Abstract
Introduction. Social prescribing (SP) is an innovative model that connects individuals to non-clinical community resources. However, its uptake and evaluation have been hindered by inconsistent role definitions for key stakeholders. Although recent studies have refined SP definitions, outside the UK, the responsibilities, educational backgrounds and training requirements of social prescribing link workers (SPLWs) remain poorly defined. Additionally, it is essential to identify which patient populations will benefit most from SP, establish specific methodologies, and standardize assessment tools and referral pathways.
Materials and methods. We will employ a 3-round Delphi protocol with an international expert panel to establish consensus on SP definitions. Approximately 60 participants from diverse disciplines and regions will be recruited to complete multiple survey rounds, providing insights into the roles of SPLWs, beneficiary populations, methodologies, and assessment tools. Consensus will be defined as at least 80% agreement on a 5-point Likert scale. Data collection and analysis will follow rigorous protocols to ensure validity, reliability and transparency, in accordance with the Guidance on Conducting and REporting DElphi Studies (CREDES) guidelines.
Objectives. The aim of this research is to unify the fragmented understanding of SP and the role of SPLWs, thereby establishing a foundation for integrating SPLWs into healthcare systems where appropriate. The Delphi technique offers key strengths – namely, participant anonymity and structured iterative feedback – to enable robust consensus building. While we acknowledge limitations such as potential participant attrition and the resource-intensive nature of the methodology, these will be mitigated through targeted engagement strategies and strict adherence to established best practices.
Conclusions. This study addresses critical gaps in SP engagement, conceptual understanding and implementation. The anticipated outcomes will reinforce SP’s role in community-based, integrated care to reduce health inequalities and foster social cohesion across Europe and beyond. Ultimately, this work aims to enhance the uptake and adoption of SP in primary care.
Key words: primary health care, integrated care, social prescribing, link workers, community health services
Introduction
Social prescribing (SP) represents an integrative approach within healthcare systems aimed at addressing social determinants of health through non-clinical interventions. Primarily delivered through referrals in primary care, SP addresses social determinants of health that are often beyond the scope of traditional biomedical interventions. This is by integrating health and social care with community activities. Social prescribing facilitates patient engagement in health-related decision-making and self-management.1, 2, 3, 4, 5
Social prescribing involves directing individuals to activities that align with their social, emotional and physical needs as well as providing social counseling to address social problems. These include, e.g., walking, gardening, cooking, crafting, fieldwork, beekeeping, group learning, and/or sports.6 While these activities have historically been integral to European community life, their health benefits are not always formally recognized. However, as SP gains traction across Europe, implementation varies significantly, shaped by country-specific healthcare systems and infrastructure.
England has pioneered SP, embedding it into its UK National Health Service (NHS) Long Term Plan as a central component of personalized care.7 This model emphasizes the role of link workers,8 who collaborate with patients to co-design individualized plans connecting them to local initiatives.9, 10, 11, 12, 13, 14, 15, 16, 17, 18 Similarly, Wales has integrated SP into its Well-being of Future Generations Act, promoting a holistic approach to health.19
Despite its potential, SP faces challenges across Europe. Variability in program design and funding, limited integration with healthcare systems and uneven European awareness hinder its widespread adaptation. Countries like Italy and Croatia, where SP remains underdeveloped, show the need for systemic support and standardized frameworks.20 Moreover, while SP demonstrates potential in improving mental health, reducing healthcare utilization and advancing community engagement, robust evidence on its long-term efficacy remains scarce.21 This lack of evidence complicates efforts to quantify SP’s impact and optimize its implementation.
Many countries struggle to establish systematic connections between healthcare and social systems. In Romania, although governmental and non-governmental organizations provide support to vulnerable populations, a formal referral system, such as SP, has not yet been established.22
In Spain, Catalonia’s SP framework, rooted in primary care and the Health Assets model, has demonstrated the potential to adapt SP to diverse healthcare contexts.23 In Germany, Herrmann et al.24 have identified significant barriers to implementing SP within the German healthcare system.
