Abstract
Background. The increasing availability of body donations and the expansion of reference centers provide forensic clinical anatomy with the opportunity to experimentally reproduce, in ex vivo settings, the effects of specific interactions on the human body, such as needle–tissue interaction in medical procedures.
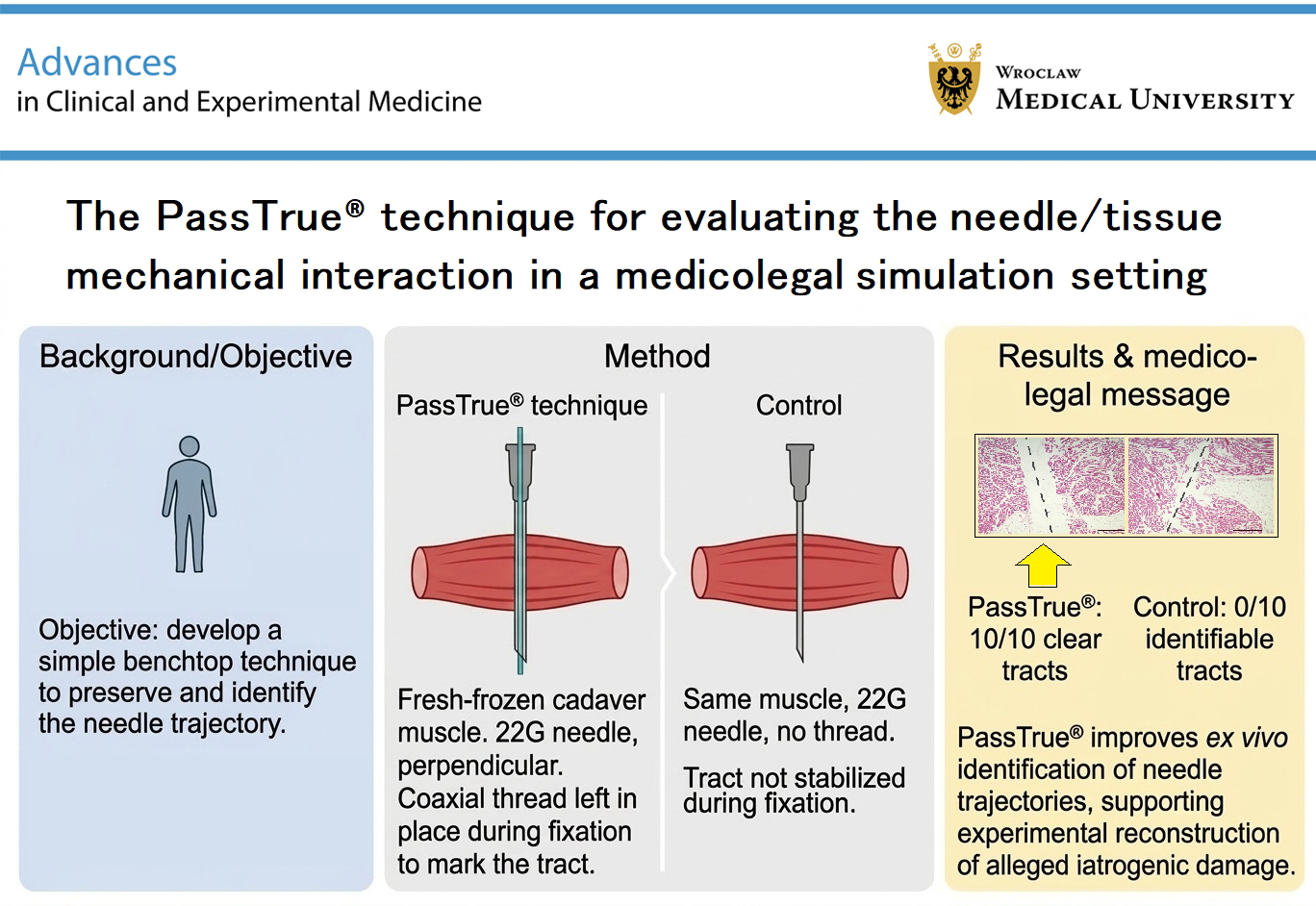
Objectives. The primary objective of this study was to develop a reproducible, standardized benchtop methodology that enables the identification of the trajectory traveled by the needle when piercing soft tissue, causing detectable iatrogenic tissue damage.
