Abstract
Background. Thyroid carcinoma (TC), the most prevalent endocrine cancer worldwide, has become progressively more common, especially in women. Most TCs are epithelial-derived differentiated TCs, specifically papillary thyroid cancer (PTC). Although there are many therapeutic drugs available, curing TC is a difficult task.
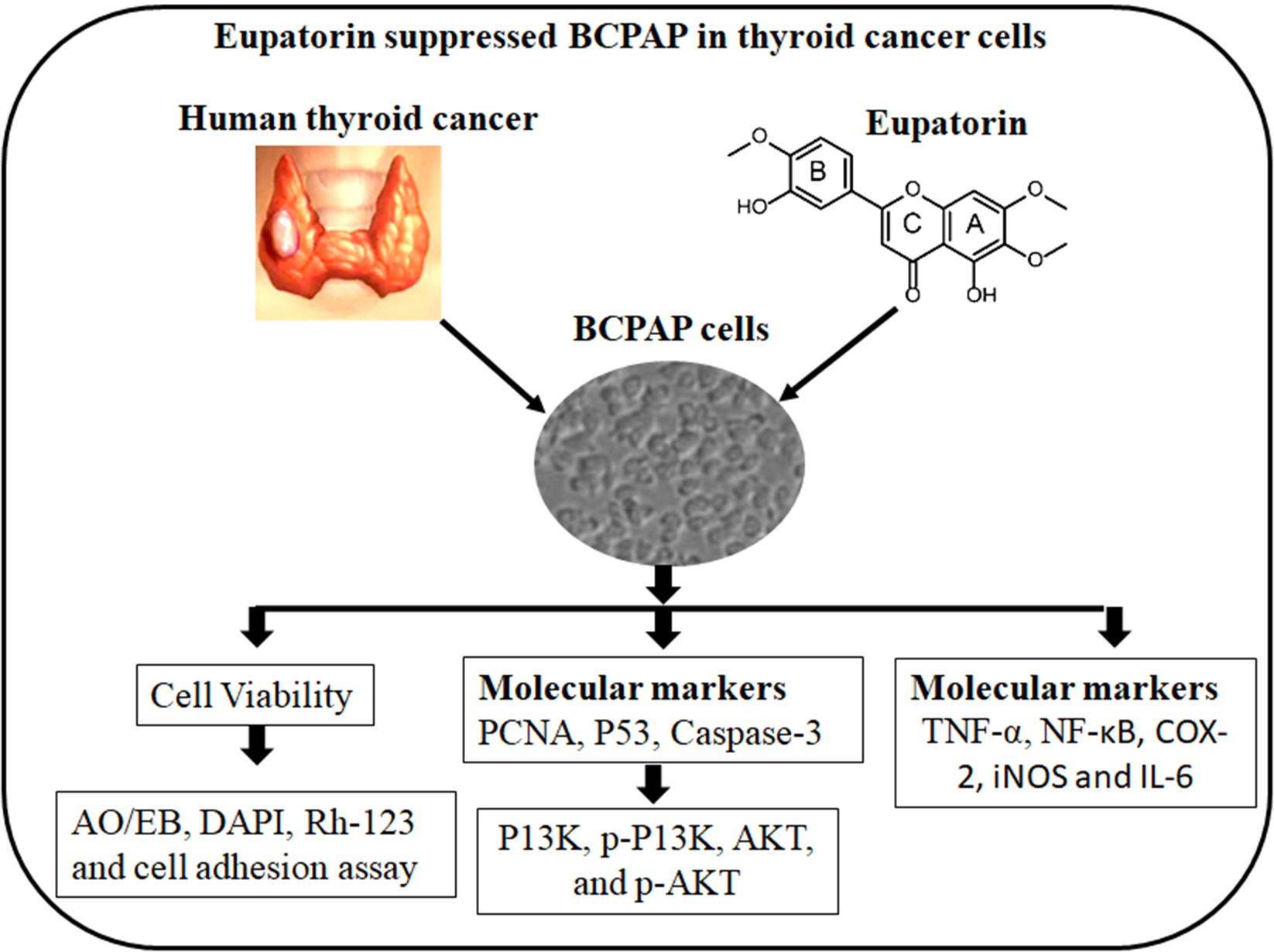
Objectives. A flavone called eupatorin (EUP) obtained from herbs can prevent the growth of many types of cancerous cells. Nonetheless, the mechanisms of EUP’s actions against PTC are still unknown.