The need to tackle these challenges has become more urgent due to the growing prevalence of chronic diseases, mental health issues and social isolation across Europe. The COVID-19 pandemic further highlighted the importance of community-centered approaches, as it worsened loneliness and disconnection. Social prescribing offers a structured approach to linking medical care with social and community-based services that prioritize comprehensive, patient-centered solutions addressing the full spectrum of health determinants.25
The World Organization for Colleges, Academies and Associations for General Practitioners (WONCA) Europe Social Prescribing and Community Orientation Special Interest Group (SPCO SIG) recognizes the potential of SP in advancing integrated care.26 Leveraging their expertise in rural health and community engagement, the group seeks to clarify the SP concept and the role of social prescribing link workers (SPLWs), map existing SP programs and identify best practices for implementation across diverse healthcare systems.
This study seeks to advance the understanding of SP by identifying key elements that support its development and integration into contemporary healthcare systems. By addressing health inequalities, improving patient outcomes and strengthening community cohesion, SP has the potential to significantly contribute to the implementation of integrated and equitable models of primary care across Europe.26, 27 The project is led by members of the SPCO SIG under the auspices of WONCA Europe, with support from colleagues in the European General Practice Research Network (EGPRN). The initiative has been awarded a research grant from the EGPRN.
Objectives
This study aims to reach expert consensus on 3 key areas: 1) the target populations most likely to benefit from SP; 2) the methodologies and assessment tools used to evaluate SP outcomes; and 3) the role, qualifications and training requirements of SPLWs.
Materials and methods
Study design
A 3-round Delphi study will be conducted to identify the key elements contributing to the success of SP programs within European primary care settings, as recognized by European experts.
The Delphi technique will be used as a structured method of achieving consensus among experts. This method involves multiple rounds of surveys, where experts respond to questions anonymously. After each round, participants receive feedback summarizing the group responses, allowing them to refine their input in subsequent rounds. This iterative process facilitates the convergence of opinions, enabling the development of agreed-upon definitions and frameworks.
Widely regarded as a robust approach in health science research, the Delphi method is particularly suited to addressing complex topics, such as SP, where diverse perspectives must be synthesized to establish consensus. This study design ensures systematic and comprehensive exploration of the critical factors underlying successful SP implementation in Europe.
This study will be conducted between April 2025 and August 2025. Given the importance of the online survey platform’s quality to this study’s success, we carefully reviewed and tested several different options before selection. We will use Welphi (www.welphi.com), which is an online survey platform that is designed explicitly for Delphi studies. The Welphi team will make coding changes when necessary to ensure that the online survey platform meets the needs of this study.
Before the start of each round, we will complete the survey development and perform pilot testing. Participants will receive a notification email 10 days before the launch of each round. At the start of each round, participants will receive an email with a link to the survey and a link to a calendar with the details of the round.
Participants will have 2 weeks to complete each round. Each survey will begin with a welcome page, which provides an overview of the survey, describes the data analysis procedures, explains what kind of feedback will be provided at the start of the next round, highlights the aim of the study, and outlines essential information about conceptual and operational definitions to ensure that there will be a common understanding of these terms. Participants will be able to return to the survey as often as they like until the round’s closure – their progress will be saved if they leave the study and return later. Even after completing the survey, they can change their submission until the round closes. Each survey will take approx. 15 min to complete. Non-responders will be removed from the study.
Participants
Delphi studies have no universally agreed-upon definition of what constitutes an expert, making participant selection a critical aspect of the research.28, 29, 30, 31 For this study, experts will be identified based on the following criteria: Individuals involved in the SP and Community Orientation Network, the Global Social Prescribing Alliance or the National Academies for Social Prescribing; authors of academic or grey literature related to SP, even if not explicitly labeled as such; researchers or healthcare providers engaged in SP activities; link workers facilitating SP, regardless of the terminology used to describe their role; patients with experience in SP; or healthcare administrators or managers overseeing the implementation of SP initiatives. The study will be conducted in English. Regarding the panel size, the literature on Delphi studies in health sciences suggests an average of approx. 40 participants, with typical ranges spanning 20–60 experts.28, 29, 30, 31 This study will recruit approx. 60 participants, ensuring adequate representation from across European countries to achieve a meaningful consensus.
Recruitment
The recruitment process for the study will commence in November 2024 with the opening of the registration survey for members of the SPCO SIG under WONCA Europe. The steering group will develop a comprehensive list of experts from each country based on their expertise in SP, and invitations will be sent to these individuals via email. Publicly available sources will be used to gather contact information.
