Abstract
Background. Sepsis is a life-threatening condition characterized by a dysregulated host immune response to infection. Currently, stress hyperglycemia is frequently associated with an unfavorable prognosis in cardiovascular and cerebrovascular disease. During sepsis, the progression of the immune response and inflammation often leads to aberrant metabolic indicators. However, the association between the stress-induced hyperglycemia ratio (SHR) and sepsis in patients admitted to the intensive care unit (ICU) remains uncertain.
Objectives. This study aimed to explore the potential correlation between SHR and sepsis.
Materials and methods. In this retrospective cohort study, data were obtained from the Medical Information Mart for Intensive Care-IV (MIMIC-IV) database. Patients with recorded glucose and glycosylated HbA1c levels within 24-h ICU admission were identified. The endpoints of the follow-up period were the occurrence of sepsis during ICU stay or ICU discharge. After adjustment for factors including demographics, vital signs and biochemical indicators, the univariate and multivariate logistic regression model was employed to examine the relationship between SHR, baseline blood glucose levels and the risk of sepsis. The associations were further explored in subgroups based on age, gender and presence/absence of type 2 diabetes.
Results. Of the total 2,161 patients, with the average age of 64.96 ±16.84 years, 205 (9.49%) had sepsis. After adjustment or confounders, high SHR levels were associated with the risk of sepsis odds ratio (OR) = 1.53, 95% confidence interval (95% CI): 1.07–2.17). Similar results were found in patients aged ≥65 years (OR = 1.91, 95% CI: 1.16–3.17), in men (OR = 1.64, 95% CI: 1.02–2.63) and patients without type 2 diabetes history (OR = 1.58, 95% CI: 1.01–2.48). The baseline blood glucose level did not exhibit a significant association with the risk of sepsis.
Conclusions. Elevated SHR levels were correlated with sepsis. Bedside monitoring of SHR may be a valuable tool for clinicians to identify patients at high risk of sepsis, and be beneficial to promptly implement clinical interventions.
Key words: sepsis, intensive care unit, glucose, stress-induced hyperglycemia ratio
Background
Sepsis is a life-threatening condition characterized by a dysregulated host immune response to infection, leading to systemic inflammatory response syndrome (SIRS), septic shock and multi-organ dysfunction.1 The manifestation of acute respiratory and kidney failure, as well as multiple organ dysfunction syndrome resulting from sepsis, impose a substantial burden on patients in the intensive care unit (ICU).2, 3 The estimated incidence of sepsis in ICU settings is approx. 58 cases per 100,000 person-years, with a pre-discharge mortality rate of 41.9%.4 Globally, the annual incidence of hospital-treated sepsis cases is estimated to exceed 30 million, with 5.3 million patients succumbing to sepsis.5 Conversely, sepsis may also serve as a predisposing factor for secondary atrial fibrillation and cardiogenic stroke.6 The early identification of high-risk patients prone to sepsis development is of paramount importance for sepsis prevention and reducing disease burden.
