has a major impact on respiratory function. In obese people, a reduction in chest compliance due to adipose tissue and a rise of the diaphragm in the resting position caused by fat accumulation in the abdominal cavity results in a significant decrease in respiratory mobility. Additionally, the excess of adipose tissue leads to the collapse of soft tissues of the upper respiratory tract, causing respiratory disturbances during sleep and obstruction in the lower respiratory tract. This may promote the exacerbation of asthma and chronic obstructive pulmonary disease (COPD), which often coexist with obesity.1 The described functional disorders manifest themselves in the form of decreased forced vital capacity (FVC), decreased functional residual capacity (FRC), decreased expiratory reserve volume (ERV), and decreased forced expiratory volume in 1 s (FEV1).2 The consequences of impaired respiratory mechanics are ventilation disorders manifested by increased physiological venous leakage. The adequate ventilation/blood perfusion ratio is responsible for gas exchange in the lungs. Under physiological conditions, the ratio is higher in the upper lung fields (more ventilation, less perfusion), optimal in the middle parts and lower in the lower fields (less ventilation, more perfusion).
The lower lung fields represent a higher percentage of the mass involved in ventilation and thus they affect average gas exchange values. In line with the above data, obesity additionally impairs the lower lung ventilation. As a consequence of the increasing imbalance between perfusion and ventilation, gas exchange disturbances may lead to hypoxemia and, over time, hypercapnia. An increased oxygen concentration in respiratory mixtures reduces physiological venous leakage. There are numerous factors influencing the effectiveness of obesity therapy and treatment of excess weight.3, 4
Oxygen as an important element in the treatment of obesity
The most important factor in the treatment of obesity seems to be the (individually dependent) metabolic rate. One of the most essential components in the metabolic process is oxygen. Moderate and long-lasting physical activity is associated with a higher metabolism rate of adipose tissue.5 Combustion is carried out in the process of oxidative phosphorylation coupled with the respiratory chain and requires the adequate supply of oxygen to the cells. It has been proven that people with obesity require larger amounts of O2 in order to carry out life functions at a basic level.6 The increased demand for oxygen by working muscles is observable during incremental physical activity.7 Kress and Hall confirmed that the use of oxygen at rest by people suffering from morbid obesity is 16% higher in comparison to people with proper body weight.8, 9 At the same time, their maximal O2 uptake (VO2max) is considerably lower. This might be due to a lower blood flow and metabolic rate at the muscular level. In patients with obesity, the factors limiting oxygen supply are respiratory dysfunctions, such as restriction and hypoventilation syndrome, leading to both hypoxia and hypoxemia. This condition is exacerbated during physical activity, leading to excessive purine nucleotide degradation, an increase in free radical transformation, and the acid-base disorder that increases fatigue symptoms. A decrease in oxygen content in arterial blood may reduce the exercise capacity in healthy individuals and in people with respiratory and circulatory diseases or anemia.10, 11, 12 A significant mechanism, which is not fully understood, is the effect of oxygen levels on peripheral skeletal muscle fatigue induced by systemic exercise. Hyperoxia during moderate exercise in patients with COPD without hypoxemia affects the dynamics of the growth rate of oxygen consumption and reduces the transient increase in lactate. Furthermore, the lower demand for ventilation caused by O2 supplementation is not correlated with the improvement of muscle function, but may result from the direct inhibition of chemoreceptors.13 Studies have shown that the prevention of vigorous exercise-induced arterial hypoxemia significantly reduced quadriceps fatigue as assessed through stimulation of the femoral nerve before and after exercise.14, 15 A study by Amann et al. assessed whether peripheral muscle fatigue is sensitive to changes in arterial oxygen content during exercise. It was shown that hypoxia, normoxia and hyperoxia during strenuous systemic exercise influenced the rate of development of peripheral muscle fatigue.16
