Abstract
Background. Leukemia may form at any age, from newborns to the elderly, and accounts for considerable mortality worldwide.
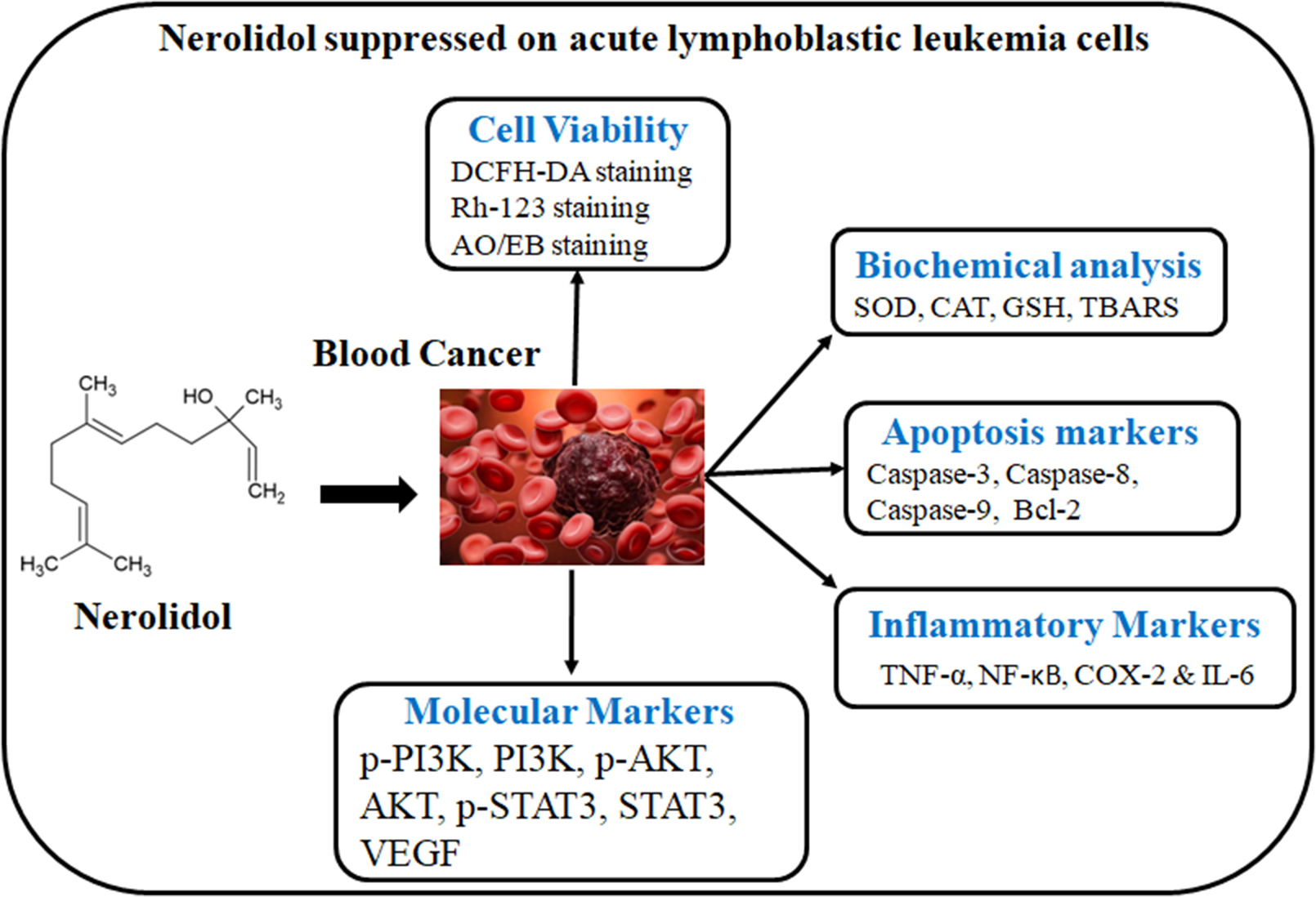
Objectives. Nerolidol (NRD) is isolated from the aromatic florae oils and was found to have anticancer activities. However, the role of NRD in antiproliferative and apoptosis actions in acute lymphoblastic leukemia (ALL) is unclear.
Materials and methods. Human ALL cell lines, MOLT-4, were used to examine the potential anticancer mechanisms of NRD on cellular proliferation, reactive oxygen species (ROS)-mediated apoptosis, oxidative stress markers, caspases, PI3K/AKT, nuclear factor kappa B (NF-κB), and STAT-3/VEGF/Bcl-2 signaling pathways.