Previous reports indicate that patients with hyperglycemia or diabetes are susceptible to infection and even sepsis, which may be attributed to chronic suboptimal glycemic control.7, 8 The presence of inflammation and neurohormonal disorders during the disease can lead to a relative increase in glucose levels, resulting in stress hyperglycemia.9 This condition significantly impacts prognosis by inducing mechanisms like endothelial dysfunction and oxidative stress.9 However, the blood glucose levels only reflect the patient’s blood glucose status at a specific moment and do not indicate glycemic control.10 To minimize the potential misinterpretation of stress-induced hyperglycemia prevalence, the stress-induced hyperglycemia ratio (SHR) has been proposed as a strategy to assess the influence of chronic glycemic factors on stress-induced glucose levels, incorporating admission blood glucose and glycosylated hemoglobin A1c (HbA1c) measurements.11 The SHR is widely recognized as a more accurate indicator of long-term glycemic control compared to glucose and HbA1c, as it considers chronic hyperglycemia levels.12 The pathophysiological mechanisms of stress-induced hyperglycemia involve hormonal and metabolic responses to stress. During acute stress, the hypothalamic–pituitary–adrenal axis becomes active, releasing stress hormones like cortisol, epinephrine and glucagon.13 These hormones exert counter-regulatory effects on insulin action, stimulating hepatic gluconeogenesis and glycogenolysis, while concurrently inhibiting peripheral glucose uptake.14 Additionally, the stress-induced activation of the sympathetic nervous system and inflammatory mediators further exacerbates insulin resistance and hyperglycemia.14 Cumulatively, these pathophysiological changes disrupt glucose homeostasis, resulting in sustained hyperglycemia in critically ill patients. Studies indicated that stress-induced hyperglycemia significantly increase the risk of postoperative infection among non-diabetic orthopedic trauma patients in the ICU.15, 16 Studies also reported an association of elevated SHR with higher mortality rates in ICU patients, regardless of their diabetes status.17, 18, 19 Moreover, it has been observed that high SHR significantly enhances the predictive value of the Global Registry of Acute Coronary Events (GRACE) score for mortality prediction.20 Currently, stress hyperglycemia is commonly associated with an adverse prognosis in cardiovascular and cerebrovascular diseases.21, 22 During sepsis, the progression of immune response and inflammation often leads to abnormal metabolic indicators. However, the relationship between SHR and sepsis in patients admitted to the ICU remains unexplored.
Objectives
This study aimed to investigate the association between SHR, baseline blood glucose levels and sepsis risk in patients admitted to the ICU. Additionally, subgroup analysis was performed among individuals aged ≥65 years, in men and in patients without type 2 diabetes.
Methods
Study design
The data for this retrospective cohort study were extracted from the Medical Information Mart for Intensive Care-IV (MIMIC-IV) database, a comprehensive longitudinal database encompassing data collected between 2008 and 2019.23 The protocol of National Health and Nutrition Examination Survey (NHANES) was approved by the National Center for Health Statistics ethics review board and all participants signed informed consent forms. In this study, data of the participants were de-identified and there is no need for approval from the hospital ethics committee.
Patients with recorded glucose and HbA1c levels within 24 h after admission to ICU were identified from the MIMIC-IV database. The SHR was determined using the following formula: SHR = [admission glucose (mg/dL)]/ [28.7 × HbA1c (%) – 46.7].11, 18 The blood glucose levels measured within 24 h of admission to the ICU were considered as the baseline blood glucose. Both SHR and baseline glucose were classified according to the tertiles.
The inclusion criteria were patients with glucose and HbA1c examinations within 24 h after admission to ICU. The exclusion criteria were as follows: patients who 1) were under 18 years of age, 2) had a duration of ICU stay shorter than 24 h, 3) were diagnosed with sepsis upon ICU admission, and 4) for whom requisite information for sepsis assessment was lacking.
Covariates
Demographic information was extracted, including age, gender and race. Additionally, quick sequential organ failure assessment (qSOFA), Charlson comorbidity index (CCI), SIRS scores, white blood cell (WBC) count, red blood cell distribution width (RDW), creatinine, prothrombin time (PT), blood urea nitrogen (BUN), calcium and sodium levels, as well as information on mechanical ventilation within 24-h ICU admission were extracted for use as covariates in our study. The SIRS measurements were: 1) tachycardia (heart rate >90 bpm), 2) tachypnea or hyperventilation (respiratory rate >20 breaths/min or partial pressure of carbon dioxide (PaCO2) <32 mm Hg), 3) fever or hypothermia (temperature >38 or <36°C) and 4) leukocytosis, leukopenia or bandemia (WBC >12×109/L, <4×109/L or bandemia ≥10%). The SIRS status was determined with the criteria (from 0 (best) to 4 (worst)).23, 24
Outcome and follow-up
Sepsis was the outcome of this study. The median follow-up duration was 2.1 (1.6, 3.4) days. Sepsis was diagnosed based on the Sepsis-3 criteria.2 In brief, patients with confirmed or suspected infection and a sudden increase in total qSOFA score of ≥2 points were classified as having sepsis. The identification of infection was determined using the International Classification of Diseases (ICD) code.
