Abstract
Background. Propofol and midazolam have been widely used in patients with sepsis. However, the effectiveness of these drugs in reducing the duration of mechanical ventilation and the risk of mortality remains controversial.
Objectives. To investigate and compare effects of propofol and midazolam on 30-day mortality in patients with sepsis-associated encephalopathy (SAE).
Materials and methods. A retrospective cohort study was conducted on data from 952 adult patients with SAE extracted from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database. Univariable and multivariable Cox proportional hazard models were utilized to investigate the associations of propofol and midazolam with 30-day mortality; and univariable and multivariable logistic regression analyses were used to explore the relationships of propofol and midazolam with ventilation duration. The outcome measures were hazard ratios (HRs), odds ratios (ORs), and 95% confidence intervals (95% CIs). In addition, subgroup analyses of age, simplified acute physiological score (SAPS)-II, Charlson Comorbidity Index (CCI), and ventilation duration were also performed to further assess the associations of propofol and midazolam with 30-day mortality.
Results. Among eligible patients, 265 (27.84%) died within 30 days. After adjusting for covariates, treatment with propofol was associated with both lower risk of 30-day mortality (HR = 0.67, 95% CI: 0.51–0.88) and lower odds of prolonged ventilation duration (OR = 0.71, 95% CI: 0.53–0.96) compared to treatment with midazolam. Moreover, the negative association between treatment with propofol and 30-day mortality was also significant in subgroups of age ≥65 years, SAPS-II score ≥47, CCI score ≥3, and ventilation duration ≥5 days (all p < 0.05).
Conclusions. Among patients with SAE, treatment with propofol was relatively more effective than treatment with midazolam in reducing the risk of 30-day mortality and the duration of mechanical ventilation. However, the causal relationships of propofol and midazolam with prognosis in patients with SAE need further clarification.
Key words: propofol, midazolam, 30-day mortality, SAE, ventilation duration
Background
Sepsis-associated encephalopathy (SAE) is a severe neurologic syndrome, namely the brain dysfunction occurring during the course of sepsis.1 Sepsis-associated encephalopathy significantly increased the length of hospital stay as well as the risk of short-term mortality in patients.2 Evidence has suggested that even mild changes in consciousness can result in a significant increase in mortality risk in patients with sepsis.3 Hence, it is important to explore factors influencing the short-term mortality risk in patients with SAE, which may help improve SAE prognoses.
Propofol and midazolam are commonly used sedatives in the intensive care units (ICUs),4 and they have been also widely used among sepsis patients.5, 6, 7 A meta-analysis showed that compared to midazolam, propofol reduced the length of ICU stay, mechanical ventilation time and extubation time among the ICU patients.4 Also, treatment with propofol has a significant advantage in the incidence of short-term postoperative cognitive dysfunction,8 e.g., patients treated with propofol had lower rates of delirium compared to those treated with midazolam.9 In addition, a recent study in children indicated that the application of midazolam could increase the risk of SAE.10 However, whether propofol or midazolam is more suitable for patients with sepsis remains controversial, especially in the presence of SAE. To date, no studies have been conducted to directly compare the effects of propofol and midazolam on the prognosis of SAE.
Objectives
This study was designed to investigate associations of propofol and midazolam with 30-day mortality in patients with SAE, comparing effects of these 2 sedatives on short-term prognosis in patients with SAE, and to provide some references for choice of sedatives for patients with SAE in clinical practice.
Methods
Study participants
This is a retrospective cohort study. Data of participants were obtained from the Medical Information Mart for Intensive Care (MIMIC)-IV database in 2008–2019. The MIMIC database is jointly published by the computational physiology laboratory of the Massachusetts Institute of Technology (MIT; Cambridge, USA), Beth Israel Deaconess Medical Center (BIDMC; Boston, USA) and Philips Medical (Amsterdam, the Netherlands) since 2001, which collects and sorts out information on clinical diagnosis and treatment of more than 40,000 ICU patients. More details about the MIMIC database can be found elsewhere: https://mimic.mit.edu/docs/iv.