Results. The MTT (3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide) assay demonstrated that NRD inhibited MOLT-4 cell proliferation in a concentration-dependent manner, with an IC50 value of 30 µM. It was found that NRD (20 and 30 µM/mL) resulted in accumulated intracellular ROS, reduced oxidative stress and loss of mitochondrial membrane potential (MMP) in MOLT-4 cells in a concentration-related way. Nerolidol was able to induce apoptosis, as evidenced by dual acridine orange/ethidium bromide (AO/EB) staining. The levels of antioxidants, caspases-3, -8 and -9 were enhanced by NRD. This research proves that NRD instantaneously triggers ROS-mediated pro-apoptotic signaling and caspases and attenuates PI3K/Akt/NF-κB and STAT3/VEGF/Bcl-2 anti-apoptotic signaling.
Conclusions. Our results suggest that NRD treatment stimulates apoptosis in MOLT-4 cells by causing the accumulation of intracellular ROS through PI3K/AKT/STAT-3 signaling pathways.
Key words: proliferation, apoptosis, blood cancer, nerolidol, PI3K/AKT/NF-κB signaling
Background
Leukemia is the heterogeneous assembly of hematopoietic cells that transform to form a malignant neoplasm,1 which is the principal source of tumor-related death in patients under 20 years of age.2 According to the GLOBOCAN 2018 statistics, 437,033 new leukemia cases and 309,006 leukemia deaths were recorded.3 Contact with environmental radiation and exposure to extreme radiation levels are recognized as leukemia risk factors.4 Leukemia can develop at any age, from newborns to the elderly, and accounts for considerable mortality worldwide. Among these, acute lymphoblastic leukemia (ALL) commonly affects infants.5 In the USA, nearly 6,000 newly diagnosed cases of ALL subtypes are detected annually,1 while the curative rate is almost ≥80%.6 Despite the high remedial rate, disease resistance and degeneration remain the foremost cause of tumor-related deaths in progenies. Additionally, the ALL cure ratio in adults is ~50%, which is relatively poor.7 Although current treatments such as stem cell transplantation and chemotherapy are emerging quickly, deterioration and medicine-associated obstacles are still endured by patients.8, 9 Another major difficulty is the progression of multi-drug resistance in these patients.10 Thus, innovative anti-leukemic medications and optimizing dominant chemotherapeutic usages are essential to rapid the diagnosis and enhancing patients’ quality of life in ALL.11 Hence, new therapeutic strategies are urgently required.
Epigenetics refers to the heritable changes in gene function with no alterations in DNA sequences, consisting of DNA methylation, histone modification and nucleosome remodeling, among others. Epigenetic modifications play a key role in regulating DNA-based processes. During the past few decades, epigenetic modifications were found to play a significant role in the occurrence and development of leukemia and were considered a promising target for treating different types of leukemia and other hematological malignancies, and sound clinical effects have been achieved.12
Several naturally occurring phytochemicals have been identified due to evidence of their anticancer properties, and they are considered possible therapeutics for regulating various malignant cells by inhibiting multiplication, incursion and metastasis.13 Nerolidol (NRD) is isolated from the aromatic florae as essential oil and identified as a sesquiterpene alcohol.14 It has numerous pharmacological properties, including anticancer, apoptotic, anti-oxidative, and anti-inflammatory effects.15 Recently, NRD has been revealed to alleviate inflammation, oxidative stress and apoptosis in cyclophosphamide-induced cardiotoxicity.16 It has also been documented that NRD blocked the inflammatory reaction in lipopolysaccharide (LPS)-stimulated acute lung injury by the elevation of antioxidants and AMPK/Nrf-2/(HO)-1 signaling.17 Furthermore, NRD has been demonstrated to be valuable as an anticancer complex, owing to its efficiency in suppressing proliferation and targeting cell survival18; it acts as a chemosensitizer in many malignancies.19, 20 Nerolidol was shown to improve the efficacy of doxorubicin (DOX) in breast carcinogenesis19 and enhance its effectiveness in lymphoblast and ovarian carcinoma cells.21 Nevertheless, to the best of our knowledge, the anticancer and apoptotic activities of NRD in ALL cells MOLT-4 have not yet been reported.
