Abstract
Background. The search for early and minimally invasive diagnostic approaches to pancreatic cancer (PC) remains an important issue. One of the most promising directions is to find a sensitive key in the metabolic changes during widespread causes of PC, i.e., chronic pancreatitis (CP) and diabetes mellitus (DM).
Objectives. The main objective of this study was to analyze the peptide pools in the blood plasma and pancreas of rats with modeling of CP and/or without type 1 DM in association with pancreas histopathological grading features.
Materials and methods. The study was conducted on white non-linear male rats, divided into 3 groups: 1st group: control, 2nd group: rats with cerulein-stimulated CP, and 3rd group: rats with CP and streptozotocin-inducible type 1 DM. Total protein and peptide content were determined in the pancreas and blood plasma. The peptide pools were fractionated using size-exclusion chromatography.
Results. Rats with CP showed a high degree of fibrosis in the pancreas and grade 1 ductal pancreatic intraepithelial neoplasia (PanIN), associated with decreased total peptides in the pancreas. In rats with CP and DM, 2nd and 3rd grade PanIN with pronounced acinar metaplasia was observed in association with decreasing total pancreatic protein and peptide pools. While there was a decrease in total protein and an increase in total peptide in blood serum, the changes were more pronounced in rats with CP and DM. A study revealed both qualitative and quantitative differences in the distribution of peptide pools in 2 groups with pathologies. Qualitatively, plasma samples from pathological groups exhibited an increased number of peaks. Quantitatively, there was a higher proportion of peptides with molecular weights exceeding 700 Da observed in both plasma and pancreas.
Conclusions. The analysis of peptide pools obtained from plasma and PanIN development demonstrated that the peptide pool can serve as an early and complementary indicator of PC emergence.
Key words: adenocarcinoma, fibrosis, pancreatic neoplasms, metaplasia, metabolome
Background
Pancreatic cancer (PC) is a very aggressive disease, with rising incidence and poor clinical outcomes, despite recent innovations in diagnosis and treatment.1 Pancreatic cancer is one of the leading causes of cancer death worldwide, with an overall 5-year survival rate of less than 6–7%.2 First of all, this is due to the difficulty of PC diagnosis in the early stages.3 The course of the disease in most patients is asymptomatic before the development of complications and decompensation with the appearance of distant metastases.4 Therefore, early detection or suspicion of the development of PC is of great importance for the treatment of patients.5 Chronic pancreatitis (CP),6 diabetes mellitus (DM),7 obesity, smoking, alcohol, and cancer in the anamnesis are all known risk factors for the development of PC.8 Coronavirus disease 2019 (COVID-19) is a global health threat, whose causative agent, severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2), is an enveloped positive single-stranded RNA virus, responsible for a pulmonary and extrapulmonary infection, by simultaneously involving multiple organs, from the heart to the pancreas, as highlighted by recent histopathological evidence. Furthermore, during the SARS-COV-2 pandemic, healthcare systems were impacted by the inflow of infected patients, with significant consequences on the care trajectories of cancer patients worldwide.9
The latest data from the GLOBOCAN 2018 program, conducted by the International Agency for Research on Cancer (IARC), indicates an increase in the number of diagnosed patients with PC to 458,918 and an increase in the number of deaths due to PC to 432,242 during the year.10 According to GLOBOCAN 2020, the number of newly diagnosed PC patients continues to grow and is 495,773, and the number of deaths is 466,003.11, 12
In Ukraine in 2018, 6,246 new cases of PC were detected. At the same time, 5,923 people died from this pathology caused by complications of PC. This indicates a negative dynamic, because in 2012, 4,728 people of both sexes were registered and diagnosed with PC. In addition, 4,168 deaths caused by PC complications were registered.13 The findings corroborate the progression of this pathology, highlighting challenges in its diagnosis and therapeutic management.
Given that both diabetes mellitus (DM) and chronic pancreatitis (CP) are recognized risk factors for pancreatic ductal adenocarcinoma (PDAC), their co-presence may significantly increase the risk and raise increased concerns about disease progression.14 Today, pancreatic intraepithelial neoplasia (PanIN) and intraductal papillary mucinous neoplasm (IPMN) are the most necessary and prognostically important in suspected PC in patients with pancreatitis in the anamnesis.15, 16 Pancreatic intraepithelial neoplasia is a noninvasive proliferation of the pancreatic epithelium, which, depending on the degree of its progression, is graded from PanIN-1 to PanIN-3, which in turn reflects the intensity of architectural and cyto-nuclear abnormalities.17, 18 Several authors have analyzed the stages of PanIN progression to carcinoma in situ; however, at this stage, it is still impossible to accurately predict the percentage of malignancy and the development of adenocarcinoma.19, 20 Currently, it is difficult to detect PanIN and its degree of progression using minimally invasive methods.21, 22 Considering the above, the search for additional markers or the improvement of existing approaches for the diagnosis of oncological and other diseases is crucial.23 The tissue peptide pool consists of a set of molecules, in particular growth factors, hormones, cytokines, and molecules that are formed as a result of protein processing.24 Consequently, peptide pools reflect the metabolic activity of an organ.25 As the composition and amount of individual peptides in pools are not static values, but change dynamically depending on the physiological state or the development of a pathological process, the analysis of the tissue peptide pool can be an additional tool for assessing the severity of disease.26
Therefore, we consider the experimental investigation of this topic to be highly relevant. It necessitates a comprehensive approach that integrates diverse methodologies, including molecular analysis. Specifically, this involves examining variations in the peptide pool and comparing their composition across different stages of complication development in these pathologies.