For participants screening, the inclusion criteria were: 1) age ≥18 years, 2) diagnosed with SAE at the ICU admission and 3) treated with midazolam or propofol exclusively as sedative. The exclusion criteria were: 1) not using mechanical ventilation, 2) having traumatic brain injury (TBI),11, 12 ischemic/hemorrhagic stroke, intracranial infection or epilepsy, 3) chronic alcohol or drug abuse, 4) pre-existing liver or kidney diseases affecting consciousness,13 and 5) missing information on survival. Finally, 952 patients met the necessary criteria for inclusion in the study. The MIMIC database has obtained ethical approval from the relevant Institutional Review Boards (IRBs) (https://mimic.mit.edu). Since the database is publicly available, ethical approval has been waived by our hospital’s IRB. Informed consent was not required due to the retrospective nature of the study.
Diagnosis of SAE
According to a previous study, SAE was defined as sepsis accompanied by a Glasgow Coma Scale (GCS) ≤14 in the first 24 h of ICU admission or delirium based on the International Classification of Diseases v. 9. (ICD-9) code (2930, 2931) and ICD-10 code (F05).14
Assessment of propofol and midazolam use
Records of the use of propofol or midazolam in patients with SAE were extracted from the MIMIC input event tables, with item ID 222168 for propofol and 221668 for midazolam.
Variables selection
We also extracted variables as potential confounding factors from the database, including race, age, gender, congestive heart failure (CHF), peripheral vascular disease (PVD), chronic obstructive pulmonary disease (COPD), renal failure (RF), liver disease, hypertension, diabetes mellitus (DM), heart rate (HR), weight, systolic blood pressure (SBP), diastolic blood pressure (DBP), temperature, respiratory rate (RR), oxygen saturation (SpO2), inspiratory oxygen concentration (FIO2), simplified acute physiological score (SAPS)-II, GCS, Charlson Comorbidity Index (CCI), red cell distribution width (RDW), white blood cell (WBC) count, platelet count, hemoglobin (HB), hematocrit, creatinine (Cr), blood urea nitrogen (BUN), glucose, lactate, sodium (Na), bicarbonate, potassium (K), pH, chloride, international normalized ratio (INR), ventilation duration, prothrombin time (PT), vasopressors use, opiates use, norepinephrine use, and antibiotics use. In addition, only information on these variables was extracted from patients when they were first admitted to the ICU.
Study outcomes
Primary study outcome was 30-day mortality, and the secondary study outcome was ventilation duration. The ventilation duration was divided into 2 categories according to the median value, including <5 days and ≥5 days. The MIMIC followed up through information recorded in electronic medical charts and hospital department, or contacting with the patients (including family members, attending healthcare workers and family physicians) via phone calls. In our study, the follow-up ended when patients died or 30 days after the ICU admission.
Statistical analyses
Non-normal data were described in median and quartiles (Me (Q₁, Q₃)), and Wilcoxon ran-sum tests were used for inter-group comparisons. The enumeration data were described in terms of number of cases and composition ratio (n (%)). The χ2 test was used for comparison between groups.
The covariates screening process included 2 steps. First, variables significantly associated with 30-day mortality in patients with SAE were selected via univariable Cox proportional hazard model. Then, the selected variables were test using the best subset method, and those with the lowest Bayesian information criterion (BIC) values were chosen as the final covariates. Finally, the selected covariables were further included in the adjustment of multivariate models.
The screening of covariates associated with ventilation duration was similar to that associated with 30-day mortality except using the univariable logistic regression.
Univariate and multivariate Cox proportional hazard models were established to explore the associations of propofol and midazolam with 30-day mortality, with hazard ratios (HRs) and 95% confidence intervals (95% CIs). Model 1 was unadjusted. Model 2 was adjusted for the selected covariates, including age, race, RR, temperature, SpO2, SAPS-II score, CCI score, RDW, WBC, BUN, lactate, chloride, INR, PT, vasopressor use, opiates use, and norepinephrine use. Additionally, subgroup analysis of age, SAPS-II score, CCI score and ventilation duration were also performed to further assess these associations.
Univariate and multivariate logistic regression analyses were used to investigate the associations of propofol and midazolam with duration of ventilation in patients with SAE, with odds ratios (ORs) and 95% CIs as assessment indices. Model 1 was unadjusted. Model 2 was adjusted for selected covariates, including SpO2, SAPS-II, vasopressor use, norepinephrine use, and antibiotics use.
