Abstract
Background. The skin, with its robust structural integrity and advanced immune defense system, serves as a critical protective barrier against environmental toxins and carcinogenic compounds. Despite this, it remains vulnerable to the harmful effects of certain hazardous agents.
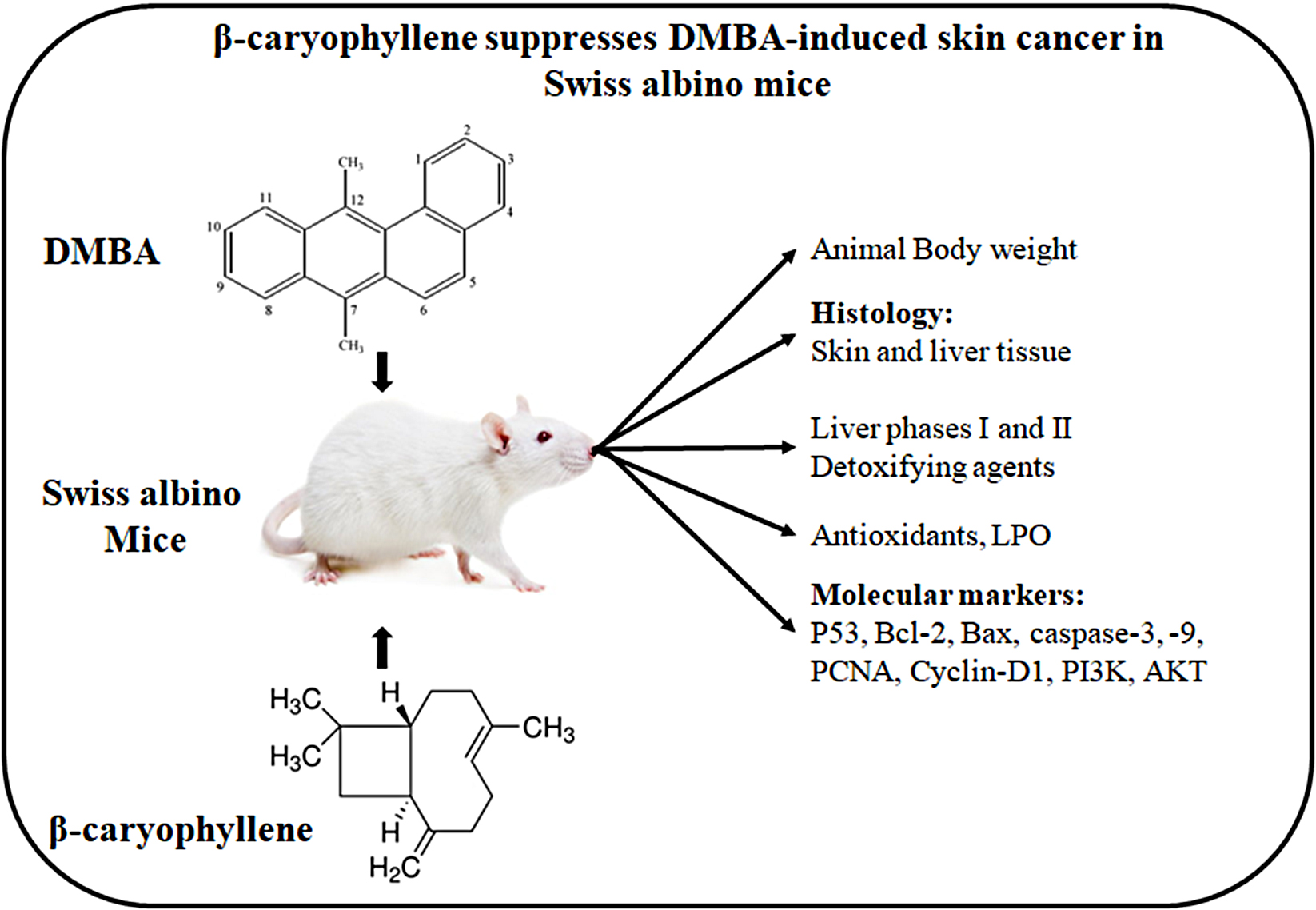
Objectives. This study aimed to investigate the chemopreventive potential of β-caryophyllene (BCP) in mitigating 7,12-dimethylbenz[a]anthracene (DMBA)-induced skin carcinogenesis, focusing on the modulation of apoptosis and PI3K/AKT signaling pathways.
Materials and methods. Swiss albino mice were utilized to assess the preventive effects of BCP in DMBA-induced skin cancer. Skin carcinogenesis was initiated by topical DMBA application, followed by promotion using croton oil. To evaluate the chemopreventive efficacy of BCP, a 50 mg/kg oral dose was administered 3 times a week for 16 weeks.
Results. The BCP treatment in DMBA-induced skin cancer mice significantly reduced tumor incidence, tumor burden and the total number of papillomas compared to untreated DMBA-exposed mice. Notably, BCP administration (p < 0.05) resulted in a marked increase in body weight and improvement in antioxidant enzyme activity. Additionally, BCP treatment led to significant reductions in lipid peroxidation and enhanced detoxification enzyme function. Histological examination of DMBA-induced skin tissues revealed the presence of keratin pearls, well-differentiated tumor cells and neutrophil infiltration. In contrast, BCP-treated mice showed only mild hyperplasia, dysplasia and moderate keratosis, suggesting a lower degree of tissue damage. Furthermore, BCP demonstrated a protective effect on liver histology, counteracting the toxic effects of DMBA exposure. Gene expression analysis revealed that BCP treatment significantly (p < 0.05) upregulated the pro-apoptotic genes Bax, p53, caspase-3 and caspase-9, while downregulating the anti-apoptotic Bcl-2 expression. Additionally, BCP treatment led to a marked reduction in the expression of proliferating cell nuclear antigen (PCNA), cyclin D1 and PI3K/Akt signaling pathways, which are key regulators of cell proliferation and survival.