Bioenergetically, O2 supplementation leads to an improvement in the phosphocreatine/inorganic phosphate ratio and the total adenosine triphosphate (ATP) content through more efficient oxidative metabolism associated with reduced lactate production and ATP decomposition with the simultaneous formation of harmful free radicals.17 Andrade et al., based on an extensive analysis of studies on the effectiveness of O2 supplementation during physical activity, showed less inflammation with a greater intensity of exercise achieved by the respondents.18 Consequently, the use of oxygen supplementation in training people with respiratory failure was implemented. Freitag et al. reached similar conclusions in their publication. He emphasized the importance of using air mixture enriched with oxygen in patients with numerous chronic diseases, such as obesity, during physical training.19
Hypothesis
The physiology of exercise is different in obese and lean people. In people with obesity, lactate accumulation and acidification occur faster during exercise. The accumulation of lactate reduces the possibility of further physical activity or its increased intensity. We hypothesize that respiration with an oxygen-enriched mixture leads to an alteration in saturation levels and positive metabolic changes in muscles during physical effort. Besides, increasing the amount of oxygen by preventing metabolic acidosis has a generally beneficial effect on the body. A study by Ehrsam et al. assessed the impact of metabolic acidosis on metabolism during exercise in healthy subjects and subjects with rheumatoid arthritis. Measurements were made at rest and during traning (at 30% and 60% of the maximum O2 uptake (VO2max), after 20 min). The study found that in hypercapnia, the metabolic effects of acidosis are modified by increased levels of circulating catecholamines, blood pressure and ventilation. The increased effect of circulating catecholamines, especially in obese patients, can cause many detrimental effects. Increasing the oxygen supply may counteract this outcome.20
Objectives
The aim of the study was to assess tissue saturation (abdominal fat around the navel and muscle tissue around the quadriceps muscle of the thigh and gastrocnemius muscle) at rest and during exercise while breathing atmospheric air or an oxygen-enriched breathing mixture.
Materials and methods
Patients were qualified by a physician (diabetologist) during a visit to the Metabolic Outpatient Clinic of the Heliodor Święcicki Clinical Hospital in Poznań, Poland. During qualifying visits, detailed interviews were conducted and the available medical documentation on concomitant illnesses was analyzed. A body composition analysis was made, as well as basic laboratory tests. In addition, during the visit, the patient signed a consent form to participate in the research. On the basis of these procedures, it was assessed whether the patient met the inclusion criteria outlined in Table 1.
Initially, 35 patients were qualified for the project. Seventeen patients were excluded from further procedures (Figure 1). Tests were carried out in a group of patients (n = 18, all female), mean age 33 years (IQR: 29–50), body mass 115.5 kg, (IQR: 97–125), body height 1.7 m (IQR: 1.6–1.7), with an average body mass index (BMI) of 42 kg/m2 (IQR: 36.1–45.2), who were hospitalized in Greater Poland Center of Pulmonology and Thoracic Surgery of Eugenia and Janusz Zeyland in Poznań, Poland. The characteristics of the study group are presented in Table 2. The study was conducted according to the Declaration of Helsinki and the National Statement and Human Research Ethics Guidelines, and was approved by Institute for Research in Biomedicine (IRB) at the Poznan University of Medical Sciences (ethis approval No. 429/17 from April 6, 2017). The study was conducted in the ergospirometry suite of the Greater Poland Center of Pulmonology and Thoracic Surgery of Eugenia and Janusz Zeyland over a period of 8 months (October 2018–June 2019). The exercise stress test laboratory was adequately equipped to provide advanced life support in the event of a cardiac arrest.
Body mass composition analysis
Component measurement of body mass in women was performed in the first phase of the cycle with bioelectrical impedance analysis (BIA).21 All measurements were taken at 7 AM, patients were fasting for at least 9 h. The patients were instructed not to consume alcohol for at least 48 h prior to the test, not perform vigorous physical exercise for at least 12 h before, to avoid saunas for at least 12 h before, and to void the bladder 30 min before the test. Patients with implanted cardiac devices (ICDs, i.e., defibrillators), pacemakers or metal implants, epilepsy, hemiparesis, or with wounds or skin lesions on hands or feet were excluded from the study.
