Abstract
Background. We still know little about the effective pharmacological treatment of heart failure (HF) associated with the Fontan circulation. One of the new options may be sodium glucose cotransporter-2 inhibitors (SGLT2i), which have been proven effective in classic forms of left ventricular HF.
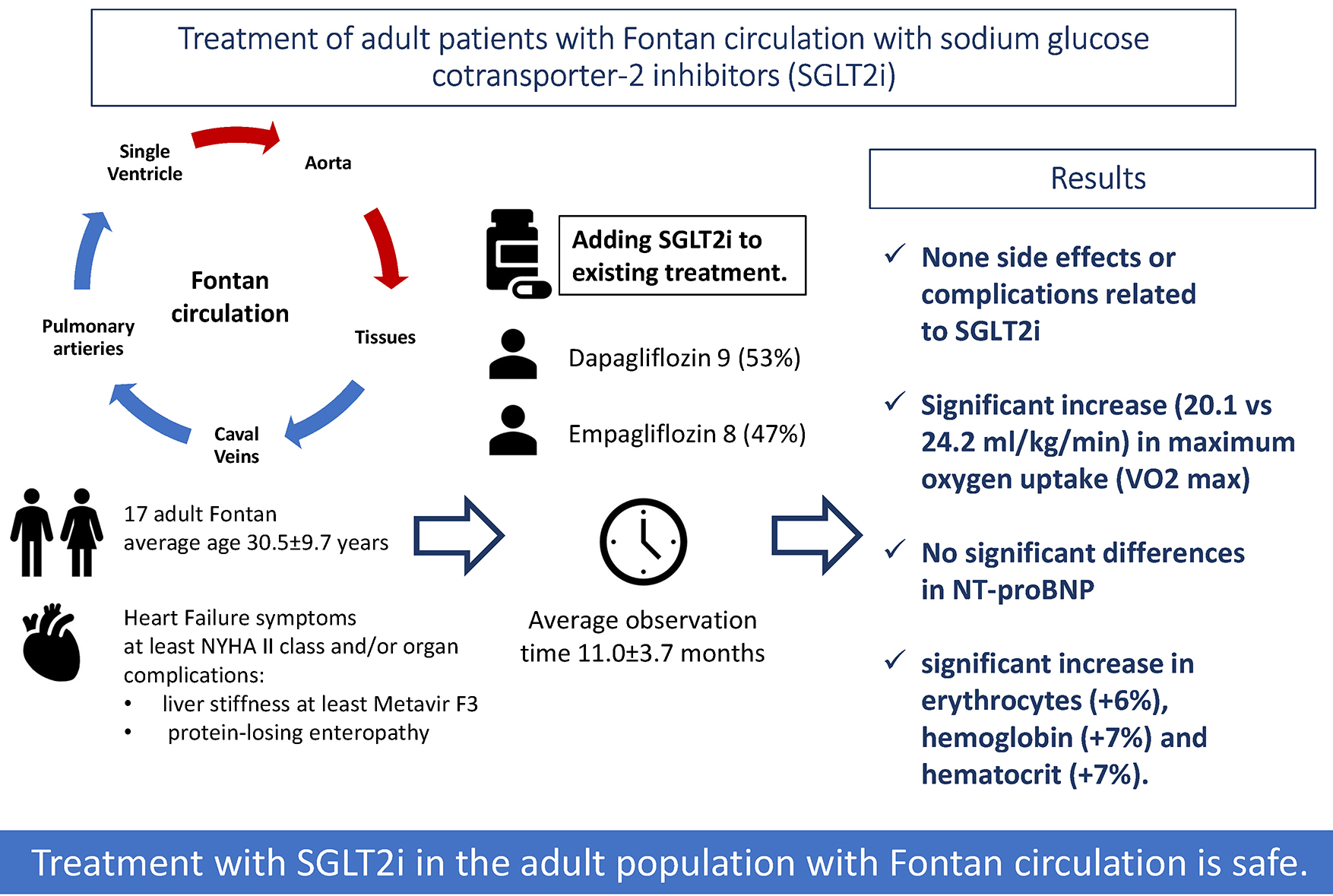
Objectives. To evaluate the effect and safety of SGLT2i inclusion in adults with Fontan circulation. To this end, we conducted observation and complex diagnostics of adult Fontan patients in whom we started treatment with flozins.