Conclusions. This study provides compelling evidence that the antioxidant and pro-apoptotic effects of β-caryophyllene contribute to its chemopreventive properties in DMBA-induced skin carcinogenesis in mice. The modulation of key apoptotic signaling pathways and the suppression of the PI3K/Akt pathway by BCP underscores its potential as a therapeutic agent for preventing skin cancer. These findings pave the way for further exploration of BCP as a promising candidate for skin cancer prevention and therapy.
Key words: chemoprevention, skin carcinogenesis, β-caryophyllene, oxidative stress, papilloma
Introduction
Skin cancer is thought to be the most common disease affecting people.1 According to a literature review, 40% of all newly diagnosed cancer cases worldwide are skin cancer instances.2 A survey conducted in India indicates that cutaneous neoplasms make up roughly 1–2% of all human cancers.3 The prevalence of skin cancer has increased due to changes in lifestyle and environmental risks. The skin’s keratin layer and immune system provide an effective barrier against potentially hazardous exposures, including those that may be carcinogenic.4 The chemical pollutant 7,12-dimethylbenz[a]anthracene (DMBA) induces DNA damage and interferes with xenobiotic metabolism.5, 6 It also breaks the bonds that bind proteins to DNA, leading to genomic instability and inflammation, which are 2 primary indicators of cancer.7 DMBA, a polycyclic aromatic hydrocarbon, is frequently used in animal models to induce cancer.8
Humans are often exposed to a variety of chemical mutagens and carcinogens, either through unintended contact, occupational hazards or lifestyle choices. The skin, due to its direct exposure to a wide range of xenobiotics, is particularly vulnerable to cancer development.9 Polycyclic aromatic hydrocarbons (PAHs), such as benzo(a)pyrene, 3-methylcholanthrene (MCA) and DMBA, are well-documented for their ability to induce skin cancer in animal models, particularly when used in one-step or two-step carcinogenic protocols (initiation and promotion).10 Recent findings from our research group have also pointed to aflatoxin B1 (AFB1) as a precursor to skin cancer formation.7 These discoveries have prompted an increased focus on identifying compounds that can inhibit the enzymes responsible for activating xenobiotic metabolism, as such inhibitors could potentially lower cancer risk and offer a safer alternative in cancer prevention.
Uncontrolled cell proliferation and the evasion of apoptosis are key factors that drive tumor growth.11 Extensive research has shown that many natural chemopreventive agents exert their effects by promoting apoptosis.12, 13, 14 The protein p53, recognized as a transcriptional activator for pro-apoptotic genes, is a crucial tumor suppressor. Mutations or inactivation of the p53 gene, which governs processes such as apoptosis, senescence and cell cycle regulation, are common in nearly all human cancers, including skin cancer.15, 16 Additionally, alterations in the Bcl-2 family of proteins, which play a vital role in regulating apoptosis, have been implicated in various cancers. Studies have demonstrated that in several human cancers, including skin cancer, Bcl-2 is overexpressed while Bax, a pro-apoptotic protein, is downregulated.17, 18 The interaction between Bcl-2 and Bax prevents Bax from dimerizing with Bad, thus impeding the apoptotic process.19 Upon Bax/Bad dimer formation, caspases-9 and -3 are activated, leading to mitochondrial membrane disruption, which allows cytochrome-c to leak from the mitochondria into the cytoplasm, triggering further apoptotic events.20, 21, 22
β-caryophyllene (BCP) is a prominent constituent of essential oils derived from various food and spice plants.23 Research in Wistar rats has highlighted the anti-inflammatory properties of BCP, particularly in the essential oil of Erymanthus erythropappus, which is rich in this compound. Moreover, BCP has been found to inhibit the expression of pro-inflammatory markers such as inducible nitric oxide synthase (iNOS), interleukin (IL)-1, IL-6, and cyclooxygenase-2 (COX-2) in C6 microglia cells when administered at a dose of 10 mg/kg.24 Additionally, BCP was shown to mitigate the neuroinflammatory response triggered by hypoxia by blocking nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) activation, and by reducing nitric oxide and PGE2 production in the BV2 mouse cell line.
Objectives
The current study aimed to explore the potential chemopreventive mechanisms of BCP against DMBA-induced skin carcinogenesis, focusing on identifying possible pathways through which BCP may exert its protective effects. We performed a comprehensive histological analysis along with assessments of lipid peroxidation, antioxidant levels and detoxification enzyme activity. We also evaluated tumor incidence, volume and burden. To investigate the molecular mechanisms, we utilized quantitative reverse transcription polymerase chain reaction (RT-qPCR), proliferating cell nuclear antigen (PCNA) and analysis of the PI3K/Akt signaling pathway to measure the mRNA expression of p53 and several key apoptotic proteins, including Bcl-2, Bax, caspase-3, and caspase-9.
Materials and methods
Chemicals
Dimethylsulfoxide (DMSO), DMBA (95% purity, Chemical Abstracts Service (CAS) No. 57-97-6, soluble in mineral oil) and β-caryophyllene (BCP, 90% purity, CAS No. 87-44-5) were purchased from Sigma-Aldrich Chemical Pvt. Ltd. (St. Louis, USA) Antibodies were obtained from BioGenex (Freemont, USA). All other analytical-grade chemicals used in the study were also supplied by Sigma-Aldrich.
Animals
Male Swiss albino mice, aged 5 to 6 weeks and weighing between 15 and 20 g, were randomly selected, transported and housed for the study. The animals were acclimated and maintained in accordance with the ethical guidelines set forth by the Animal Care Committee of Changchun University of Chinese Medicine, China. The experimental protocol was approved by the Institutional Animal Ethics Committee of Changchun University of Chinese Medicine (registration No. 2023551).