The body composition analyzer used for the measurements was a Tanita BC 980 (Tanita Corp., Tokyo, Japan). The body composition analyzer MC-980 MA (Tanita Corp.) is intended for professional use in specialized health centers, hospitals and medical institutions, as well as universities for research. The analyzer has the MDD CLASS IIa and NAVI CLASS III medical certificate required for the use of medical equipment in clinical setting. Eight integrated electrodes allow for performing analysis with segment reading. The current flows at 6 frequencies: 1 kHz, 5 kHz, 50 kHz, 250 kHz, 500 kHz, and 1000 kHz, which maximizes the accuracy of the measurements.
Among the body mass components, the following values were estimated: fat mass (FAT; kg and %) and muscle mass (MM; kg and %). Patient norms have been established based on previously published reports.22 In order to perform the body composition test, it was necessary to measure 2 basic somatic features (body height in cm and body weight in kg), based on which the BMI was calculated.23 The waist-to-hip ratio (WHR; ratio of waist circumference to hip circumference) was measured using anthropometric measuring tape. The measurement was taken 3 times and the average value was drawn.
Assessment of aerobic fitness
The exercise tests were conducted on the 1st day of the experiment between 8:00 AM and noon, in an air-conditioned laboratory, 2 h after consuming a light breakfast. The duration of the physical exercise was 20 min. The test was performed on a cycloergometer (Kettler® DX1 Pro; Kettler GmbH, Ense-Parsit, Germany), as follows: after a 3-minute warm-up with a load of 25 W, the load was increased by 10 W (60 RPM) every 90 s. The test lasted until refusal or inability to maintain the given cadence. On this day, the threshold load was determined for each patient with cardiopulmonary exercise testing.
Physiological measurements
Expired gases, minute ventilations (Ve) and heart rate (HR) during graded exercise test (GXT) were monitored continuously with an automated START 2000 M system (MES Sp. z o.o., Kraków, Poland). Oxygen intake (VO2) and carbon dioxide output (VCO2) were measured breath-by-breath and were averaged at 15-second periods. Before each trial, the system was calibrated according to the manufacturer’s instructions. Ambient conditions, including temperature, humidity and barometric pressure, were recorded by the sensors. With a 2-point volume calibration (0.2 L/s and 2 L/s), flow values were measured automatically at the set measuring points. The gas analyzer calibration was done using standard gas mixture containing 5% CO2 and 16% O2. To determine the threshold load (VT), the V-slope method was used, applying computerized regression analysis of the slopes of the CO2 output compared to O2 uptake plot, which detects the beginning of excess CO2 output generated from the buffering of [H+]. This method involves the analysis of the behavior of VCO2 as a function of VO2 during GXT with a consequent increase in VCO2. During the direct test, peak oxygen consumption (peakVO2) was determined for each examined person.