Apoptosis is an active cell death program that occurs in normal physiological and pathological environments. It has been established that triggering apoptotic cell death is a highly beneficial approach by which anti-tumor compounds can target malignant cells.22 Chemotherapeutic mediators may stimulate apoptotic signaling via 2 crucial pathways: an intrinsic mitochondrial-facilitated pathway and a death receptor-facilitated extrinsic pathway. An intrinsic cascade is activated by specific molecules, ultimately inducing downstream caspase-3 stimulation, which is one of the vital apoptotic mediators.23 Caspases are a series of cysteine proteases that play a critical part in the molecular pathways of cell death.24 A set of oxygen-containing chemicals comprise reactive oxygen species (ROS), generally produced from the mitochondria.25, 26 Tumor cells usually accumulate more ROS than typical cells.27 Reactive oxygen species-stimulated apoptosis is greatly dependent on the triggering of pro-apoptotic signaling pathways such as PI3K/AKT, STAT-3, NF-κB, and MAPKs. The NF-κB is a well-known chemo-resistance-related anti-apoptotic factor.28 Acute lymphoblastic leukemia cells’ elevated constitutive NF-κB activity caused anti-apoptosis and inflammation.29 Therefore, regulation of NF-κB is a strong molecular target for therapeutics. Trials have revealed cellular communications relating to ROS and NF-κB.28 While ROS might activate the tumor cell apoptosis, the ROS-prompted anti-apoptotic component, NF-κB, can reduce the apoptotic activity of ROS. PI3K/Akt is the most activated network, observed in 70–85% of ALL patients.30 Dysregulation of PI3K/Akt signaling is a major event occurring in both T-cell and B-cell ALL, resulting in increased proliferation, survival and drug resistance.31 Due to the significant physiological communication between STAT-3, Bcl-2 and PI3K/Akt networks in acute leukemia, these signaling networks are strong targets for collective inhibition.32, 33 The vascular endothelial growth factor (VEGF) protein family has been implicated in vascular angiogenesis regulation.34 More importantly, increasing evidence has established that aberrant regulation of VEGF signaling triggering stimulation of the PI3K/Akt pathway is connected to the poor prognosis in ALL.35 Hence, identifying medications that can concurrently trigger persistent ROS-facilitated pro-apoptotic signaling and inhibit PI3K/Akt/NF-κB and STAT3/VEGF/Bcl-2 activities may advance cancer chemotherapy.
Even though NRD has been described as having anti-tumorigenesis activities in numerous human malignant cell lines, its anticancer benefits on blood cancer have barely been researched. However, the possible use of NRD in anti-tumor therapy in ALL cells is relatively unexplored, especially in MOLT-4 cells. Therefore, the current work is focused on assessing NRD for inhibiting cell multiplication and stimulating apoptosis in the ALL cell line, MOLT-4.
Objectives
This research aimed to demonstrate that NRD instantaneously triggers ROS-mediated pro-apoptotic signaling and caspases and attenuates PI3K/Akt/NF-κB and STAT3/VEGF/Bcl-2 anti-apoptotic signaling.
Materials and methods
Reagents and chemicals
Nerolidol (NRD), Dulbecco’s modified Eagle’s medium (DMEM), antibiotics, fetal bovine serum (FBS) , phosphate-buffered saline (PBS), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), dimethyl sulfoxide (DMSO), Rhodamine-123 (Rh-123), sodium dodecyl sulfate (SDS), acridine orange/ethidium bromide (AO/EB), and 2′−7′-dichlorodihydrofluorescein diacetate (DCFH-DA) were obtained from Ruicong Ltd (Shanghai, China). The primary and secondary antibodies were acquired from Elabscience Biotechnology (Wuhan, China).
Culturing of MOLT-4 cells
Human blood cancer MOLT-4 cells were procured from the German Collection of Microorganisms and Cell Cultures ((DSMZ); Braunschweig, Germany). They were matured and maintained in DMEM, with the addition of antibiotics (100 U/mL) and FBS (10%), at 37°C, 5% CO2 atmosphere and less than 95% humidified air.
Cell cytotoxicity assay
Using the MTT test, the effect of NRD on MOLT-4 cells was measured36 and was used to determine cell proliferation. MOLT-4 cells were sown at 6 × 103 cells/well in 96-well plates kept overnight in DMEM media. Treated cells were given various doses of NRD (5–50 μM/mL) at 37°C. A volume of 20 µl of MTT was administered to an individual well and kept at 37°C for 4 h to permit the MTT conversion into formazan insoluble crystals through mitochondrial dehydrogenase. The subsequent formazan crystals were softened by adding 150 μL DMSO to the culture media. Each experiment was repeated thrice, and cells grown in a culture medium containing DMSO were used as a control. To assess cell proliferation, the optical density (OD) was determined at 570 nm employing a microplate reader (Pope Scientific, Inc, Saukville, USA). Cell proliferation was determined as the ratio of cell viability against untreated MOLT-4 cells (100%). The IC50 (drug concentration that caused a 50% reduction in MTT assay) was determined.