Two-sided p < 0.05 was considered statistically significant. Variables with missing values were removed if the proportion was >20%; otherwise, they were interpolated using a random forest interpolation method (Supplementary Table 1).15 Sensitivity analyses of patients’ characteristics before and after interpolation of missing data are shown in Supplementary Table 2. The associations of midazolam and propofol with 30-day mortality (Supplementary Table 3) and duration of mechanical ventilation (Supplementary Table 4) in patients with SAE were also assessed using the original unimplemented data. Statistical analyses were performed using Python 3.9.12 (Python Software Foundation, Wilmington, USA) and R v. 4.3.1 (2023-06-16 ucrt; R Foundation for Statistical Computing, Vienna, Austria).
Results
Characteristics of participants
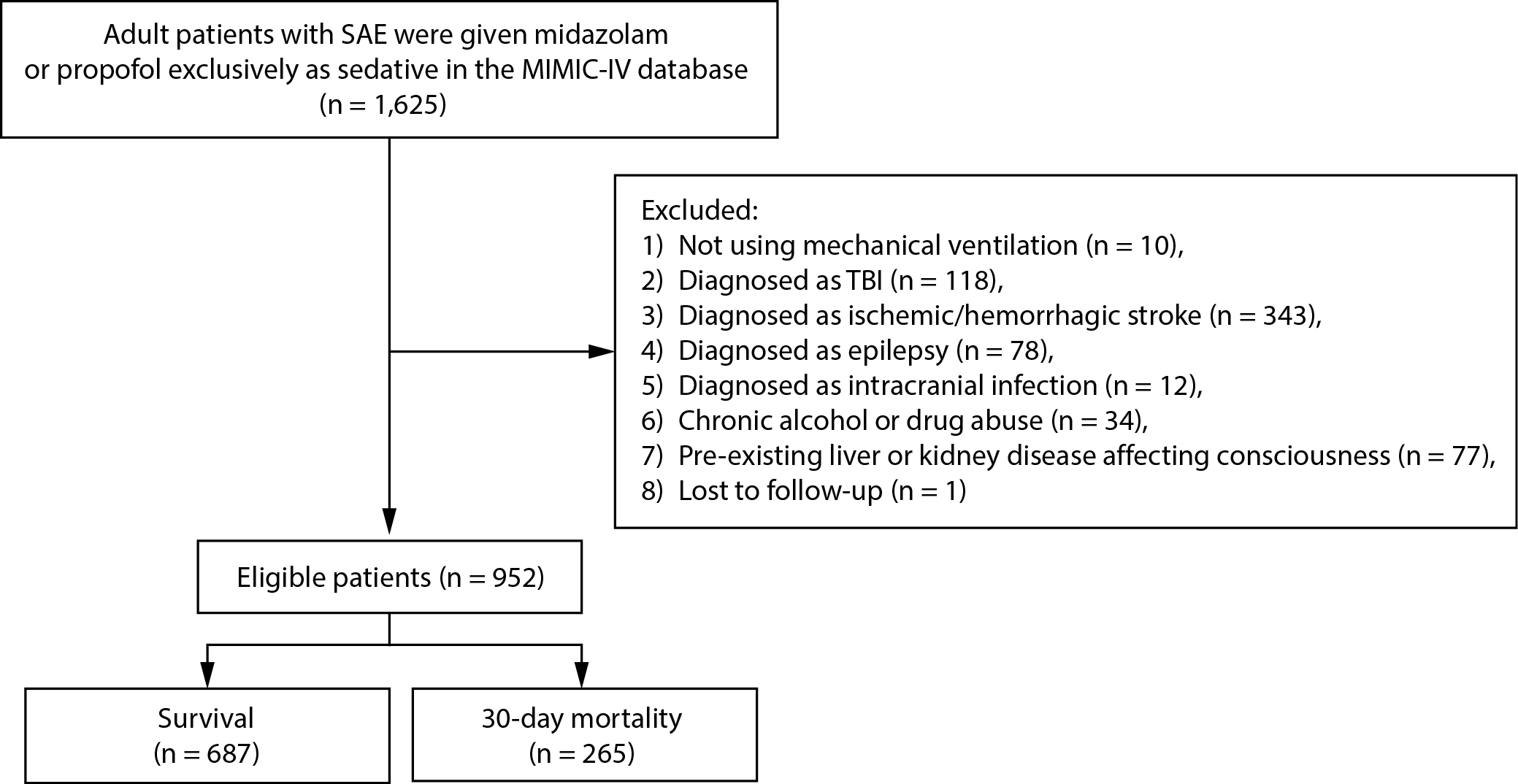
Figure 1 shows inclusion and exclusion of research subjects. There were 1,625 adult SAE patients who received treatment with propofol or midazolam in the database. Those who without mechanical ventilation use (n = 10), diagnosed as TBI (n = 118), ischemic/hemorrhagic stroke (n = 343), epilepsy (n = 78), or intracranial infection (n = 12), with chronic alcohol or drug abuse (n = 34), with pre-existing liver/kidney disease affecting consciousness (n = 77), or lost to the follow-up (n = 1) were excluded. Finally, 952 were eligible.