Objectives
The main objective of this study was to analyze the formation and accumulation of the peptide pools in blood serum and pancreas of rats with modeling of CP and/or without type 1 DM in association with pancreas histopathological grading features.
Materials and methods
Study design
Experiments were conducted on male white Wistar rats weighing 200 ±10 g. When working with laboratory animals, we followed generally accepted international recommendations regarding the conduct of medical and biological research using animals by the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, France, 1986). The work with animals has been regulated by the Rules for the Conduct of Experimental Work with Laboratory Animals, which have been approved by the Bioethics Committee of the Educational and Scientific Centre “Institute of Biology and Medicine” of Taras Shevchenko National University of Kyiv, Ukraine (protocol registration No. 4, issued May 25, 2020).
The following conditions were maintained in the room for keeping animals: temperature 20–24°C, humidity 43 ±2%, 12-h/12-h day/night cycle. The animals were fed standard chow (VІТА, Obukhiv, Ukraine). Rats had free access to water and food.
The CP was induced by intraperitoneal (ip.) injection of cerulein (Sigma-Aldrich GmbH, Steinheim, Germany) diluted in physiological solution (5 μg × kg–1 body weight), 5 times a day with an interval of 1 h.27 Control rats received equal volumes of 0.9% NaCl administered ip. Injections were carried out for 5 consecutive days. There were 25 injections in total. After the last day of cerulein injection, the rats were kept under standard conditions, with free access to water, for the following 9 days. The development of pancreatitis was confirmed by a high level of amylase in the blood serum, as well as by the study of pathohistological changes in the parenchyma of the pancreas of experimental rats, which confirmed the above. On the 14th day from the start of the experiment, half of the animals from the CP group were randomly selected and used to model DM. Diabetes mellitus was induced in rats fasted for 16 h by a single ip. injection of streptozotocin (Sigma-Aldrich, St. Louis, USA) at a dose of 65 mg × kg–1 of body weight, dissolved in 0.5 mL of freshly prepared 0.01 M citrate buffer, pH 4.5. Other animals in the CP group and all control rats received an equal amount of placebo. The validity of DM was confirmed by the high blood glucose concentration (above 15 mM×l–1) and a high level of glycosylated hemoglobin. Blood glucose concentration was determined using a glucometer Hlyukofot II (Norma, Kyiv, Ukraine) according to the manufacturer’s instructions. The glycosylated hemoglobin kit (Spectrum, El Ubour, Egypt) was used for the determination of glycosylated hemoglobin.
Finally, there were 3 experimental groups: 1) control (n = 10); 2) CP (n = 20); and 3) CP + DM (n = 20). On the last day of the experiment (44th day), the animals were killed by decapitation, and then blood samples and pancreatic tissue were collected.
Histopathological analysis
Histopathological examination was performed to characterize the morphology and functional status of the pancreas. Pancreatic fragments were fixed in 4% neutral buffered paraformaldehyde and embedded into the paraffin according to standard procedures. Slides were stained with Bemer’s hematoxylin and eosin (H&E). Even though there are other methods of staining pancreatic sections, mainly for fibrosis, Bemer’s method with H&E remains widespread, as evidenced in modern literature.28, 29
All microphotographs were obtained using a light microscope Axio Imager A2 (Carl Zeiss Microscopy GmbH, Jena, Germany) with a ×10, ×20 and ×40 objective lens. Microphotographs were taken using the Axiocam 105 color (Carl Zeiss Microscopy GmbH) digital camera and the Zen2 (blue edition) software (Carl Zeiss Microscopy GmbH).
Obtaining peptide pools
The peptide pool was prepared according to the generally accepted methods.30 Blood plasma or tissue homogenate (10% homogenate in 50 mM Tris-HCl buffer (pH 7.4) containing 130 mM NaCl) was mixed with 1.2 M HClO4 at a 1:1 (v/v) ratio to precipitate the proteins. After centrifugation (10,000×g, 20 min, +4°C), the supernatant was neutralized with 5 M potassium hydroxide (KOH) to pH 7.0, and the samples were centrifuged again. At the final stage, the oligopeptides were precipitated with ethyl alcohol at a final concentration of 67% and the supernatant was lyophilized. The peptide concentration was determined at 210 nm after dissolving of lyophilized samples in 50 mM Tris-HCl buffer (pH 7.4) containing 130 mM NaCl. Dipeptide N-carboxy-glycyl-glycine was used to construct the calibration graph. The protein concentration was determined according to the method described by Bradford,31 using crystalline bovine serum albumin as a standard protein, and the absorbance was measured at 595 nm with a Smart SpecTMPlus spectrophotometer (BioRad Laboratories Inc., Hercules, USA).