An international, multidisciplinary panel of experts will be prioritized, ensuring diversity in country, job title, SP expertise, and years of experience. A monitoring system will track recruitment to identify and address underrepresented groups. To this end, targeted invitations will be extended through relevant communication channels, including the Social Prescribing Network newsletter, the Social Interventions Research and Evaluation Network listserv, and the Canadian Social Prescribing Community of Practice listserv. Invitations will also be advertised on social media platforms like X (Twitter) and LinkedIn. Following the principles of snowball sampling, experts will be encouraged to share the recruitment call with their contacts.
Experts invited to participate will receive an email containing a link to the registration survey. The survey’s first page will feature an information letter outlining the study’s objectives and emphasizing that participation is voluntary. By proceeding beyond this page, experts will provide their informed consent. Consent will be ongoing and voluntary.
The registration survey will collect sociodemographic data, including the participant’s name, email address, country, job title, organization, expertise in SP, and years of experience in the field. Once sufficient experts have registered, participants will be notified about the start of the study’s first round. The recruitment process will ensure a robust and representative panel of experts to achieve meaningful consensus on the topics of this study in European primary care settings.
Consensus
Without standardized guidelines for defining consensus in Delphi studies, this study will adopt a widely accepted threshold of ≥80% agreement as the criterion for consensus. Participants will indicate their level of agreement on a 5-point Likert scale: 1 = “Strongly disagree”, 2 = “Disagree”, 3 = “Neutral”, 4 = “Agree”, and 5 = “Strongly agree”. An item will achieve consensus if at least 80% of participants select “Agree” (4) or “Strongly agree” (5). This rigorous threshold aligns with best practices in health science research, where consensus is typically defined at levels of ≥70%, ≥75% or ≥80%, with the latter representing the most stringent level of agreement.28, 29, 30, 31, 32, 33
Rounds
The study will include 3 rounds, reflecting the structure commonly employed in Delphi studies. This format is based on prior research demonstrating that most Delphi studies to establish agreed definitions are conducted in 3–5 rounds.34, 35, 36, 37 Each round will refine and build upon the findings of the previous one, progressively working towards consensus among participants.
Data collection
The Delphi surveys will span 7 months. To ensure confidentiality and impartiality, participants will be assigned randomly generated participant codes by the online survey platform. These codes will not be shared with participants, nor will the research team have a key to link participants to their codes. This anonymization safeguards the integrity of responses and mitigates potential bias.
The study’s online survey platform has been carefully selected to facilitate seamless data collection and management while ensuring participant anonymity throughout the study. The anonymized approach will foster an environment conducive to candid and unbiased input, supporting the study’s objective of achieving robust expert consensus on SP in European primary care settings.