Measurement of intracellular level ROS
The level of ROS was determined with the DCFH-DA staining method.37 Blood cancer cells, MOLT-4, were seeded in 6 culture dishes each and maintained for 24 h, and various concentrations of NRD (control and 20 and 30 μM/mL) were administered. Following this, all groups were stained with DCFH-DA (10 μM) and subsequently kept at 37°C. Treated cells were gathered and then cleaned twice with ice-cold PBS to remove extra dye. The fluorescence was measured by excitation at 485 ±10 and emission at 530 ±12.50 nm, compatible with a multimode reader (PerkinElmer EnVision; PerkinElmer, Waltham, USA).
Analysis of mitochondrial membrane potential
The mitochondrial membrane potential (MMP) was estimated to understand early apoptotic phases by conducting Rh-123 staining.37 Human ALL adult cells, MOLT-4, were seeded in 6 wells and sustained with CO2 (5%) at 37°C in a humidified CO2 incubator for 24 h. Subsequently, MOLT-4 cells were exposed to a control solution and 20 and 30 μM of NRD for 1 day, wet twice with cold PBS, fixed with paraformaldehyde (4%) for 20 min and swabbed. Successively, all groups were stained with Rh-123 (10 μg/mL) in darkness at 37°C for 30 min. Following this, they were washed twice with washing buffer to remove any extra stain, and the MMP difference was determined using fluorescent microscopy (Nikon Eclipse TS100; Nikon Corp., Tokyo, Japan). The intensity of fluorescence in the captured images was examined using ImageJ v. 1.48 software (National Institutes of Health (NIH), Bethesda, USA).
Evaluation of apoptosis demonstrated by AO/EB staining
Morphological and nuclear changes were assessed in MOLT-4 human ALL cells, to which NRD was added, and were distinguished by staining with AO/EB.37 MOLT-4 cells were supplemented with control and NRD (20 and 30 µM/mL) conditions and sustained for 24 h. All groups were administered the dye mixture of AO/EB (100 µg/mL per each). Treated cells were maintained in the dark for 20 min, ensuring unbound dye was eliminated by washing with PBS and viewing through a fluorescence microscope (Nikon Eclipse TS100; Nikon Corp.).
Analysis of oxidative stress markers
The oxidative stress markers assessed were glutathione (GSH), catalase (CAT), superoxide dismutase (SOD), and thiobarbituric acid reactive substances (TBARS), using assay kits provided by Biomed (Badr City, Egypt).
Measurement of caspase -3, -9 and -8
The activity of the caspases caspase-3 (cat. No. 32203), caspase-9 (cat. No. 13915) and caspase-8 (cat. No. 13913) was determined with an enzyme-linked immunosorbent assay (ELISA) kit procured from Cayman Chemical (Ann Arbor, USA). These experiments were conducted according to the manufacturer’s protocol, and 3 independent replicates were carried out.