Materials and methods. The study population consisted of 17 adult Fontan patients with average age 30.5 (9.7) years, 59% in II New York Heart Association (NYHA) class, among whom 53% received dapagliflozin and rest empagliflozin.
Results. The average observation time was 11.0 (3.7) months. None of the patients have reported side effects or complications related to treatment. We observed a significant increase (20.1 mL/kg/min vs 24.2 mL/kg/min, p = 0.008) in the median of maximum oxygen uptake (VO2 max) among participants (9) who completed at least 2 reliable cardiopulmonary exercise tests. We did not notice any significant differences in N-terminal prohormone of brain natriuretic peptide concentration (641.35 (923.7) vs 741.47 (1,139.02), p = 0.12) after the inclusion. Interestingly, we observed a significant increase in erythrocytes (+6%, p = 0.003), hemoglobin (+7%, p = 0.03) and hematocrit (+7%, p = 0.02).
Conclusions. To the best of our knowledge, this is the first study to demonstrate that the implementation of SGLT2i may have a positive effect on exercise capacity among adults with Fontan circulation. Our experience confirms the high safety of using these drugs in Fontan adults.
Key words: heart failure, congenital heart disease, Fontan circulation, flozins
Background
The increasing number of adult patients with Fontan circulation (FC) is one of the greatest successes and at the same time challenges of modern cardiology of congenital heart disease (CHD). Recent studies indicate that in 2020, among 11 analyzed countries, the number of people after Fontan procedure was 47,881, of which 55% were adults, and in 2030 it will increase to 59,777 (64% adults).1
Unfortunately, the number of multi-organ complications increases during follow-up, with heart failure (HF) being one of the most significant problems. In a single-center cohort study, clinical symptoms of HF were observed in 40% of FC patients over an average follow-up of 15.7 years.2 Fontan circulation pathophysiology is not only complex but also poorly understood and includes, i.a., systolic and/or diastolic dysfunction of systemic ventricle, fibrosis and increased pulmonary resistance. The presence of HF symptoms in Fontan patients is associated with a very poor prognosis and reduced quality of life, and is the leading cause of death.3
Despite significant advances in care, there is still a lack of effective and guideline-directed options for managing FC failure, especially pharmacological ones.3 Previous attempts have largely been adapted from the recommendations dedicated strictly to the classic forms of HF,4, 5 but these have generally yielded unsatisfactory results. Nevertheless, there are also promising reports of attempts to introduce new drugs, such as sacubitril/valsartan (ARNI) or sodium glucose cotransporter-2 inhibitors (SGLT2i), into the population of CHD. Fusco et al. in their study showed that the implementation of ARNI was associated with significant clinical improvement, including New York Heart Association (NYHA) class change and/or increased 6-minute walking distance after 1 year of follow-up among 50 adult patients with systemic right ventricle.6 There are also the first reports on the use of SGLT2i (commonly flozins) in the population with CHD. Despite the fact that SGLT2i are one of the newest additions to medical therapy in HF, their position is already well-established in HF guidelines and their update.4, 5 According to these documents, SGLT2i (dapagliflozin or empagliflozin) are recommended for patients with HF with preserved ejection fraction, mildly reduced ejection fraction and with reduced ejection fraction to decrease the risk of HF hospitalization and/or death.4, 5 These recommendations are based on the reduction of the primary composite endpoint used, e.g., in DAPA-HF, EMPEROR-Reduced, EMPEROR-Preserved and DELIVER trials.7, 8, 9, 10 The above “universality” is even more important because patients with a single-ventricle heart after the Fontan procedure present a very heterogeneous picture of HF, with diastolic dysfunction dominating.1, 2, 3
Objectives
Although SGLT2i are a cornerstone of HF pharmacotherapy, the literature on the use of flozins in the treatment of FC failure is still very limited.11 This encouraged us to share our experience with the use of SGLT2i in the treatment of adult patients after Fontan surgery. The aim of this study was to evaluate the effect and safety of SGLT2i inclusion in adults with Fontan circulation.