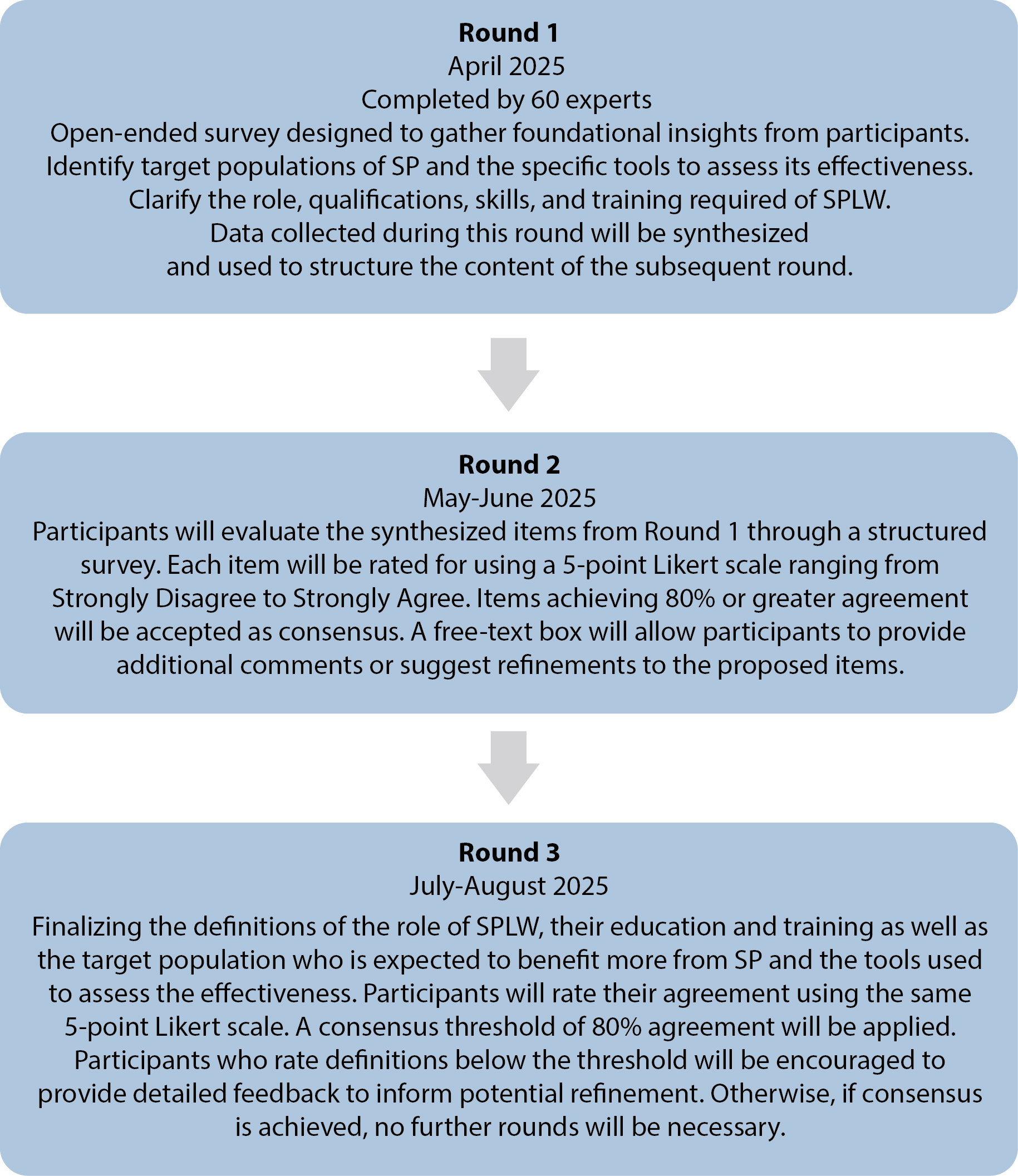
Round 1 will be conducted in April 2025, involving an open-ended survey designed to gather foundational insights from participants. They will be prompted to clarify 2 main aspects: identify the target populations of SP (i.e., those who are expected to benefit more from SP), the specific methodologies used and tools to assess their effectiveness, and the role, qualifications, skills, and training required of SPLW. Participants will have 2 weeks to complete the survey, with an additional 3-day extension provided for non-responders. Data collected during this round will be synthesized and used to structure the content of the subsequent round.
The research team leaders will conduct a qualitative content analysis to analyze participants’ responses. Two senior researchers, F.P. and D.K., will independently review and categorize the qualitative data to identify key themes. The analysis will be performed manually, following a systematic approach to ensure rigor and reliability.
The findings from this round will be used to develop a structured survey for the next round of the Delphi process. At the start of the next round, participants will receive an anonymized summary of the responses from this round, ensuring transparency and allowing for informed consensus-building in the subsequent phase.
Round 2 will take place between May and June 2025. Participants will evaluate the synthesized items from Round 1 through a structured survey. Each item will be rated using a 5-point Likert scale ranging from “Strongly disagree” to “Strongly agree”. Items achieving 80% or greater agreement will be accepted as consensus. A free-text box will allow participants to provide additional comments or suggest refinements to the proposed items.
Round 3 is scheduled for July to August 2025 and will focus on finalizing the definitions of the role of SPLW, their education and training as well as the target population who is expected to benefit more from SP and the tools used to assess the effectiveness. Participants will rate their agreement using the same 5-point Likert scale. A consensus threshold of 80% agreement will be applied. Participants who rate definitions below the threshold will be encouraged to provide detailed feedback to inform potential refinement. Otherwise, if consensus is achieved, no further rounds will be necessary.
Data analysis
The data collected from the registration survey will be analyzed with IBM SPSS Statistics for Windows v. 29 (IBM Corp., Armonk, USA). Qualitative data will be presented for each survey item and analyzed through qualitative content analysis.38
Quality and transparency
This study will be conducted and reported by the Conducting and REporting DElphi Studies (CREDES) guidelines.39
Ethics
At all time, the ethical principles of the Declaration of Helsinki of 1964, revised by the World Medical Association (WMA) in 2013 in Fortaleza (Brazil), will be followed. Compliance with the European Data Protection Regulation 2016/679 of the European Parliament and of the Council, dated April 27, 2016, regarding the protection of individuals concerning the processing of personal data and the free movement of such data, will be always ensured.