Experimental design
Six experimental groups, each consisting of 6 animals, were established from the animal population. Group 1 served as the normal control, while group 2 received a single dose of 25 mg/kg DMBA dissolved in 100 µL of acetone. In group 3, animals were administered a 1% DMSO solution containing both 50 mg/kg of BCP and 50 mg/kg of DMBA. Group 4 animals received a 50 mg/kg body weight dose of BCP, administered orally 3 times per week, starting 1 week prior to DMBA application and continuing through the 25th week. The study concluded after 25 weeks, at which point all animals were euthanized. The skin and liver tissues were promptly harvested, cleaned and weighed for further analysis.
Tumor induction and assessment
Over the course of 8 weeks, DMBA was applied topically, and tumor progression was closely monitored. Throughout the study, body weight of all animals was recorded weekly across all experimental groups. To prepare for treatment, the dorsal area of each mouse was shaved using a depilatory cream, and the skin was left undisturbed for 2 days. Mice with no hair regrowth during this period were selected for inclusion in the study. At the conclusion of the experiment, all animals were euthanized by cervical dislocation. Tumor burden was calculated by multiplying the number of tumors by the tumor volume per animal. Tumor volume was determined using the formula v = (4/3) [D1/2] [D2/2] [D3/2], where D1, D2 and D3 represent the 3 tumor diameters (in mm³).
Histopathological examinations
Skin and liver tissues containing tumors were promptly excised from the animals after the rats were sacrificed, and washed with saline solution. Following this, all animals were humanely euthanized by cervical dislocation. Tissue specimens, sectioned at 3–5 µm thickness using a rotating microtome, were stained with hematoxylin and eosin (H&E). The stained slides were then examined under a light microscope (Nikon Eclipse TS100; Nikon Corp., Tokyo, Japan), and photomicrographs were captured from both the experimental and control groups.
Biochemical analysis
The liver tissues of the same animals were further analyzed to assess the expression of xenobiotic-metabolizing enzymes (XMEs) involved in both phase I and phase II detoxification pathways. The skin and liver tissues were homogenized using an all-glass homogenizer equipped with a Teflon pestle (DuPont, Wilmington, USA) along with the appropriate buffer solution, following a rinse with ice-cold saline.25 Protein content was quantified using the established method. To evaluate the enzymatic activities of cytochrome P450 (Cyt-P450), cytochrome b5 (Cyt-b5), glutathione S-transferase (GST), and glutathione reductase (GR) in the liver, we applied the procedure described by Esteves et al.26 The same methodology was used to measure the concentrations of thiobarbituric acid reactive substances (TBARS) in the skin tissue samples. Enzymatic activities for superoxide dismutase (SOD), glutathione peroxidase (GPx), catalase (CAT), and glutathione peroxidase (GPx) were determined according to standard protocols.27 Additionally, the levels of glutathione (GSH), as well as oxidized GSH, were assessed in the skin tissues.27
mRNA expressions detected with quantitative reverse transcription polymerase chain reaction (RT-qPCR)
Total RNA was extracted from skin tissues using TRIzol reagent (Thermo Fisher Scientific, Waltham, USA) according to the manufacturer’s protocol. The isolated RNA was subsequently converted into complementary DNA (cDNA) using the cDNA Reverse Transcription Kit. For gene expression analysis, the cDNAs were evaluated using the Faststart SYBR Green Master Mix (Thermo Fisher Scientific), following the manufacturer’s instructions. Electrophoresis was performed on 1.5% agarose gels to visualize the RNA bands, and the band intensity was quantified using ImageJ v. 1.48 software (National Institutes of Health (NIH), Bethesda, USA).
Western blotting
To prepare cell lysates for western blot analysis, lysis buffer was stored at freezing temperatures. Protein concentration was determined using the BCA Protein Assay Kit (Thermo Fisher Scientific). The proteins were then separated by electrophoresis and transferred onto a polyvinylidene difluoride (PVDF) membrane. The membrane was blocked with bovine serum albumin (BSA) for approx. 1 h at room temperature. Next, primary antibodies were applied at a 1:1,000 dilution, and the membrane was incubated overnight at 4°C. Following this, secondary antibodies were applied for 2 h. Enhanced chemiluminescence was used to visualize the protein bands. Quantification of the bands was performed through densitometric analysis using ImageJ software.
Statistical analyses
The data from each group were statistically analyzed using GraphPad Prism v. 8.0.2 (GraphPad Software, San Diego, USA) and IBM SPSS software v. 25 (IBM Corp., Armonk, USA). The results are presented as the median with the interquartile range (Q1 and Q3). The normality of the data distributions was assessed using the Kolmogorov–Smirnov test. Since all distributions were normal, the Brown–Forsythe test was employed to assess the equality of variances. Significant differences between multiple groups were determined using the Kruskal–Wallis test, followed by Dunn’s post hoc test for pairwise comparisons. A p-value of less than 0.05 was considered statistically significant. All statistical tests were two-tailed.
Results
The results of the Kruskal–Wallis test and Dunn’s post hoc test are presented in Table 1
Effect of BCP on body weight changes
To evaluate the chemopreventive potential of BCP, a DMBA-induced mouse skin carcinogenesis model was first established in vivo (Figure 1A). The body weight of the mice in the DMBA group showed a significantly lower gain compared to the BCP-only group (p > 0.02). However, the DMBA group exhibited a marked reduction in body weight compared to the control group (p > 0.001). Experimental groups treated with DMBA + BCP showed a significantly greater reduction in body weight compared to the BCP-only group (p < 1.000). Moreover, the body weight of the DMBA + BCP group was significantly lower than the control group (p < 0.615). Kruskal–Wallis statistical analysis revealed significant differences across the experimental groups (H = 15.62; degrees of freedom (df) = 3; p > 0.001) as shown in Table 1.
