Abstract
Background. Vestibular rehabilitation therapy (VRT) is widely utilized to enhance balance and mitigate dizziness in patients with vestibular disorders. However, its overall effectiveness remains to be comprehensively assessed, particularly in the context of variability among studies.
Objectives. This study aimed to address the current need for a systematic evaluation of VRT’s efficacy.
Materials and methods. A meta-analysis was conducted using the “meta” and “dmetar” R packages to evaluate VRT’s efficacy. The analysis included statistical tools, such as Begg’s test, Egger’s test, Baujat plots, Galbraith plots, and influence analysis. Additionally, heterogeneity and outliers were assessed using generalized scatterplot smoothing (GOSH) diagnostics and clustering methods, including K-means, density-based spatial clustering of applications with noise (DBSCAN) and Gaussian mixture model (GMM).
Results. The meta-analysis examined the impact of VRT on the dizziness handicap inventory (DHI) and Berg balance scale (BBS). For the DHI, VRT resulted in a significant mean improvement of 7.63 points, despite high heterogeneity (I2 = 88%). Similarly, the BBS exhibited significant improvement, with a mean difference (MD) of –2.31 points in the fixed effects model, while the random effects model also suggested improvement, though with greater variability (I2 = 92%). Subgroup analysis identified outliers significantly influencing the results.
Conclusions. We showed that VRT significantly enhanced patient outcomes as measured with both the DHI and BBS. These findings provide strong evidence supporting VRT’s effectiveness, though the substantial heterogeneity underscores the need for further research to refine patient selection and intervention protocols. This study advances the understanding of VRT’s role in managing vestibular disorders and highlights the importance of addressing variability in future studies.
Key words: meta-analysis, Berg Balance Scale, Dizziness Handicap Inventory, vestibular rehabilitation training
Background
Vestibular rehabilitation therapy (VRT) is an essential intervention for individuals experiencing balance disorders and dizziness, particularly those recovering from conditions, such as stroke and multiple sclerosis (MS).1, 2, 3 Balance disturbances and dizziness are common complications for stroke survivors,4, 5 and VRT has been shown to play a significant role in alleviating these symptoms.6 Sana et al.7 demonstrated that VRT effectively enhances balance, reduces dizziness and improves gait in patients with subacute stroke. Furthermore, Ekvall Hansson et al.8 provided evidence through a pilot study that VRT can also benefit stroke patients with concomitant dizziness. In a comprehensive review, Meng et al.9 confirmed the positive effects of VRT on post-stroke balance and gait through a systematic review and meta-analysis.
Understanding the underlying neuroscience of vestibular compensation is critical to understating how VRT contributes to recovery.10, 11 Key neural structures, including the vestibular nuclei, cerebellum, thalamus, and cortical areas, play crucial roles in processing and compensating for vestibular deficits.12, 13, 14 The vestibular nuclei, located in the brainstem, are the primary centers for processing vestibular information and initiating the compensation process.15, 16 The cerebellum further refines these signals, which is essential for adjusting motor responses and maintaining balance.17 The thalamus serves as a relay station, transmitting vestibular information to the cerebral cortex, where higher-order processing takes place, facilitating spatial orientation and movement perception.18, 19 Neuroplasticity mechanisms involved in vestibular compensation include both peripheral and central changes. Peripheral adaptation refers to adjustments in the sensitivity of the hair cells in the inner ear, while central adaptation involves synaptic plasticity and reorganization within the vestibular nuclei and associated neural circuits.20, 21 Evidence suggests that repeated vestibular stimuli can enhance synaptic efficacy and promote new neural connections, aiding in functional recovery.22
Patients with MS also benefit from VRT. García-Muñoz et al.23 demonstrated that vestibular therapy significantly improves balance and reduces dizziness in MS patients through a systematic review and meta-analysis. Additionally, García-Muñoz et al.24 explored the combined use of immersive virtual reality and VRT, showing notable improvements in balance and dizziness rehabilitation for MS patients. Despite numerous studies on the effectiveness of VRT, including those conducted by Sulway and Whitney,25 Bush and Dougherty26 and Wang et al.,27 there remains a need for a more comprehensive evaluation of the effectiveness of VRT in different patient populations. Studies carried out by Moore et al.,28 Loftin et al.29 and Smółka et al.30 have reported promising results for VRT in treating persistent post-concussion symptoms and chronic unilateral vestibular dysfunction. However, there is still a lack of systematic assessment across diverse patient populations.
Objectives
The primary objective of this study was to address this gap by conducting a meta-analysis to systematically evaluate the effectiveness of VRT. This analysis aimed to consolidate findings from various studies, address heterogeneity and mitigate potential publication biases.
The aim was to identify relevant studies focusing on the Berg Balance Scale (BBS), Dizziness Handicap Inventory (DHI), and vestibular rehabilitation training, specifically within the context of randomized controlled trials (RCTs)
Methods
Search strategy
The search for this systematic review and meta-analysis was conducted over a period from January 2015 to May 2024. The primary databases searched included PubMed, Web of Science and Embase. The search terms used were a combination of key words and Medical Subject Headings (MeSH) terms to ensure comprehensive coverage. The key words included “Berg Balance Scale”, “Dizziness Handicap Inventory ”, “vestibular rehabilitation training”, “randomized”, and “RCT”. Boolean operators such as AND and OR were employed to refine and expand the search results appropriately. In addition to the database searches, supplementary records were identified through other reputable sources including Cochrane Library, China National Knowledge Infrastructure (CNKI) and WanFang databases. This multi-database approach was designed to capture a broad range of studies, including those published in languages other than English and those that might not have been indexed in the primary databases. We conducted a systematic review and meta-analysis following the Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISMA) guidelines to assess the efficacy of VRT. Studies were evaluated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach to rate the certainty of evidence for key outcomes, including balance improvement and reduction in dizziness severity. The systematic review and meta-analysis were independently conducted by 2 authors, J.S. and A.M., who carried out the literature search. In the event of any conflict during the process, M.R. acted as a 3rd author to make the final decision, ensuring consistency and accuracy in study selection.
Inclusion criteria
Study design: Only RCTs were considered eligible, regardless of participants’ gender, age, race, and country, as well as time of study. This broad inclusion ensured a diverse and comprehensive data analysis.
Intervention methods: Included studies had to use one of the following intervention methods:
– Balance and eye-movement exercises for persons with multiple sclerosis (BEEMS);
– Vestibulo-ocular reflex (VOR);
– Gaze and postural stability (GPS) retraining intervention;
– Dynamic motion instability system training (DMIST);
– Gaze stabilization exercises (GSEs);
– Turning-based treadmill training;
– Robot-assisted stair climbing training;
– Epley maneuver (canalith repositioning procedure);
– Gaze stability exercises combined with postural stability exercises;
– Outcome measures: The studies needed to report outcomes using either the DHI or the BBS, which are validated tools for assessing the effectiveness of vestibular rehabilitation training.
Exclusion criteria
– Non-RCTs: Any study that was not an RCT was excluded to maintain the rigor and reliability of the analysis.
– Small sample size: Studies with a patient number of 10 or less were excluded to ensure sufficient statistical power and the generalizability of the findings.
– Incomplete data: Studies where the full text was unavailable or the article data were incomplete were excluded from the analysis. This was done to ensure a comprehensive analysis.
– Non-English or Non-Chinese studies were not included.
– Similar intervention methods: Studies in which both interventions involved vestibular rehabilitation therapy were excluded to avoid redundancy and ensure the diversity of the interventions analyzed.
Data extraction
Data extraction was performed accurately and independently by J.S. and A.M., with M.R. resolving any discrepancies to ensure that the extraction process remained unbiased and valid, thereby reducing the potential for error.