Measurement of tissue saturation
The near-infrared spectroscopy (NIRS) method is based on the transparency of human tissue to light in the near-infrared range (700–1000 nm), which is dependent on the oxidation of oxyhemoglobin (HbO2) and deoxyhemoglobin (Hb).24 The depth of NIRS light penetration amounts to approx. 3 mm, which allows a camera to measure the level of skeletal muscle O2 saturation (StO2) in subcutaneous tissues.25 The NIRS was used in our research to assess tissue oxygen saturation in 3 areas of the body. It was performed on days 2 and 3 of the experiment. On day 1, using the ergospirometric method, individual load levels were set for each patient. The exercise protocol included riding a cycloergometer (Kettler® DX1 Pro) with a 3-minute warm-up and a 25 W load, followed by an effort with a threshold load assigned individually for each patient (60 RPM) without oxygen (day 2) and after supplementation with oxygen-enriched air mixture with a flow rate of 6 L/min (day 3).26, 27, 28, 29 A description of the tests performed on the following days of the experiment is presented in Figure 2. Rest measurements were taken on both day 2 and day 3 during a 5-minute rest before starting the exercise test, breathing the atmospheric air on day 2 and the oxygen-enriched air on day 3. For each person, the effort phase always lasted 20 min. The threshold load was on average 50.0 (IQR: 60.0–80.0) W. The oxygen-enriched air mixture with a flow rate of 6 L/min on day 3 was administered to the patient through a nasal catheter during exercise. Simultaneously, CO2 retention was measured. In obese people, ventilation disorders such as restrictions are observed, resulting in decreased ventilation and a tendency to accumulate carbon dioxide. The decrease of StO2 associated with exercise is a mechanism stimulating hyperventilation. If this mechanism is limited by oxygen supplementation in obese patients, hidden respiratory ventilation failure may appear and hypercapnia may be observed.
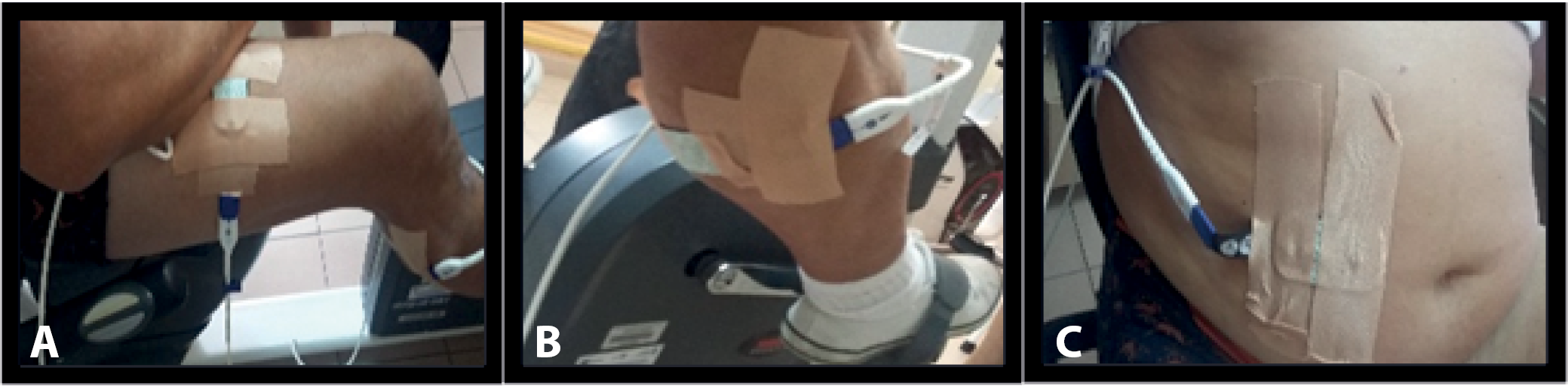
The saturation assessment was performed on each patient 2 times per day on days 2 and 3, at rest and during exercise after stabilization. Saturation measurements were performed in 3 places on the patient’s body by measuring absorbance via NIRS with the INOVOS 5100C device (Somanetics/Covidien, Mansfield, USA). The arrangement of the electrodes is shown in Figure 3. Fat tissue thickness was assessed using ultrasound with a Siemens Acuson device (Siemens AG, Munich, Germany). This measurement was used to determine the place of saturation measurement. Blood lactate level, saturation and HR were measured at rest before and after the actual exercise test. During each trial, HR and blood lactate levels were also measured. Capillary blood lactate measurement was determined using the EDGE Blood Lactate Test Strip (ApexBio; Hsinchu, Taiwan), while HR was measured with a Nonin 8500 pulse oximeter (Nonin Medical, Inc., Plymouth, USA).