Statistical analyses
The data cleaning, imputation of missing values, covariate screening, logistic regression analysis, and subgroup analysis were conducted using R v. 4.2.3 (The R Foundation for Statistical Computing, Vienna, Austria).
The variables with a missing rate ≤20% underwent multiple imputations. Subsequently, a sensitivity analysis was conducted to compare the variables before and after imputation. Assuming that the data is Missing Completely at Random (MCAR), missing values can be predicted and interpolated from observed values. The measurement data with a normal distribution were presented as the mean ± standard deviation (mean ±SD). The normality of continuous variables was tested by skewness and kurtosis, while homogeneity was detected using the Levene test. For data with homogeneity of variance, a t-test was employed to compare between 2 groups, while for data with non-homogeneity of variance, Satterthwaite’s t-test was utilized. Measurement data that did not conform to a normal distribution were described using the median (Me) and quartile, and differences between any 2 groups were compared using the Wilcoxon rank sum tests. Count data were described as the number of cases and constituent ratio, and group differences were assessed using the χ2 test. The confounding factors were screened using univariate logistic regression models. After adjusting for confounders including age, gender, race, qSOFA score, CCI, SIRS, calcium levels, sodium levels, and mechanical ventilation status, the multivariate logistic regression model was employed to examine the relationship between SHR, baseline blood glucose levels and the risk of sepsis. Model 1 represents a univariate logistic regression model. Model 2 incorporates adjustments for qSOFA, CCI, SIRS, calcium, sodium, and mechanical ventilation. Model 3 further adjusts for age, gender, race, qSOFA, CCI, SIRS, calcium, sodium, and mechanical ventilation. The present study adopted the Box–Tidwell test to verify whether the predictors and the logit of the response variable were linear. The line test results show a linear relationship between all continuous predictor variables and sepsis. The generalized variance inflation factor (gVIF) test was adopted to detect multicollinearity among the explanatory variables, with all gVIFs < 10 indicating that no multicollinearity was observed. Cook’s distances were conducted to detect the presence of extreme outliers. Subgroup analyses were conducted based on age, gender, and presence or absence of history of type 2 diabetes. A p-value of 0.05 was considered significant.
