Abstract
Background. Pyroptosis has been implicated in the progression of chronic prostatitis (CP)/chronic pelvic pain syndrome (CPPS).
Objectives. The present study was performed to explore the diagnostic value of the levels of the pyroptosis-related protein nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain-containing 3 (NLRP3) inflammasome in the expressed prostatic secretions (EPS) of patients with CP.
Materials and methods. A total of 167 CP patients, including 85 National Institutes of Health (NIH)-IIIA CP patients and 82 NIH-IIIB CP patients, as well as 80 benign prostatic hyperplasia (BPH) patients and 80 healthy controls, were enrolled. The levels of NLRP3, interleukin 1 beta (IL-1β), and interleukin 18 (IL-18) in EPS were detected using an enzyme-linked immunosorbent assay (ELISA). Disease severity was assessed using the Bergman CP scale. Differences in EPS NLRP3 inflammasome levels between the groups were analyzed, and receiver operating characteristic (ROC) curves were used to investigate the clinical value of the NLRP3 inflammasome in the diagnosis of CP. The numerical rating scale (NRS), the National Institutes of Health Chronic Prostatitis Symptom Index (NIH-CPSI) and the Danish Prostatic Symptom Score (DAN-PSS-1) were applied to evaluate symptom severity. The cutoff value of NLRP3 expression was calculated using R language.
Results. NLRP3 inflammasome levels in EPS were significantly higher in CP patients of NIH-IIIA and NIH-IIIB compared to the BPH patients and controls. NLRP3 levels in EPS were positively associated with Bergman grade. In addition, NRS levels were in a positive relationship with NIH-CPSI and DAN-PSS-1. The ROC curve analysis demonstrated that NLRP3 in EPS may act as a decent indicator for the diagnosis of CP/CPPS. The cutoff value of EPS NLRP3 expression was ≥55.25 ng/mL.
Conclusions. NLRP3 levels in EPS were significantly higher in NIH-IIIA and NIH-IIIB patients compared to BPH patients and healthy controls. NLRP3 inflammasome levels in EPS may be valuable as diagnostic indicators, and targeting chemokines may present a promising approach to treatment for those suffering from CPPS.
Key words: chronic prostatitis, chronic pelvic pain syndrome, NLRP3 inflammasome, pyroptosis
Background
Chronic prostatitis (CP) has a prevalence of nearly 5–10% among young and middle-aged individuals, significantly affecting their daily lives.1 Chronic prostatitis is categorized into 3 types by National Institutes of Health (NIH): NIH-II (chronic bacterial prostatitis), NIH-III (chronic prostatitis/chronic pelvic pain syndrome (CPPS)) and NIH-IV (asymptomatic prostatitis).2 Chronic prostatitis can manifest with a variety of symptoms that significantly impact quality of life, such as pelvic pain, lower urinary tract symptoms, psychological distress, and sexual dysfunction.3
The NIH-III, also referred to as CP/CPPS, is characterized by pelvic pain without any detectable bacterial infection. It is further classified into 2 subcategories: category IIIA, which is the inflammatory type, indicated by the presence of white blood cells in the expressed prostatic secretion (EPS), and category IIIB, the non-inflammatory type, where leukocytes are not present in the EPS or seminal fluid.4 Chronic prostatitis/CPPS, falling under NIH category III, is frequently encountered in clinical settings, with its prevalence in the general population estimated to be between 5% and 14.2%.5 Therefore, CP/CPPS accounts for approx. 60–80% of all prostatitis cases.
Cytokines and chemokines, which are inflammatory mediators, have been linked to the development of CPPS.6 Cytokines are signaling proteins that are secreted by different types of cells to facilitate intercellular communication and regulate immune responses.