Estimation of mRNA levels using quantitative reveserse transcription polymerase chain reaction
Total RNA was isolated from human ALL cells, MOLT-4, according to the procedures indicated for TRIzol® reagent (Bio-Rad, Hercules, USA). The isolated RNA was reverse transcribed into cDNA using the High-Capacity cDNA Reverse Transcription kit (Thermo Fisher Scientific, Waltham, USA) following the kit protocols. Next, the Fast Start SYBR Green master mix (Bio-Rad) was used to investigate the cDNAs according to the company’s protocols. The samples were subjected to electrophoresis, and the resulting band intensities were measured using ImageJ v. 1.48 software (National Institutes of Health (NIH), Bethesda, USA). Fold conversions for the individual gene expressions were determined using a comparative threshold cycle (Ct) method, according to the formula 2−(ΔΔCt). The series of primers for using quantitative reveserse transcription polymerase chain reaction (RT-qPCR) were as follows:
TNF-α F: 5′-CTTCTGCCTGCTG CACTTTGGA-3′
R: 5′-TCCCAAAGTAGACCTGCCCAGA-3′
NF-κB F: 5′-GCAAAGGGAACATTCCGATAT-3′
R: 5′-GCGACATCACATGGAAATCTA-3′
COX-2 F: 5′-CCGGGTACAATCGCACTTAT-3′
R: 5′-GGCGCTCAGCCATACAG-3′
IL-6 F: 5′-GACTGATGTTGTTGACAGCCACTGC-3′
R: 5′-TAGCCACTCCTTCTGTGACTCTAACT-3′
GAPDH F: 5′-CTTCTTTTGCGTC GCCAGCCGA-3′
R:5′-ACCAGGCGCCCAATACGACCAA-3′
Western blot study
The human blood cancer cells, MOLT-4, were given NRD (control and 20 and 30 µM/mL) and grown for 1 day. The cell lysates were prepared with ice-cold lysis buffer, including protease inhibitors, and a western blot experiment was carried out. The measurement of protein was performed using a Protein BCA Assay Kit (Pierce Chemical Co, Rockford, USA). Briefly, the proteins were electrophoretically dispersed and relocated to a polyvinylidene difluoride (PVDF) film. Then, the film was blocked using a probe overnight at 4°C, and primary antibodies were administered in 1:1,000 dilutions, preserved overnight at 4°C; secondary antibodies (1:5,000) were subsequently added. Beta-actin was employed as an internal control. After introducing the specific antibodies, the proteins were visualized with an LI-COR Odessy imaging system (LI-COR, Belfast, UK).
Statistical analyses
The statistical analysis of data from each group was conducted using GraphPad Prism v. 8.0.2 (GraphPad Software, San Diego, USA) and IBM SPSS software v. 25 (IBM Corp., Armonk, USA). Data are presented as the median (min–max). As the sample size was too small to verify normal data distribution, the differences between the groups were analyzed using the nonparametric Kruskal–Wallis test with Dunn’s post hoc test. Subsequently, significant differences among multiple groups were examined using the Kruskal–Wallis test, and Dunn’s post hoc test was employed for multiple comparisons. Dunn’s post hoc test was employed for multiple comparisons to control for the false positive rate due to multiple testing. Dunn’s test adjusts the significance levels to account for multiple comparisons, ensuring the control of type I error rates across the 16 comparisons. A statistically notable data divergence was considered when p < 0.05. All tests in this study were bilateral.
Results
The results of the Kruskal–Wallis test and Dunn’s post hoc test are presented in Table 1, Table 2, Table 3.
Cytotoxicity effect of NRD on human ALL cells
The MTT test estimated human MOLT-4 cell proliferation at various quantities (control, 5, 10, 20, 30, 40, and 50 μM/mL) of NRD (Table 1, Figure 1A). It was revealed that NRD had antiproliferative effects on MOLT-4 cells in a concentration-dependent manner. Untreated control cells did not exhibit altered MOLT-4 cell proliferation and showed 100% viability. However, NRD at dosages of 10, 20 and 30 μM/mL considerably (p < 0.05) inhibited the viability of MOLT-4 cells compared to control. Viability was extremely inhibited by the administration of a high dosage of NRD (40 and 50 μM/mL). From the MTT test, the IC50 value was determined to be 30 μM for MOLT-4 cells. To avoid the destruction caused by high dosages of NRD in ALL cells, we added various dosages of NRD to the MOLT-4 cells, assessing their sensitivity to NRD administration. Based on data from the IC50 value of NRD (20 and 30 μM/mL), we opted for further experiments. The MOLT-4 human ALL cells’ morphological variations were detected with a phase-contrast microscope (Nikon Eclipse TS100; Nikon Corp.) in the bright field. Administration of 20 μM NRD resulted in a substantial loss of cell viability and morphological modifications, and augmented morphological changes were observed with higher amounts of NRD. The IC50 value of 30 μM NRD reduced cell proliferation and additional morphological modifications of MOLT-4 cells (Figure 1B).
Influence of NRD-induced intracellular ROS generation in MOLT-4 cells
The formation of ROS in cells is related to different stimuli and can induce apoptosis and block the cell cycle (Figure 2A). To assess ROS accumulation, DCFH-DA-labeled MOLT-4 cells were examined with a fluorescence microscope (Nikon Eclipse TS100). High-intensity green fluorescence correlated with the level of ROS. Administration of NRD (20 μM/mL) in MOLT-4 cells resulted in a weak fluorescent background, whereas NRD (30 μM/mL) treatment resulted in bright green fluorescence, which signified an elevated ROS level. The formation of ROS in the MOLT-4 human ALL cells was very low, while the level of ROS increased in the NRD-supplemented (20 and 30 μM/mL) groups in a concentration-dependent manner. Treatment with 30 μM NRD significantly (p < 0.05) enhanced ROS generation in MOLT-4 cells compared to control and 20 μM NRD.