Quality assessment
The quality of controlled intervention studies was evaluated using the Quality Assessment of Controlled Intervention Studies tool developed by the National Heart, Lung, and Blood Institute (NHLBI; National Institutes of Health (NIH), Bethesda, USA). This tool involves 14 criteria, including randomization, allocation concealment, blinding of participants, providers, outcome assessors, and baseline comparability between groups. It also helps evaluate drop-out rates, adherence to intervention protocols, consistency in applying other treatments, and the use of valid and reliable outcome measures. We used the tool to assess whether the studies clearly defined outcomes and subgroups at the time of study initiation, whether the sample size was sufficiently powered to achieved at least 80% statistical power, whether the studies used intention-to-treat populations, and whether the target population was representative. To aid in determining the quality of these studies, each was evaluated and classified as good, fair or poor according to these criteria to prevent overreaching conclusions that are based on weak and possibly biased research.
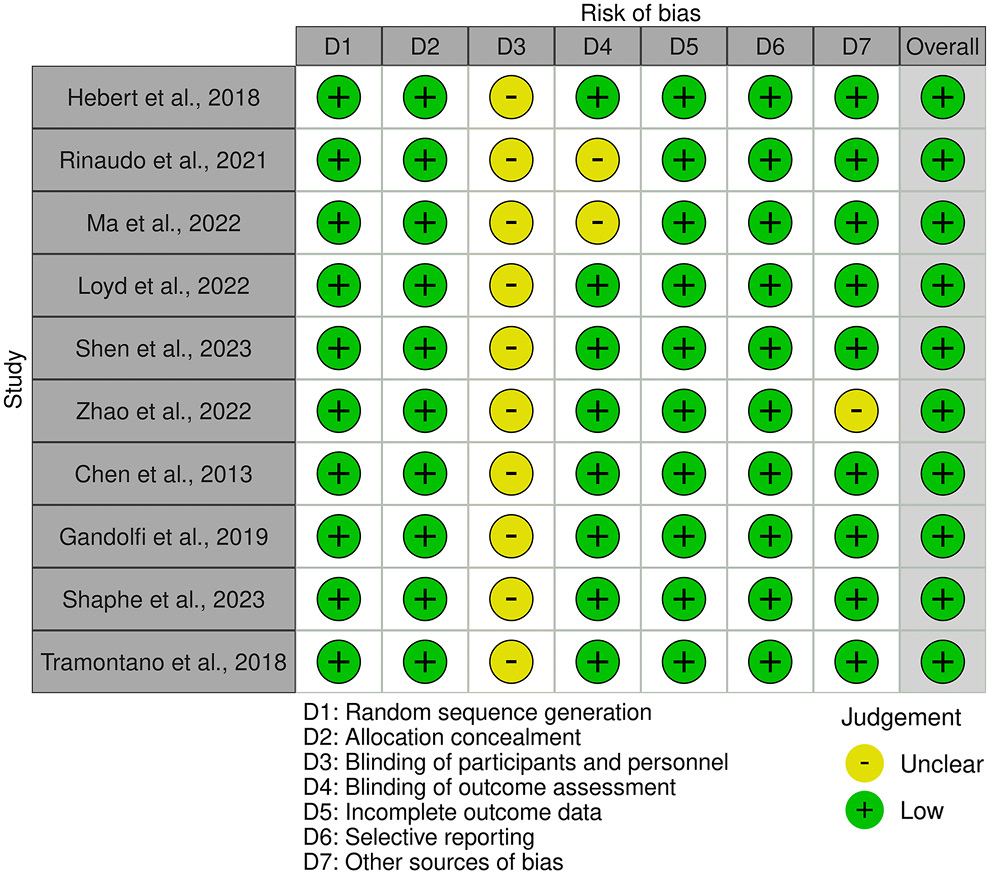
The risk of bias plot was generated using the online tool available at robvis (https://mcguinlu.shinyapps.io/robvis) by selecting the Generic_example template and uploading the relevant data. The review was performed by 2 reviewers with the use of recommendations from Cochrane Handbook for Systematic Review of Interventions.31 Items assessed included: 1) random sequence generation (selection bias), 2) allocation concealment (selection bias), 3) blinding of participants and personnel (performance bias), 4) blinding of outcome assessment (detection bias), 5) incomplete outcome data (attrition bias), 6) selective reporting (reporting bias), and 7) other biases. The risk of bias of the included studies was noted as low, unclear or high. The question of risk bias suggested that most of the studies were at low risk in most categories, although there was an optional risk of performance bias due to lack of information on blinding of participants and personnel.
Statistical analyses
The meta-analysis was conducted using R software v. 4.3.2 (R Foundation for Statistical Computing, Vienna, Austria) with specific packages and statistical methods designed to handle meta-analytic data. The primary packages used were R tools “meta”32 and “dmetar”.33
Begg’s test was employed to evaluate publication bias by examining the correlation between effect sizes and their variances.34 Egger’s test was used to assess funnel plot asymmetry through linear regression of the intervention effect estimates on their standard errors (SEs).35 It is important to note that Begg’s and Egger’s tests are typically recommended for use when the number of studies in a meta-analysis exceeds 10. This recommendation is based on the increased statistical power and reliability of these tests with larger sample sizes. In this study, these tests were used, but we recognized that with fewer studies, the results may have limited reliability and should be interpreted with caution.
Baujat plots were used to identify studies contributing most to the heterogeneity in the meta-analysis. Galbraith plots were utilized to detect outliers and influential studies by plotting standardized effect sizes against the inverse of their SEs.36 To supplement the findings from Begg’s and Egger’s tests, qualitative assessments were also performed using funnel plots, Baujat plots and Galbraith plots. These additional methods helped to identify potential sources of heterogeneity and the impact of individual studies on the overall results, providing a more comprehensive evaluation of potential biases.
Influence analysis was performed to determine the impact of each individual study on the overall meta-analysis results. This included leave-one-out analysis and various influence diagnostics such as rstudent, dffits and Cook’s distance.
Generalized scatterplot smoothing (GOSH) diagnostics were used to explore the robustness of the meta-analytic findings by assessing the distribution of effect sizes and heterogeneity across a large number of potential meta-analytic models.37
Clustering methods including K-means,38 DBSCAN39 and GMM40 were applied to identify patterns and outliers within the dataset. For the clustering analysis, specific hyperparameters were selected for each method. In the K-means algorithm, the number of clusters was determined using the Elbow method and Silhouette score, with “k-means++” initialization and 10 initializations to avoid local minima. For DBSCAN, the epsilon (ε, eps) parameter was selected using the k-distance graph to find the “elbow” point, with a minimum sample parameter set to 5. In the GMM, the number of components was based on the Bayesian information criterion (BIC) and Akaike information criterion (AIC), and the covariance type was set to “full”. These parameters were optimized using cross-validation and specific data characteristics. In case of the high heterogeneity in the outcomes, further analysis was conducted to explore potential sources of this variability. While the K-means algorithm indicated a different clustering pattern compared with DBSCAN and GMM, this discrepancy was attributed to K-means’ assumption of spherical clusters with uniform sizes, which was not suitable for our dataset’s characteristics. The final clustering interpretation prioritized the results from DBSCAN and GMM, which better accounted for clusters of varying densities and shapes. While GMM provided a probabilistic framework for cluster assignment, DBSCAN was particularly effective at identifying noise and outliers.
Notably, the meta-analysis utilized both fixed-effect and random-effects models. However, the final model was the random-effects model, aligning with the guidelines from the Cochrane Handbook for Systematic Reviews of Interventions.31 The decision to use the random-effects model was made a priori, based on the assumption that the included studies represented a random sample from a larger population, acknowledging the variability in effect sizes due to differences in study populations, interventions and other factors. This approach ensured that the meta-analytic findings were generalizable and robust. Meta-regression and subgroup analyses were employed to identify factors contributing to heterogeneity. Meta-regression was performed to assess the impact of study-level covariates, such as intervention type, follow-up time and patients’ characteristics on effect sizes. Subgroup analysis was conducted based on intervention methods, geographical locations of studies and quality assessment ratings.