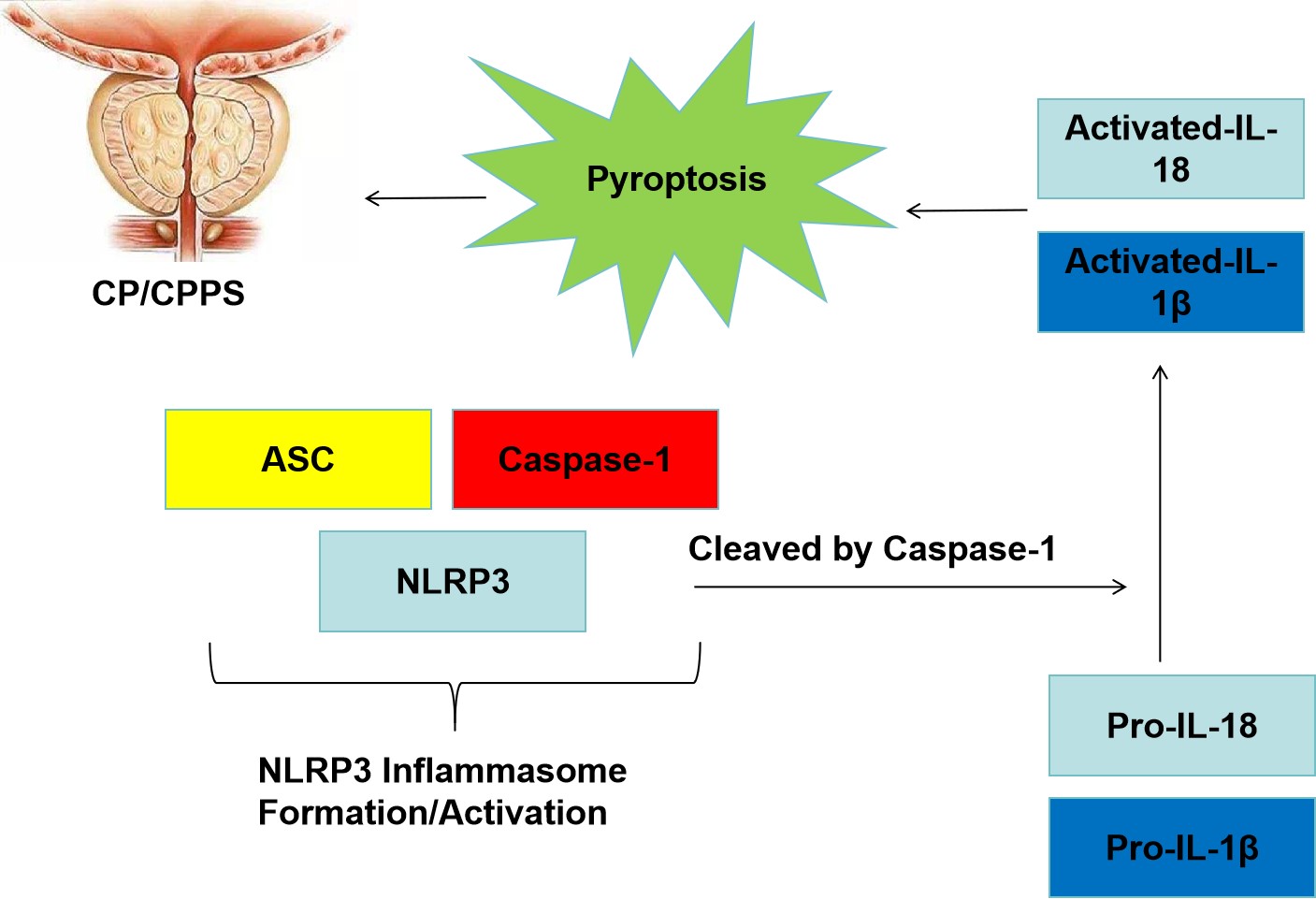
In recent years, pyroptosis has been implicated in the progression of CP/CPPS.7 The NLRP3 inflammasome, a multi-component protein complex, is composed of the NOD-like receptor family, pyrin domain containing 3 (NLRP3), the adaptor protein ASC, and the precursor enzyme pro-caspase-1.8 The NLRP3 protein is composed of 3 homologous domains: The C-terminal features a leucine-rich repeat, the central region houses a nucleotide-binding oligomerization domain, and the N-terminal is characterized by a pyrin domain.9 Intracellular NLRP3 levels are markedly low under resting conditions. Upon activation, NLRP3 inflammasomes serve as scaffolds that initiate the activation of caspase-1, promote the secretion of cytokines and lead to the induction of pyroptosis.10
The initiation of pyroptosis, a form of cell death, is primarily driven by the activation of the NLRP3 inflammasome. Research by Lu et al. on a rat model with hormone imbalance-induced chronic non-bacterial prostatitis revealed that the activation of the NLRP3 inflammasome sets off a cascade of inflammatory reactions within the prostate glands of these rats.11 Another study indicated that the regulation of cell autophagy in central neuropathic pain (CNP) might be governed by the NLRP3 inflammasome, which exerts its effects through the IL-6/STAT3 signaling pathway.12
Objectives
The collective findings from the aforementioned research suggest that pyroptosis, mediated by the NLRP3 inflammasome, could play a significant role in the development of CP/CPPS. However, no studies have investigated the presence of the NLRP3 inflammasome and its correlation with CP severity. Therefore, this research is the initial investigation into the expression of the NLRP3 inflammasome within the EPS of patients categorized under both CPPS IIIA and IIIB.