Fractionation of peptide pools by size-exclusion chromatography
Size exclusion chromatography on Sephadex G 15 (Bio-Rad Laboratories) was used to fractionate the peptide pool.32 To estimate the molecular weight of the peptides, the column was precalibrated with substances of known molecular weight – lysozyme (14.3 kDa); insulin (5.7 kDa); vitamin B12 (1.35 kDa).
Statistical analyses
The distribution of the data was assessed using the Shapiro–Wilk W-test for normality and the test for homogeneity of variance (Supplementary Table 1). Nonparametric tests (Kruskal–Wallis as overall test and post hoc Dunn’s test with Bonferroni correction) were used to assess the significance of the observed changes. A statistically significant difference was evaluated at p < 0.05 using Origin 8 Pro (OriginLab, Northampton, USA). Histograms were created using Microsoft Excel 2010 software (Microsoft Corp., Redmond, USA) and Origin 8 Pro. The obtained results are presented as the median, 25% and 75% quartile and minimum and maximum.
Results
Histopathological analysis
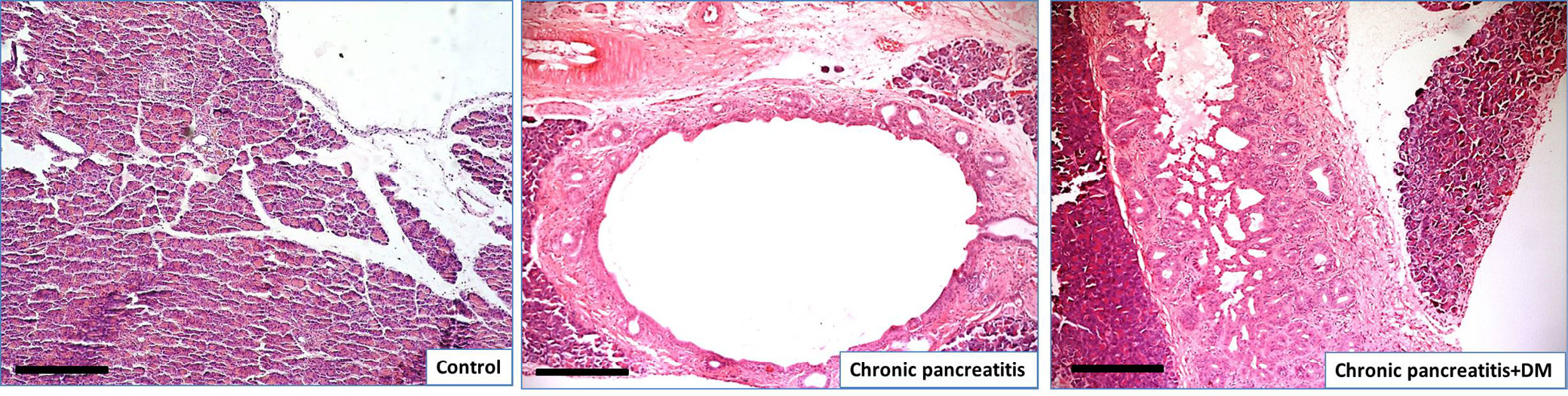
Histopathological observation included both ductal and acinar parts of the pancreas. Changes in the ductal part were classified according to the PanIN grading system.33 In the control group, the ductal part (Figure 1) was characterized by the typical structure of a simple cuboidal epithelium. In the CP group (Figure 1), most ducts were surrounded by developed connective tissue stromal elements, which in some samples could be classified as an early stage of fibrosis (also around the vascular part). The presence of immune cell infiltration (the most common cells are neutrophils and macrophages) in the expanded stromal part of the round ducts also indicated the intervention of a low degree of chronic inflammation. Ductal cells in the CP group (Figure 2) in all samples changed their shape from cuboidal to prismatic; in some of them, active proliferation with a dysplastic process was observed, which led to the appearance of glandular epithelium in the thickened wall of the duct. These changes stimulated the epithelium for invagination. This type of pacreas lesion manifestation is classified as a PanIN-1A - PanIN-1B according to the established grading system.