Sensitivity analysis
To assess the robustness of meta-analysis results and the potential impact of high-risk bias studies, sensitivity analysis was conducted. This involved excluding studies that were rated as having a high risk of bias in any of the key domains assessed. The risk of bias for each study was evaluated using the Cochrane Collaboration’s tool, considering factors such as random sequence generation, allocation concealment, blinding, incomplete outcome data, and selective reporting. For the sensitivity analysis, we re-ran the meta-analysis excluding studies categorized with a high risk of bias in the domains of random sequence generation, allocation concealment, and blinding of participants and personnel. This approach aimed to determine whether the exclusion of these studies significantly altered the overall effect estimates and the conclusions drawn from the primary analysis.
Results
Selection and inclusion of studies
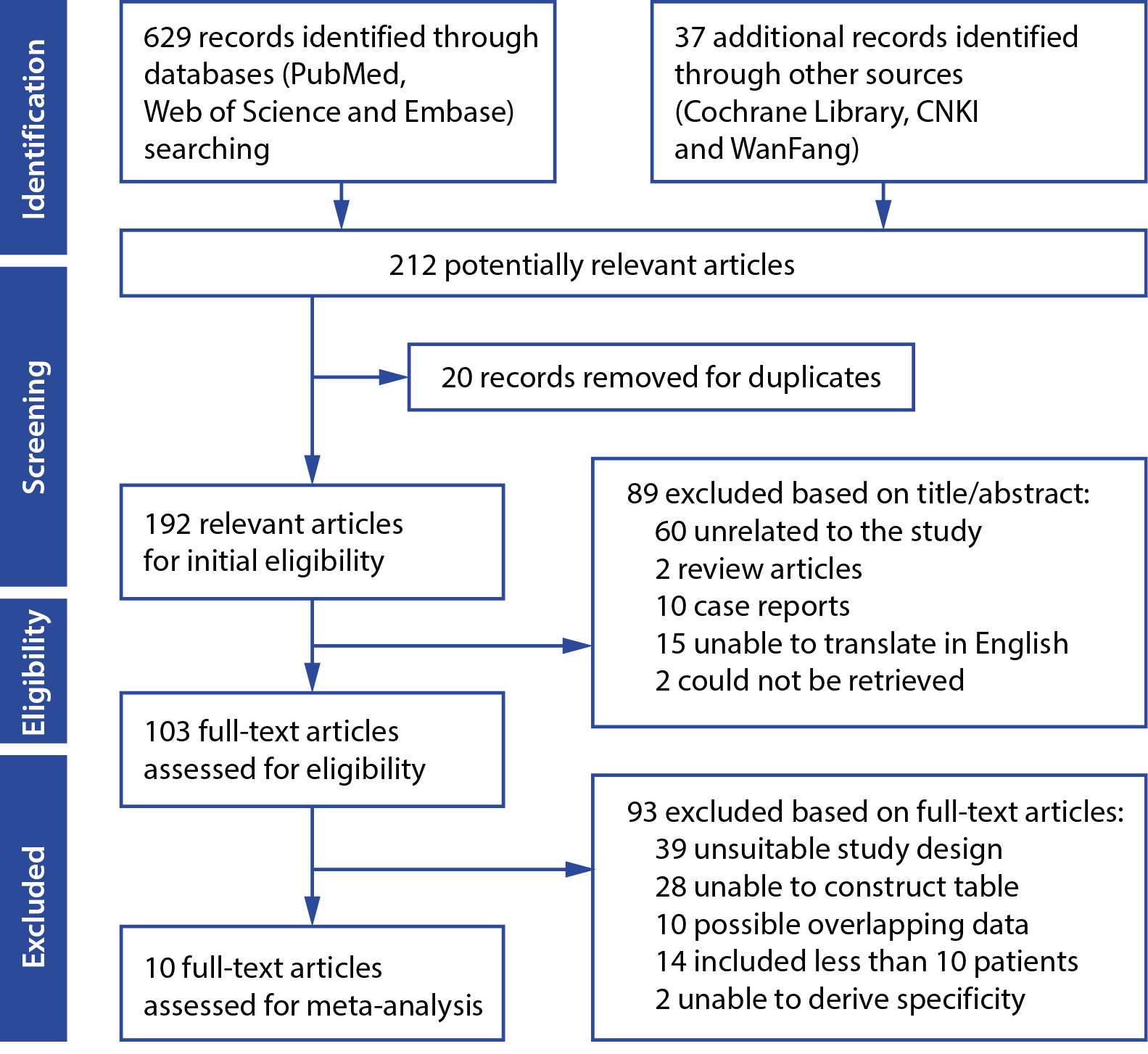
A comprehensive search strategy identified 629 records through database searches in PubMed, Web of Science and Embase. An additional 37 records were found through other sources, including Cochrane Library, CNKI and WanFang databases, leading to a total of 666 potential articles. After removing 20 duplicate records, 212 articles were considered for further examination. Title and abstract screening resulted in the exclusion of 89 articles for reasons such as irrelevance to the study, review articles, case reports, non-English or non-Chinese studies, and retrieval issues. This left 103 full-text articles for eligibility assessment. During this assessment, 93 articles were excluded due to unsuitable study designs, overlapping data, small sample sizes, or inability to derive specificity. Ultimately, 10 full-text articles met the inclusion criteria and were included in the meta-analysis (Figure 1). A summary of the study selection and inclusion process is provided in Table 1.24, 25, 26, 27, 28, 29, 30, 31, 32
Characteristics of the included studies
The included studies examined various clinical conditions related to vestibular and balance disorders and were conducted across multiple countries, including the USA,41, 42 Australia,43 China,44, 45, 46 Taiwan,47 Italy,48, 49, 50 and Saudi Arabia.51 All studies were RCTs, addressing conditions such as multifaceted vestibular disorders, peripheral vestibular hypofunction, recurrent vertigo, multiple sclerosis, stroke, and diabetic patients with posterior benign paroxysmal positional vertigo.
Participants ranged in age from their late 30s to late 60s, with follow-up periods extending from 1 week to 14 weeks, allowing for evaluation of both short-term and longer-term effects. The interventions studied included specialized exercises and training programs, such as balance and eye-movement exercises for persons with multiple sclerosis (BEEMS), vestibulo-ocular reflex (VOR) exercises, etc. These were assessed using validated scales such as the DHI and BBS, ensuring reliable outcome measurement.
Most studies demonstrated good methodological quality, with a few rated as fair, and several provided clinical trial identifiers, adding to the credibility of the research. The GRADE assessment indicated moderate certainty of evidence for reducing dizziness severity and low certainty for balance improvement, primarily due to inconsistencies and imprecision in study findings. Overall, the studies collectively provide strong evidence supporting the effectiveness of various interventions for vestibular and balance disorders, although there is some variability in the certainty of the evidence, particularly concerning balance improvement outcomes. The overall quality of the research is high, with most studies demonstrating rigorous design and execution. Table 1 presents characteristics of the included studies.
Risk of bias in the included studies
The bias risk analysis of the studies showed that the majority had a low risk of bias across most categories. However, there was an unclear risk of performance bias due to the lack of information on the blinding of participants and personnel in all studies (Figure 2).
