Abstract
Background. Glycated hemoglobin A1c (HbA1c) is a well-established marker for glycemic control; recent studies suggest its potential role in cancer prognosis. Understanding the relationship between preoperative HbA1c levels and lymph node metastasis (LNM) in diabetic women with endometrial cancer (EC) can enhance prognostic assessments and treatment strategies.
Objectives. This study aimed to evaluate the predictive value of preoperative HbA1c levels for LNM in diabetic women with EC.
Materials and methods. A retrospective analysis was conducted on 233 diabetic women who underwent surgery for endometrioid-type EC at a tertiary referral hospital between 2010 and 2021. Data collected included demographic information, fasting plasma glucose, HbA1c levels, ultrasound findings, and tumor characteristics. Receiver operating characteristic (ROC) analysis was used to assess the predictive power of HbA1c levels for LNM. Univariate and multivariate regression analyses were performed to identify independent risk factors for LNM.
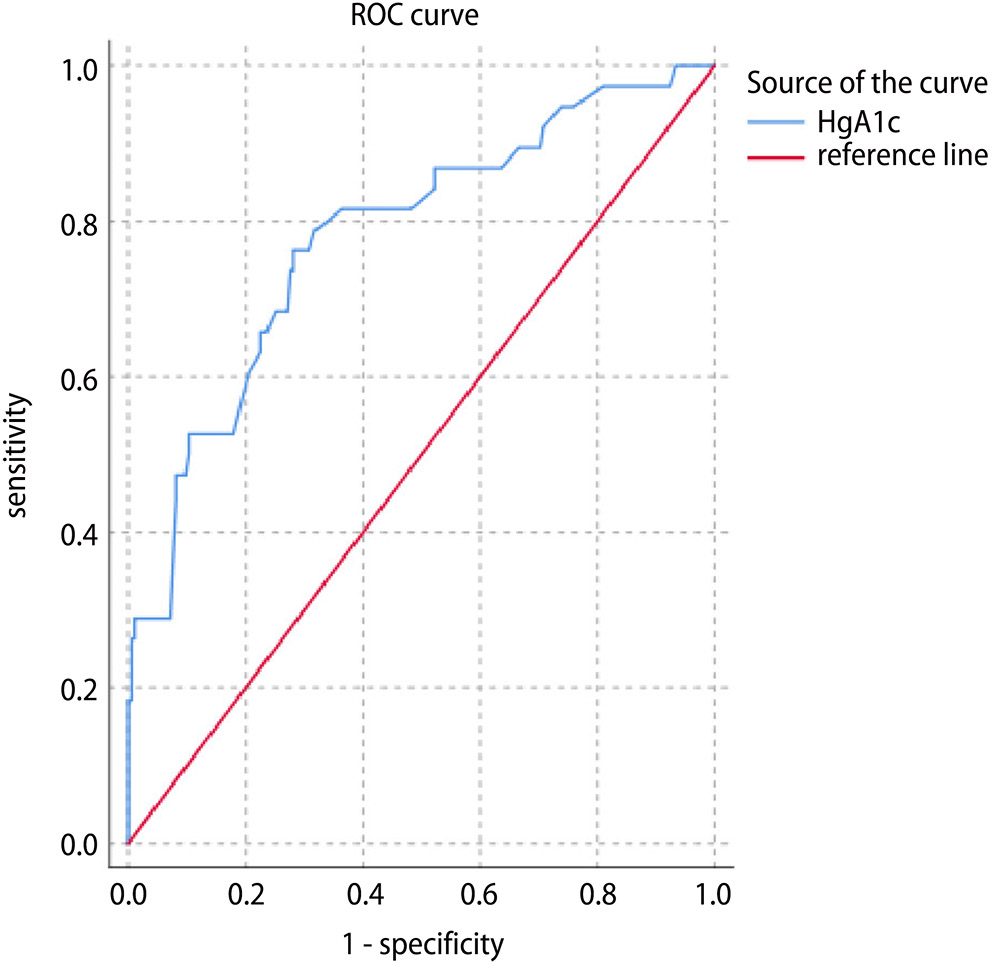
Results. The mean preoperative HbA1c level was 7.03 ±1.37%. A cutoff HbA1c level ≥7.26% demonstrated a sensitivity of 73.7%, a specificity of 72.3% and an area under the curve (AUC) of 0.781 for predicting LNM (p < 0.001). Significant correlations were found between HbA1c levels and endometrial thickness (r = 0.231, p < 0.001), primary tumor diameter (PTD) (r = 0.173, p = 0.008) and duration of diabetes (r = 0.203, p = 0.002). Multivariate analysis identified HbA1c level (odds ratio (OR) = 2.621, 95% confidence interval (95% CI): 1.722–3.987, p < 0.001), lymphovascular space involvement (LVSI) (OR = 19.193, 95% CI: 5.805–63.458, p < 0.001), body mass index (BMI) (OR = 1.095, 95% CI: 1.010–1.188, p = 0.029), and duration of diabetes (OR = 1.019, 95% CI: 1.001–1.301, p = 0.039) as independent risk factors for LNM.