Statistical analyses
We calculated the sample size in the present study based on a study by Maltais et al.12 They determined the effects of oxygen supplementation on the peak exercise capacity during exercise in COPD. After calculation power analysis of Mann–Whitney test (adopting a power as 1-beta error probability: 80%, effect size (ES): 0.98, and error assumed as alpha: 0.05 (2-sided)), 11 participants were indicated for allocation equally for each test. We decided to accrue more participants in each test due to a possible dropout. Therefore, the present study was initiated with 18 women. The distribution of normality was measured using the Shapiro–Wilk test. Because the hypothesis of normality of distribution was rejected (at p < 0.05) for many variables (13 of 52), nonparametric tests were used for analysis of all variables. A significance test for 2 mean values was performed using the Wilcoxon signed rank test, according to the assumption that population data do not follow a normal distribution and tested differences are symmetric with respect to a common median. The Spearman’s rank analysis was used to calculate correlation coefficients. The level of statistical significance was set at p ≤ 0.05. The obtained results were analyzed statistically using the Dell STATISTICA data analysis software system v. 13 (Dell Inc., Round Rock, USA).
Nonparametric ES was calculated according to the formula (where n is the total number of observations, and Z correlation coefficient is the same as the r coefficient between –1.00 to 1.00).30
The difference between means divided by the pooled standard deviation (SD) was calculated. Using Cohen’s (1988) criteria, an effect size ≥0.20 and <0.50 was considered small, ≥0.50 and <0.80 medium, and ≥0.80 large.
Results
Table 2 presents the characteristics of the study group. The subjects displayed a high BMI of 42.0 kg/m2 (36.1; 45.2). The level of cardiopulmonary fitness (VO2max) was 17.5 mL/kg/min (15.1; 19.0) in the subjects, amounting to only approx. 50% of the expected value and being significantly reduced. The mean values of the threshold load for the study group are shown in Table 2.
The administration of additional breathing mixtures in obese patients resulted in a significant decrease in HR only at rest (Z = 2.1339, p = 0.0328, r = 0.3556). In response to an exercise with a threshold load, a lower effort increase was observed in the examined indicator without statistical significance (Z = 1.6805, p = 0.0928) (Table 3).
Measurements of saturation were performed in 3 areas of the body (point 1 – midpoint of the quadriceps muscle, in the abdominal plane, on the right side; point 2 – 1/3 proximal part of the gastrocnemius muscle on the right calf; point 3 – 2 cm to the right of the umbilicus).31, 32 They were conducted at rest and during exercise, both without and with supplementation of an oxygen-enriched air mixture. A significant decrease in saturation was shown only for the midpoint of the quadriceps muscle (Z = 2.1572, p = 0.0309, r = 0.3595). No significant difference in oxygenation between the 2 experimental models was found (Table 3).
The experimental model did not demonstrate an effect of the administration of additional O2 in the respiratory mixture on the resting lactate concentration in blood. However, the difference was statistically significant in the experimental model in which patients performed an effort at the level of individual threshold load, with and without administration of oxygen in the respiratory mixture (Z = 2.9394, p = 0.0032, r = 0.4899). In addition, medium effect sizes were shown in the difference in resting and exercising lactate concentration change between the experimental models studied (Z = 2.5041, p = 0.0122, r = 0.4174) (Table 3).
The Spearman’s rank correlation showed a negative relationship between the exercise change in blood lactate concentration and tissue saturation at the midpoint of the quadriceps muscle only in the experiment without supplementation with an oxygen-enriched air mixture (p = 0.0185, r = −0.5631).