Effectiveness of VRT in improving patient outcomes on the DHI scale
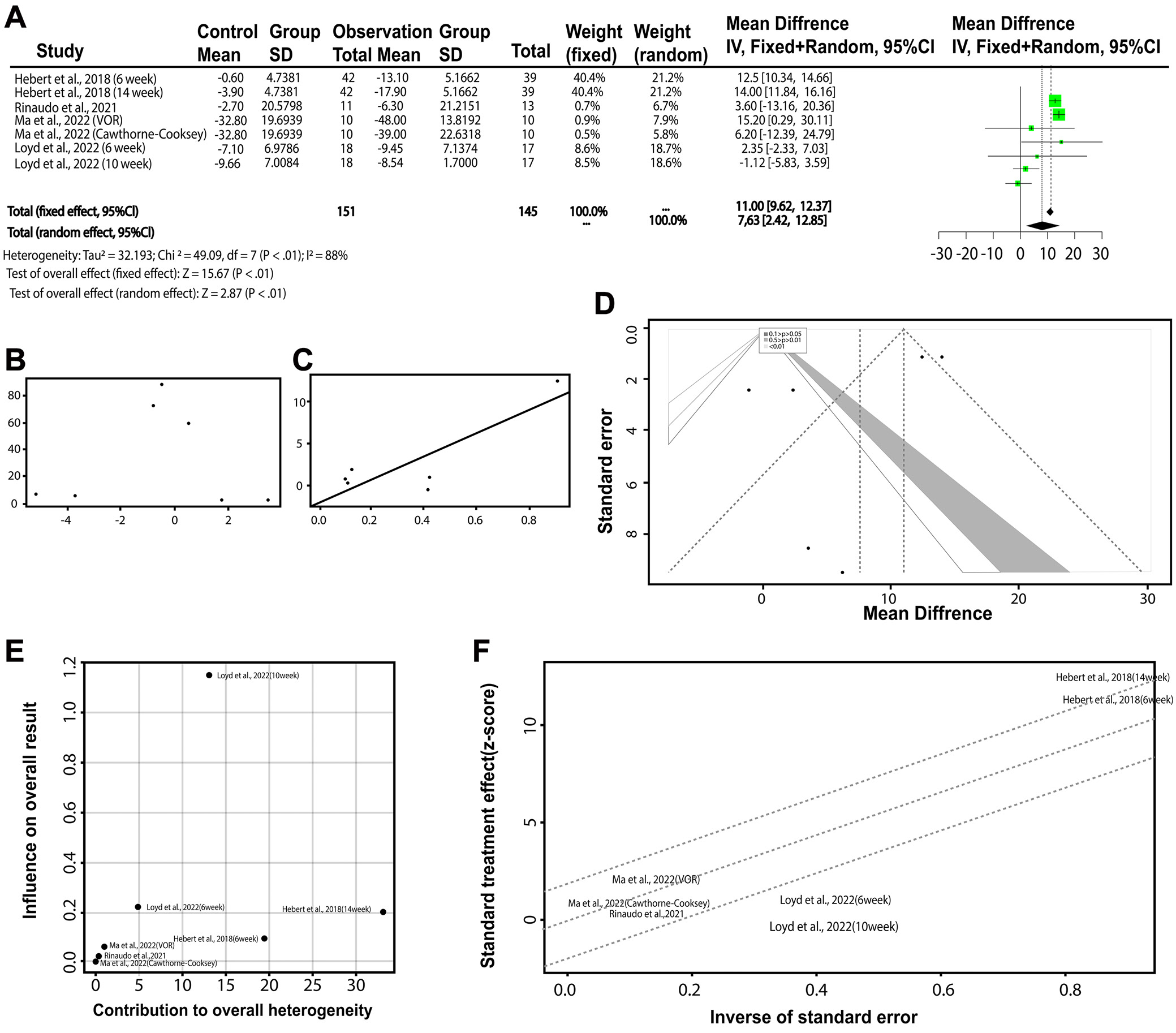
The primary outcome assessed was the effectiveness of VRT in improving patient outcomes using the DHI scale. Due to the substantial variability across the studies, a random effects model was used for analysis. The results indicated that VRT significantly improved patient outcomes compared to controls (Figure 3).
To assess publication bias, both Begg’s and Egger’s tests were conducted, neither of which suggested significant publication bias. However, the analysis of the funnel plot and Baujat plot highlighted potential influences on the distribution of study effects, with some studies showing distinct contributions to the overall heterogeneity. The Galbraith plot further illustrated patterns in the SEs and effect sizes among the included studies (Figure 3). Overall, the findings suggest that VRT is effective in improving patient outcomes as measured with the DHI scale. While there is significant heterogeneity across the studies, the results consistently support the benefits of VRT. Further analyses also indicate minimal risk of publication bias, although certain studies have a disproportionate influence on the overall results. Summary of VRT’s effectiveness (DHI scale) is presented in Table 2.
GOSH diagnostics analysis with DHI datasets
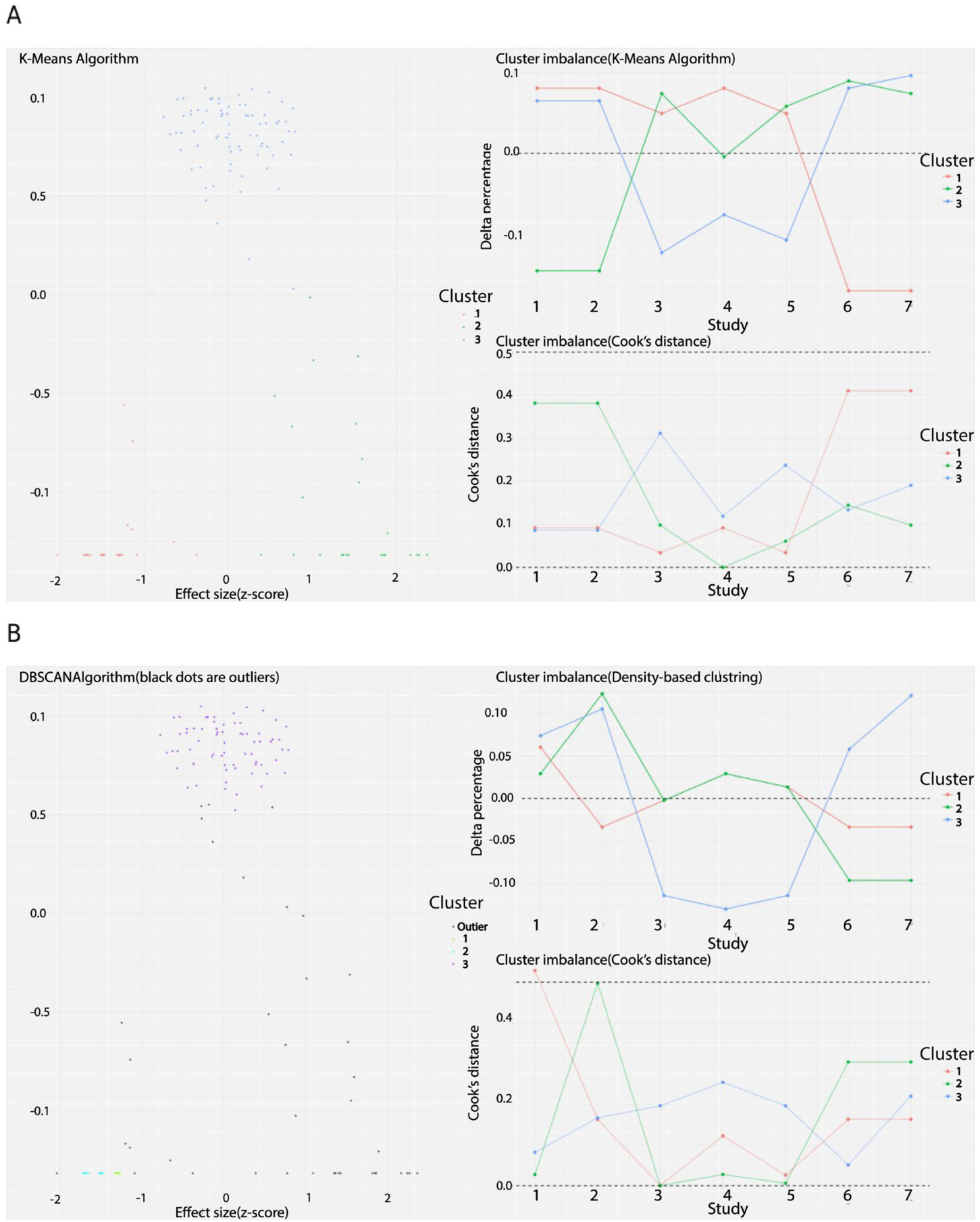
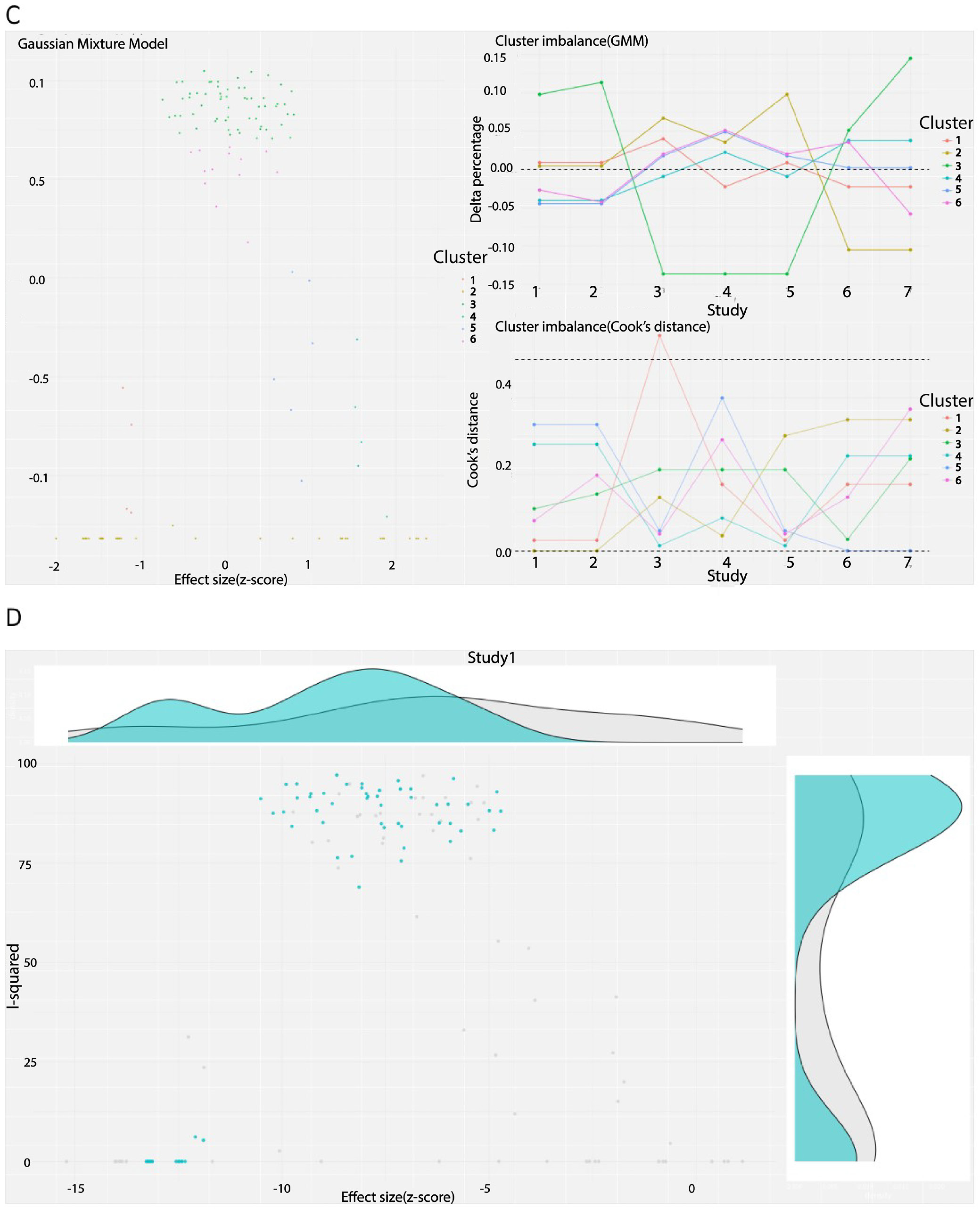