Methods
Study participsnts
From May 2022 to February 2024, we selected 167 patients with NIH-III CP, who exhibited pronounced symptoms of CP syndrome and fulfilled the NIH criteria for CP diagnosis, as participants.13 Based on the NIH classification system,13 85 cases were NIH-IIIA while the other 82 cases were NIH-IIIB. In addition, 80 benign prostatic hyperplasia (BPH) patients were also enrolled, and 80 healthy men receiving physical examinations without any clinical symptoms served as the control group. Within the healthy control group, no abnormalities were found during physical examinations, nor were any irregularities detected in the examination of EPS, urine routine tests, or in the bacterial cultures of urine samples taken before and after prostatic massage.
Each patient submitted a comprehensive medical profile that encompassed their history of medication use and aspects of their sexual health. Individuals with conditions similar to the presenting symptoms, such as diabetes, urethritis, epididymitis, varicocele, and diseases of the rectum and perianal area, were excluded from the study.
This study was approved by the ethical committee of Tianjin Medical University Baodi Hospital (Tianjin, China; approval No. 20220015). All patients signed written consent forms.
Sample collection and ELISA
Participants were instructed to refrain from sexual activity for a period of 3 to 5 days and to thoroughly cleanse and disinfect the glans penis and external urethral area prior to sample collection. Urine samples were collected both before and after prostate massage to conduct standard urinalysis and to assess urinary bacterial cultures.
The EPS was extracted by massaging the prostate gland through digital rectal examination. The patient adopted a knee-chest position and a bent position. The doctor wore gloves and applied paraffin oil, then slowly extended his fingers into the anus, touched the prostate, and then gently massaged the prostate. When EPS was secreted into the urethral opening, sterile slides were used to collect prostate fluid. The sample was placed into a cryovial and initially stored at –20°C for about 2 h. Subsequently, it was moved to a –70°C environment for long-term preservation until it was time to thaw it for examination.
Double-blinded quantitative measurements of EPS NLRP3 levels were assessed using a commercial enzyme-linked immunosorbent assay (ELISA) kit (cat. No. ab274401; Abcam, Cambridge, UK). Following the manufacturer’s protocol, standard samples of recombinant human NOD-like receptor family, pyrin domain containing 3 (NLRP3) were pipetted into each well of a microplate that had been precoated with an antibody specific to NLRP3. The wells were left to incubate at room temperature for 2 h before being washed 3 times with the washing reagent. Following this, each well received a pipetted amount of NLRP3 conjugate and was then incubated for an additional 2 h at room temperature. Following 3 wash cycles, the substrate solution was dispensed into every well. The microplate was subsequently incubated for 20 min at room temperature and shielded from light. To halt the reactions, stop solution was added, and the optical density (OD) of the contents was measured at 450 nm using a microplate reader (Infinite M200 PRO; Tecan, Männedorf, Switzerland).
The coefficient of variation (CV) within the assay for NLRP3 was 2.5%, while the variation between assays was less than 5.1%. The sensitivity of the ELISA kit for detecting NLRP3 is 1.63 ng/mL. Additionally, the detection range for the kit is between 4.38 ng/mL and 280 ng/mL. An optical density–concentration standard curve was created to ascertain the value of NLRP3. Expressed prostatic secretions interleukin (IL)-1β (cat. No. DLB50; R&D Systems, Minneapolis, USA) and IL-18 (cat. No. DL180; R&D Systems) concentrations were also examined. The CV within the assay was 5.5% for IL-1β and 5.9% for IL-18, while the CV between assays was 7.5% for IL-1β and 8.8% for IL-18.
Evaluation of disease severity
The disease severity of CP was evaluated using the Bergman scale as follows14: grade 0: No symptoms, normal sexual function; grade 1: Occasional lower abdominal pain, frequent urination at night or when feeling cold, frequent ejaculation after waking up or defecating, relief of symptoms after ejaculation, and normal sexual desire; grade 2: Persistent bloating in the lower abdomen, accompanied by abnormal urination and decreased libido, can induce ejaculation due to sexual intercourse, and symptoms may worsen after ejaculation; grade 3: Severe urination disorder, even acute urinary retention, accompanied by obvious decline in sexual desire, unable to conduct normal sexual behavior. Only patients with grade 1 or above were included in the study.