Materials and methods
Study design and participants
The study was prepared in accordance with the appropriate Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist.12 It was conducted from June 2022 to February 2024. The investigation included adult patients with symptoms of Fontan failure, defined as at least NYHA class II and/or chronic organ complications related to FC without any other objective cause (liver stiffness assessed with elastography to at least Metavir F3 score or proteinlosing enteropathy), hospitalized in our center and receiving SGLT2i in addition to their previous treatment. Each participant underwent detailed diagnostics to exclude potentially reversible or treatable causes of Fontan failure, including: tunnel stenosis, the presence of significant vascular anastomoses/malformations, pulmonary artery stenosis, and significant coarctation of the aorta, prior to initiation of SGLT2i. The exclusion criteria were: inability to give informed consent, cancer, pregnancy, current urinary tract infection, or a history of severe urinary tract infection in the last 12 months.
The investigation conforms with the principles outlined in the Declaration of Helsinki. The Ethics Committee at the Jagiellonian University (Cracow, Poland) approved the study (approval No. 118.6120.57.2023), and each participant provided written informed consent.
The following variables were collected from medical records: patient demographics, exact anatomy of CHD, medical therapy, past interventions and clinical complications, and hemodynamic data from the last invasive diagnostics. Before and after the initiation of SGLT2i treatment, each patient underwent a comprehensive diagnostic procedure used in our center for patients with FC.
The study was not blinded. Follow-up examinations were performed according to the clinical indications of the attending physician, but at intervals of at least 12 months.
Each patient was instructed in detail about the mechanism of action of SGLT2i, possible side effects, complications, and ways to reduce the risk of their occurrence. The above information was provided verbally and in writing in the discharge documentation.
Data sources and measurements
Venous blood was drawn from an antecubital vein with minimal stasis after an overnight fast. Laboratory tests such as the N-terminal prohormone of brain natriuretic peptide (NT-proBNP), blood cell count, total protein, creatinine, and aspartate aminotransferase (AST) levels were assayed using routine laboratory techniques.
The exercise tolerance was evaluated by the cardiopulmonary exercise testing (CPET) on a treadmill in the modified Bruce protocol (Reynolds Medical System, ZAN-600; Lode BV, Groningen, the Netherlands). Maximum oxygen uptake (VO2 max) was defined as the highest value at peak workload in mL/kg/min and percentage of predicted value. The ventilatory equivalent for carbon dioxide (VE/VCO2) was defined as the amount of ventilation needed for elimination of a given amount of CO2. The respiratory exchange ratio (RER) was calculated by dividing the VO2 by VCO2. The test was considered reliable if the RER was at least 1.0. However, if the patient was unable to perform 2 reliable tests (initial and follow-up), their data were not included in the analysis of changes in exercise tolerance. Most of these cases involved musculoskeletal problems, both chronic and acute. Less frequently, the inability to perform this test was related to neurological disorders, fear of performing the test and, in only 1 case, poor clinical condition.
The NYHA functional class was estimated by an experienced adult CHD specialist based on the patient’s self-reported symptoms and exercise tolerance. Abdominal ultrasound examination with elastography was conducted on all participants by a highly experienced researcher using Philips iU22 XMatrix Ultrasound System (Koninklijke Philips N.V., Amsterdam, the Netherlands). The transthoracic echocardiographic was performed (Philips CVx EPIQ model; Koninklijke Philips N.V.) by the same echocardiographer according to a uniform scheme. The following parameters were analyzed: ventricle morphology, peak velocity of blood flow through the dominant atrioventricular valve from left ventricular relaxation in early diastole (E) to peak flow velocity in late diastole due to atrial contraction (A), E/A ratio, velocity of annulus tissue of the dominant atrioventricular valve in early diastole (E’), E/E’, annular plane systolic excursion of dominant atrioventricular valve (TAPSE/MAPSE), and, in a similar way, also Doppler tissue imaging-derived annular systolic velocity (S’). In patients with FC, it is difficult to assess the ejection fraction using echocardiography precisely and in accordance with classical terminology. However, various techniques were used to estimate systemic ventricular systolic function (primarily visual assessment by an experienced investigator), trying to replicate the classic ejection fraction (as preserved ≥50%, mildly reduced 41–49% and reduced ≤40%).
Statistical analyses
IBM SPSS Statistics v. 29.0 (IBM Corp., Armonk, USA) software was used for statistical analysis. Values of continuous data were presented as mean ± standard deviation (±SD) or median [interquartile range (IQR)], while the qualitative data were shown as the number and percentages. Continuous variables were first checked for normal distribution using the Shapiro–Wilk test. For repeated-measures analysis, a dedicated two-sided t-test for dependent groups or the Wilcoxon test was used. For all tests, p-value less than 0.05 was considered significant. Subgroup analysis was adjusted with Bonferroni’s method. Study power was estimated at 0.93 for VO2 max.