Discussion
People with obesity may present an impaired ability to use fat as fuel during moderate-intensity exercise. This may be associated with impaired mobilization of fat reserves, low activity of β-oxidation enzymes and low activity of skeletal muscle lipoprotein lipase.33, 34 Thus, obesity treatment should include interventions that can increase the ability of skeletal muscles to utilize fat. Endurance training in lean individuals causes increased fat oxidation during submaximal exercise at constant load.35, 36 Increased oxidation of resting fat after endurance training has also been shown.37 Impaired ability to mobilize and utilize fat in obese individuals may suggest that physical training has a different effect on fat oxidation in obese and lean individuals. A study by van Aggel-Leijssen et al. evaluated the effect of physical exercise of different intensities on fat oxidation in obese men.38 At rest, total fat oxidation did not change in any group. During exercise, after low intensity training, fat oxidation increased by 40% (p < 0.05). This was due to the increased oxidation of fatty acids outside the plasma (p < 0.05). Based on their research, they concluded that the total fat oxidation during exercise did not change during high-intensity training. It seems that in obese patients, only low-intensity training increases fat oxidation during exercise. A long-term exercise of an intensity corresponding to 60–80% of VO2max leads to the depletion of glycogen stores in the muscle, which is associated with fatigue. Insufficient energy supply results from the limited supply of the substrate (acetyl-CoA) to the Krebs cycle and increases fatigue during prolonged exercise.39, 40 The duration of exercise is also closely related to the pre-exercise glycogen content in the muscles. However, the effort tasks used in our study were not that intense, but tailored to the capabilities of a given patient. An important marker of fatigue in this type of exercises is lactic acid.
The energy of metabolic changes during low-intensity exercise is mainly based on O2 mitochondrial changes. Unlimited access to oxygen contributes to the effectiveness of lipolytic changes. One of the products of metabolic changes having an inhibitory effect on the rate of lipolytic changes is lactate.
Low-intensity physical exercise does not cause a sharp increase in blood lactate concentration.41 In healthy people, during exercise with a load of about 50–70% VO2max, rapid accumulation of lactate in the blood is observed. High lactate concentration leads to the development of metabolic acidosis in the working muscle, thus limiting its exercise capacity.42 This process, which in people who exercise regularly is a desirable phenomenon, in patients with reduced exercise tolerance leads to the development of changes due to fatigue at a low exercise load. As a result of the aforementioned changes in homeostasis, obese people are not able to fully implement training programs. It can be assumed that the supplementation of an oxygen-enriched air mixture proposed by us, administered intranasally during physical exercise, resulted in a decrease in blood lactate levels after an exercise test. A lower accumulation of lactate extends the duration of exercise due to the delayed development of metabolic acidosis in the working muscle. Thus, owing to a decrease in the production of lactate in the working muscle, a person is able to exercise for an extended period of time with an intensity below the individual threshold load. In addition, acidosis may also affect contraction efficiency by disrupting ATP production processes. Hydrogen ions also influence the activity of creatine kinase and other glycolytic enzymes – hence the indirect influence of acidosis on energy metabolism and the contraction process.39 Decreasing intracellular pH may influence the level of oxidative enzyme activity and ryanodine receptor activity.43 The best-known markers of muscle fatigue related to ATP metabolism are serum lactate, ammonia and oxypurines (xanthine and hypoxanthine). Elevated serum lactate is associated with insufficient aerobic ATP production, which must be supplemented by its anaerobic production. In non-fit people, the lactate threshold is at 50–60% of the maximum load, whereas in athletes it is as much as 70–90% of that load.
Serum lactate concentration does not increase with age or gender but with exercise intensity in healthy and non-healthy (e.g., with comorbidities) subjects.44 During prolonged endurance exercise with constant stress, lactate levels usually rise by <5 mmol/L. However, this parameter especially increases in patients with impaired oxidative phosphorylation (mitochondrial disorders) and lung diseases. It also increases significantly in people with COPD during a gradual exercise test on a cycloergometer.45 Therefore, lactate level and an increase in calcium with a concomitant decrease in magnesium are very useful markers of fatigue following moderate-intensity exercise.46
After analyzing the literature, no studies on the validity and effectiveness of supplementation with an oxygen-enriched mixture in obese patients during physical training were found. On the other hand, the high effectiveness of the use of an oxygen-enriched breathing mixture in the training of patients with impaired respiratory efficiency has been demonstrated.