Effect of BCP on tumor incidence
DMBA-induced mice exhibited a significantly higher incidence of papillomas, which was notably reduced in the BCP-treated group. Additionally, DMBA exposure resulted in a greater number of skin papillomas, which was significantly suppressed (p < 0.001) in the mice receiving BCP treatment, reflecting a reduction in the number of tumors per mouse. The suppressive effect of BCP on tumorigenesis was further corroborated by the size distribution of the papillomas. Notably, while DMBA treatment increased tumor area, BCP administration significantly reduced the tumor area as the treatment progressed over time (Table 2)
Levels of phase I and II detoxification enzymes
The levels of phase I detoxification enzymes, Cyt-P450 and Cyt-b5, were significantly higher in the livers of mice treated with BCP alone compared to those exposed to DMBA alone (Cyt-P450: p < 0.001; Cyt-b5: p = 0.000). In contrast, the phase I enzymes were significantly lower in the livers of control mice than in the DMBA group (Cyt-P450: p < 0.004; Cyt-b5: p < 0.01). The Kruskal–Wallis statistical test provided the following significance values: Cyt-P450 (H = 19.731, df = 3, p = 0.000) and Cyt-b5 (H = 20.629, df = 3, p = 0.000), indicating significant differences among the experimental groups. Regarding phase II detoxifying enzymes, GSH, GST, and GR, their levels were significantly reduced in the DMBA group compared to both the control group (GSH: p < 0.000; GST: p < 0.001; GR: p < 0.001) and the BCP alone group (GSH: p < 0.017; GST: p < 0.004; GR: p < 0.007). The Kruskal–Wallis test for these enzymes revealed the following significance values: GSH (H = 20.087, df = 3, p = 0.000), GST (H = 19.680, df = 3, p = 0.000) and GR (H = 17.120, df = 3, p = 0.001), highlighting substantial differences among the groups. The administration of BCP at 50 mg/kg bw in DMBA-treated animals effectively restored phase I and phase II detoxifying agents to near normal levels. However, no significant changes were observed when 50 mg/kg of BCP was given alone. The BCP alone treatment resulted in reduced levels of GSH, with no notable effect on phase II enzyme activities (Table 1, Figure 1B).
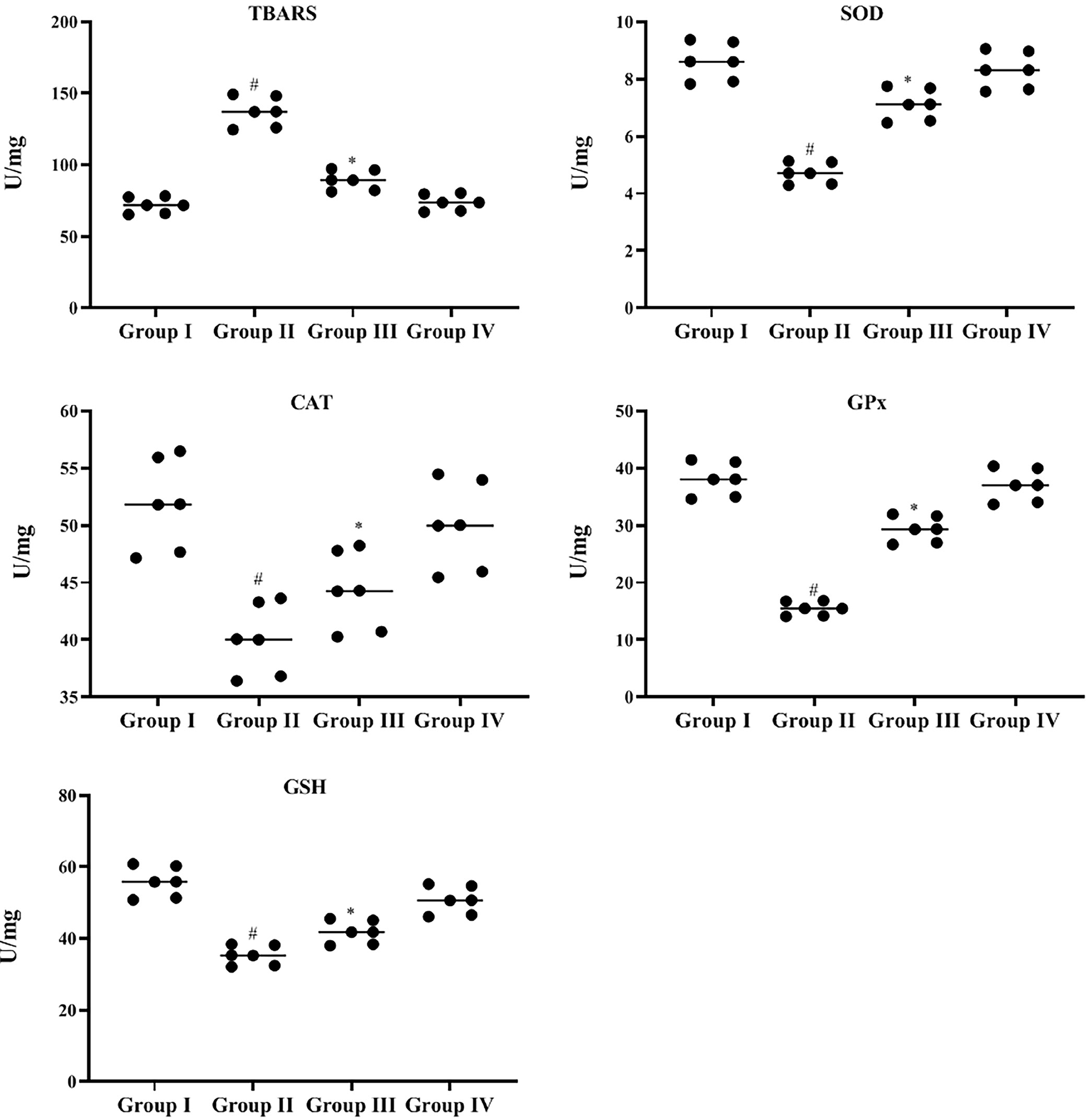
Estimation of lipid peroxidation and antioxidants