Results
Clinical characteristics of ICU patients
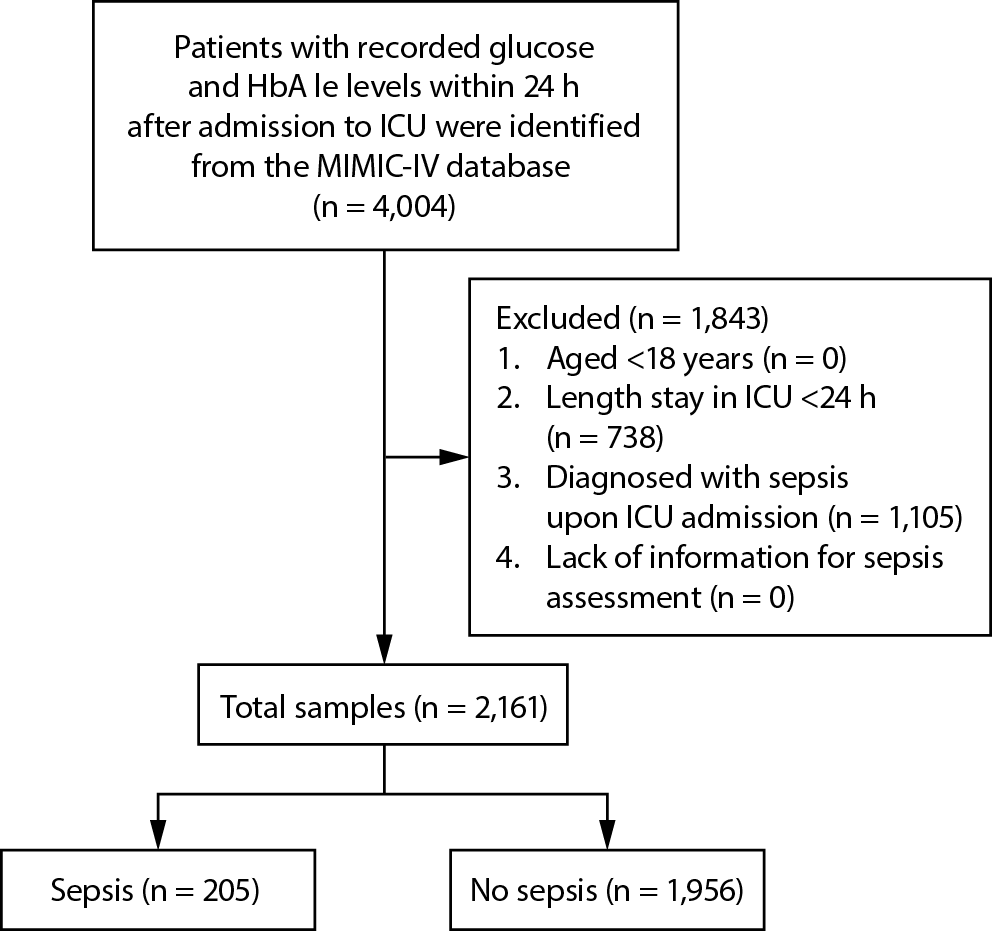
The data extraction and screening process were conducted following the workflow depicted in Figure 1. No significant difference was observed before and after data interpolation (Supplementary Tables 1 and 2).
A total of 4,004 patients with documented blood glucose and HbA1c levels within 24 h of admission to the ICU were included in this study. After excluding 1,105 patients diagnosed with sepsis upon ICU admission and 738 patients who stayed in the ICU for less than 24 h, the final cohort consisted of 2,161 patints, of which 205 (9.49%) patients had sepsis during the ICU stay. The average age of all patients was 64.96 ±16.84 years, with men accounting for 56%. The characteristics of the included patients were presented in Table 1. In comparison to the non-sepsis group, the sepsis group exhibited higher levels of temperature (p = 0.001). Additionally, the sepsis group exhibited elevated SAPS-II score (p < 0.001), SOFA score (p < 0.001), qSOFA score (p < 0.001), GCS score (p = 0.029), CCI score (p < 0.001), and SIRS score (p < 0.001). Moreover, compared to the non-sepsis group, the sepsis group showed significantly higher levels of WBC count (p < 0.001), red blood cell distribution width-coefficient of variation (RDW-CV) (p = 0.011), creatinine (p = 0.027), PT (p = 0.038), BUN (p = 0.015), and sodium (p = 0.040) concentration; whereas calcium (p = 0.008) and HbA1c levels (p < 0.001) were significantly lower in this group. The proportion of mechanical ventilation and antibiotic usage was higher among patients with sepsis. Additionally, the distribution of elevated glucose levels (≥110, p < 0.001) and SHR (≥0.9, p = 0.007) were significantly higher in the sepsis group.
Association of SHR and baseline glucose levels with sepsis
Ultimately, the qSOFA score, CCI score, SIRS score, calcium, and sodium were identified as confounding factors. Additionally, demographic factors such as age, gender and race were included in model 3 as covariates to account for their potential influence (Supplementary Table 3).
After adjustment for confounders (Table 2), SHR ≥0.9 (odds ratio (OR): = 1.53, 95% confidence interval (95% CI): 1.07–2.17, p = 0.020) was also associated with the risk of sepsis (model 3). Conversely, glucose levels ≥110 did not exhibit any significant association with the risk of sepsis (p = 0.588).