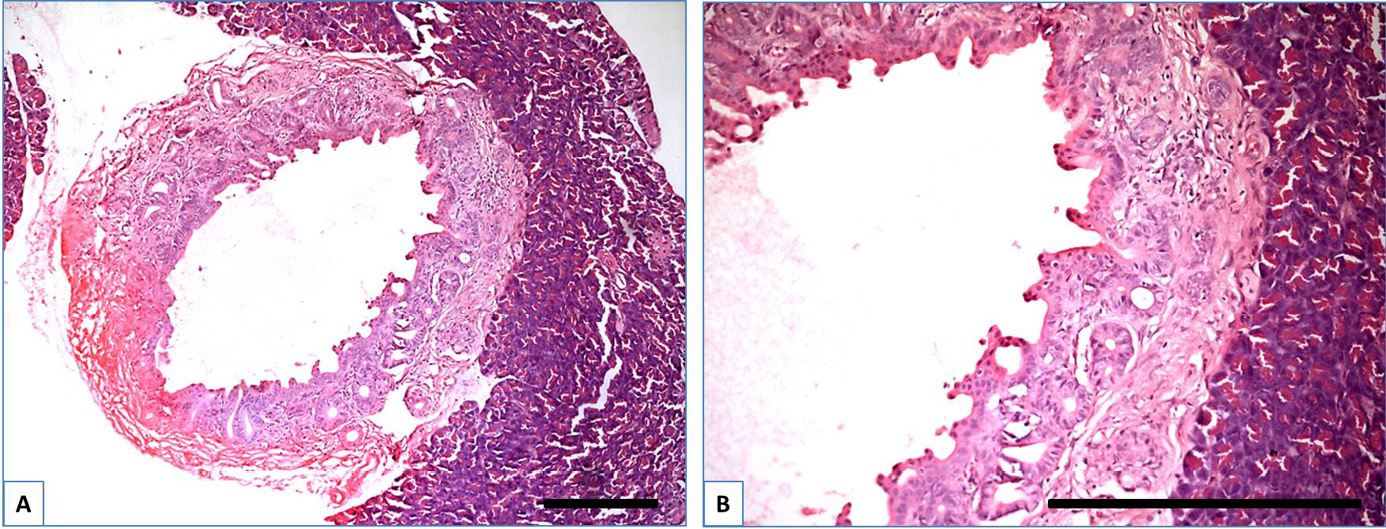
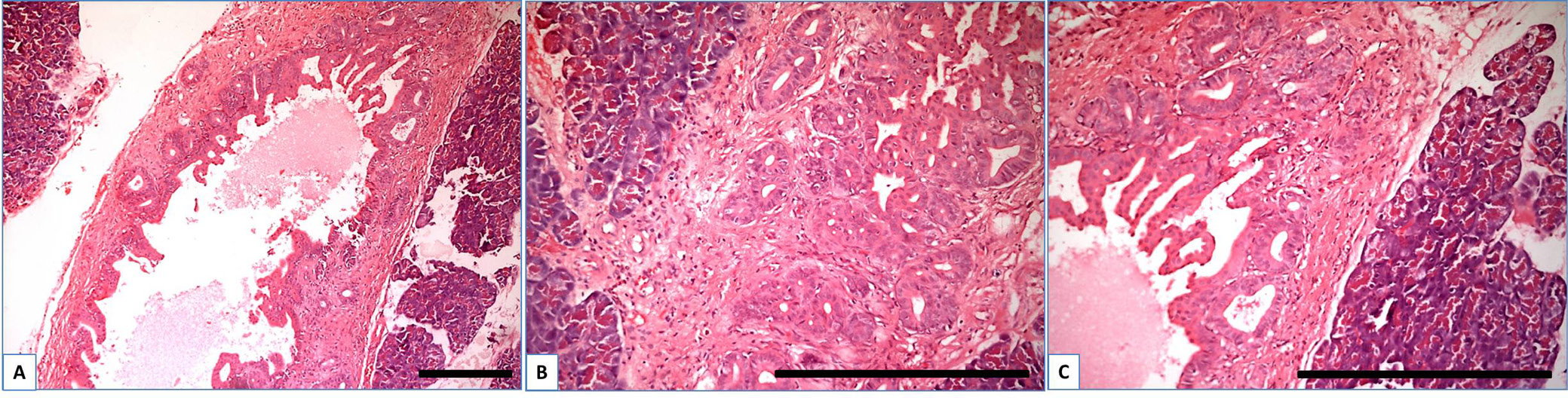
In the CP + DM group, in some samples with pronounced dysplasia, the ductal part exhibited the previously described features (Figure 1). The ductal epithelium was frequently transformed into a pseudostratified type (Figure 3) and pseudopapillary structures were formed within the wall (Figure 3B). Additionally, villous-like structures were observed in the duct lumen (Figure 3C), indicating a high-grade PanIN-2 and PanIN-3.