In the study by Nixon at al., a group of cystic fibrosis patients performed progressive exercise tests until exhaustion on a bicycle ergometer while breathing normoxic air (21% O2) during one test and hyperoxic air (30% O2) during another one. It has been shown that O2 supplementation minimizes O2 desaturation and allows cystic fibrosis patients to exercise with reduced ventilation and cardiovascular effort.47 At rest, the body uses about 22% of oxygen, which is supplied to the tissues in arterial blood. During exercise, the O2 consumption of working muscles increases, i.e., venous blood leaving the working muscles is far less oxygenated than when at rest. This difference can even be as high as 90%. The increase in oxygen extraction is highly beneficial upon analyzing changes at the mitochondrial level. The contracting muscle receives much more O2 during a single blood passage, and deoxygenated hemoglobin is again saturated with oxygen in the lungs, which does not cause any side effects. The introduction of additional O2 supplementation in the group of untrained, obese patients resulted in a decrease in saturation within the blood vessels of the muscle due to its increased uptake by mitochondria. This may be indicated by the metabolic response measured with blood lactate concentration in response to providing patients with an oxygen-enriched air mixture.48 The meta-analysis conducted by Fiogbé et al. showed that physical exercise alone, depending on the patient’s condition, improves the oxygenation of various muscle areas.49 Oxygen supplementation may intensify this effect.
Oxygen supplementation has a major beneficial effect on the effectiveness of exercise in patients with COPD. A study by Rooyackers et al. examined whether training with O2 supplementation enhances training effects during indoor air breathing in patients with severe COPD. The study showed that O2 supplementation increases exercise tolerance in the maximum incremental test. Oxygen supplementation has an important ergogenic effect, and muscle damage caused by overload is positively correlated with overall muscle performance.50
The degree of muscle tissue deoxygenation during physical exercise was also assessed with NIRS in a study by Koga et al. They found that this method can accurately assess the temporal and spatial diversity of oxygenation.51 This can be considered particularly consequential since earlier studies conducted with less complicated methods of measurement indicated that, at the onset of exercise, only a few seconds are required for the increase of oxygen uptake in exercising, while O2 extraction peaks after 50 s of exercise. Bangsbo et al. claimed that insufficient oxygen availability is not the reason for limited O2 utilization in the initial phase of intense exercise.52 On a similar note, Segal et al. suggested that several motor units are supplied by individual activated microvascular units.53 In these, increased blood flow can increase O2 supply to muscle fibers, resulting in unchanged or increased O2 saturation, as measured using NIRS. In addition, differences in tissue saturation may also apply to different muscle groups and their regions. This will depend on the blood flow, as well as contractions in blood vessels during exercise.
Over 95% of body fat is accumulated in adipose tissue and stored as triglycerides.54 The metabolism of adipose tissue is influenced by hormones such as insulin and catecholamines, autocrine and paracrine factors, as well as nutritional status and exercise.55 Physical effort causes an increase in lipolysis through a marked increase in catecholamine concentration; however, pronounced changes are most often observed only 30 min after the exercise commences.56 Lipolysis intensification and acceleration is a particularly beneficial process, especially in people with obesity. In the obese patients examined in our research, it was observed that in the fat tissue in the abdomen at rest, there is an increase in saturation depending on O2 supplementation. However, there is no change in tissue saturation during effort. Conversely, the 20-minute effort may be too short for significant changes in the metabolism of this tissue.