Among the eligible participants, 265 (27.84%) died within 30 days. The median age of total population was 70 years, and 546 (57.35%) were male. We compared the characteristics of patients between the midazolam (n = 259) and propofol group (n = 693) (Table 1). Disease conditions (CHF and COPD), clinical indexes (HR, SBP, RR, SAPS-II, GCS, platelet, HB, hematocrit, Cr, BUN, glucose, pH, and PT) and treatments (ventilation duration, opiates use and norepinephrine use) were all significantly different between the 2 groups (all p < 0.05).
Associations of midazolam and propofol with 30-day mortality
Before investigating relationships between different sedatives and 30-day mortality in patients with SAE, covariates have been screened (Supplementary Table 3). As shown in Table 2, treatment with propofol was linked to lower 30-day mortality risk (HR = 0.67, 95% CI: 0.51–0.88), compared to treatment with midazolam after covariates adjustment. Moreover, we assessed this relationship in subgroups of age, SAPS-II score, CCI, and ventilation duration (Figure 2). The results showed that in aged ≥65 years (HR = 0.61, 95% CI: 0.45–0.84), SAPS-II score ≥47 (HR = 0.55, 95% CI: 0.40–0.75), CCI score ≥3 (HR = 0.63, 95% CI: 0.46–0.87) and ventilation duration ≥5 (HR = 0.58, 95% CI: 0.41–0.84) subgroups, treatment with propofol was also significantly linked to lower 30-day mortality risk comparing to treatment with midazolam.
Relationship between different sedatives and ventilation duration in SAE patients
In addition, we explored the associations of midazolam and propofol with ventilation duration in patients with SAE. The process of covariates screening was shown in Supplementary Table 4. We found that treatment with propofol was associated with lower odds of long ventilation duration comparing to that with midazolam (OR = 0.71, 95% CI: 0.53–0.96), indicating that treatment with propofol may be more effective than that with midazolam in improving excessive ventilation duration among SAE patients (Table 3).
Discussion
The current study explored the associations of propofol and midazolam with 30-day mortality in patients with SAE. The results suggested that patients with SAE treated with propofol had a lower risk of 30-day mortality compared to those treated with midazolam. This association was also significant in the subgroups of adults aged ≥65 years, SAPS-II score ≥47, CCI score ≥3, and ventilation duration ≥5. In addition, treatment with propofol was significantly associated with lower odds of long duration of ventilation in patients with SAE compared to patients treated with midazolam.
We believe our study is the first to explore and compare associations of propofol and midazolam with short-term mortality and ventilation duration in patients with SAE. Previous studies have discussed the effects of propofol and midazolam on outcomes in ICU patients. For instance, a post hoc analysis of the DESIRE trial conducted by Miyagawa et al.16 found that patients with sepsis required mechanical ventilation during the acute phase. That study also found that sedation with midazolam was associated with an increased risk of coma and delirium compared to propofol. Another study in adult ICU patients showed that propofol was associated with improved clinical outcomes compared to midazolam sedation, reducing extubation time and mechanical ventilation time in acute surgical patients and extubation time in critically ill patients.4 Yet another observational, propensity-matched study suggested that, compared with midazolam, sedation with propofol reduced mortality and bleeding rates in patients with cardiogenic shock.17 However, the associations of propofol and midazolam with short-term mortality in SAE have not been clarified. By observing negative associations of propofol treatment with 30-day mortality risk and prolonged duration of mechanical ventilation, our findings relatively filled a gap in the literature on patients with SAE. However, further research is needed on the causal associations of midazolam and propofol with ICU outcomes in this population.