Results
To date, our study cohort includes 17 patients, whose clinical data are summarized in Table 1. The mean age of the group and time since the Fontan procedure were 30.5 ±9.7 years and 24.8 ±9.8 years, respectively. In a slight majority of cases, the systemic ventricle had the structure of the right ventricle 9 (53%). The average height was 171.2 ±8.9 cm and the average body weight was 62.6 ±9.5 kg. Initially, most patients were in class II of the NYHA classification of HF symptoms (10 patients, 59%). The pharmacological treatment used before and during SGLT2i therapy is presented in Table 2.
Of the total number of participants, 9 (53%) received dapagliflozin, while the remaining participants were administered empagliflozin. The mean time from the start of treatment to the last follow-up visit was 11.0 ±3.7 months. None of the patients reported any treatment-related adverse events or complications.
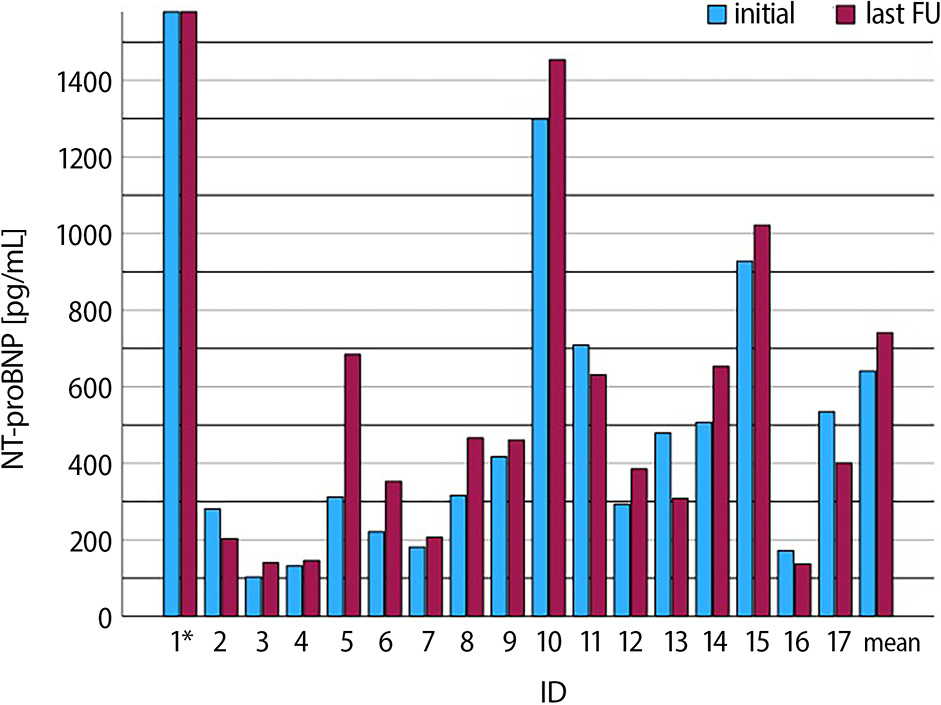
In the analysis of the entire study group, we did not observe any significant differences in NT-proBNP concentration before and after the inclusion of SGLT2i (Table 3, Figure 1). The quantitatively analyzed echocardiographic parameters indirectly indicating systolic function, such as S’ (7.7 ±2.5) vs 7.5 ±2.1 cm/s), p = 0.78) and TAPSE/MAPSE 13.6 ±3.3 vs 14.3 ±5.7 mm, p = 0.64), did not change significantly during the observation. Importantly, the E/A (1.6 [1.1–1.8] vs 1.5 [1.1–1.8], p = 0.55) and E/E’ (8.3 [5.7–10.2] vs 8.2 [5.9–11] ratios, which are parameters of diastolic function, also remained similar. At the same time, we did not note any significant changes in the function of kidneys, liver or other analyzed laboratory tests (Table 3). The clinical condition of almost the entire group generally remained stable during follow-up. The analysis also showed no significant changes in the NYHA class (p = 0.96).