Evaluation of symptom severity
NLRP3 inflammasome levels were linked to clinical severity, as measured using 3 different scales: The numerical rating scale (NRS), the National Institutes of Health Chronic Prostatitis Symptom Index (NIH-CPSI) and the Danish Prostatic Symptom Score (DAN-PSS-1).
The NRS is a numerical rating standard. It is a common method of using numbers to represent pain intensity that is used in pain assessment and clinical research. The NRS typically ranges from 0 to 10, with 0 indicating the absence of pain and 10 signifying the most intense pain imaginable.15 When using the NRS, patients are usually asked to select a number to reflect their current level of pain. This can be achieved by verbally asking the patient or asking them to choose a number on their own. Typically, the definition of pain score may be as follows: 0: no pain; 1–3: mild pain; 4–6: moderate pain; 7–10: severe pain.
The NIH-CPSI is a dependable and user-friendly self-reporting tool extensively utilized in both scientific research and clinical trials. It evaluates pain symptoms (sum of items 1–4), urinary symptoms (sum of items 5 and 6) and impact on quality of life (QOL) (sum of items 7–9).16
The DAN-PSS-1 is derived from the patient’s self-reported data regarding the existence and intensity of 12 symptoms associated with the functions of bladder storage and emptying, known as the symptom score. In addition, the extent to which these symptoms are troublesome to the patient is also documented. The degree of bother and severity of each symptom are rated on a scale ranging from 0 to 3. A weighted score is derived by taking the product of the symptom score and bother score, resulting in an overall score for each symptom that could vary between 0 and 9. Therefore, a symptom is considered irrelevant if it does not cause the patient any distress. This occurs when the patient rates a symptom with a score of 0 (absent) or assigns a bother score of 0 (not bothersome).17
Statistical analyses
Data were expressed as mean ± standard deviation (SD) for normally distributed variables and as median with interquartile range (IQR) for variables not normally distributed. Variance homogeneity was examined using the F-test. The distribution data were tested using the Kolmogorov–Smirnov test. Student’s t-test or the Mann–Whitney U test was carried out between 2 groups, where appropriate. One-way analysis of variance (ANOVA) or the Kruskal–Wallis test was applied to statistically test the differences among ≥3 groups. Spearman’s correlation analysis was used to examine the association between NLRP3 expression and Bergman grade, NRS, NIH-CPSI, and DAN-PSS-1 scores, as well as IL-1β and IL-18 levels. Receiver operating characteristic (ROC) curve analysis was conducted to test the potential diagnostic value of NLRP3 expression with regard to Bergman grade. All statistical evaluations were carried out using GraphPad Prism v. 9.0 (GraphPad Software, San Diego, USA). The cutoff value of NLRP3 expression was calculated using the R language (R Foundation for Statistical Computing, Vienna, Austria). A p-value < 0.05 was regarded to be statistically significant.
Results
Demographic statistics
Table 1 displays the demographic data for the study cohorts. The distribution of age and body mass index (BMI) and showed no significant differences between the CP group and the controls. However, NLRP3 levels were significantly elevated in the CP subgroup (NIH-IIIA: 52.1 ±7.1 ng/mL; NIH-IIIB: 51.7 ±8.0 ng/mL) when compared to both the control group (15.4 ±2.9 ng/mL) and the BPH group (15.7 ±2.8 ng/mL) (one-way ANOVA, F = 69.35, degrees of freedom (df) = 326, p < 0.001 vs control, p < 0.001 vs BPH), as shown in Table 1 and Figure 1.