Conclusions. Preoperative HbA1c levels serve as a significant predictor for LNM in diabetic women with EC. A cutoff HbA1c level ≥7.26% indicates higher risk of LNM. These findings underscore the importance of glycemic control in reducing cancer progression risks and improving the prognosis of diabetic patients with EC. Integrating HbA1c monitoring into preoperative assessments can help tailor personalized treatment strategies for better outcomes.
Key words: endometrial neoplasms, diabetes mellitus, glycated hemoglobin
Background
Diabetes mellitus is a significant global health problem, closely linked to the development and progression of various cancers.1 Among these, endometrial cancer (EC) stands out as the most common gynecological malignancy in industrialized countries.2 Epidemiological studies consistently report an increased risk of developing EC in diabetic women.3 The interplay between diabetes and cancer is complex, with hyperinsulinemia, hyperglycemia and chronic inflammation believed to be common pathways that may contribute to both conditions.4, 5, 6, 7, 8 Hyperinsulinemia, in particular, is thought to promote tumor growth through increased insulin-like growth factor (IGF) signaling, which can enhance cell proliferation and inhibit apoptosis.9, 10 Additionally, elevated blood glucose levels may lead to the formation of advanced glycation end-products (AGEs), which can trigger inflammatory pathways that promote carcinogenesis.11
Glycated hemoglobin A1c (HbA1c) is a well-established marker used to assess long-term glycemic control.12 Beyond its role in diabetes management, elevated HbA1c levels have gained attention in the context of cancer prognosis. Recent studies suggest that high HbA1c levels are associated with poorer outcomes in a variety of cancers, including breast, colorectal and pancreatic cancers.13, 14, 15, 16 In EC, studies have shown that elevated HbA1c levels may be associated with an increased risk of lymphovascular space invasion (LVSI) and a more advanced cancer stage at the time of diagnosis.5, 17, 18, 19 Additionally, HbA1c levels can be used in patients with endometrial intraepithelial neoplasia (EIN) to help diagnose concurrent EC, as higher HbA1c may indicate the presence of malignancy or a higher likelihood of progression to invasive cancer.20
Due to the shared pathophysiological mechanisms between diabetes and EC, it is crucial to explore how HbA1c levels might influence cancer prognosis. Higher HbA1c levels could be indicative of more aggressive disease and a higher likelihood of lymph node metastasis (LNM), a critical factor in determining disease stage and guiding treatment decisions.10, 12, 13, 14
Objectives
This study aimed to investigate the potential role of preoperative HbA1c levels as a predictor of LNM in diabetic women with EC. Given the established link between diabetes and the increased risk of EC, understanding how HbA1c levels correlate with tumor progression and metastasis could provide valuable insights into patient prognosis.
Clinically, this research aims to enhance risk assessment and treatment planning for diabetic women with EC. By identifying elevated HbA1c as a predictive marker for more aggressive disease and a higher likelihood of LNM, clinicians could better stratify patients, allowing for tailored treatment approaches. This could lead to more intensive monitoring and earlier interventions, ultimately improving survival rates and reducing the risk of recurrence.
Materials and methods
Study design
A retrospective analysis was conducted on 1,163 EC patients who received primary treatment at the Gynecologic Oncology Clinic of Ankara Etlik City Hospital, Turkey, between March 2011 and August 2023.
Participants
From this cohort, 233 patients were selected based on specific criteria, focusing on diabetic individuals with endometrioid-type EC according to final pathological results who had their HbA1c levels measured within 3 months before surgery. Patients with non-diabetic, non-endometrioid histology, irregular follow-up or incomplete data were excluded from the study.