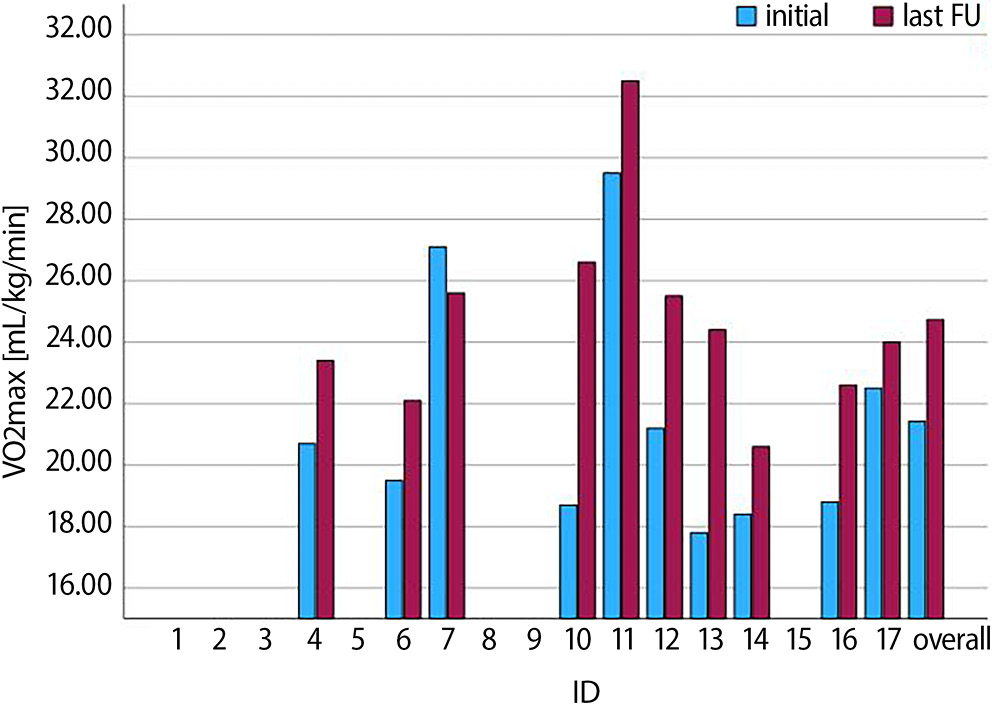
Initial and follow-up ergospirometry was performed in only 9 cases, for the reasons mentioned above. Interestingly, we observed a significant increase (20.1 [18.6–23.7] vs 24.2 [22.5–25.9] mL/kg/min, p = 0.008) in VO2 max among those who underwent the test (Figure 2). However, we did not observe any significant difference (42.4 [37.8–50.8] vs 40.9 [35.6–44.1], p = 0.51) in VE/VCO2. In additional analysis of this subgroup, there was also no significant difference in NT-proBNP levels before and after treatment (452.8 ±353.9 vs 467.4 ±389.4 pg/mL, p = 0.70).
Interestingly, we observed a significant increase in red blood cell count (+6%, p = 0.003), hemoglobin level (+7%, p = 0.03) and hematocrit (+7%, p = 0.02) compared to baseline values in the entire cohort (Table 3). At the same time, the other analyzed erythrocytes parameters as well as the number of leukocytes and platelets did not differ significantly.
Additional subgroup analyzed in terms of systemic ventricular morphology (left vs right) showed no significant differences in initial values of NT-proBNP (479.0 [257–608] vs 181.0 [152–917], p = 0.54) and VO2 max (19.5 [18.1–25.4] vs 20.7 [18.8–24.8], p = 0.66). The subgroups did not differ in the final values of the above parameters (respectively, 385.0 [331– 642] vs 207.0 [142–927], p = 0.74; 24.4 [20.6–29.0] vs 24.0 [23.0–26.1], p = 0.84).
Discussion
This is one of the first reports on the use of SGLT2i in the treatment of adult patients with a single-ventricle heart after the Fontan procedure.11 Our experience confirms the high safety profile of using these drugs. We hope that our efforts to educate patients about the mechanism of action of SGLT2i have contributed to the lack of complications. However, our preliminary study also revealed interesting and important effects of flozins in patients with FC.