Nerolidol stimulated the loss of MMP in human ALL cells
The MMP depolarization was assessed measuring the cell’s ability to take up Rh-123 dye (Figure 2B). MOLT-4 human acute lymphoblastic leukemia control cells presented high Rh-123 intensity, depicted as green fluorescence and almost 99% MMP. Rh-123 fluorescence accumulation was reduced to a weak green fluorescence signal in NRD-treated (20 and 30 µM/mL) MOLT-4 cells in a dosage-related manner. Nerolidol (30 μM) supplemented with MOLT-4 cells resulted in an extensive increase (p < 0.05) in MMP loss due to strengthened mitochondrial depolarization.
Nerolidol-triggered apoptosis in human ALL cells as demonstrated with AO/EB staining
Apoptotic cells were revealed due to the unique morphological changes identified using AO/EB dual staining (Figure 3). Human MOLT-4 cells were visible as homogeneously dyed green live cells. Nerolidol (20 and 30 μM/mL) supplementation increased cell apoptosis dose-dependently. When intercalated into dsDNA, AO is collected by both viable and non-viable cells and exhibits green fluorescence, whereas EB only accumulates in non-viable cells, where it intercalates into DNA to produce red fluorescence. Therefore, early apoptotic cells have green-fluorescent nucleus with dense green spots where chromatin is condensing, in contrast with the shorter or fragmented orange chromatin. Early apoptotic cells showed abridged chromatin and membrane blebbing with 20 μM NRD administration, which appeared as pale greenish-yellow marks. Late apoptotic cells exhibited an orange color, indicating a decrease in their membrane integrity owing to the ethidium bromide co-stain in 30 μM NRD-administered MOLT-4 cells.
Nerolidol enhances antioxidant status and reduces lipid peroxidation
To observe the influence of NRD on oxidative stress, we analyzed the antioxidant status and lipid peroxidation levels in MOLT-4 cells. Human MOLT-4 cells showed elevated levels of TBARS with decreased SOD, CAT and GSH compared to NRD-treated cells. However, the antioxidant status and lipid peroxidation levels were reversed by the administration of NRD. Treatment with NRD (20 and 30 μM/mL) significantly reduced (p < 0.05) the intensity of TBARS, whereas SOD, CAT and GSH increased compared to control dose-dependently (Table 2, Table 3 and Figure 4).
Measurement of caspase -3, -8
and -9 activities with ELISA
MOLT-4 cells administered NRD showed elevated activity of caspase-3, -8 and -9 when compared to untreated control cells (Table 2, Table 3 and Figure 5). Nerolidol (30 μM) significantly (p < 0.05) enhanced the caspase protein levels compared to 20 μM NRD-treated MOLT-4 cells. These results reveal that NRD stimulated the pro-apoptotic markers, such as caspase proteins, in MOLT-4 cells.
Influence of NRD on the mRNA expression of anti-apoptotic inflammatory cytokines
Figure 6 depicts the influence of NRD on the mRNA level of anti-apoptotic pro-inflammatory mediators in human ALL cells (Table 2, Table 3). MOLT-4 cells exhibited upregulated tumor necrosis factor alpha (TNF-α), nuclear factor kappa B (NF-κB), interleukin 6 (IL-6), and cyclooxygenase-2 (COX-2) mRNA levels. Nerolidol (20 and 30 μM/mL) treated-MOLT-4 cells significantly (p < 0.05) attenuated the levels of these cytokines dose-dependently. These results suggest that NF-κB inhibition is involved in NRD-triggered MOLT-4 cell growth inhibition.
Effect of NRD on the PI3K/AKT pathway
Administration of NRD (20 and 30 μM/mL) resulted in dose-dependent downregulation of phosphorylated PI3K and AKT protein expression compared to untreated controls (Table 2, Table 3 and Figure 7). MOLT-4 human ALL cells treated with NRD showed reduced p-PI3K, PI3K, p-AKT, and AKT protein expression; thus, NRD induced apoptosis in MOLT-4 cells through inhibition of PI3K/AKT signaling.