Materials and methods. The goal of our work was to evaluate the mechanisms of EUP (20 and 30 µM/mL) and examine its antiproliferative and apoptotic effects on human PTC cells BCPAP. The MTT test; dual acridine orange/ethidium bromide (AO/EB), rhodamine-123 (Rh-123), and 4′,6-diamidino-2-phenylindole (DAPI) staining; adherence assays; and western blot analyses were used to evaluate the antiproliferative and apoptotic properties of EUP on BCPAP cells.
Results. Our research showed that the quantity-dependent administration of EUP inhibited the proliferation of BCPAP cells, which in turn caused apoptosis through the increase in caspase-9 and p53 protein expression and the reduction of proliferating cell nuclear antigen (PCNA) levels. Additionally, when P13K/AKT signaling is inhibited by nuclear factor kappa B (NF-κB), EUP reduces inflammation and BCPAP proliferation.
Conclusions. By blocking the NF-κB and P13K/AKT pathways, EUP can reduce the growth of BCPAP cells and promote cell death.
Key words: thyroid cancer, papillary thyroid cancer, apoptosis, P13K/AKT signaling, eupatorin
Background
Thyroid cancer (TC) is a fatal tumor of the endocrine system, and its prevalence has been progressively increasing over the past 30 years.1, 2 The major etiology of TC is environmental factors, radiation exposure, gender, family history, and lifestyle choices.3 A recent study documented that intense living environments accompanied by working with gravitational variations may impair the function of the thyroid and affect various carcinomas.4 The frequently occurring form of malignant TC is papillary thyroid carcinoma (PTC), which accounts for 80–85% of all human TCs.5 Currently, the available treatments of TC rely on radioiodine ablation, suppression of thyroid-stimulating hormone and surgery.6 These remedies are generally effective for the majority of TCs. However, advanced TC, including recurrent, 131I-refractory and metastatic or medullary TCs, remains a therapeutic challenge. Therefore, it is crucial to develop an effective therapeutic agent for these patients.
Herbal-based natural bioactive components have anticancer efficacy in numerous tumors, which can also be combined with traditional chemotherapy medications to enhance their anticancer properties.7, 8 Eupatorin (EUP) is a natural flavone isolated from several herbal plants.9 Eupatorin has been established as an antiproliferative agent in numerous carcinoma cells, including gastric neoplasms, cervical adenocarcinoma, breast neoplasm cells, melanoma, and colon cancer cells.10, 11, 12 Eupatorin activates apoptosis and concurrently inhibits the angiogenesis, invasion and migration of MCF-7 and MDAMB-231 cells by inactivation of the p-Akt pathway.13 It has been documented that co-administring EUP with DOX in colon cancer triggered mitochondrial-facilitated apoptosis with an elevated ratio of Bax/Bcl-2, increased levels of caspase-3 and increased PARP cleavage.14 An earlier study recognized that EUP halts the cell cycle at the G2–M stage in leukemia cells and prompts apoptosis by the stimulation of numerous caspases, discharge of cytochrome-c (cyt-c), and subsequently PARP cleavage through the instigation of the MAPK signaling. It was proposed that EUP-induced leukemia cell apoptosis is facilitated by the extrinsic and intrinsic apoptotic pathways.15 Currently, no report is available on the apoptotic and cytotoxic efficacy of EUP on PTC cells.