The K-means clustering method identified 3 distinct clusters within the dataset, as shown in Figure 4A. This method did not detect any outliers, suggesting that the data points were well-separated into the 3 clusters without any significant anomalies. In contrast, the DBSCAN method also identified 3 clusters, as depicted in Figure 4B. However, in contrast to K-means, DBSCAN detected 1 outlier, Hebert et al.46 (6-week interval). The GMM provided a more granular clustering solution, identifying 6 distinct clusters as illustrated in Figure 5A. Similar to the DBSCAN method, the GMM also flagged the study by Hebert et al.46 (6-week interval) as a potential outlier. The GOSH Diagnostics analysis also visualized in this Figure 5B highlights that Hebert et al.46 (6-week interval) is identified as an outlier. The scatter plot shows the distribution of studies with respect to effect size and I2, with the results of Hebert et al. deviating significantly from the main cluster. The density plots above and to the right further emphasize the anomalous distribution of the results of Hebert et al. compared to other studies.46 Consequently, the clustering analysis revealed consistent grouping across different methods, with some variability in the granularity of the clusters. Notably, the study by Hebert et al.46 (6-week interval) emerged as an outlier in multiple analyses, suggesting it may have unique characteristics that set it apart from the other studies in the dataset. These findings underscore the importance of using multiple clustering methods to capture different dimensions of data structure and to identify potential outliers effectively.