Materials and methods. Skeletal muscle tissue samples were harvested from a fresh-frozen 78-year-old male body housed at our Reference Center, Center for Body Donation. Needle transfixions were then tested using a novel technique designed for this purpose (PassTrue® methodology): full-thickness tissue transfixion by a needle, insertion into the needle of a coaxial thread, grasping the thread with forceps, and needle withdrawal, so that the in-place thread indicates the route of transfixion. This procedure was tested against the control procedure without a thread.
Results. Microscopic analysis revealed a pattern consistent with the needle’s course during transfixion in all cases with the PassTrue® technique (10/10), but not in controls (0/10).
Conclusions. Our novel methodology improves the efficiency of assessing needle–tissue interactions, enabling identification of the needle’s trajectory within biological tissues.
Key words: medical malpractice, legal medicine, body donation, cadaver lab, clinical anatomy
Background
A substantial proportion of medical procedures entail the use of needles to inject or aspirate fluids, and this practice inherently constitutes a potential source of tissue trauma.1 The ensuing tissue injury may plausibly have clinically relevant consequences and, as an adverse effect of medical interventions, can prompt patients to pursue compensation claims or initiate criminal proceedings.2 In response, there has been a marked and growing concern among clinicians regarding the medicolegal ramifications of their professional activities on patients’ health, extending even to seemingly routine needle-based procedures involving tissue penetration. This phenomenon has frequently been described as a “medicolegal epidemic,” a characterization mirrored by the expanding body of literature on these issues internationally.3, 4 Nonetheless, with specific regard to tissue damage caused by needle use, the current evidence base remains limited. Although several research papers have examined needle–tissue interaction during medical procedures, particularly focusing on parameters such as velocity of needle insertion and pressure of fluid injection, no consistent, experimentally robust, and reproducible findings have yet been established.
More recently, some authors have focused on iatrogenic tissue injury arising from needle insertion, employing heterogeneous, non-standardized methodologies with limited demonstrable validity and substantial variability in microscopic outcomes, with the fundamental problem of the loss of the tract during histological fixation. These developments have significant implications for clinical risk management and for the formulation of policies guiding the selection of specific invasive techniques among multiple available options. Given the extraordinarily high volume of procedures involving needle–tissue interaction across all medical specialties, this topic is of considerable relevance and warrants systematic, standardized investigation.
Objectives
The primary objective of this pilot study was to develop a reproducible, standardized benchtop methodology that would allow the trajectory of the needle to be identified in tissue.
Materials and methods
Research ethics
All procedures were conducted on human bodies from the “Donation to Science” body donation program of the University of Padova (Italy) and the Veneto Region/National Reference Center for the preservation and use of donated bodies. These procedures adhered to national laws and the ethical standards set by regional and national research committees, as well as the Declaration of Helsinki of 1964, including its later amendments or comparable ethical standards. Participants provided their written informed consent to join the Body Donation Program, which is consistent with ISO 9001:2015 (registration No. IT-62435-16764). The privacy rights of all human subjects were and will always be respected. Informed consent was obtained from all participants involved in the study, in accordance with the procedure for collecting and donating anatomical material.
In accordance with the regulations of the Body Donation Program of the Institute of Human Anatomy (University of Padova), a skeletal muscle tissue specimen (2 × 2 × 1 cm) was harvested from a fresh-frozen male body aged 78 years, housed at our Reference Center.5, 6, 7 As a certified standard, we use a routine protocol according to which we receive the bodies 48 h after death, and on the same day, they are frozen at −20°C with a remote temperature control system until they are used with programmed thawing, approx. 1 month after death.
The donor had no documented diseases that could interfere with the study’s objectives. In particular, the donor did not suffer from age-related pathological or physiological cachexia and had a normal build. He was not bedridden and had documented normal mobility consistent with his age. The tissue was fully thawed to room temperature prior to transfixion. On the bench, transfixion of the sample was performed 10 times using the PassTrue® technique.