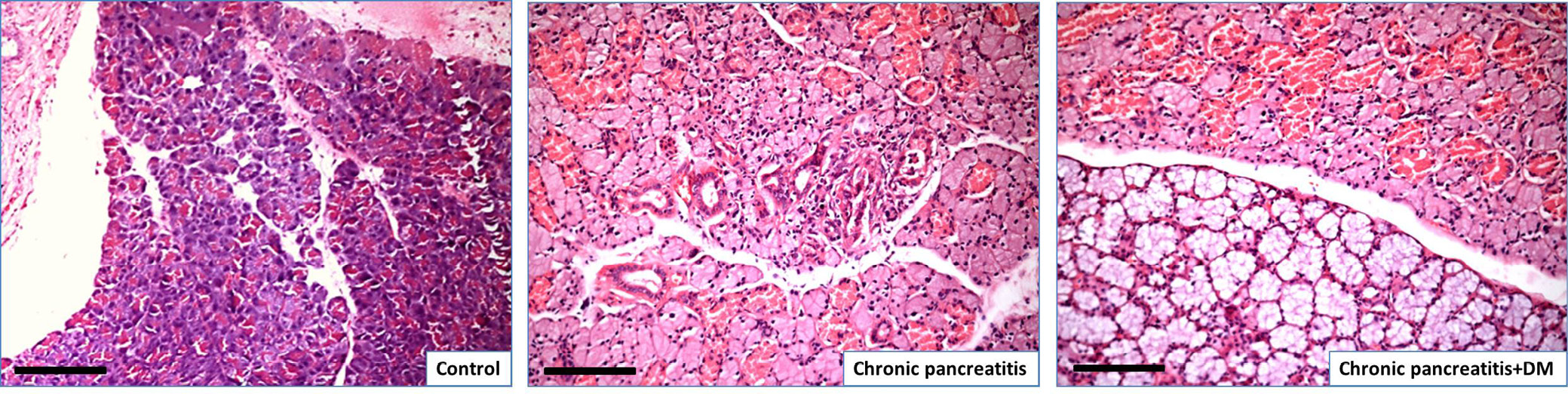
The changes observed in the ductal part were accompanied by corresponding changes in the exocrine acinar part (Figure 4). The CP group (Figure 4B) exhibited significant morphological heterogeneity in the acini, including loss of polarization in some acini and a notable difference between the eosinophilic apical part and the basophilic basal part. Conversely, the CP + DM group demonstrated acinar-ductal metaplasia through intermediate mucinous cells (Figure 4C).
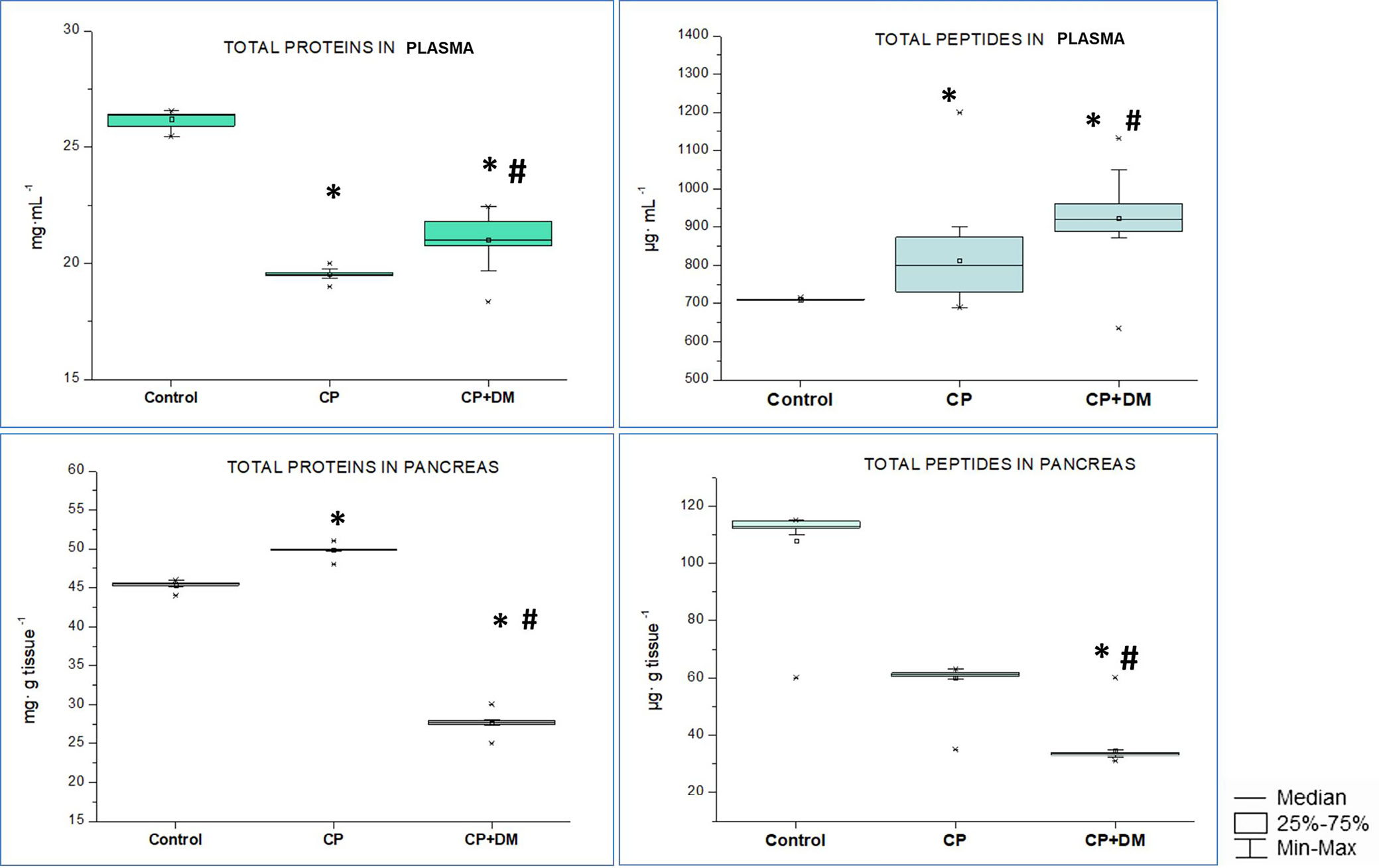
The total protein and peptide concentration in plasma and pancreas
Biochemical analysis showed a significant decrease in total plasma protein of 26% and 20% in the CP and CP + DM groups, respectively, compared to the control level (Figure 5). In the pancreas, the level of total proteins exhibited a more pronounced decrease under conditions of CP in the context of diabetes (by 40% compared to the control and by 45% compared to the CP group). In addition, the CP + DM group had a 30% increase in total peptides in blood plasma compared to the control group (any significant changes were not fixed in the CP group). The alterations in