Influence analysis with DHI datasets
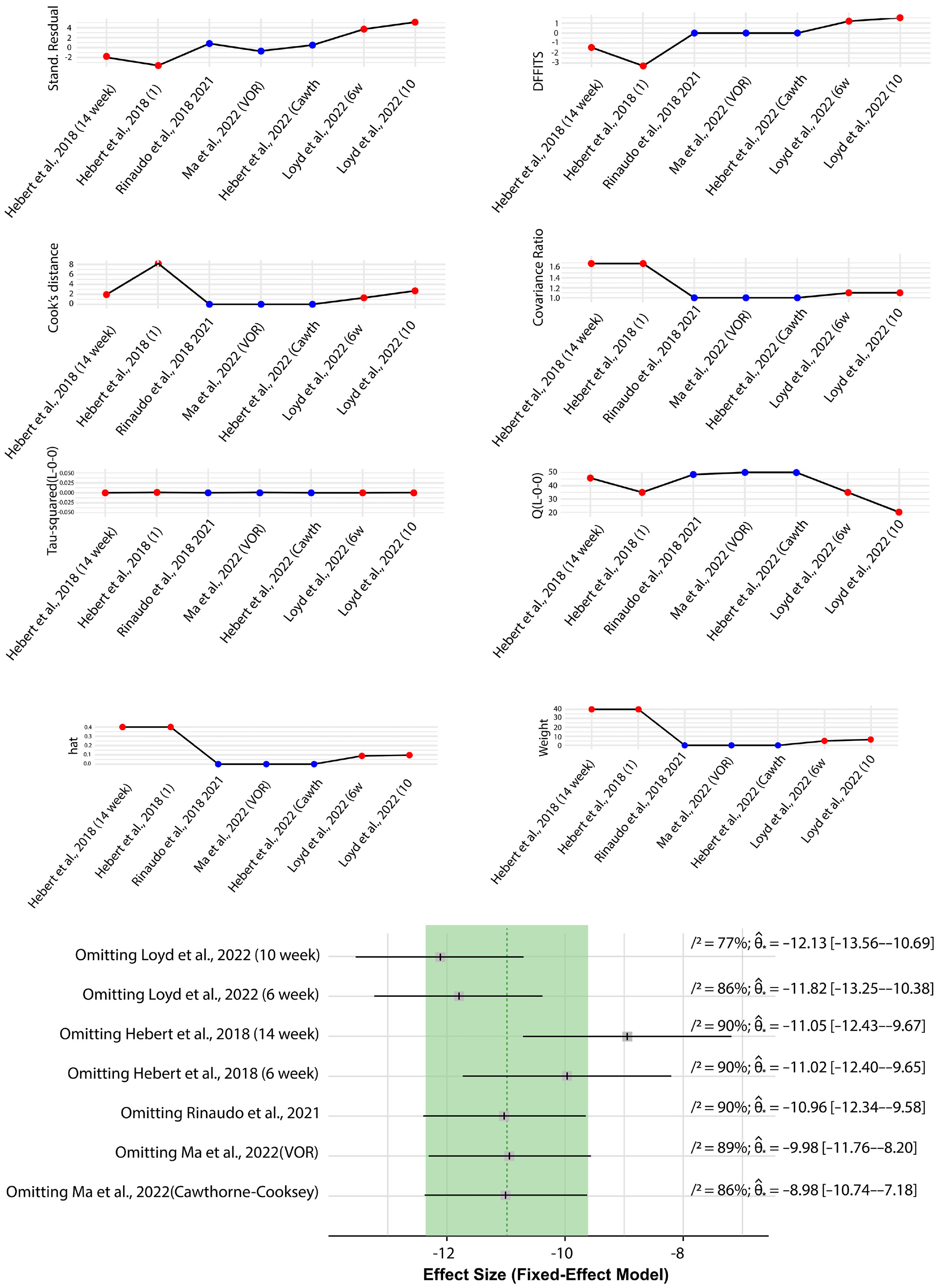
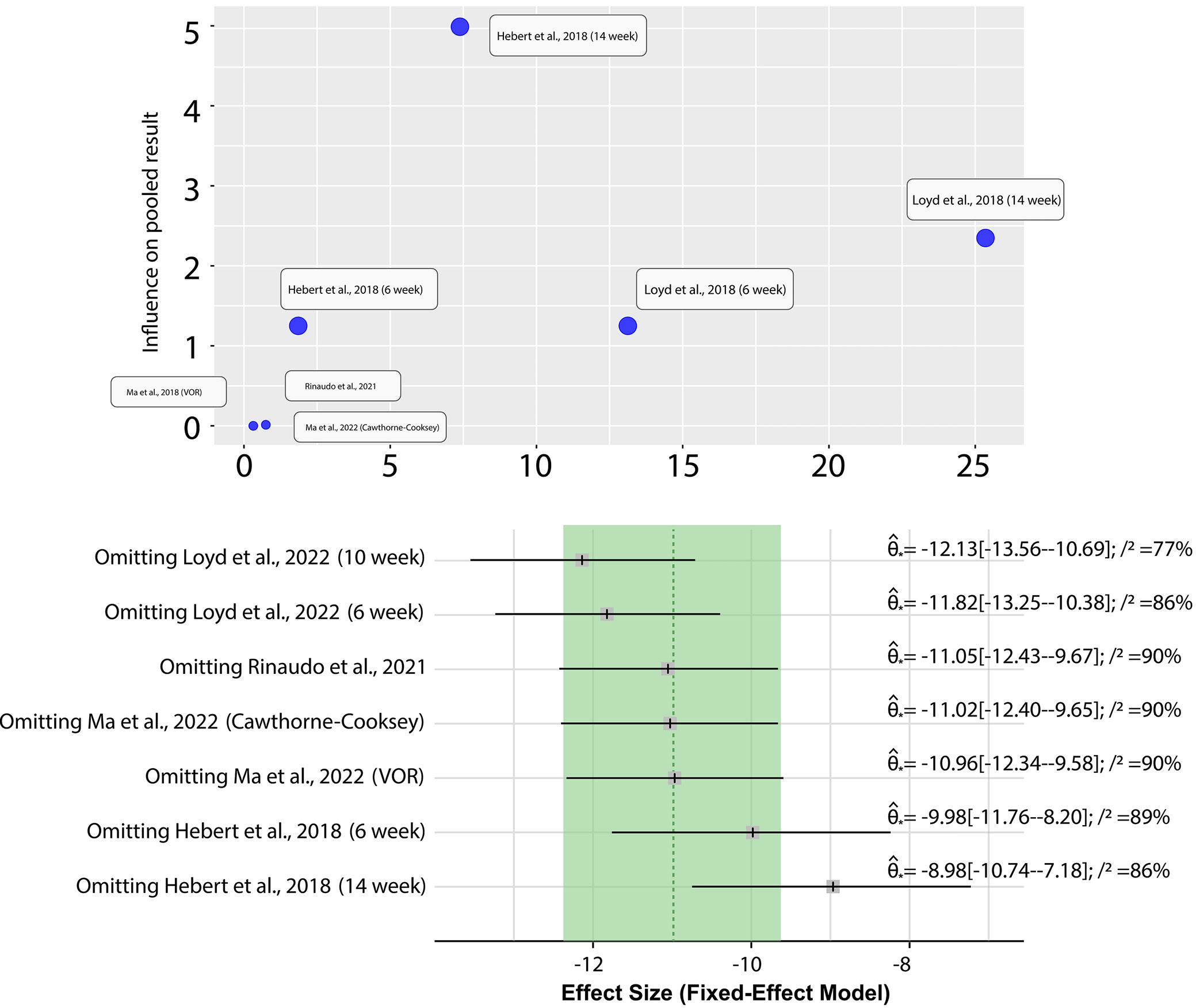
The leave-one-out analysis explored how the omission of individual studies affected the overall effect size and heterogeneity in the dataset. Notably, omitting studies such as Loyd et al.49 (10-week and 6-week intervals) and Hebert et al.46 (14-week and 6-week intervals) led to significant changes in both effect size and heterogeneity (Figure 6, Figure 7). These findings indicate that these studies have a substantial impact on the overall meta-analysis results. The Influence Diagnostics further highlighted the considerable influence of these studies, with specific metrics showing their effect on the stability of the regression model. The Baujat Diagnostics also supported these findings, revealing that omitting these key studies contributed to significant reductions in heterogeneity (Figure 6). Overall, the influence analysis underscores the pivotal role of certain studies, particularly those by Loyd et al.49 and Hebert et al.,46 in shaping the outcomes of the meta-analysis. Their omission leads to notable shifts in both effect size and heterogeneity, suggesting that these studies are influential in determining the overall conclusions of the analysis. These results highlight the importance of carefully considering the impact of individual studies in meta-analyses, especially when they have a significant influence on the overall findings (Figure 7).