All data included in the study will be anonymized and treated confidentially. In the database, participants will be identified only by a code, without including any information that could allow for their identification.
The data obtained in the study will not be transferred to 3rd parties outside the members of the research team of this study at any time. All implemented security measures will be ensured to be sufficient to prevent any breaches of data confidentiality and privacy. Under no circumstances will the research team members have access to participant identification, either directly or indirectly.
Once the study is complete, the research team reserves the right to use the anonymized database solely for scientific purposes (journal articles, scientific papers, book chapters, etc.). The research team will not be permitted to access, use, transfer, or publish the project’s database under any circumstances.
This study has been reviewed and approved for ethical compliance to the Research Ethics Committee of the IDIAP Jordi Gol Primary Care Research Institute in Catalonia, Spain: V2, 08/03/2025 – 01/04/2025.
Data protection
The Principal Investigator, Ferdinando Petrazzuoli, will be responsible for the data storage and handling (ferdinando.petrazzuoli@gmail.com). The anonymized raw data that will support the findings of this study will be available from the corresponding author, upon reasonable request.
Discussion
By engaging diverse stakeholders and utilizing a structured consensus methodology, this study addresses the variability and lack of clarity surrounding the SPLW role, education, skills, and training across Europe, providing a unified framework that can inform research, policy and practice. Clarification and agreement on the target populations of SP (i.e., those who are expected to benefit more from SP), along with the methods and tools used for outcome evaluation, are also among the intended outcomes.
The findings of this Delphi study are expected to extend beyond conceptual clarification. By establishing consensus on the role, education and training of SPLWs, as well as the target populations and assessment tools, the results will inform the development of practical guidelines for health policy, educational curricula and clinical protocols. Specifically, they may inform national and regional policies on workforce development, serve as the basis for designing standardized educational curricula for SPLWs,4, 7 and support clinicians in making more targeted referrals.8 Moreover, the recommendations developed through this process will be adaptable across diverse European healthcare systems24, 25, 26 – whether centralized, decentralized or in developmental stages – facilitating context-sensitive adoption. Finally, the consensus definitions and frameworks may facilitate the design of future comparative and interventional studies evaluating the effectiveness of SP,2, 3 thereby strengthening its evidence base and promoting integration into holistic models of care.
The Delphi method has distinct strengths make it particularly suited for this endeavor.40 Its core features – participant anonymity, iterative rounds of questioning and structured feedback between rounds – help minimize bias and encourage independent and thoughtful contributions from experts. The flexibility of this approach allows for the integration of diverse perspectives on complex topics, such as SP, where consensus must be built across varied healthcare systems and cultural context.29, 34 Furthermore, using an online platform ensures geographic inclusivity and logistical feasibility, allowing experts from across Europe and beyond to participate without the constraints of physical meetings.
Limitations
Despite its strengths, the Delphi technique is not without limitations. It is known to be resource-intensive and time-consuming, requiring careful management to ensure the process remains efficient and that panel attrition is minimized. This study employs regular reminder emails, clearly defined timelines and engaging communication strategies to sustain participant engagement over all 3 rounds. Another challenge is the lack of standardized guidelines in literature regarding the Delphi process, particularly in determining what constitutes expert status and consensus thresholds. This protocol has addressed these concerns through adherence to the CREDES guidelines, which provide a structured approach to mitigate variability and ensure methodological rigor.39 We also acknowledge other potential limitations, which are common to all Delphi studies, such as the risk of participants dropping out in later rounds or the overrepresentation of certain occupational groups or countries.
Conclusions
This Delphi study constitutes an important step toward establishing a shared conceptual and operational framework for SP in European primary care. By achieving expert consensus on the role, qualifications and training of SPLWs, as well as identifying appropriate target populations and tools for outcome assessment, the study will generate practical recommendations applicable across diverse healthcare systems. The findings are expected to support the systematic integration of SP into patient-centered and community-oriented models of care, enhance the coherence of future research, and inform educational and policy development. In doing so, this work addresses key gaps in the standardization and implementation of SP across Europe.
Use of AI and AI-assisted technologies
Not applicable.