Association between SHR and sepsis in subgroups of ages, gender and type 2 diabetes
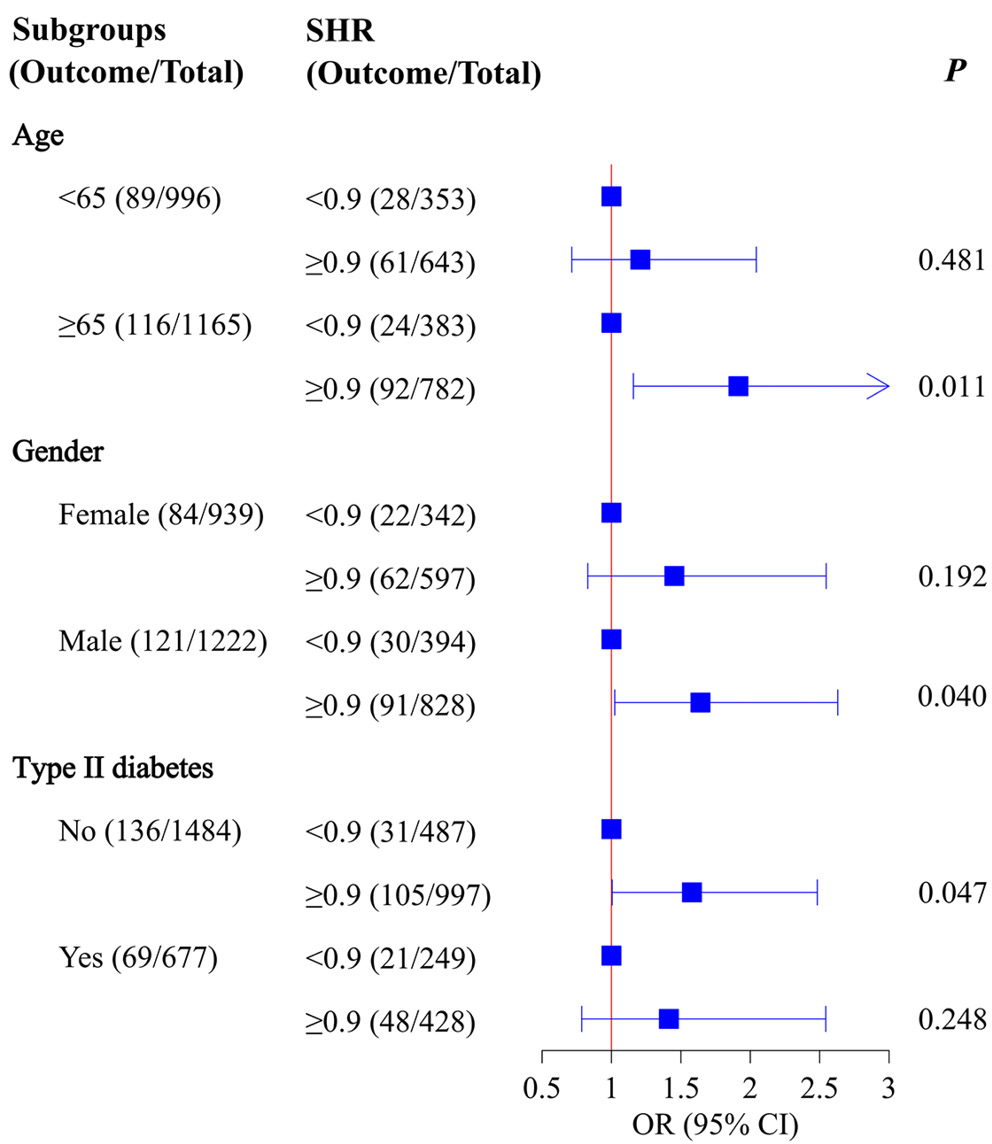
In patients aged ≥65 years (OR: 1.91, 95% CI: 1.16–3.17, p = 0.011), among men (OR: 1.64, 95% CI: 1.02–2.63, p = 0.040) and in patients without history of type 2 diabetes (OR: 1.58, 95% CI: 1.01–2.48, p = 0.047), elevated levels of SHR were associated with higher odds of sepsis (Figure 2). The baseline blood glucose level did not exhibit a significant association with the risk of sepsis in these patients (p > 0.05) (Table 3).