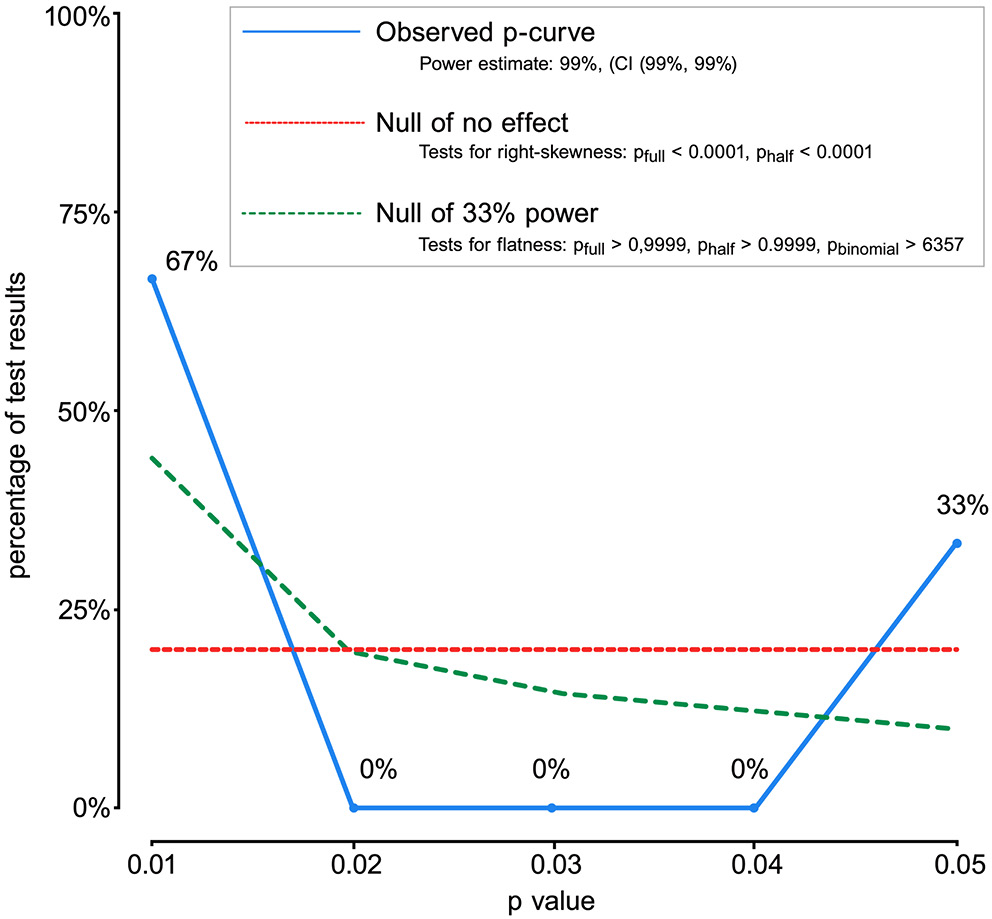
P-curve analysis with DHI datasets
The P-curve analysis, depicted in Figure 8, was conducted to assess the evidential value of the findings across the included studies. The analysis revealed a significant right-skewness in the P-curve, indicating that the results are not only statistically significant but also possess strong evidential value. The distribution of p-values further supports the robustness of the findings, with a high-power estimate suggesting that the dataset is well-suited to detect true effects. In summary, the P-curve analysis confirms the presence of substantial evidential value in the dataset, reinforcing the reliability of the findings. The significant right-skewness and high-power estimate indicate that the results are not driven by chance, but rather reflect genuine effects. This analysis contributes to the overall robustness of the meta-analysis, underscoring the credibility of the conclusions drawn from the DHI datasets.
Results of meta-regression and subgroup analyses
The meta-regression analysis identified key study characteristics that influenced the effectiveness of interventions on DHI and BBS outcomes. Specifically, the type of intervention and follow-up time significantly affected DHI outcomes, with gaze stabilization exercises showing greater effectiveness, while longer follow-up periods were associated with reduced treatment effects. For BBS outcomes, geographical location and quality assessment ratings emerged as significant factors, with studies from Asia and those rated as “good” showing more substantial improvements in balance. Subgroup analyses corroborated these findings, revealing that studies using gaze stabilization exercises had lower heterogeneity and higher mean differences compared to other interventions. Additionally, studies conducted in Asia demonstrated greater improvements in balance than those from Western countries. Collectively, the meta-regression and subgroup analyses highlight the importance of specific study characteristics in determining the effectiveness of interventions for DHI and BBS outcomes. Interventions such as gaze stabilization exercises and studies conducted in Asia or rated as “good” quality were associated with better outcomes. These findings emphasize the need to consider these factors when designing and interpreting future studies.
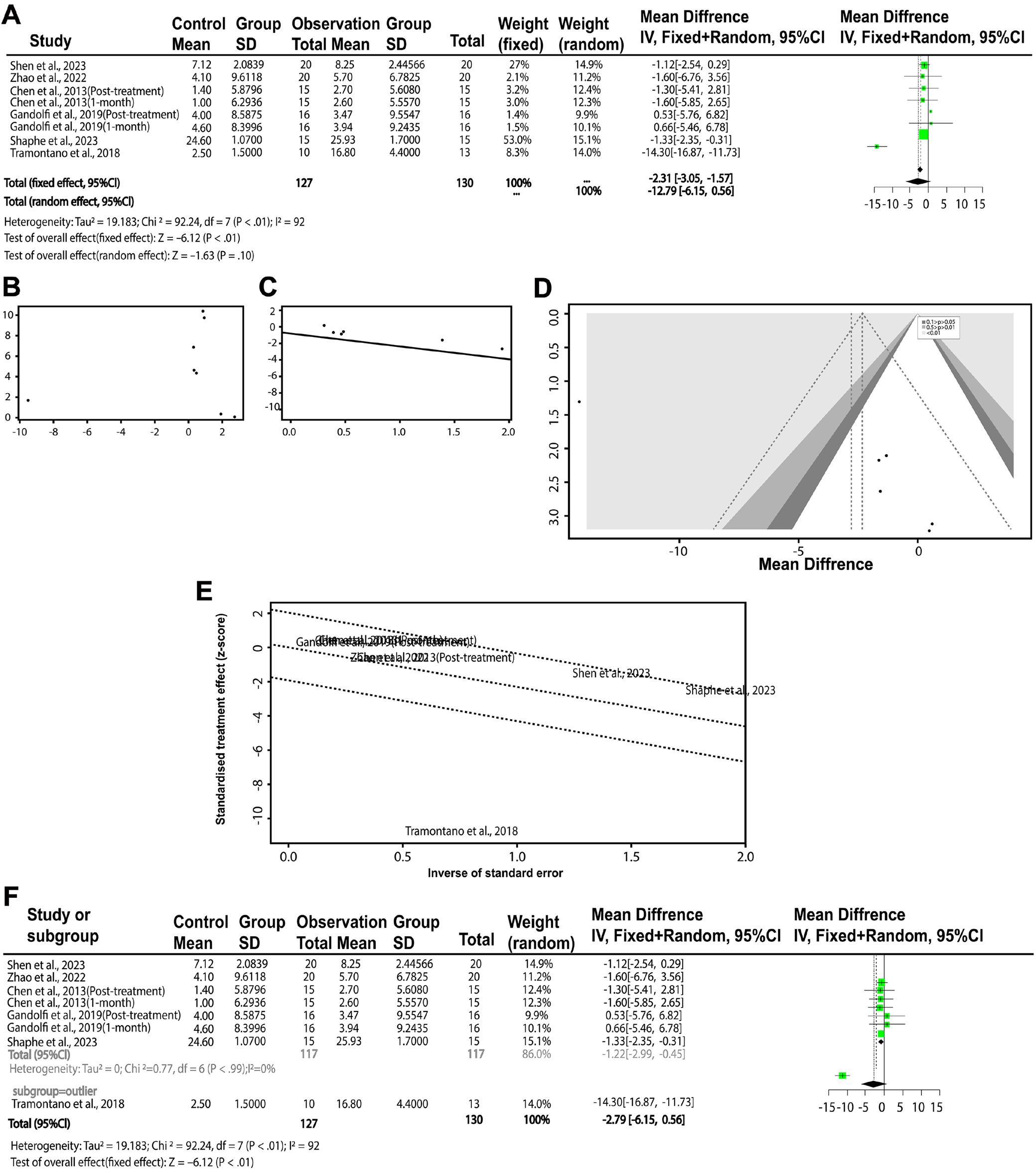
Effectiveness of VRT in improving patient outcomes with BBS
Two statistical models, the fixed effect and random effects models, were used to evaluate the data. Due to significant heterogeneity among the studies, the random effects model was determined to be more suitable. The fixed effect model initially indicated a significant improvement in balance scores for the VRT group compared to the control group (Figure 9). However, the random effects model, which accounts for variability across studies, suggested that the difference between groups was not statistically significant (Figure 9). This finding underscores the importance of considering study heterogeneity in meta-analyses.
Further analysis using the Begg’s and Egger’s tests indicated no significant publication bias, as both tests showed symmetry in the distribution of study effect sizes. However, the funnel plot did reveal a nonstandard pattern, suggesting potential concerns that warrant further investigation. The Galbraith analysis revealed distinct patterns of heterogeneity, with certain studies contributing more to the variability in effect sizes than others (Figure 9).