EPS NLRP3 levels with disease severity
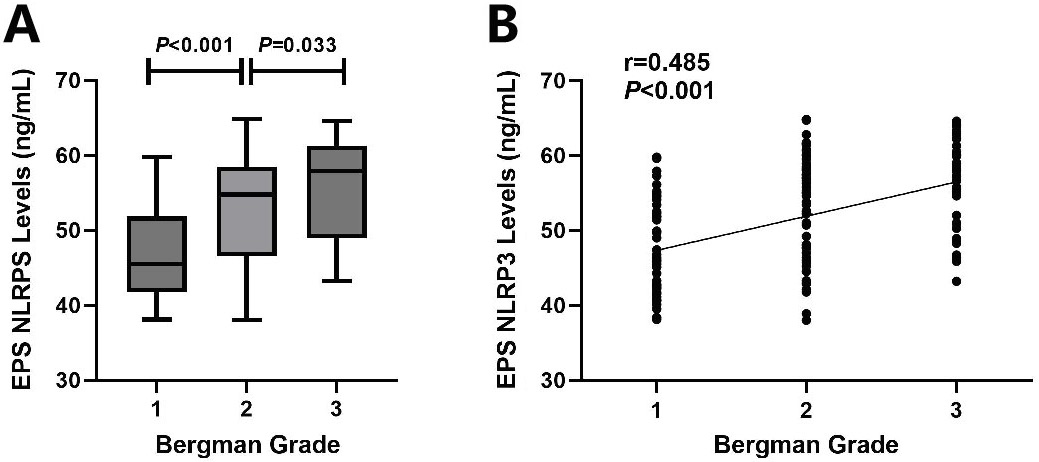
The EPS NLRP3 levels of the 167 NIH-III CP patients with different Bergman grades are shown in Table 1. NIH-III CP patients were divided into 3 groups based on Bergman grade. NIH-III CP patients included 56 patients with Bergman grade 1, 60 with Bergman grade 2 and 51 with Bergman grade 3. NIH-III CP patients with Bergman grade 3 showed significantly higher EPS NLRP3 concentrations compared to those with Bergman grade 2 (55.8 ±6.5 ng/mL vs 53.0 ±7.3 ng/mL, Mann–Whitney U test, U = 827.5, df = 109, p = 0.033) (Figure 2A). NIH-III CP patients with Bergman grade 2 demonstrated significantly higher EPS NLRP3 levels compared with Bergman grade 1 (53.0 ±7.3 ng/mL vs.46.8 ±5.9 ng/mL, Mann–Whitney U test, U = 1,168, df = 114, p < 0.001) (Figure 2A). The analysis using Spearman’s correlation revealed a positive relationship between NLRP3 levels in EPS and Bergman grade, with a correlation coefficient of 0.485, indicating a statistically significant association (Spearman’s correlation, r = 0.485, T(N-2) = 165, p < 0.001) (Figure 2B).
ROC curve analysis
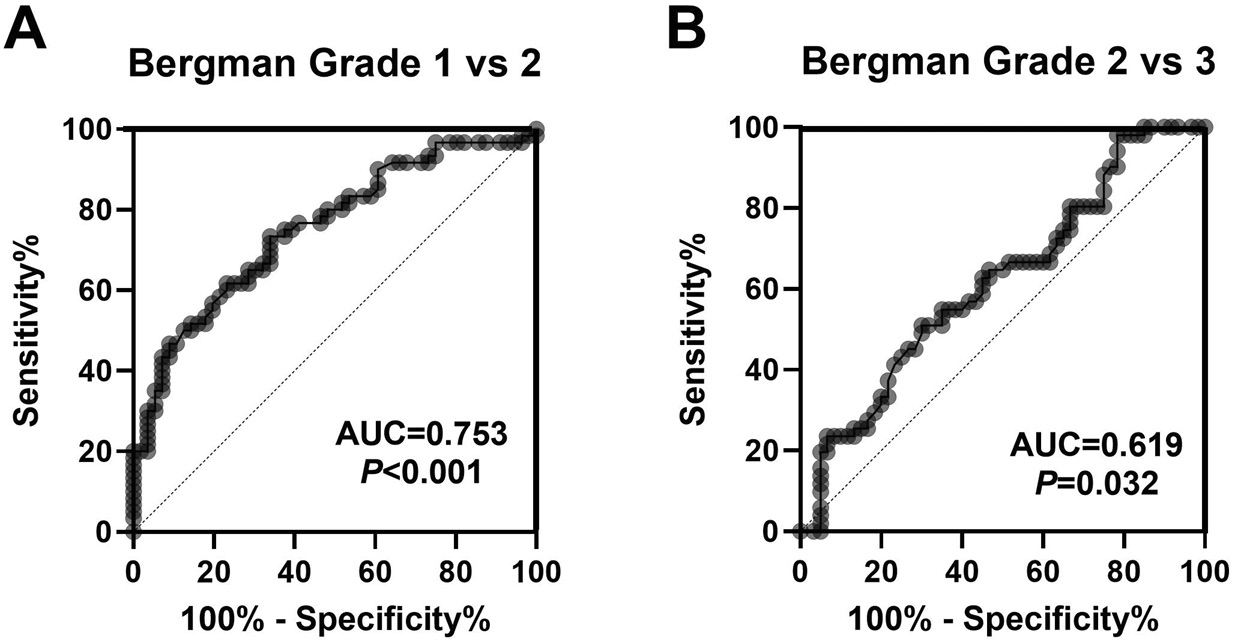
The ROC curve analysis was conducted to evaluate the diagnostic potential of NLRP3 in EPS as a possible biomarker for CP/CPPS. As shown in Figure 3, elevated NLRP3 levels showed a significant AUC in discriminating between Bergman grade 1 and Bergman grade 2 (AUC = 0.753, p ≤ 0.001) (Figure 3A) as well as Bergman grade 2 compared to Bergman grade 3 (AUC = 0.619, p = 0.032) (Figure 3B). These findings suggest that elevated EPS NLRP3 level may serve as a potential diagnostic marker to evaluate disease severity in CP/CPPS. The cutoff value of EPS NLRP3 expression was ≥55.25 ng/mL.