Setting
Diabetes was diagnosed according to the American Diabetes Association (ADA) criteria: fasting plasma glucose level ≥126 mg/dL, HbA1c level ≥6.5% or a plasma glucose level ≥200 mg/dL 2 h post-glucose load.21
Cancer staging was classified based on the 2009 FIGO (Fédération Internationale de Gynécologie et d’Obstétrique) staging system.22 Surgical procedures for early-stage EC included: hysterectomy, bilateral salpingo-oophorectomy (adjusted for patient age), infracolic omental biopsy, and peritoneal washings tailored to specific histological subtypes. During the study period, lymphadenectomy cases in our clinic were determined using a frozen-section-based approach, and no sentinel lymph node procedures were performed on any EC cases. The decision to perform lymph node dissection was guided by the criteria described by Mariani et al.23 Specifically, patients with a greatest surface dimension ≤2 cm, myometrial invasion ≤50% and no intraoperative evidence of macroscopic disease were classified as low risk and treated with hysterectomy only, without lymph node dissection. For cases that did not meet these criteria, lymph node dissection was performed. All operations were performed by gynecologic oncologists.
All EC patients underwent intraoperative frozen section evaluation to assess histopathologic type, grade and depth of myometrial invasion. Surgical specimens were examined by gynecologic pathologists. Lymphovascular space invasion was characterized by the presence of tumor cells or clusters within the vessel walls, as identified through hematoxylin and eosin (H&E) staining.24
Adjuvant treatment followed the guidelines of the European Society for Medical Oncology (ESMO), the European Society of Gynecological Oncology (ESGO) and the European Society for Radiotherapy and Oncology (ESTRO).25 Disease-free survival (DFS) was calculated from the start of treatment to recurrence or last follow-up for non-recurrent cases, or to the date of death. Overall survival (OS) was measured from the time of diagnosis until either death or the most recent hospital admission.
Data sources
Patient follow-up records included: age, body mass index (BMI), menopausal status, fasting plasma glucose and HbA1c levels, ultrasound findings, surgery dates, LVSI status (based on postoperative pathology), cancer stage and grade, myometrial invasion, risk group classification, lymph node involvement, adjuvant therapy details, recurrence (location and timing), and deaths during follow-up.
Patients were monitored every 3 months during the 1st year and every 6 months during the 2nd year. Each visit included a physical examination, ultrasonography and review of laboratory parameters. Data were extracted from medical records.
Statistical analyses
All statistical analyses were conducted using IBM SPSS v. 23.0 (IBM Corp., Armonk, USA). Normality of the data was assessed using histograms and the skewness and kurtosis values (see Supplementary data). Normally distributed quantitative data presented as mean ± standard deviation (±SD) and qualitative data as frequency (percentage). Given the relatively large sample size of our study, we proceeded with t-tests for group comparisons. The use of t-tests was further justified by the central limit theorem (CLT), which supports the robustness of parametric tests under these conditions even with minor deviations from normality. To ensure reliability, we conducted sensitivity analyses to evaluate the potential impact of non-normality, confirming the appropriateness of the approach. Pearson’s χ2 test of independence was used for comparing categorical variables. Pearson correlation analysis was employed to examine relationships between normally distributed variables with linear relationships. Receiver operating characteristic (ROC) analysis was performed to assess the predictivity of HbA1c levels for LNM, calculating sensitivity, specificity and cutoff values. According to the literature, increased age, BMI, lymphovascular space involvement, myometrial invasion, and primary tumor diameter (PTD) are recognized risk factors for lymph node metastasis in EC patients. Based on this, we included these variables, along with HbA1c, in our multivariate logistic regression analysis. This selection aimed to address potential confounding factors and enhance the explanatory strength of the model. All data were analyzed with 95% confidence intervals (95% CIs), and statistical significance was assumed at p < 0.05.