The levels of antioxidant indicators — SOD, CAT and GPx — in the skin tissue of experimental and control rats are summarized in Table 1 and Figure 2. In the skin tissues, the enzymatic antioxidant content was significantly lower in the rats treated with DMBA alone compared to those in the BCP alone group (SOD: p < 0.007; CAT: p < 0.012; GPx: p < 0.004) and the control group (SOD: p < 0.001; CAT: p < 0.002; GPx: p < 0.001). In contrast, administration of BCP (50 mg/kg bw) alongside DMBA notably improved antioxidant levels (p < 0.001), outperforming both the DMBA group (SOD: p < 0.133; CAT: p < 0.203; GPx: p < 0.086) and the BCP alone group (SOD: p < 0.614; CAT: p < 0.615; GPx: p < 0.300). Moreover, the levels of lipid peroxidation (LPO) indicators, specifically TBARS, were significantly lower in the skin tissue of control rats compared to the DMBA-only group (p < 0.001). Rats treated with BCP alone exhibited significantly higher LPO by-products (p < 0.004) compared to controls. However, the oral administration of BCP at 50 mg/kg bw significantly reduced the LPO by-product levels in the skin tissue (p < 0.086) when compared to the DMBA-only group. No significant differences were observed between the rats in the control group (group 1) and the BCP-alone group (group 4). The statistical analysis, using the Kruskal–Wallis test, revealed the following significance values among the experimental groups: TBARS (H = 19.680, df = 3, p = 0.000), SOD (H = 18.523, df = 3, p = 0.000), CAT (H = 16.053, df = 3, p < 0.001), and GPx (H = 19.680, df = 3, p = 0.000).
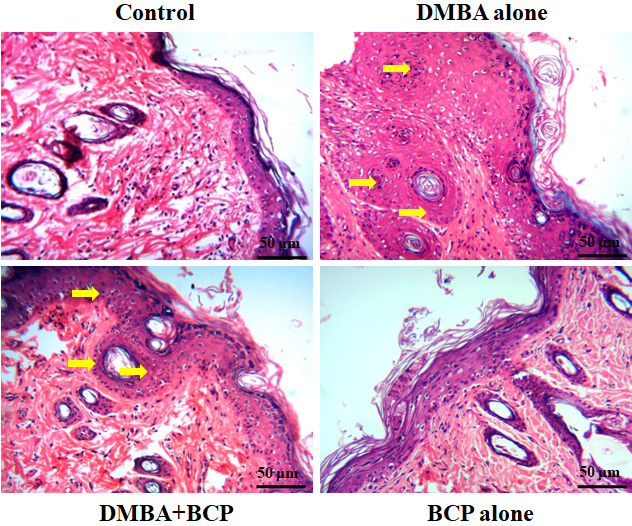
BCP impact on the DMBA-induced in skin tissues
Hematoxylin and eosin (H&E) staining of skin tissue illustrates the progression of squamous cell carcinoma (SCC) in mice. In the normal control (NC) group, skin tissues exhibited a typical, intact epithelial layer. In contrast, the DMBA-treated mice showed significant epidermal thickening, hyperplasia and the development of well-formed SCC with characteristic keratin pearls. Notably, oral administration of BCP at 50 mg/kg bw effectively suppressed these DMBA-induced skin alterations, including hyperplasia, hyperkeratosis and the progression of SCC, in a dose-dependent manner. These findings collectively demonstrate the potent inhibitory effect of BCP on DMBA-induced skin carcinogenesis in mice (Figure 3).
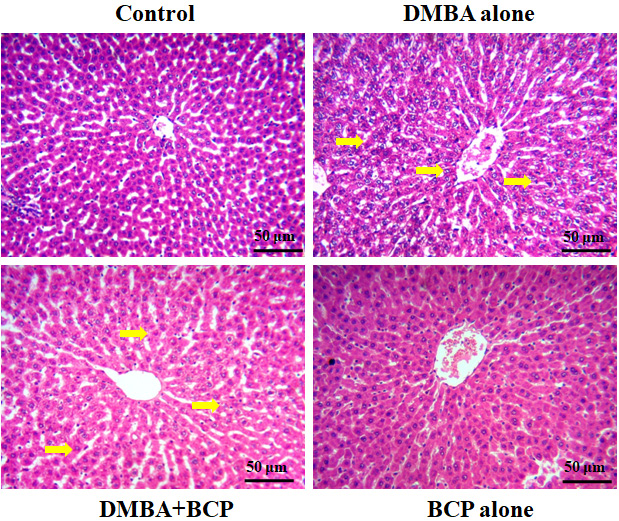
BCP impact on the DMBA-induced in liver tissues
In the DMBA-induced model mice, hepatic tissue sections exhibited pronounced signs of inflammation, including neutrophil infiltration, lymphocyte presence and liver cell damage. Liver cell injury was evident through necrotic changes, such as membrane collapse, cytoplasmic damage, alterations in nuclear pyknosis, and dilation of Bowman’s capsule. Remarkably, these pathological alterations were significantly alleviated with the administration of BCP at 50 mg/kg bw, in a dose-dependent manner (Figure 4).