total protein and peptide levels in blood plasma observed in the CP + DM group may serve as an early indicator of proteolysis in metabolic processes. The total peptide level in the pancreas was reduced by 46% and 70% in the CP and CP + DM groups, respectively, compared to the control level. In addition, the CP + DM group showed a significant 46% decrease in this parameter compared to CP.
Fractionation of peptide pools using size-exclusion chromatography
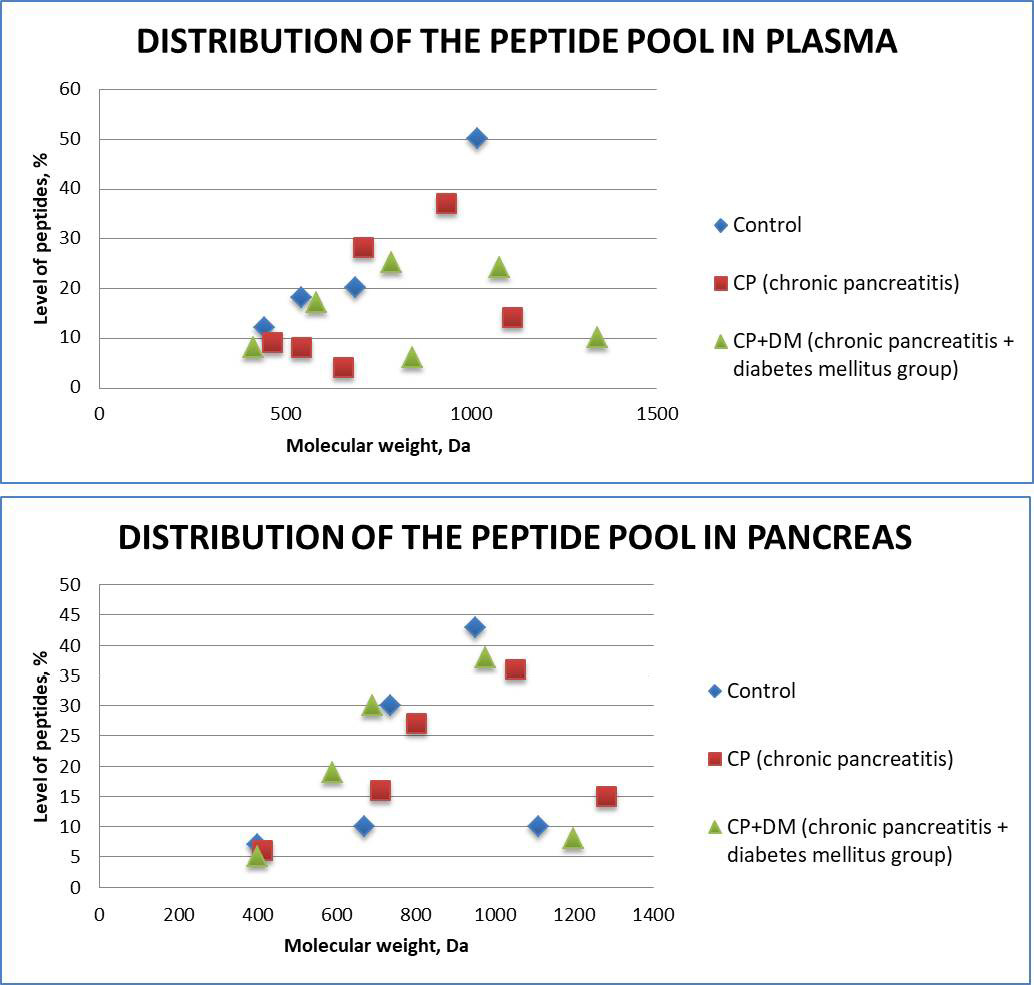
Chromatographic analysis of the peptide pools of patients in the CP and CP + DM groups revealed deviations in the quantitative and qualitative composition of peptides when compared to the control group (Figure 6). The appearance of peptides in a wider range of molecular weights, as well as changes in the concentration of some peptides in the blood plasma of patients were found. The peptide pool derived from the blood plasma of the study participants in the control group consisted of 4 main peptide fractions with a molecular weight of 1,017 Da, 689 Da, 545 Da, and 444 Da. At the same time, the number of peptide fractions in both pathologies increased to 6 main peptide fractions.