Results
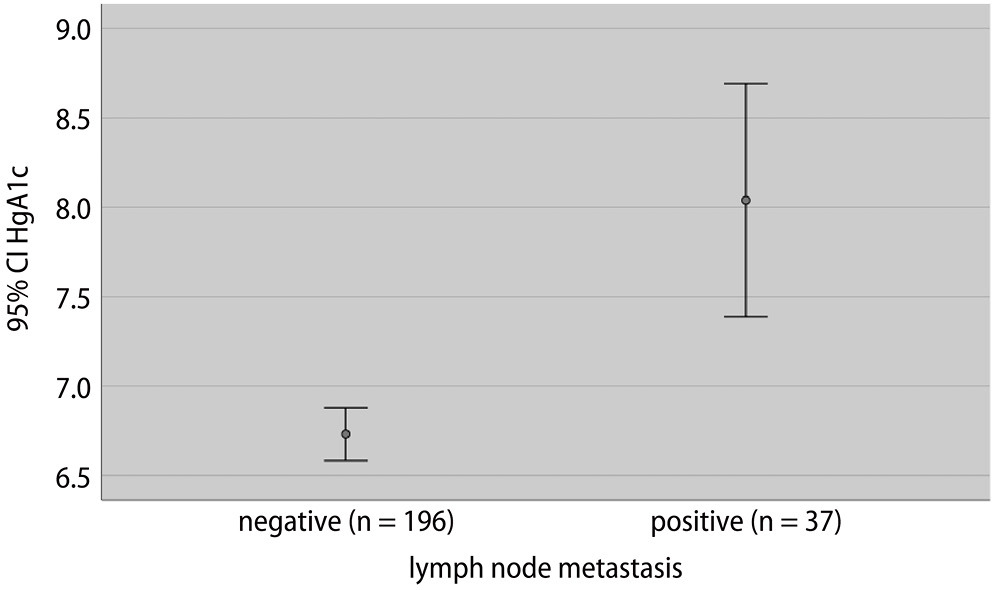
The median age at diagnosis was 57 years (range: 26–80 years), with a majority of women being postmenopausal (68.2%). Among these women, 195 (83.6%) exhibited high fasting glucose levels, while 213 (93.5%) were classified as overweight and obese based on their BMI. Table 1 provides a comprehensive summary of demographic characteristics, laboratory results and clinical outcomes of the study population. Additionally, the overview of tumor characteristics is presented in Table 1. Figure 1 shows the comparison of HbA1c levels according to LNM.
Table 2 compares patients based on preoperative HbA1c levels. Two groups were created according to ADA diabetes criteria of HbA1c (≥6.5%). There were no significant differences in age, BMI and fasting glucose values between the groups (p > 0.05). However, the group with HbA1c ≥ 6.5% showed a higher prevalence of LVSI and MMI ≥ 50% (44 vs 7, 57 vs 19; p < 0.001, p = 0.004, respectively) (Table 2). Notably, all patients with lymph node involvement and recurrence were in the HbA1c ≥ 6.5% group.
To evaluate the predictive value of preoperative HbA1c levels for LNM, a ROC analysis was performed. It revealed a sensitivity of 73.7% and a specificity of 72.3% at an optimal cutoff HbA1c value of 7.26%. The area under the curve (AUC) was 0.781 (p < 0.001), indicating good discriminatory power. These results suggest that preoperative HbA1c levels can reliably differentiate between patients with and without LNM (Figure 2).
Table 3 illustrates the correlation between HbA1c levels and various variables. Significant positive correlations were observed between HbA1c and endometrial thickness, PTD, tumor stage and grade, number of pelvic and para-aortic lymph nodes, and duration of diabetes (Table 3).
In multivariate logistic regression analysis, variables including age, BMI, PTD, HbA1c level, LVSI, and myometrial invasion were assessed as potential risk factors for LNM. The findings of the study indicated that HbA1c level, LVSI, and BMI were independent risk factors for LNM (odds ratio (OR) = 2.215, 95% CI: 1.657–3.539, p < 0.001; χ2 = 19.373, 95% CI: 5.892–63.699, p < 0.001; χ2 = 1.085, 95% CI: 1.006–1.171, p = 0.036; respectively). The results are summarized in Table 4.
Discussion
Our analysis of 233 cases of endometrioid EC revealed a significant association between elevated preoperative HbA1c levels and the risk of LNM, as well as between LVSI and the risk of LNM. Although the relationship between LVSI and LNM has been emphasized in numerous previous studies,23, 25, 26, 27, 28 our study suggests that diabetic EC patients with poor glycemic control, as reflected by higher preoperative HbA1c levels, are at an increased risk for LNM.
Several studies have shown that elevated HbA1c levels may increase the risk of various cancers, including EC.18, 29, 30, 31 A study by Senkaya et al. analyzed HbA1c levels and fasting glucose in 138 diabetic EC patients and found a significant association between HbA1c and LVSI, but no correlation with endometrial thickness. However, a low positive correlation between fasting glucose and endometrial thickness was noted.32 In contrast, our study identified significant correlations between HbA1c and several clinical parameters, including endometrial thickness, PTD, tumor stage and grade, lymph node involvement, and duration of diabetes, further supporting the role of glycemic control in influencing the invasive potential of EC.