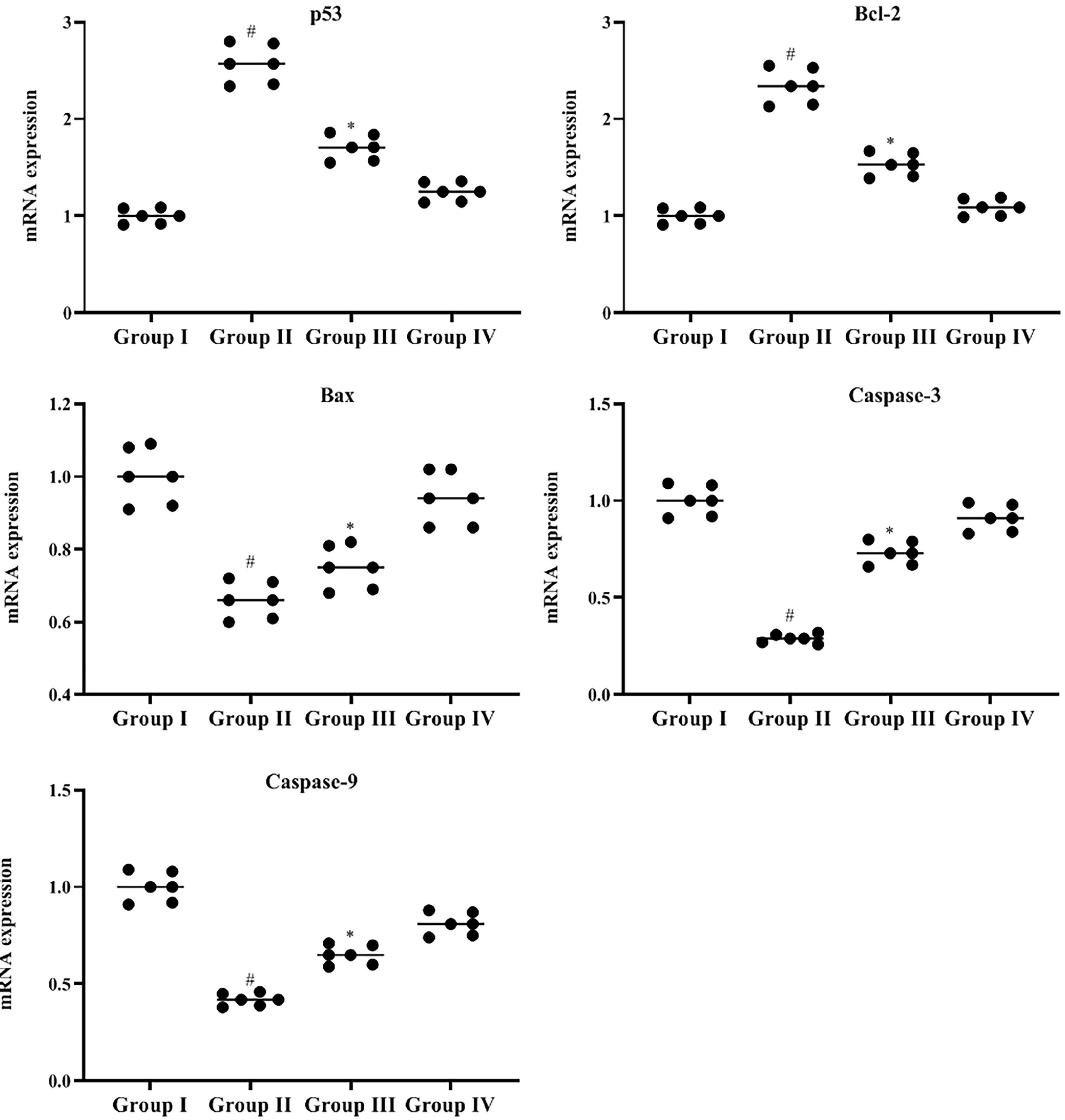
Analysis of apoptotic mRNA expressions in skin region tissue using RT-qPCR
Table 1 and Figure 5 illustrate the analysis of apoptotic markers including p53, Bcl-2, Bax, caspase-3, and caspase-9 in skin tissue samples from both experimental and control groups. In mice from group 2 (tumor-bearing), the levels of Bax, caspase-3 and caspase-9 were significantly reduced compared to both the control (Bax: p > 0.001; caspase-3: p = 0.000; caspase-9: p = 0.000) and BCP-alone groups (Bax: p > 0.006; caspase-3: p > 0.010; caspase-9: p > 0.020). Treatment with BCP in group 3 (DMBA-painted) effectively restored these markers to near-normal levels, significantly differing from group 1 (Bax: p > 0.053; caspase-3: p > 0.037; caspase-9: p > 0.020). There was no notable difference in the expression of these markers between mice treated with BCP (group 3) and those in groups 1 and 4. Statistical significance was confirmed with the Kruskal–Wallis test, with the following results: Bax: H = 18.876, df = 3, p = 0.000; caspase-3: H = 20.629, df = 3, p = 0.000; caspase-9: H = 21.638, df = 3, p = 0.000.
In contrast, p53 and Bcl-2 expressions were notably lower in group 1 than in both the DMBA-only group (p53: p = 0.000; Bcl-2: p = 0.000) and the DMBA + BCP group (p53: p > 0.020; Bcl-2: p > 0.005). When BCP was administered alone, its effect was also significant in reducing these markers compared to group 2 (p53: p > 0.020; Bcl-2: p > 0.007). There were no significant differences in p53 and Bcl-2 levels between the group 3 (BCP) and groups 1 and 4. Kruskal–Wallis analysis revealed strong statistical significance: p53: H = 21.628, df = 3, p = 0.000; Bcl-2: H = 20.228, df = 3, p = 0.000.