Apoptosis is a specific kind of programmed cell death, which is characterized by variations in cellular morphology and nuclear alterations that commonly occur without inflammation. The crucial events that occur in the development of tumor progression are the inhibition of apoptotic cell death and organized de-restricted cellular propagation.16 Malignant cells exhibit numerous systems that allow them to evade apoptosis triggered by chemotherapeutic medications, including p53 mutations, which is an important status.17 Enhancing responses might be beneficially achieved by either growing a p53-self-regulating, pro-apoptotic target or altering the appearance and transcriptional function of p53 to chemotherapy agents.18 Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) dynamically functions in many cancer cells and controls a sequence of essential proceedings in cancer progression including cell proliferation, cell survival, invasion, and angiogenesis.19 The triggered NF-κB signalosome complex primes downstream signaling, which stimulates the transcription of proinflammatory intermediaries, including cyclooxygenase-2 (COX-2), inducible nitric oxide synthase (iNOS), interleukin 6 (IL-6), and tumor necrosis factor alpha (TNF-α).20 These mediators triggered various immune reactions that are critical for carcinoma initiation in tissues.21 For cell existence, NF-κB generally represses cell death by enhancing the transcription of anti-apoptotic genes encoding proteins such as Bcl-2 and Bcl-XL.22 Hence, inhibition of NF-κB may be efficient in triggering apoptosis of neoplasms. The PI3K/Akt signaling has been shown to play a crucial role in cell proliferation, cell adhesion, invasion, migration, and apoptosis in diverse human cancers. The overexpression of the PI3K/AKT pathway appears to be linked to malignancies and poor prognosis in numerous human carcinomas.23 Also, the PI3K/AKT pathway has been determined to be a characteristic of follicular carcinomas and ATCs, but it is less prevalent in PTC.24 Furthermore, targeting the PI3K/AKT pathway has been supported to be an effective medicinal approach for treating both solid human tumors and TC.25, 26
Objectives
In the current research, we inspected the efficacy of EUP on apoptosis and the inhibition of PTC cell BCPAP proliferation and explored the molecular mechanism of the anticancer effects through an analysis of the regulation of NF-κB and P13K/AKT pathways.
Materials and methods
Chemicals
Eupatorin (purity: ≥98%), antibiotics, phosphate-buffered saline (PBS), RPMI 1640 medium, fetal bovine serum (FBS), MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay, acridine orange/ethidium bromide (AO/EB), dimethyl sulfoxide (DMSO), Rh-123, and DAPI(4′,6-diamidino-2-phenylindole) were purchased from Gibco (Waltham, USA). The primary and secondary antibodies were bought from Abcam (Cambridge, UK).
Cell culture
BCPAP human PTC cells were bought from the Peking Union Cell Resource Centre (Beijing, China). Cultured the BCPAP cells using RPMI 1640 media along with FBS (10%), penicillin/streptomycin antibiotics (1%) at 37°C, in a 5% CO2 environment, and humidified air below 95%.
Cell proliferation assay
Human BCPAP cell proliferation was performed according to MTT assay.27 The BCPAP cells were disseminated into 96 wells and cultured at 37°C using a 5% CO2 wet incubator. After an overnight incubation period, the BCPAP cells were cultured for 24 h with various quantities of EUP (5–50 μM/mL). Later, the sustained BCPAP human PTC cells underwent MTT (10 μL) testing and maintained for an extra 4 h to allow MTT conversion into insoluble crystals of formazan. The DMSO (150 μL) was then used to dissolve the resultant insoluble crystals. The estimated viability was performed at 490 nm using Agilent BioTek Synergy Neo2 hybrid multimode reader (BioTek Instruments, Colmar, France). Cell cytotoxicity was determined as the ratio of viability compared to control DMSO (150 μL) cells (100%). The value of IC50 was obtained using the below equation:
cell viability inhibition (%) = (control OD – test OD) × 100,
where OD is optical density.