NLRP3 levels with symptom severity
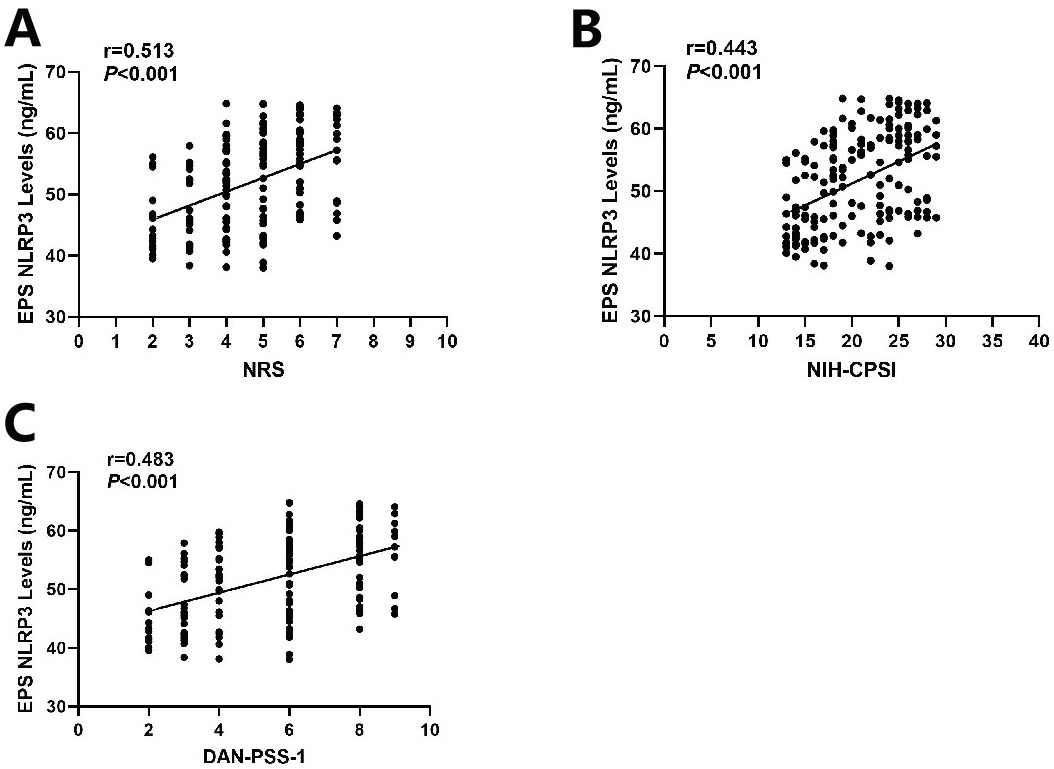
The association between NLRP3 levels in EPS and clinical outcomes was examined, as measured with NRS, NIH-CPSI and DAN-PSS-1 scores. We found EPS NLRP3 levels were positively associated with NRS scores (Spearman’s correlation, r = 0.513, T(N-2) = 165, p < 0.001) (Figure 4A), NIH-CPSI scores (Spearman’s correlation, r = 0.443, T(N-2) = 65, p < 0.001) (Figure 4B) and DAN-PSS-1 scores (Spearman’s correlation, r = 0.483, T(N-2) = 165, p < 0.001) (Figure 4C). These findings imply that CP/CPPS patients with higher EPS NLRP3 levels may suffer more severe clinical symptoms.