In the examined tissues (muscle and adipose tissue), there was a smaller effort increase in saturation in patients supplemented with an oxygen-enriched mixture. Thus, it can be concluded that there is an increase in the use of O2 in the metabolism of working muscle and adipose tissues, which results in lower physical acidification of the body while performing the same effort. Such exercise exerts beneficial changes in metabolism and has an impact on reducing cardiovascular risk.57
The study also observed the effect of supplementation with an oxygen-enriched mixture on tissue saturation of fat located over working muscle (quadriceps muscle). A decrease in the saturation of this fat over working muscle was observed when supplementing an oxygen-enriched air mixture during exercise, while there was no change in fat oxygenation at rest. This confirms that better oxygenated fat tissue releases triglycerides to a greater extent, enhances lipid metabolism and fat burning. However, physical effort in this situation must last longer because of the increase in the intensity of lipolytic changes during the first 30 min of exercise in people with low aerobic capacity.58
The study group consisted of people with obesity or morbid obesity who trained rarely. In such persons, lipolysis in adipose tissue occurs only 20–25 min after the start of exercise, while the average time of exercise in the study was 20 min. Oxygen supplied additionally to adipose tissue was not used up before the end of exercise, which indicates an increase in saturation in this tissue. On the other hand, the supply of additional oxygen to muscle tissue caused an enhanced O2 use for metabolic changes, which increased the amount of energy obtained from oxygen processes and caused a decrease in blood lactate levels. The use of O2 supplementation during exercise may be clinically significant. Oxygen supplementation can be used during physical activity, especially in patients preparing for bariatric surgery, when people who are often extremely obese must reduce their weight within a short period of time. It can be assumed that in these patients, the administration of O2 during exercise can extend the time before significant acidification of the muscles is reached, and thus, enable them to exercise longer and more willingly, at the same time helping them to faster achieve the weight expected before operation.
According to the study by Andrade et al., the positive effect of O2 supplementation cannot be attributed only to the improvement of the oxygen content in the blood and the reduction of HR.18
In addition, in terms of the analysis of cardiological parameters, a reduction in the resting frequency of heart contractions was found as a result of breathing an oxygen-enriched mixture. Arterial chemoreceptors located in the cervical glomeruli are the main recipients responding to changes in O2 and CO2 pressure in a hypoxic situation, increasing myocardial activity and causing tachycardia. Increased saturation of arterial blood leads to the inhibition of chemoreceptor stimulation and a decrease in HR at rest and during exercise. In the present study, people supplemented with an oxygen-enriched air mixture could therefore exercise for a prolonged period of time without a rapid increase in HR. Similarly, in a study by Nonoyama et al.,59 oxygen supplementation in patients with COPD increased mean exercise time from 6 min to 14 min and caused significant improvement in Borg scale score after the shuttle walk test.
Based on the results of this study, it can be concluded that O2 supplementation increases the use of oxygen in the mitochondria and intensifies the oxygen metabolic processes in the working muscle.
The use of this type of therapy in combination with optimized exercise can contribute to improve physical activity, reduce changes due to fatigue, and, as a consequence, lead to weight loss. The NIRS measurement method used in the study is a highly effective noninvasive method for monitoring tissue saturation in real time. In contrast to pulse oximetry, which indicates the saturation of hemoglobin in pulsating arterial blood, the onset of hypoxia may be delayed by up to 90 s. Unlike NIRS, arterial blood gas measurement is an invasive assessment that is delayed by the time needed to perform the laboratory analysis of the sample taken.
Limitation of the study
A limitation of this pilot study was the small number of patients qualified for the project and the absence of obese patients with restrictive respiratory disorders.
Conclusions
Lower increase in exercise saturation in muscle and fat tissues in patients supplemented with oxygen-enriched mixture may occur due to an increased use of oxygen during mitochondrial metabolic changes and lower acidification of the muscle under stress. Increased saturation of arterial blood due to supplementation with oxygen-enriched mixture leads to the inhibition of chemoreceptor stimulation and a decrease in HR at rest. Physical effort with peri-threshold intensity in conditions of increased O2 supply led to less fatigue, and thus it may result in longer exercise routines in obese persons who get tired more easily. The supplementation of this respiratory mixture in addition to optimized exercise can result in improved physical fitness.