Other studies have examined the impact of elevated HbA1c levels on the prognosis of various cancers. For instance, Cheon et al. demonstrated that elevated HbA1c levels were associated with worse survival in patients with advanced pancreatic cancer and diabetes.33 Similarly, HbA1c has been identified as a potential preoperative predictor of aggressive tumor profiles in diabetic patients with clinically localized prostate cancer.34 Siddiqi et al. found that elevated HbA1c is an independent predictor of aggressive colorectal cancer patterns.18 In contrast, Nief et al. found that in diabetic patients with endometrioid EC, glycemic control markers such as HbA1c, BMI and antihyperglycemic medications did not correlate with progression-free survival (PFS), while diabetic neuropathy was associated with an increased risk of recurrence.35 In a review of 55,475 EC patients, diabetes was linked to worse cancer-specific and OS, reinforcing the notion that poor glycemic control may influence EC prognosis.36 However, discrepancies in findings, such as those observed by Nief et al., highlight the need for further exploration of the role of HbA1c in cancer progression, particularly in EC.
Despite these findings, the relationship between HbA1c and cancer stage, grade and histological type in diabetic patients remains underexplored. Karaman et al. reported significantly higher HbA1c levels in EC patients compared to controls, but no significant correlation with tumor stage, grade or histological type.19 Similarly, Stevens et al. observed a trend of more advanced stages in patients with elevated HbA1c; however, HbA1c was not a strong predictor of EC prognosis.5 Folsom et al. showed that diabetes mellitus is associated with worse survival after EC, independent of tumor stage and grade.37 However, their study did not examine HbA1c or glucose levels. In contrast, our study identified a clear correlation between elevated preoperative HbA1c levels and both tumor stage and grade, as well as lymph node involvement, suggesting that HbA1c may serve as a valuable prognostic biomarker in diabetic EC patients.
The potential mechanisms linking diabetes, poor glycemic control and cancer progression are multifactorial.3 Hyperglycemia has been shown to create a favorable microenvironment for tumor growth by inducing oxidative stress, inflammation and angiogenesis.4, 5, 6, 7, 8 Glycolysis enhances glucose metabolism in cancer cells, supplying substrates essential for rapid proliferation.11 Han et al. suggested that glucose stimulates cell proliferation through several complex signaling pathways, supporting the hypothesis that elevated glucose levels may increase cancer risk.8 Furthermore, chronic hyperglycemia can elevate pro-inflammatory cytokine levels, facilitating tumor cell invasion and metastasis.6, 7, 8, 9 Hyperinsulinemia and insulin resistance, both hallmarks of diabetes, may also play pivotal roles by activating the insulin/IGF-1 pathway, which promotes cell growth and survival.9, 10 These mechanisms have been implicated in other malignancies, including breast, genitourinary and gastrointestinal cancers, and may also contribute to the aggressive behavior of EC in diabetic patients.38 Mitsuhashi et al. reported common impaired glucose metabolism and insulin resistance (IR) in patients with endometrial intraepithelial neoplasia (EIN) and EC.39
Limitations
The retrospective design and reliance on medical records for data collection are the main limitations of our study. As a result, the assessment of preoperative HbA1c’s impact on long-term survival was not feasible. Additionally, the follow-up of patients has not been completed yet, preventing the evaluation of HbA1c’s prognostic significance on survival. Future investigations are planned to address these limitations and provide a more comprehensive understanding of the relationship between preoperative HbA1c levels and long-term outcomes in diabetic EC patients.
Conclusions
The findings indicate that inadequate glycemic control, as demonstrated by elevated HbA1c levels, could be a significant prognostic factor, impacting tumor behavior and metastasis. This highlights the potential clinical application of HbA1c as a biomarker to guide treatment decisions, such as identifying patients at higher risk for aggressive disease who may benefit from more intensive surgical or adjuvant therapies. Additionally, the study contributes to the growing body of literature on the role of diabetes and glycemic control in cancer prognosis.
Declarations
Ethical approval was obtained from the relevant institutional review board (approval No. 26.07.2022-09/25), and informed consent was waived due to the retrospective nature of the study and the use of anonymized data. The study adhered to the principles outlined in the Declaration of Helsinki.
Supplementary data
The Supplementary materials are available at https://doi.org/10.6084/m9.figshare.28382411. The package includes the following files:
Supplementary Fig. 1. Histogram analysis of the age.
Supplementary Fig. 2. Histogram analysis of the BMI.
Supplementary Fig. 3. Histogram analysis of the glucose level.
Supplementary Fig. 4. Histogram analysis of the HbA1c.
Supplementary Fig. 5. Histogram analysis of the endometrial thickness.
Supplementary Fig. 6. Histogram analysis of the tumor diameter.
Supplementary File 1. T-test with variance homogeneity.
Supplementary File 2. Correlations and multicollinearity.
Supplementary File 3. Normality test results.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
ChatGPT was solely used to improve the language and readability of the text. After utilizing this tool, the authors thoroughly reviewed and edited the content as needed and take full responsibility for the content of the publication.