The acute phase of SAE is characterized primarily by delirium symptoms,18 which are associated with worse outcomes.3 In ICU patients, maintaining light sedation was linked to increased survival and decreased delirium.19 Researches suggested that midazolam could prolong the time to light sedation in comparison with propofol.20, 21 Also, in the study by Miyagawa et al., midazolam produced significantly deeper and more inappropriate sedation than propofol in the acute phase (day 3 of hospitalization), despite the use of light sedation protocols.16 Similarly, in our study population, the GCS score of patients in the midazolam group was significantly lower than that in the propofol group (10.61 vs 11.62). In fact, it has been reported that even mild alterations of the mental status (GCS of 13–14) have prognostic potential towards a worse outcome in sepsis.3 Nevertheless, in the present study, we could not clarify the mechanism of the potential superior effect of propofol to that of midazolam on short-term mortality in SAE, and whether it was related to the mental status or delirium of the patients. In addition, neuroinflammation induced by microglial activation is closely linked to the development of SAE.22 Guan et al.23 suggested that propofol attenuated the inflammatory response by inhibiting metabolic reprogramming via downregulation of the ROS/PI3K/Akt/mTOR/HIF-1α signaling pathway. On the contrary, higher doses of midazolam may be an independent risk factor for sepsis-associated delirium among mechanically ventilated patients with sepsis.24 Additionally, Sasabuchi et al.25 retrospectively analyzed 30-day mortality and intubation duration in children ventilated for ≥3 days and sedated with midazolam or propofol and showed that weaning from mechanical ventilation was slower in children sedated with midazolam than in those sedated with propofol. Further investigation is necessary to determine the specific mechanism that led to the selection of propofol sedation as the preferred option for patients with SAE over midazolam sedation.
Results of subgroup analysis showed that among patients with SAE aged ≥65 years, SAPS-II score ≥47, CCI score ≥3, or with ventilation duration ≥5, the association of propofol with lower 30-day mortality risk was also significant. In fact, age (≥75 years) and SAPS-II score (≥23) were common risk factors for SAE.26 The progression of SAE and neurological deficits resulted from age-related reconstruction of the brain tissue with senescence of astroglia.27 In patients admitted to the ICU for sepsis, SAPS-II also showed good performance in predicting mortality. Our results suggest that in patients with SAE with older age or higher SAPS-II score, treatment with propofol may be a better choice to reduce short-term mortality than that with midazolam. The CCI seemed to be helpful for early identification of septic shock patients with poorer outcomes.28 In our research, although the CCI score had no significant difference between the midazolam group and the propofol group (3.65 vs 3.56), it seemed that clinicians should consider propofol as the first choice of sedative. Mechanical ventilation is a cornerstone of sepsis treatment, and prolonged mechanical ventilation (exceeding 21 days) is associated with increased mortality rates of both in-hospital and post-discharge.29 In the present study, 57.53% of patients in the midazolam group had a ventilation duration ≥5 days, whereas the proportion in the propofol group was 48.34%. Duration of ventilation was not found to be significantly associated with 30-day mortality, but in the subgroup with duration of ventilation ≥5, the association between propofol and lower 30-day mortality risk was significant, suggesting that this potentially high-risk population should receive more attention.
Limitations
The current study was the first to explore effects of treatment with 2 different sedatives on short-term mortality risk in patients with SAE. In statistical analyses, we have considered and evaluated important variables associated with sedation in SAE, including scores reflecting severity and prognosis of patients, comorbidities and multiple laboratory indicators. However, there were still some limitations. As a retrospective cohort study, it is difficult to avoid the inherent bias of this study design. Data in the MIMIC database were collected from a single medical center, which may limit the representativeness of the study sample, and therefore, large-scale multi-center studies are needed to further verify our findings. Besides, due to limitations of the MIMIC database, we could not obtain the detailed sedation information, such as different drug dosages, treatment duration and daily sedation level data, which could cause some bias.
Conclusions
Sedation with propofol was a potentially better choice than midazolam for patients with SAE in clinical practice, which may reduce the risk of short-term mortality and prolonged duration of ventilation. However, the causal associations of propofol and midazolam with prognosis in SAE patients need to be further clarified.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.15061951. The package includes the following files:
Supplementary Table 1. Variables with missing data and processing.
Supplementary Table 2. Sensitivity analysis of characteristics of patients before and after interpolation of missing data.
Supplementary Table 3. Covariates associated with 30-day mortality in SAE patients.
Supplementary Table 4. Covariates associated with ventilation duration in SAE patients.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.


















.jpg)