To address the observed heterogeneity, a subgroup analysis was conducted, dividing studies into outlier and non-outlier groups. The non-outlier group demonstrated minimal heterogeneity and a more consistent effect size, while the outlier group showed a significantly larger effect size (Figure 9). These results highlight the importance of identifying and accounting for outliers in meta-analyses to ensure more accurate and reliable findings. Summary of VRT’s effectiveness (BBS) is presented in Table 3.
In summary, this analysis demonstrates the complexity of evaluating interventions across diverse studies. While the random effects model provides a more nuanced understanding by accounting for study variability, the presence of outliers significantly impacts the overall results. The findings suggest that while VRT shows promise, its effectiveness may vary depending on specific study characteristics, and careful consideration of heterogeneity is essential in interpreting meta-analytic results.
Results of sensitivity analysis
The sensitivity analysis was conducted by excluding studies identified as having a high risk of bias. For the primary outcome assessed using the DHI scale, the results remained statistically significant, with only a slight reduction in heterogeneity. This suggests that studies with a high risk of bias had a minimal impact on the overall findings. Similarly, for the BBS, the exclusion of high-risk bias studies did not significantly change the overall effect size or the statistical significance, though the heterogeneity among the studies remained substantial. Sensitivity analysis summary is presented in Table 4.
Taken together, the sensitivity analysis reinforces the robustness of the primary outcomes, particularly for the DHI scale, even when studies with a high risk of bias were excluded. While some heterogeneity persists, the overall effect sizes and conclusions remain consistent, suggesting that the results are reliable despite variations in study quality. This analysis underscores the importance of accounting for study quality in meta-analyses to ensure accurate and meaningful conclusions.
Discussion
The present investigation provided a comprehensive evaluation of the effectiveness of VRT in improving patient outcomes related to balance and dizziness, as measured using the BBS and the DHI. The findings strongly supported the efficacy of VRT in enhancing these outcomes, aligning with those of previous research.51 The significant improvements observed in both BBS and DHI scores across various studies highlight VRT’s potential as a valuable intervention for patients suffering from vestibular disorders. However, the analysis also revealed remarkable heterogeneity among the included studies, pointing to the variability in study designs, patient populations and intervention protocols as key factors influencing the results.
The results of the present study build upon and extend previous work in the area of vestibular rehabilitation. For instance, the improvements in DHI scores observed in this meta-analysis align with the findings of previous studies, such as those reported by Başoğlu et al.,41 who demonstrated the effectiveness of virtual reality-based vestibular rehabilitation in patients with peripheral vestibular hypofunction. Similarly, our analysis echoes the conclusions of Mitsutake et al.,43 who highlighted the positive impact of VRT on gait performance in stroke patients, a group that often experiences significant balance and mobility issues. These consistencies with prior research underscore the robustness of VRT as a therapeutic approach, while the high heterogeneity observed suggests that future studies need to address standardization in intervention protocols to achieve more uniform outcomes.
The implications of our findings are multifaceted. First and foremost, they underscore the importance of VRT as a noninvasive, cost-effective treatment option for patients with vestibular disorders. The observed improvements in both balance and dizziness-related outcomes suggest that VRT can significantly enhance the quality of life for these patients, reducing their risk of falls and improving their ability to perform daily activities. Additionally, the identification of key factors contributing to heterogeneity, such as geographical location, intervention type and study quality, provides valuable insights for future research. By concentrating on these variables, researchers can design more targeted and effective VRT interventions, ultimately improving patient outcomes.
For instance, findings from Chen et al.52 and Gandolfi et al.53 highlighted the importance of task-specific interventions in improving balance, gait, and postural control in individuals with chronic stroke. Chen et al.52 demonstrated that turning-based treadmill training significantly improved turning speed, gait symmetry, muscle strength, and balance control compared to conventional treadmill training. Similarly, Gandolfi et al.53 found that robot-assisted stair climbing training showed improvements in postural control and sensory integration processes, specifically in challenging balance conditions. These findings underscore the importance of having personalized rehabilitation strategies that incorporate both dynamic gait training and sensory integration exercises to optimize functional recovery in chronic stroke patients.
Moreover, this study highlighted the potential for VRT to be tailored to specific patient populations, depending on factors such as age, underlying health conditions and geographical location. For instance, the subgroup analysis revealed that studies54, 55, 56 conducted in Asia reported more substantial improvements in balance, suggesting that cultural or regional differences may play a role in the effectiveness of VRT. Similarly, the analysis unveiled that studies57, 58 employing gaze stabilization exercises as part of their VRT protocols yielded larger effect sizes, indicating that this specific intervention may be particularly beneficial for certain patient groups. These findings point to the need for personalized treatment plans that take into account individual patient characteristics and preferences.
The ultimate objective of VRT research and clinical practice is to develop standardized, evidence-based protocols that can be widely implemented across diverse healthcare settings. Achieving this goal will require a concerted effort to address the challenges identified in this meta-analysis, including the need for longer follow-up periods, the standardization of intervention protocols and the inclusion of diverse patient populations. Additionally, the development of new technologies and methodologies, such as virtual reality and telerehabilitation, has the potential to further enhance the effectiveness and accessibility of VRT. By incorporating these innovations into clinical practice, healthcare providers can provide more comprehensive and individualized care to patients with vestibular disorders.
The significance of this line of research cannot be overstated. Vestibular disorders are common and debilitating conditions that affect millions of people worldwide, leading to significant morbidity and reduced quality of life. By advancing our understanding of the most effective treatment strategies for these conditions, we can improve patient outcomes and reduce the burden on healthcare systems. Furthermore, the insights gained from this research can inform the development of new therapeutic approaches for other balance-related disorders, such as those associated with aging, neurological conditions or traumatic brain injuries.
This meta-analysis has several merits. It provided a comprehensive synthesis of the available evidence on the effectiveness of VRT, highlighting valuable insights into the factors that influence treatment outcomes. The inclusion of a wide range of studies, covering diverse patient populations and intervention protocols, could enhance the generalizability of the findings and provide a solid foundation for future research. Moreover, the identification of key factors contributing to heterogeneity provides a roadmap for researchers to design more targeted and effective VRT interventions.
The potential clinical applications of VRT are vast. In addition to its use in managing vestibular disorders, VRT can be adapted for other patient populations who experience balance and mobility issues, such as those recovering from stroke or traumatic brain59, 60 injury, or older adults at risk of falls.60, 61, 62 By integrating VRT into multidisciplinary treatment plans, healthcare providers can offer a more holistic approach to patient care, addressing both the physical and psychological aspects of balance disorders.63, 64, 65 Furthermore, the development of telerehabilitation platforms for VRT delivery can enhance access to care, particularly for patients in remote or underserved areas.
Limitations
However, it is essential to acknowledge the limitations of this meta-analysis when interpreting the results. The high heterogeneity observed in both the DHI and BBS outcomes suggests significant variability in study designs, patient populations and intervention protocols.
This variability complicates the interpretation of the findings and underscores the need for more rigorous study designs in future research. Additionally, the relatively short follow-up periods in many of the included studies limit our ability to assess the long-term effectiveness of VRT. Future research should prioritize longitudinal studies with extended follow-up periods to better understand the sustainability of the observed treatment effects.
Conclusions
Collectively, this meta-analysis provided strong evidence, supporting the effectiveness of VRT in improving balance and reducing dizziness in patients with vestibular disorders. While the findings are consistent with previous studies,66, 67, 68, 69, 70 the high heterogeneity observed highlights the need for standardization in intervention protocols and study designs. Future research should concentrate on addressing these challenges, with an emphasis on long-term follow-up, personalized treatment approaches and the integration of new technologies. By advancing our understanding of VRT and its clinical applications, we can improve patient outcomes and contribute to the broader field of rehabilitation science.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.