First of all, the average VO2 max increased significantly during the observation period. This seems to be an important finding, whereas improvement in functional capacity is a component (besides improvement in clinical status and quality of life) of the 3 major goals of treatment of patients with HF. Our observations are similar to those from a large randomized controlled trial (RCT) in heart failure with reduced ejection fraction (HFrEF) patients, where VO2 max significantly improved in the empagliflozin-treated group compared to placebo-treated patients.13
Like in many other studies, we used ergospirometry to assess the effectiveness of the therapeutic intervention.14, 15, 16, 17, 18, 19 As we mentioned earlier, many of the previous attempts to apply recommendations from classic forms of HF to Fontan population have unfortunately not brought the expected results. Therefore, in trials using enalapril or carvedilol, as typical groups of drugs in HF, no improvement in exercise capacity was observed in the Fontan population.14, 15, 16 The effect of treatment with pulmonary vasodilators on changes in VO2 max among Fontan patients remains unclear and controversial.17 Thus, this encourages further research on SGLT2i, especially considering the recently published positive results of using this drug group in adult patients with systemic right ventricle.18
The 2nd important finding from our study is a significant increase in erythrocytes, hemoglobin and hematocrit during follow-up. These changes are consistent with theories explaining many positive pleiotropic effects of flozins, not only due to reduced plasma volume secondary to increased diuresis.19, 20, 21 The organ primarily responsible for erythropoietin synthesis in adults is the kidney. The most basic hypotheses suggest a beneficial effect of SGLTi on renal hemodynamics (with rejuvenation and protection of erythropoietin-producing cells) and on local hypoxia triggered by increased tubular workload.19, 20, 21 However, the above theories also have limitations, preventing a full explanation of this phenomenon.19 Packer suggests that SGLT2i may increase the role of the liver in erythropoietin regulation. Hepatocytes are the main source of erythropoietin during fetal life, but they retain a latentability to synthesize it also in adulthood. SGLT2i may influence inflammatory and cellular stress pathways, as well aserythropoietin gene transcription, especially in the liver.19 This is particularly interesting in Fontan population, as liver damage is an inherent part of long-term complications.22, 23 However, frequent central cyanosis and the presence of additional vascular collaterals complicate the interpretation of these findings related to the red blood cell system in FC patients.The importance of the above processes in the cardioprotective effect remains unclear. Additionally, other pleiotropic effects associated with SGLT2i that may benefit FC and warrant further investigation.24
In our study, we did not find significant changes in analyzed echocardiographic parameters. Patients with a single-ventricular heart after Fontan surgery constitute a very heterogeneous group of complex CHD. Echocardiographic examinations are very demanding, and the high risk of measurement error makes analysis of these parameters particularly challenging. However, there are reports in patients with normal cardiac anatomy where empagliflozin significantly improved left ventricular filling pressure.25, 26
In our study, we did not observe a significant decrease NT-proBNP levels after treatment, as in study by Neijenhuis et al.18 It should be emphasized that our study group reflects a real-life population of Fontan patients. Patients with various complications related to Fontan physiology and comorbidities were taken into consideration. For example, the impact of protein-losing enteropathy on the SGLT2i effect is unknown.2 It was very common in our study population and may significantly interfere with absorption and metabolic processes.2, 3 The very complex impact of protein-losing enteropathy on the body deserves to be taken into account in further studies of SGLT2i in patients with Fontan circulation. However, we did not notice that the inclusion of SGLT2i led to an intensification of symptoms, including protein-losing enteropathy. It is worth considering whether not only improvement but also the absence of deterioration is satisfactory, especially in such a demanding form of HF. So far, we have not analyzed the impact on the number of hospitalizations and HF exacerbations, which was the main focus of one of the largest studies on SGLT2i.4, 5, 7, 8, 9, 10 However, the observation of our patients will be continued and flozins will be introduced in subsequent participants.
Limitations
There are several limitations to this study. First, this is only an observational study in a small group of patients. Large, high-quality studies are needed to confirm our findings. Second, there was no control group in our study. However, the high heterogeneity of patients with FC and their clinical condition makes it very difficult to select a suitable control group, and we decided that it would be best to observe the fate of specific patients. Third, the interpretation of the VO2 max increase should be done with caution, while not all patients were able to perform this test. Finally, our cohort of Fontan patients is heterogeneous in terms of the medications used, but represents real patients.
Conclusions
This study showed for the first time that the use of SGLT2i can improve VO2 max in adult Fontan patients. We also observed significant changes that may support the hypothesis of a significant effect of SGLT2i on erythropoiesis. Finally, our experience shows that the use of SGLT2i in adult patients with FC is safe. Multicenter collaborations and large, high-quality trials are needed to more reliably assess the clinical effects of flozins in the adult FC population.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.