Discussion
Our study aimed to explore the association between baseline blood glucose levels, SHR and the risk of sepsis in ICU patients. We found that elevated SHR levels were related to higher odds of sepsis. Moreover, our findings showed that elevated SHR levels were associated with sepsis among patients aged ≥65 years, who were men and in patients without a history of type 2 diabetes. The baseline blood glucose level did not exhibit a significant association with sepsis risk.
Sepsis represents a significant global health challenge and is associated with substantial mortality rates.1 The incidence of sepsis varies considerably across different regions worldwide, ranging from 14% to 39%.25 Patients requiring intensive care demonstrate an elevated susceptibility to sepsis development compared to those admitted to general wards.26 Currently reported factors associated with sepsis occurrence include bone mineral density,27 qSOFA,28 PCT,28 and emergency surgery.29 However, in a study investigating risk factors for sepsis following geriatric surgery,29 blood glucose was found to be associated with sepsis in patients. In contrast to our findings, we did not observe the association between glucose levels and sepsis risk, which may be attributed to differences in the study population. Generally, blood glucose fluctuations are controlled during early hospitalization in elderly surgical patients, reflecting the patient’s long-term blood glucose status with some degree of stability. Given the high incidence of diabetes in the ICU, evaluating chronic hyperglycemia based solely on absolute glucose concentration is deemed ineffective.10 The measurement of HbA1c serves as an indicator of chronic hyperglycemia in patients with both overt and recessive diabetes. Conversely, the admission glucose concentration in critically ill patients indicates the severity of acute hyperglycemia.30 Similarly, elevated glucose levels (≥110 mmol/L) and SHR (≥0.9) were more prevalent in the sepsis group in our study.
The calculation of SHR considers admission glucose and HbA1c concentrations, estimating stress-induced hyperglycemia while accounting for chronic hyperglycemia in patients with or without diabetes.11 Previous research suggests that SHR may be associated with more severe adverse outcomes compared to chronic hyperglycemia, particularly in severe conditions such as acute myocardial infarction, trauma and acute ischemic stroke.18, 22, 31 Similarly, we also observed the relationship between SHR and sepsis in ICU patients.
Stress-induced hyperglycemia is characterized by increased blood glucose levels due to the activation of stress hormones such as cortisol and catecholamines.14 This hyperglycemic state can lead to immune dysfunction through several interconnected pathways. First, hyperglycemia has been shown to impair various components of the immune response, including neutrophil function, macrophage activity and lymphocyte proliferation.32 Neutrophils, key mediators of the innate immune response, exhibit reduced chemotaxis and phagocytic activity in hyperglycemic conditions, compromising their ability to effectively clear pathogens.33 Additionally, hyperglycemia impairs the function of macrophages, inhibiting antigen presentation and cytokine production essential for coordinating the immune response.34 Second, hyperglycemia promotes a pro-inflammatory state characterized by increased production of inflammatory cytokines such as interleukin 6 (IL-6) and tumor necrosis factor alpha (TNF-α).35 These cytokines not only contribute to tissue damage but also disrupt the delicate balance between pro-inflammatory and anti-inflammatory responses, potentially exacerbating the systemic inflammatory response associated with sepsis. Furthermore, hyperglycemia-induced oxidative stress may play a role in immune dysregulation and tissue damage. Elevated glucose levels promote the production of reactive oxygen species (ROS) and reactive nitrogen species (RNS), leading to oxidative damage to cells and tissues.36 Oxidative stress not only impairs immune cell function but also exacerbates endothelial dysfunction and microvascular damage, contributing to organ dysfunction commonly seen in sepsis.36