The differences were noticeable in blood serum more prominently than in the pancreas, and for peptide pools with a molecular weight greater than 700 Da. Peptide pool peaks were detected in the blood plasma of CP and CP + DM, but not in the control samples.
Thus, it was shown that the degree of PanIN 1A–PanIN-1B histopathologic changes was accompanied by decreased total plasma proteins and total pancreatic peptides, while total pancreatic proteins and total plasma peptides (with increased part of peptides 1,100 Da) increased in CP group. In the CP + DM group, we observed another pattern of changes. Specifically, high-grade PanIN-2 and PanIN-3 histopathological changes were followed by decreased total proteins in plasma and pancreas, and total peptides in the pancreas. Conversely, only total peptides in plasma (with elevated levels of peptides increased from 1,100 Da to 1,300 Da. The total amount of proteins in the pancreas and the peak distribution of the peptides in the serum changed differently in the CP and CP + GM groups.
Discussion
It is now widely accepted that the structural and functional disorders of the pancreas that result in the development of pancreatitis are caused by a cascade of molecular events initiated by the uncontrolled activation of pancreatic enzymes.34 Even though this factor is actively involved in the initial stages of the disease, the pathogenesis of CP is also closely associated with changes in the functioning of the proteolytic link,35 which is a prerequisite for the development of several accompanying manifestations of pancreatitis. These manifestations are associated with clinical and pathophysiological characteristics.36 One of the complications is the development of fibrosis and the subsequent exhaustion of the exocrine and endocrine functions of the pancreas, which leads to the further development of diabetes, as well as maldigestion and malabsorption syndrome in patients.37, 38
This study was the first to report the relationship between pancreatic histopathological features and peptide pool changes in a rat model of CP and CP combined with DM. The full mechanisms underlying and ensuring this connection remain unclear.39 Our previous study showed a significant increase in the content of interleukin 6 (IL-6), tumor necrosis factor alpha (TNF-α) and matrix metalloproteinase (MMP)-2 and -9 in blood serum, liver and pancreas of animals with CP separately and under DM conditions.40 We determined the levels of these factors in animals with CP and CP + DM because the main enzymes involved in maintaining the metabolism of extracellular matrix (ECM) proteins are MMPs, which contribute to the appearance of changes in peptide levels.41 Therefore, the difference in the distribution of peptides on criteria such as molecular weight and amounts of individual peptide fractions observed in the control and CP and CP + DM groups may represent the disorders of proteolytic homeostasis and deregulation of protease activity under the development of pathology. It is possible that these peptides may be among the first features involved in the development of different grades of dysplasia (including PanIN).42 On the other hand, changes in the peptide pools can be a signal of the severity of the disease, and in our study, a sign of pancreatic lesions. According to modern concepts, tissue peptide pools are quite stable under physiological conditions, based on criteria such as qualitative and quantitative composition, so they can be considered an important indicator characterizing the functional state of organs. One of the explanations for the significant decrease in the level of peptides in the pancreas in patients with CP and CP + DM is their release into the bloodstream due to cell damage.43 This assumption is partly confirmed by the increase in the concentration of blood plasma peptides. Changes in peptide concentrations were more pronounced in the CP + DM group, which may be a direct consequence of more severe cell metabolism disorders in patients suffering from both pathologies simultaneously. Additionally, increased plasma peptide levels may in part result from the proteolytic breakdown of plasma proteins mediated by enzymes that appear in the plasma as a result of disruption of proteolytic homeostasis. Our previous findings highlighted changes in proteolytic balance, namely a significant increase in the level of MMPs, particularly MMP-2 and MMP-9, in the serum of rats with CP.40 Analysis of the peptide pools using chromatography confirmed the hypothesis that the accumulation of peptides of different molecular weights in the blood plasma of patients with CP and CP + DM may be associated with protein degradation due to the activation of proteolytic reactions.