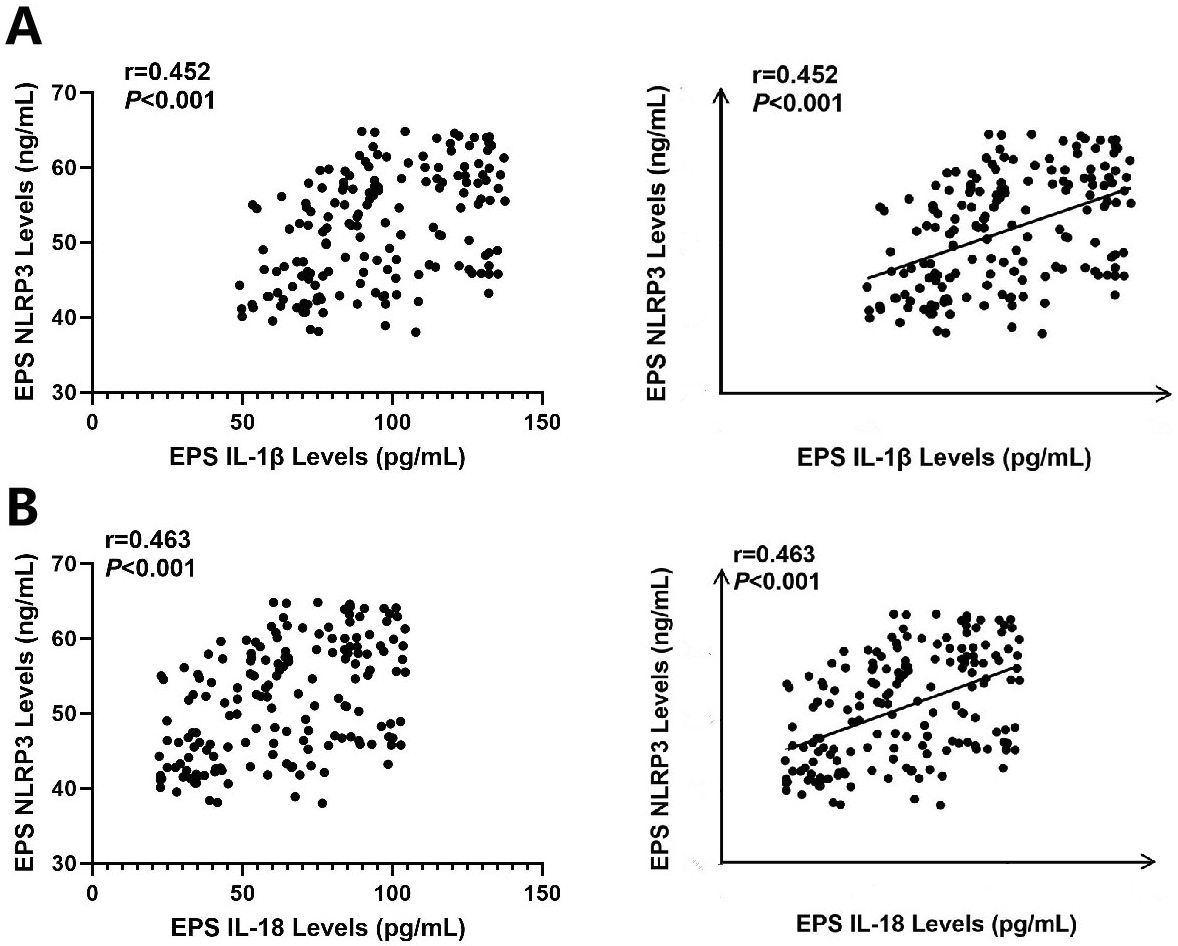
Interleukin 1 beta and IL-18 have been shown to be important markers in CP/CPPS. Interleukin 1 beta18 and IL-1819 have been implicated as potential triggers of CP progression. In this study, EPS NLRP3 levels were positively correlated with EPS IL-1β levels (Spearman’s correlation, r = 0.345, T(N-2) = 165, p < 0.001) (Figure 5A) and IL-18 levels (Spearman’s correlation, r = 0.326, T(N-2) = 165, p < 0.001) (Figure 5B).
Discussion
Our research examined the correlation between the expression of NLRP3 in EPS and the severity of CP/CPPS. For the first time, we have shown that NLRP3 levels in EPS are markedly higher in patients with CP/CPPS than in those with BPH or in healthy individuals. In addition, elevated NLRP3 levels in EPS exhibited a positive correlation with Bergman grade. The ROC curve analysis showed that an elevated EPS NLRP3 level may act as a potential indicator of CP/CPPS disease severity. In addition, EPS NLRP3 concentrations were positively correlated with NRS, NIH-CPSI and DAN-PSS-1. Finally, we found a positive association between the concentrations of NLRP3 in EPS and the levels of IL-1β and IL-18 in EPS. These results suggest that an increase in NLRP3 levels in EPS could indicate the severity of CP/CPPS.