In the subgroup of individuals aged ≥65 years, among men and in individuals without type 2 diabetes, elevated levels of SHR were associated with sepsis in our study. The association can be ascribed to the diminished immune function observed in elderly patients compared to their younger counterparts, rendering them more susceptible to infections.37 Additionally, the delayed clearance of pathogens at local infection sites further exacerbates the likelihood of sepsis development among elderly individuals.38 Elevated SHR levels indicate an imminent surge in blood glucose levels, which can inflict additional harm on the immune system and escalate the progression of localized infections into septic conditions.17 Furthermore, it is worth noting that male patients demonstrate a higher propensity for sepsis development compared to women in similar circumstances. Numerous studies have substantiated this observation by demonstrating that men display heightened susceptibility to bacterial, fungal and viral infections, and other diseases owing to their robust physiological constitution and infrequent occurrence of regular illnesses or systematic immune clearance processes.39, 40 Consequently, they tend to accumulate prolonged immunological debt which amplifies their vulnerability to sepsis. In patients with elevated SHR levels but without a diagnosis of type 2 diabetes, stress-induced hyperglycemia manifests as a consequence of traumatic events, infections and other acute precipitating factors.41 The elevated blood glucose levels exert varying degrees of deleterious effects on the nervous, cardiovascular and immune systems.9 The sudden increase in blood glucose levels compromises the immune system rapidly, allowing insufficient time for adaptation and resulting in exacerbation of pre-existing infections or direct progression to severe infection followed by sepsis.8 In the subgroup analysis investigating the relationship between baseline glucose levels and sepsis, we did not observe a statistically significant correlation. This may be attributed to several confounding factors that can influence patients’ glucose levels within 24 h of admission to the ICU, such as concurrent administration of therapeutic medications, impaired renal function and hepatic failure.16 It appears that immediate glucose levels have limited prognostic relevance for patients.
The present study represents the potential effort to investigate the correlation between SHR and sepsis risk in ICU, aiming to offer valuable insights for prognostic marker screening and identification of high-risk patients, as well as potential applications of SHR through subgroup analysis. The association between SHR and sepsis suggests that timely and regular monitoring of SHR in ICU patients by clinicians can readily identify high-risk groups for sepsis and enable promptly implementing clinical interventions to enhance patient prognosis.
Limitations
First, due to the retrospective design and single-center setting, there is an inherent selection bias. Moreover, the limited exposure to HbA1c further exacerbates this bias. Additionally, as it was an observational study, we can only establish associations rather than determine a causal relationship between SHR and sepsis risk in ICU patients. Future prospective studies are warranted to validate our findings.
Conclusions
The elevated level of SHR was identified to be significantly correlated with sepsis. Bedside monitoring of SHR is a valuable tool for clinicians to identify patients at high risk of sepsis and promptly implement clinical interventions, thereby improving patient outcomes.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.13901764. The package includes the following files:
Supplementary Table 1. The distribution of missing values.
Supplementary Table 2. Sensitivity analysis before and after interpolation of missing values.
Supplementary Table 3. Covariates screening in regression analysis.
Supplementary Table 4. Box–Tidwell tests between variables and sepsis (model 2).
Supplementary Table 5. Box–Tidwell tests between variables and sepsis (nodel 3).
Supplementary Table 6. The gVIF among variables.
Supplementary Table 7. The tests of normality and homogeneity assumptions.
Supplementary Table 8. The confusion matrix.
Supplementary Table 9. The accuracy, precision and recall values of the study.
Supplementary Fig. 1. Cook’s distance plot of 3 models.
Supplementary Fig. 2. Receiver operating characteristic (ROC) curve of the study.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.