It consisted of a multistep procedure. Initially, a full-thickness, side-to-side tissue transfixion was performed using a 22-gauge needle (BD Microlance; Becton Dickinson (BD), Oslo, Norway) oriented perpendicular to the tissue plane (Figure 1A). A coaxial polypropylene monofilament suture (Prolene 3/0; Ethicon, Raritan, USA) was subsequently advanced through the lumen of the needle and grasped on the opposite side with forceps, which maintained traction on the thread while the needle was carefully withdrawn from the tissue along a trajectory opposite to that of insertion (Figure 1A). This resulted in the placement of a reference thread indicating the tissue route created by the needle during transfixion (Figure 1B). A control procedure was performed with 10 free transfixions using the same materials and steps on a comparable control specimen, except for the use of the reference thread passed inside the needle.
Following completion of the procedures, the skeletal muscle tissue specimens were immersed in a 4% m/v formalin solution and stored at room temperature. After 5 days of formalin fixation, they were sectioned at 5-mm intervals, processed according to standard microscopic protocols, and embedded in paraffin. From each sample, sequential 5 μm-thick sections were systematically cut and stained with hematoxylin and eosin (H&E) (W01030708; BioOptica, Milan, Italy) for microscopic characterization.
To perform both the 5-mm and 5 μm-thick sections, the cut plane was consistently aligned with the needle path, which followed a trajectory perpendicular to the skeletal muscle tissue specimen.
The sections were dipped in xylene for deparaffinization, immersed in a graded series of alcohol solutions for hydration, and stained with the kit dyes. Subsequently, dehydration through immersion in an increasing series of alcohol solutions, clarification in xylene, and mounting with Eukitt medium (09-00250; BioOptica) were performed on all tissue sections. Hematoxylin and eosin staining was performed to observe the tissue morphology and architecture of each section of the skeletal muscle tissue samples.
Discussion
Over the past several years, forensic medicine has undergone significant development, particularly in relation to the evaluation of alleged cases of medical malpractice. Within this framework, the subspecialty of forensic clinical anatomy has emerged to investigate the forensic implications of anatomical alterations and variants in the context of assessing iatrogenic injuries and quantifying personal damage.8, 9 The increasing availability of body donations and the expansion of reference centers provide forensic clinical anatomy with the opportunity to experimentally reproduce, in ex vivo settings, the effects of specific interactions on the human body, including those involving medical procedures (e.g., needle–tissue interaction, novel techniques, and innovative tools).10, 11, 12 The interest of legal medicine in this procedure within a hypothetical medical malpractice setting could, e.g., be experimental in nature, relating to the study of the injury a needle could cause when inserted into tissue. In this case, the procedure described in the present paper allows for further investigation of this aspect by precisely tracking the needle’s path within the tissue and identifying any consequent vascular or nerve injuries. This allows for better interpretation and analysis of specific clinical conditions, e.g., in the field of anesthesiology, where, in the case of peripheral nerve blocks, nerve injury caused by needle insertion during an anesthetic procedure is possible. Such investigations were previously feasible only in an anecdotal manner in living subjects and were substantially limited by ethical constraints.
The literature exhibits substantial heterogeneity regarding the types of procedures performed, the materials employed, and the biological specimens utilized to investigate needle–tissue interactions. Our novel methodology improves the efficiency of assessing needle–tissue interactions, both in detecting them and in analyzing the needle’s trajectory within biological tissues and the consequent structural alterations. Indeed, in the control experiments, no needle tracts (0/10) were identified, even when carefully examining tissue slices corresponding to surface changes that suggested the exact point of needle penetration into the sample (Figure 1C,D). By contrast, using the PassTrue® procedure, the thread acts as a “wick” that prevents the natural elastic recoil of tissue during fixation, which would otherwise lead to fading of the needle path during transfixion. For this reason, the thread simply keeps the passageway created by the needle open and does not cause any structural alteration or distortion of the tissue. Consequently, a microscopic pattern consistent with needle transfixion was identified in all cases (10/10) in which the PassTrue® procedure was used (Figure 1E,F). Furthermore, the entire trajectory of the needle was visualized in substantial portions of its course, together with the associated partial tissue removal. The procedure has therefore proven to be valid for testing the muscles of an elderly individual. We expect that in younger subjects, the effect is even greater than in the muscles of an elderly individual. The trophism and the greater presence of elastic fibers in younger subjects would result in a greater probability of obscuring the needle tracks after the needle has passed if a reference thread is not used, as proposed in this procedure.