Prostatitis is the 3rd most common disease of the prostate gland after BPH and prostate cancer.20 Though not a direct threat to life, prostatitis manifests through symptoms like erectile dysfunction, issues with the lower urinary tract and localized discomfort in the perineal or testicular regions, all of which can greatly influence a patient’s quality of life.21
The primary methods for diagnosing prostatitis include evaluating clinical signs, performing a thorough physical examination, assessing the white blood cell count (WBC), and conducting bacterial cultures of expressed prostatic secretions.22 However, the value of routinely analyzing leukocytes is a matter of debate, chiefly due to the lack of correlation between leukocyte count and symptom severity. Consequently, there is a pressing need to pinpoint specific biomarkers for the diagnosis of CP/CPPS.
Research in clinical settings has revealed that factors that modulate inflammation play a role in numerous pathological processes.23 Chronic inflammation typically correlates with intensified symptoms of the lower urinary tract in cases of prostate pathology.24
The NLRP3 inflammasome has been recognized as a crucial mediator of pathogenic inflammation across a range of inflammatory disorders and is linked to persistent inflammatory responses. Imbalances in the NLRP3 inflammasome have been implicated in an array of illnesses, such as multiple sclerosis,25 diabetes,26 atherosclerosis,27 Alzheimer’s disease,28 inflammatory bowel disease,29 and numerous other autoimmune conditions.30 In this study, we found that EPS NLRP3 levels were significantly upregulated in CP/CPPS patients, indicating that NLRP3 may participate in CP progression.
Pain is the main symptom of CP/CPPS. An increasing amount of evidence indicates that dysregulation of the NLRP3 inflammasome is associated with chronic pain conditions and plays a role in the development of chronic pain.31 Previous studies have demonstrated the involvement of the NLRP3 inflammasome in various pain conditions, such as complex regional pain syndrome type-I,32 CNP,33 inflammatory pain,34 cancer-related pain,35 and muscle pain,36 among others. Our findings also reveal a positive correlation between EPS NLRP3 levels and the severity of pain and dysfunction in CP/CPPS patients, indicating that higher EPS NLRP3 levels may be associated with more severe symptoms.
Mature IL-1β acts as a strong proinflammatory agent in various immune responses, playing a key role in attracting innate immune cells to infection sites and adjusting the activity of adaptive immune cells. On the other hand, mature IL-18 is crucial for stimulating interferon gamma (IFN-γ) production and enhancing the cell-killing functions of natural killer cells and T cells.37 The NLRP3 inflammasome is recognized as a vital element of the innate immune system, responsible for the maturation process of IL-1β and IL-18.38 Additionally, we observed a correlation between NLRP3 levels in EPS and the concentrations of IL-1β and IL-18 in the EPS of patients with CP/CPPS.
Limitations
There are some limitations that should be taken into account. First, this study was conducted at a single center and involved a relatively small cohort of patients, all of whom were of Han Chinese ethnicity. Therefore, multicenter studies with larger samples are needed in further research. Furthermore, the cross-sectional design of the study prevented the longitudinal tracking of NLRP3 inflammasome levels in correlation with disease progression, and as a result, the potential causal link between NLRP3 inflammasome activity and the severity of CP could not be comprehensively demonstrated. In addition, we evaluated only the NLRP3 inflammasome (pyroptosis classical pathway) in this study; investigation of other pyroptosis-related proteins involved in CP/CPPS may reveal more valuable information. Additionally, we did not investigate the potential mechanism that the NLRP3 inflammasome plays in CP/CPPS; other proteins that interact with the NLRP3 inflammasome may provide additional insight for understanding the pathogenesis of CP/CPPS.
Conclusions
The study represented a pioneering effort in demonstrating that levels of the NLRP3 inflammasome in EPS were significantly elevated in patients with CP/CPPS when compared to those with BPH and healthy controls. Moreover, the presence of NLRP3 in EPS showed a positive correlation with the onset of CP/CPPS. There was also a direct correlation between the concentration of NLRP3 and the intensity of symptoms, as well as with the levels of the inflammatory cytokines IL-1β and IL-18 in EPS. These findings support the hypothesis that elevated levels of NLRP3 could indicate the progression of CP/CPPS and suggest the potential of NLRP3 to serve as a biomarker for diagnosing this condition. However, further research is needed to elucidate the underlying mechanisms of NLRP3-mediated pyroptosis.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.13911503. The package includes the following files:
Supplementary Fig. 1. Cutoff value calculation of NLRP3 process using R.
Supplementary File 1. Normality check and variance homogeneity check.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.