Influence of NRD on STAT-3/VEGF/Bcl-2 protein levels
Administration of NRD (20 and 30 μM/mL) resulted in dose-dependent downregulation of phosphorylated STAT-3 as well as VEGF and Bcl-2 protein expression levels compared to untreated controls (Table 2, Table 3 and Figure 8). MOLT-4 human ALL cells treated with NRD attenuated p-STAT-3, VEGF and Bcl-2 protein expression, showing that NRD induced apoptosis and inhibited proliferation and angiogenesis in MOLT-4 cells through the suppression of STAT-3/VEGF/Bcl-2 signaling.
Discussion
While there are several remedies available for ALL, there is a need for more effective and less toxic treatments due to harmful effects and drug resistance. This objective has resulted in an increased focus on investigating and improving natural agents used for chemoprevention or in cases of tumor relapses. Recently, NRD has sparked interest due to its immense array of pharmacological actions, including anti-tumor and apoptotic activities.15 The anticancer efficacy of NRD has been widely examined in various types of cancer cells, including oral cancer,38, 39 ovarian cancer,21 osteosarcomas,40 breast cancer,19 and hepatocellular carcinomas,41 among others. There are no previous reports on the induction of apoptosis in MOLT-4 cells by NRD. The current study demonstrates that NRD exhibited an antiproliferative and apoptotic effect on human ALL cell lines MOLT-4. Our results revealed that NRD (20 and 30 µM/mL) efficiently inhibited MOLT-4 cellular proliferation, elevated intracellular ROS accumulation, reduced MMP, and induced apoptosis dose-dependently. In this study, we have shown for the first time that NRD dose-dependently induces significant cytotoxicity in MOLT-4 human ALL cells. Nerolidol reduced cell viability chiefly due to the induction of apoptosis. Numerous anti-tumor medications have been shown to cause death of susceptible cells by stimulating apoptosis. This type of cell death is regarded as a significant reaction to the best chemotherapeutic drugs for leukemia.22, 23 According to the data, this current research is the first description of the antiproliferative and apoptotic actions of NRD on MOLT-4 human ALL cells.
Apoptosis is a highly controlled kind of cell death, through which cells with substantial damage are eradicated.22 Apoptotic cell death can be induced by either an intrinsic mitochondria-facilitated or an extrinsic death receptor-facilitated pathway.23 Hence, medicines are needed that selectively prompt apoptosis in tumor cells by inducing tumor-specific upstream activity. Tumors have recurrently utilized these pathways, and it may be promising to selectively stimulate apoptosis in malignant cells or sensitize them to well-known cytotoxic agents.22 Our experiments prove that NRD stimulated apoptosis in ALL cells through the mitochondria-mediated intrinsic pathway and suggest it may translate as a potential drug candidate for a leukemia remedy. We noticed that NRD effectively induced ROS generation and subsequently reduced MMP, along with high levels of caspase-3, -8 and -9 in ALL cells in a quantity-reliant mode. Reactive oxygen species are intermediaries of several intracellular signaling cascades; however, upon hyperproduction, they may promote MMP collapse, which activates a series of mitochondria-related events comprising apoptosis.25, 26 Related findings were previously reported that NRD exerts antiproliferative and apoptotic activities in osteosarcoma40 and hepatoprotective activities.16 Our study demonstrated that NRD supplementation enhanced ROS generation and MMP loss, correlating with high caspase activity in mitochondrial-mediated ALL cell apoptosis. Thus, NRD triggered ROS-mediated mitochondrial apoptosis in MOLT-4 cells.
Apart from inducing apoptotic features, ROS can also prompt anti-apoptotic elements. The influence of ROS on tumor cells depends on the balance between ROS-promoted pro- and anti-apoptotic factors. Human ALL cells constitutively express enhanced levels of a vital anti-apoptotic factor, nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB).29 However, Nakano et al. established that NF-κB-controlled genes exhibit a crucial action in the accumulation of ROS in the cells.28 It has been shown that ROS have diverse stimulatory or inhibitory actions in the NF-κB pathway.28, 29 Our results point out that NRD significantly suppressed the mRNA expression of COX-2, TNF-α, NF-κB, and IL-6 in a concentration-dependent manner. Therefore, NRD can inhibit NF-κB activation and may stimulate apoptosis in ALL cells. Prior research has established that NRD constrains the intensities of TNF-α and IL-1β in rats with LPS-prompted acute kidney injury and peritoneal macrophages.41, 42 Another study revealed that NRD inhibited rotenone-promoted IL-6, COX-2, IL-1β, and TNF-α expression in rat brain.43 Prior research reported that NRD sequestered from Lindera erythrocarpa essential oil alleviated IκB kinase (IκB) disintegration and NF-κB phosphorylation by MAPK phosphorylation in LPS-triggered macrophages.42 These studies showed that the protective actions of NRD were involved in the attenuation of pro-inflammatory mediators. Our current research found that NRD was able to suppress tumor growth and anti-proliferation by the induction of apoptosis through the inhibition of inflammatory mediators in MOLT-4 cells.