BCPAP cell apoptosis was evaluated by AO/EB staining
Human BCPAP cell morphology of apoptosis was assessed with AO/EB staining.28 BCPAP cells were exposed to 20 and 30 µM/mL of EUP and incubated for 24 h. All groups were administered AO/EB dye (100 µg/mL of each dye) and preserved for 30 min at room temperature under dark conditions. The remaining unbound dye was removed with PBS washing and viewed through a fluorescence microscope (Nikon Eclipse TS100; Nikon Corp., Tokyo, Japan).
Assessment of BCPAP cell apoptosis
Apoptosis is a functional form of cell death that is distinguished by significant DNA fragmentation and physical features.29 Human BCPAP cells were dispersed in well plates and kept in a humidified CO2 (5%) incubator at 37°C for 24 h. Various amounts (control, 20 μM/mL and 30 μM/mL) of EUP were administered to the cells. Subsequent control and treated cells were washed twice with PBS, fixed by paraformaldehyde (4%), rinsed, DAPI-stained, and then incubated for 20 min. Then, DAPI-treated BCPAP cells were exposed to staining with Rh-123 and incubated at 37°C for 30 min. The stained cells were methanol washed 2-fold to eliminate extra dyes and immersed with PBS, and the mitochondrial membrane potential (MMP) variant was viewed using a fluorescence microscope (Nikon Eclipse TS100; Nikon Corp.).
Assay for cell adhesion analysis
Human BCPAP cells were immersed in RPMI 1640 culture media and pre-treated with EUP (control, 20 μM/mL and 30 μM/mL) and sowed in a 24-well fibronectin-coated (500 μL/well) plate. The whole well plate was preserved at 37°C for 60–100 min; then, non-adherent cells were removed and taken apart evenly twice with PBS cleaning. Next, 94% paraformaldehyde fixation in each well was kept at 4°C incubation for 5 min. After incubation, TB (0.4%) staining of the adherent cells was counted using fluorescence microscopy (Nikon Eclipse TS100; Nikon Corp.).29
Western blotting assay
Untreated BCPAP cells and 20 and 30 µM/mL of EUP-treated human BCPAP cells that had been cultured for 24 hours were used to create the cellular lysates for Western blot analysis. To guarantee protease inhibitory activity, lysis buffer was employed in extremely cold temperatures. Briefly, the entire protein was estimated by consuming the protein BCA Assay Kit (Pierce Chemical Co, Rockford, USA). Subsequently, quantified proteins were dispersed electrophoretically and moved to a polyvinylidene difluoride (PVDF) film. It was blocked by using a probe at room temperature for 1 h using the primary antibodies (PCNA, P53, caspase-3, TNF-α, NF-κB, COX-2, iNOS, IL-6, PI3K, p-PI3K, Akt, and p-Akt) in 1:1,000 dilutions and kept at 4°C overnight. Then, horseradish peroxidase (HRP)-conjugated secondary antibodies were administered. Afterward, the protein bands were stained and imagined for protein identification, quantifying the protein bands through densitometry with ImageJ software (National Institutes of Health (NIH), Bethesda, USA) and homogeneous to β-actin expression.
Statistical analyses
The data from each group were analyzed statistically using GraphPad Prism v. 8.0.2 (GraphPad Software, San Diego, USA) and IBM SPSS software v. 25 (IBM Corp., Armonk, USA). The measurement data were reported as medians (min–max). The normality of the distribution was tested using the Kolmogorov–Smirnov test. Since all the distributions were normal, the Brown–Forsythe test was used to establish the equality of variances, and then significant differences between multiple groups were analyzed using the Kruskal–Wallis test, and Dunn’s post hoc test was used for multiple comparisons. If the p-value was <0.05, the data divergence was statistically notable. All tests in this study were bilateral tests.
Results
The Results of the Kruskal–Wallis test and Dunn’s post hoc test are presented in Table 1 and Table 2.