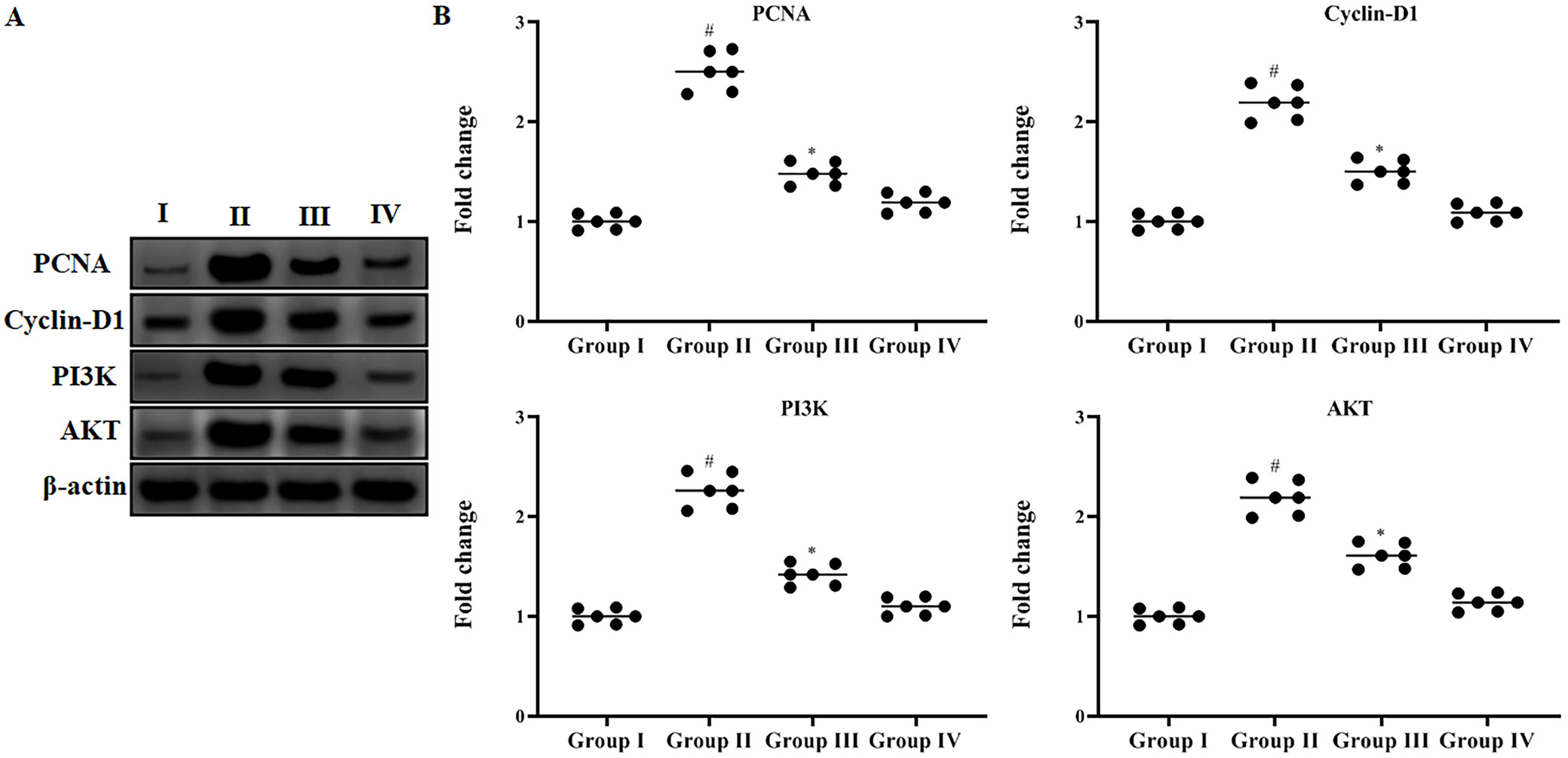
Influence of BCP on P13K/Akt pathway
One of the key factors influencing cell proliferation pathways in skin cancer development is the expression of proteins such as PCNA, cyclin-D1 and PI3K/Akt (Table 1, Figure 6). In the normal control group, the expression of cyclin-D1, PI3K and Akt was significantly lower compared to the DMBA-only group (PCNA: p = 0.000; cyclin-D1: p = 0.000; PI3K: p = 0.000) and the DMBA + BCP group (PCNA: p > 0.025; cyclin-D1: p > 0.053; PI3K: p > 0.037). Mice treated with BCP alone showed expression levels of PCNA, cyclin-D1 and PI3K/Akt similar to normal, with minimal differences compared to the control group (PCNA: p > 0.015; cyclin-D1: p > 0.006; PI3K: p > 0.010). Based on these results, it is suggested that BCP may effectively inhibit tumor growth by modulating these key cell proliferation proteins. Statistical significance was confirmed through Kruskal–Wallis testing, with the following results: PCNA: H = 21.202, df = 3, p = 0.000; cyclin-D1: H = 20.186, df = 3, p = 0.000; PI3K: H = 20.629, df = 3, p = 0.000.