NF-κB, PI3K/AKT and STAT3/VEGF/Bcl-2 signaling pathways are involved in cell proliferation, differentiation, angiogenesis, metastasis, and apoptosis signal transduction, along with their regulatory role in human ALL.28, 32, 33 Aberrant PI3K/AKT and STAT 3 protein expression leads to various aspects of malignant cell growth, differentiation, survival, transformation, metabolism, and cell cycle regulation.32, 33 In this study, NRD (20 and 30 µM/mL) was able to alter PI3K/AKT and STAT3/VEGF/Bcl-2 protein expression dose-dependently. Previously, similar results were reported that NRD induced apoptosis and cell cycle arrest in MG-63 osteosarcoma cells via PI3K/AKT/JNK regulation.40 Several molecular pathways are involved in malignant metastasis, including the VEGF signaling pathway.34 VEGF signaling regulates trans-endothelial leukemia cell migration, contributes to extramedullary infiltration of ALL cells, and is implicated in vascular angiogenesis regulation.34 More importantly, increasing evidence has established that aberrant regulation of VEGF signaling triggering the stimulation through the PI3K/Akt pathway is connected to a poor prognosis in ALL.35 Based on the physiological communication between the STAT-3, BCL-2 and PI3K/Akt signaling networks, which are significant to acute leukemias, these pathways comprise strong targets for collective inhibition.32, 33 Our current findings showed that NRD inhibited PI3K/AKT/NF-κB and STAT-3/VEGF/Bcl-2 signaling and induced apoptotic, anti-inflammatory, anti-oxidative, anti-angiogenic, and antiproliferative actions in MOLT-4 cells. This may be the first report that NRD inhibits the PI3K/AKT/NF-κB and STAT3/VEGF/Bcl-2 signaling pathways in MOLT-4 human ALL cells and provides additional proof of the underlying anticancer mechanisms of NRD. Important to this study, the development of microscopic techniques has revolutionized the morphological sciences, progressively providing new levels of magnification and resolution for exploring biological and non-biological samples. Microscopy, in fact, provides an understanding of the structure and ultrastructure and of the organization of the main cellular components.44
Limitations
In vivo method could not be conducted in this study; however, it will be utilized in future research. PI3K, AKT, STAT3, VEGF, Bcl-2, caspase-3, caspase-9, and caspase-8 were analyzed in protein levels, but need to be analyzed also in RNA levels in future research. Moreover, detailed mechanisms of action still need to be clarified.
Conclusions
Nerolidol was shown to effectively suppress the viability and inflammation and dose-dependently induce apoptosis in MOLT-4 human ALL cells. By increasing the antioxidant status, NRD demonstrated its lethal effects by forming intracellular ROS, which may be caused by MMP collapse, caspase activation or the reduction of NF-κB-associated inflammatory mediators. By blocking the PI3K/AKT/NF-κB pathways, NRD caused ROS-mediated mitochondrial death. Moreover, by blocking STAT-3/VEGF/Bcl-2 signaling, NRD decreased the viability of MOLT-4 cells and angiogenesis. These results suggest that NRD may be developed as a potential preventative therapeutic for ALL in humans. Additional investigations should confirm the anticancer activity using in vivo animal models.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.13268443. The package includes the following files:
Supplementary Fig. 1. Results of Kruskal–Wallis test as presented in Figure 1.
Supplementary Fig. 2. Results of Kruskal–Wallis test as presented in Figure 4.
Supplementary Fig. 3. Results of Kruskal–Wallis test as presented in Figure 5.
Supplementary Fig. 4. Results of Kruskal–Wallis test as presented in Figure 6.
Supplementary Fig. 5. Results of Kruskal–Wallis test as presented in Figure 7.
Supplementary Fig. 6. Results of Kruskal–Wallis test as presented in Figure 8.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.

















.png)
.jpg)
.jpg)
-VAR1.png)
-VAR2.png)
-VAR2.jpg)
-VAR2.jpg)
-VAR2.jpg)