Antiproliferative efficacy of EUP on BCPAP cells
The antiproliferative assay of BCPAP cells was estimated using the MTT experiment in a dose-dependent manner (5–50 μM/mL) of EUP. Results showed that EUP diminished cell proliferation through its antiproliferative and cytotoxic deeds on BCPAP cells in a dosage-dependent way. Administration of EUP at a quantity less than 15 μM did not considerably amend BCPAP cell proliferation. However, moderate dosages of EUP (20 and 30 μM/mL) significantly decreased (p < 0.05) BCPAP cell viability compared to controls. A high dose of EUP (50 μM) impaired the BCPAP cells. After the MTT test, the IC50 assessment of EUP was 30 μM for BCPAP cells. The inhibitory concentration values of 20 and 30 μM/mL of EUP have been obtained for additional examination (Table 1, Figure 1).
Effect of EUP on BCPAP cell apoptosis presented using the AO/EB dual method
Human BCPAP cell apoptosis was envisioned as distinct aberrations in morphology attributed to the AO/EB staining method. Untreated BCPAP control cells were visible as consistently live green-stained cells. Administration of EUP (control, 20 μM/mL and 30 μM/mL) in the BCPAP cells caused significant (p < 0.05) cell apoptosis in a quantity-reliant manner. Eupatorin (20 μM) treatment revealed primary apoptotic cells comprised of condensation of chromatin and membrane eruption, depicted as dots of light greenish and yellow. Supplementation of EUP (30 μM) exposed late apoptotic cells presenting an orange color because of ethidium bromide co-stain (Figure 2).
Effect of EUP on the BCPAP cells, MMP loss and nuclear morphology
The effect of EUP on human BCPAP cells’ morphological features, membrane integrity, nuclear condensation, nuclear fragmentation, and MMP were evaluated. The technique of DAPI staining can be utilized to identify DNA fragmentation in apoptotic bodies. Supplementation of the EUP (20 and 30 µM/mL) to the BCPAP cells triggers apoptosis, developing nuclear condensation and disintegration of nuclear bodies compared to controls. This outcome exposed the antiproliferative efficacy of EUP on BCPAP cells in a quantity-dependent way. The MMP is permeable to ions and yields a promising electrical abnormality. Fluorescence microscopic images showed the accumulation of Rh-123 fluorescence dye from orange red to green compared in vehicle control and the accumulation found to be decreased in EUP treated BCPAP cells at various concentrations (20 µM/mL and 30 µM/mL). Rh-123 staining exhibited mitochondrial strength in apoptotic cell death with an intensified MMP. The untreated BCPAP exhibited higher fluorescence intensity due to the Rh-123 red accumulation. The fluorescence mass of Rh-123 was reduced when EUP (20 and 30 µM) was administered in a concentration-dependent way. These outcomes show that EUP-administered human BCPAP cells avert the MMP, thus increasing the mitochondrial-mediated apoptosis (Figure 3).
Influence of EUP on BCPAP cell adhesion
Extracellular matrix (ECM) and BCPAP cell adhesion played a substantial action in initiating cancer cell metastasis (Figure 4). BCPAP cell adhesion and cell signaling are controlled by the ECM protein integrin. This cell adhesion assay analyzes the action of EUP silencing on BCPAP cell adhesion to the proteins present in the ECM. When EUP inhibition was administered for 60 or 100 min, the number of BCPAP adherent cells decreased in comparison to controls. The modulatory influences of cell adherence generated with EUP silencing might provide the suppression of cancer cell propagation and metastasis.
Effect of EUP on PCNA, caspase-3 and p53 protein levels in BCPAP cells
Administration of EUP (control, 20 μM/mL and 30 μM/mL) on BCPAP cells presented an extreme (p < 0.05) repression of PCNA while upregulating p53 and caspase-3 protein expression in a quantity-reliant way. These findings specified that EUP inhibited BCPAP cells during cell proliferation and elevated apoptosis via these signaling pathways (Table 2, Figure 5).