The pool of plasma peptides has the potential to serve as a valuable source of diagnostic information, as it represents a comprehensive record of cellular and extracellular enzymatic events characteristic of the pathological process. Consequently, the pool of blood plasma peptides can be considered an integral indicator reflecting the general metabolic state of the body.44 To some extent, an increase in plasma peptide concentration may be a consequence of a decrease in plasma detoxification potential. One mechanism for the elimination of low-molecular-weight substances involves their absorption into abundant blood proteins such as albumin and further excretion through the liver and kidneys. Therefore, a decrease in protein concentration in the blood of patients in both the CP and the CP + DM groups may be an additional factor leading to the accumulation of peptides. Moreover, given the ability of the glomeruli to effectively remove low molecular weight substances, thereby reducing the concentration of potentially toxic molecules, the accumulation of peptides that we detected in the plasma of patients may be an indirect evidence of renal dysfunction, especially in patients in the CP + DM group.
It should be noted that a violation of the composition of peptide pools, as well as an increase in the concentration of some peptides, not only indicates the severity of the pathological process, but may be an additional factor causing PC progression and provoking the development of a number of complications. It can be assumed that pathological peptide pools may contain molecules structurally similar to biologically active peptides, or molecules with activity atypical for “physiological” peptides. Such molecules can alter cellular metabolism by binding to the active and/or regulatory sites of enzymes. In addition, pathological peptides can occupy cellular receptors, thereby disrupting intracellular signaling pathways and affecting intercellular communication.
As shown above, in the pancreas of rats of the CP + DM group, we observed the development of changes (ductal dysplasia, pronounced acinar metaplasia, formation of glands in the fibrotic tissue of the duct, proliferation of the epithelium of glands with severe dysplasia, appearance of papillary and cribriform structures) corresponding to high and moderate ductal dysplasia (PanIN-2 and PanIN-3). These changes are direct predictors of pancreas adenocarcinoma. The impairment of PanIN represents a pathway to the development of PC and directly to ductal adenocarcinoma. This conclusion has been corroborated in several studies.45, 46
Diabetes mellitus is another common comorbidity known to contribute to changes in the serum metabolome.47 In ductal adenocarcinoma, DM may promote tumor progression through changes in the transcriptome and metabolome.48 Its close association with chronic inflammation adds an additional risk for complicating and advancing PC.49, 50
Limitations
The design of the study lacks a group of animals with only 1 model of diabetes. The distribution of the peptide pool is a nonspecific parameter. In any case, it may vary with another type of pathology development. Therefore, an additional specific test for pathology identity should always be performed. The cerulein-induced model of pancreatitis demonstrated significant regenerative capacity in the rat organism. In some cases, only mild morphological alterations were observed in the pancreas. However, in the CP + DM group, lesions ranged from PanIN-1 to PanIN-3, indicating a wide spectrum of pancreatic intraepithelial neoplasia. Also, the exact direct mechanism of the reciprocal relationship between the histophysiological changes of the pancreas and its peptide pool remains unclear.
Conclusions
Blood collection for plasma peptide pool analysis, in comparison to pancreatic biopsy, provides a less invasive alternative for diagnostic evaluation. In the case of cerulein-induced CP, alterations in the ductal region of the pancreas classified as PanIN-1A to PanIN-1B were observed, accompanied by increased total protein in the pancreas and low prevalence of peptide pool molecular weight of 450–770 Da in plasma compared to controls. In the presence of comorbidities such as DM (streptozotocin-induced) changes in the ductal part of the pancreas were classified as PanIN-2 and PanIN-3 grade, accompanied by a decrease in total pancreatic protein and the appearance of 2 new fractions of the high molecular weight peptide pool (more than 1,000 Da) in plasma compared to controls. Overall, chronic pancreatitis (CP) with diabetes mellitus (DM) is marked by significant histopathological alterations in the pancreas, characterized by pronounced neoplastic changes. These changes are accompanied by a reduction in total pancreatic protein levels and an increase in peptide fractions of 1,100 Da and 1,300 Da compared to CP lesions without diabetes mellitus. These results suggest that combined plasma and pancreatic peptide pool analysis has great diagnostic potential for assessing the severity of PC. It is a fundamental promise for finding possible associations, predicting the progression and early detection of the threat of PC development in patients with a difficult medical history, namely: surgery for CP, obesity, diabetes, coronavirus disease, alcohol consumption, and smoking.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.13630059. The package includes the following files:
Supplementary Table 1. Data of Shapiro–Wilk (W) normality test and homogeneity of variance test (Levene’s F test).
Supplementary Table 2. Kruskal–Wallis as the overall test (H-values) and post hoc Dunn’s test with Bonferroni correction. The corrected α using the Bonferroni correction method is 0.017.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.