Overall, based on a literature review, we identified a number of biomechanical studies based on computational simulations, relying on mathematical models and mechanical representations, which we intentionally excluded, restricting our analysis exclusively to studies employing biological material. In several instances, the investigations employed biological material derived from various animal species, including bovine tissue,13, 14, 15 porcine tissue,16 rabbit tissue,17 rat tissue,18, 19 and canine tissue.20 Human biological material (placental tissue) was utilized in only a single study.21 Moreover, the tissues collected for the experimental analyses were heterogeneous across species, as in each case a different anatomical structure was utilized, including segments of the carotid artery,13 brain,16, 18, 19 sciatic nerve,17 skeletal muscle,14 liver,15 kidney,20 and placenta.21 The comparability and reproducibility of experimental results are further compromised by the heterogeneity of microscopic assessment protocols applied to tissues obtained following mechanical interaction with the needle. For accurate identification of the needle tract and a more reliable evaluation of the associated tissue injury, histological sectioning should be performed in a longitudinal plane parallel to the needle’s major axis,16, 20 rather than in a transverse orientation.14, 15, 17, 18, 19, 21 Notably, in at least 1 study, morphological characterization using light microscopy was not conducted.13
In any case, among all these experimental investigations on needle–tissue interactions, no established method has yielded a defined and reproducible standard that approximates, even remotely, the features of the current PassTrue® technique, and in particular the positioning of a reference thread within the needle trajectory that allows the route to be kept open during the fixation and embedding steps of the tissue sample.
Our findings indicate that the PassTrue® technique constitutes an efficient, standardized, rapid, and cost-effective method that facilitates inter-study comparability, provided that investigations are conducted on the same tissue type within the same species. Within this framework, research involving human biological material assumes particular importance for addressing both clinical and forensic questions, and in this setting, forensic clinical anatomy occupies a pivotal position.
Limitations of the study
Our pilot study presents limitations that warrant consideration. First, while the control demonstrates the usefulness of the thread for visualization, it does not confirm whether the thread maintains the fidelity of the original injury geometry, although it has a smaller diameter than the needle used for transfixion, and we do not expect any significant inconsistencies. Second, this technique has currently only been tested in muscle tissue harvested from a single donor, and the needle interaction observed here using the PassTrue® technique may differ in tissues with different collagen densities, for which specific experimental tests will be necessary. Third, the use of skeletal muscle samples obtained from a deceased individual allows us to demonstrate only the trajectory traveled by the needle when piercing soft tissue, causing iatrogenic mechanical damage to the tissue, without capturing potential in vivo biological or immunological responses to injury. This may be of clinical interest but is beyond the scope of this paper, which focuses on the development of an innovative experimental model. Fourth, the exclusive use of sharp needles does not fully encompass the spectrum of clinical scenarios, given that non-cutting needles are sometimes preferred for specific procedures. Similarly, different needle diameters and tissue thicknesses could yield slightly different results, although the effectiveness of the innovative approach is expected to be independent of these circumstantial factors, which are probably irrelevant but need to be verified.
Conclusions
The present pilot study showed that the novel PassTrue® technique improves the efficiency of assessing needle–tissue interactions, allowing the needle trajectory to be identified within biological tissues. The methodology is standardized, rapid, and cost-effective. It provides a potential common framework for future medicolegal simulations on cadavers and facilitates comparability across studies addressing similar research questions. However, further studies will be needed to evaluate the suitability of this new procedure across different combinations of needle type, needle diameter, tissue type, and tissue thickness.
Registered trademark
PassTrue® is a trademark registered by the University of Padova on behalf of Prof. Rafael Boscolo-Berto, using his personal funds.
Consent for publication of personal information
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.

















.jpg)