Discussion
Skin cancer is widely regarded as one of the most prevalent diseases affecting humans. According to estimates, cutaneous neoplasms account for approx. 1–2% of all human malignancies in India, based on a 2014 survey.1, 3 The increasing incidence of skin cancer has been associated with changes in lifestyle and environmental factors. The oral cavity, due to its direct exposure to phytochemicals, presents a unique opportunity for studying the chemopreventive potential of diet.28 One of the key constituents of essential oils from culinary and spice plants is BCP, which has garnered attention for its potential anticancer properties.23 Animal models of cancer, particularly those induced by the polycyclic aromatic hydrocarbon DMBA, are commonly used to explore the disease’s development.7 The most effective pharmaceutical strategies against carcinogenesis are thought to involve a combination of both natural and synthetic agents.29 In our study, oral administration of BCP (50 mg/kg/bw) in DMBA-induced rats effectively inhibited tumor growth. No significant differences in cancer incidence or other metrics were observed between the 2 doses tested. Based on these findings, we proceeded with further research using a BCP dose of 50 mg/kg/bw. Similarly, oral supplementation with 10 mg/kg bw of sanggenol L in DMBA-painted hamsters effectively prevented tumor development, enhanced biochemical markers, suppressed inflammatory markers, and promoted apoptosis.30
Dietary antioxidants play a crucial role in regulating the activity of cytochrome P450 enzymes, which are essential for the metabolic breakdown of xenobiotics (including carcinogens), while phase I enzymes initiate the activation of these substances. To assess the chemopreventive potential of test compounds, it is important to understand the levels of both phase I and phase II enzymes.31, 32 In this study, the administration of BCP (50 mg/kg/bw) to DMBA-induced rats effectively reversed the marked alterations in phase I enzyme activities. This suggests that BCP inhibits the metabolic activation of DMBA, thus offering potential protective effects against carcinogenesis. Furthermore, BCP has been found to exert cardioprotective benefits against doxorubicin-induced injury, attributed to its anti-inflammatory and antioxidant properties. Sesquiterpenes, such as BCP, have demonstrated the ability to scavenge reactive oxygen species (ROS), including superoxide anions and hydroxyl radicals. Additionally, BCP significantly reduces the levels of pro-inflammatory cytokines, COX-2 and iNOS. Therefore, BCP is proposed as a potent chain-breaking antioxidant, offering protection against oxidative stress-related damage.33
Glutathione reductase plays a crucial role in maintaining stable GSH levels by facilitating the NADPH-dependent reduction of glutathione disulfide (GSSG). Glutathione, a non-enzymatic tripeptide antioxidant containing a cysteine residue, is vital for protecting cells against ROS and free radicals. In this study, we observed that DMBA-induced liver damage in mice led to reduced levels of GST, GR and GSH. However, administration of BCP (50 mg/kg/bw) effectively restored these levels to near-normal values, highlighting its potent antioxidant activity. The chemopreventive effects of BCP (50 mg/kg/bw) in counteracting DMBA-induced skin carcinogenesis were further confirmed by histopathological analysis. β-caryophyllene has also demonstrated its ability to mitigate toxicity from various harmful agents, including carbon tetrachloride, glutamate, 1-methyl-4-phenylpyridinium, and d-galactose, in both in vitro and in vivo models.34 Additionally, Chang et al. demonstrated that BCP exhibits a stronger cytoprotective effect than the epoxide metabolite, reinforcing its potential therapeutic value.35
Mutant p53 accumulates in cells as a result of stress and DNA damage, disrupting normal cellular processes.36 In fact, the inactivation of wild-type p53 is a hallmark of several cancers, often leading to poor clinical outcomes.37 Alterations in the Bax/Bcl-2 ratio have been reported in various malignancies, including skin cancer. In this study, we observed that DMBA-induced rats exhibited elevated levels of mutant p53 and Bcl-2, coupled with reduced levels of Bax. However, oral administration of BCP (50 mg/kg/bw) effectively induced apoptosis, as evidenced by an increase in Bax expression and a reduction in both mutant p53 and Bcl-2 levels.
Apoptotic caspases are generally classified into 2 categories: caspase-9, which acts as an upstream initiator, and caspase-3, a downstream effector. However, mitochondria can contribute to the activation of both caspases. Notably, significant oxidative stress is often linked to the activation of death receptors or the mitochondrial pathway. Bcl-2, a pro-apoptotic B-cell lymphoma protein, has been shown to mediate mitochondrial-driven apoptosis. Furthermore, when cellular stress becomes severe or prolonged, disruptions in the endoplasmic reticulum (ER) trigger the unfolded protein response (UPR), which can culminate in an apoptotic response.38 In the DMBA-induced skin cancer model, downregulation of caspase-3 and caspase-9 was observed.39 In this study, DMBA-induced mice exhibited a compromised apoptotic response, but oral administration of BCP (50 mg/kg/bw) restored the expression of caspase-3 and -9, effectively preventing the development of skin cancer.
The expression levels of PCNA mRNA, cyclin-D1 protein, and other key markers can serve as valuable indicators for predicting malignant transformation. In the present study, we observed that DMBA-induced mice exhibited elevated levels of PI3K, Akt, PCNA, and cyclin-D1 in their skin tissues.39 Notably, the overexpression of PCNA has been associated with enhanced tumorigenesis and increased aggressiveness of tumors, especially during the progression from hyperplasia to dysplasia, and eventually to skin cancer. Assessing the tissue distribution of cyclin-D1 may prove instrumental in evaluating the extent of cancer progression, particularly in oral tumors. Furthermore, cyclin-D1 plays a critical role as a sensor of extracellular signals, especially during the early to mid-G1 phase of cell division.40 These findings are consistent with our research, which demonstrates that BCP effectively inhibits cell proliferation and division by downregulating the expression of PI3K, Akt, cyclin-D1, and PCNA.
Limitations
Our study suggests that the BCP major role in preventing skin cancer, but the molecular mechanism of inflammatory markers and molecular modeling will be studied in future research. Detailed mechanisms of action still need to be clarified.
Conclusions
Altogether, the study suggested that BCP has garnered considerable pharmacological attention for its potential therapeutic benefits. When administered to mice, BCP effectively controls tumor progression by inducing apoptotic signaling, as evidenced by the upregulation of Bax, Bcl-2 and caspase-3 and -9. Additionally, BCP inhibits cell proliferation by downregulating PCNA, cyclin-D1, p53, and the PI3K/Akt signaling pathway. These findings highlight the potential of BCP and similar natural compounds as promising chemopreventive agents, warranting further pharmacological and pharmaceutical studies to fully explore their therapeutic applications.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.13836654. The package includes the following files:
Supplementary Fig. 1. Results of Kruskal–Wallis test as presented in Figure 1.
Supplementary Fig. 2. Results of Kruskal–Wallis test as presented in Figure 2.
Supplementary Fig. 3. Results of Kruskal–Wallis test as presented in Figure 5.
Supplementary Fig. 4. Results of Kruskal–Wallis test as presented in Figure 6.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.