Influence of EUP on apoptotic protein expression levels
Human BCPAP cells treated with EUP (control, 20 μM/mL and 30 μM/mL) attenuated TNF-α, NF-κB, COX-2, iNOS, and IL-6 protein expression. Our findings documented the anti-inflammatory action of EUP on human PTC cells BCPAP in a dose-dependent manner (Table 2, Figure 6).
EUP attenuates the signaling of the P13K/AKT pathway
Our results show that PI3K/AKT signaling is involved in EUP-associated prevention of BCPAP cell metastasis. Our outcomes revealed that PI3K/AKT protein level expressions were remarkably (p < 0.05) alleviated in the BCPAP cancer cells treated with EUP (control, 20 μM/mL and 30 μM/mL) in a concentration-dependent way. These findings indicated that EUP triggered BCPAP cancer cell apoptosis through the suppression of the PI3K/AKT activation in the cell survival pathway (Table 2, Figure 7).
Discussion
Thyroid carcinoma is an endocrine malignancy, of which PTC accounts for nearly 80% of all TCs that have been well differentiated by the variation of distinct protein kinases.2, 5 Phytochemicals are substances of plant origin with biological activity, which have beneficial effects on human health.30 Two studies proposed that flavones and flavonoids exert powerful actions of anti-proliferation on TC cells and suggested it as a healing agent for controlling TC.7, 8Other studies reported that EUP has antiproliferative and apoptotic effects in numerous tumor cells.10, 11, 12 However, the anticancer potential of EUP and its corresponding apoptotic latent mechanisms of TC are still unknown. In this investigation, we proved the repressive influence of cell growth displayed by apoptosis at various concentrations of EUP on BCPAP malignant cells. This research also explored the EUP activity on the regulation of NF-κB and P13K/AKT pathways in BCPAP human PTC cells.
One of the main ways that anti-tumor drugs work is by using their cytotoxic properties to stop tumor cells from proliferating. Cytotoxicity is distinguished as a cell necrotizing function of a compound relieved from the pathways of cell death.31 The MTT assay is an appropriate method for testing innovative bioactive compounds to establish cytotoxicity on malignant cells over a short duration.32 The MTT analysis implied that EUP reduced the BCPAP cell’s growth in a quantity-dependent mode. Earlier studies illustrated that EUP inhibits many tumor cell proliferation involving gastric cancer, cervical adenocarcinoma cells, breast neoplasm cells, melanoma, and colon carcinoma.10, 11, 12 It has been reported that EUP inhibits cancer cell viability and concurrently curbs the invasion, migration and angiogenesis of MCF-7 and MDAMB-231 mammary carcinoma cells through the attenuation of the p-Akt signaling.13 In our study, EUP exhibited substantial inhibitory growth potential with IC50 values of 20 μM, which is within the range value stated for many flavonoids. As per our data, this is the primary report directing that EUP (20 and 30 µM/mL) greatly averts BCPAP cell proliferation in a dosage-dependent way.
Apoptosis is the main type of automated cell death that has a profound action in eliminating peculiar cells, which can be morphologically depicted by cell shrinkage, fragmentation of DNA and nuclear bodies, and, accordingly, being swallowed by macrophages.16 A typical anti-tumor agent should target cancer cells without affecting normal cells. Hence, numerous anti-tumor agents rely on commencing apoptosis to generate malignant cell death.17 An earlier study has investigated that co-administration of EUP with DOX on colon cancer triggered mitochondrial-facilitated apoptosis with an elevated ratio of Bax/Bcl-2, level of caspase-3, and poly(ADP-ribose) polymerase (PARP) cleavage.14 It has been recognized that EUP halts the G2–M stage of the cell cycle in leukemia cells and prompts apoptosis in the stimulation of numerous caspases, cytochrome-c (cyto-c) discharge and PARP cleavage over the activation of the MAPK pathway. It has been suggested that EUP-induced leukemia cell apoptosis is facilitated by both extrinsic and intrinsic apoptotic pathways.15
Cell proliferation is involved in a multistep process of typical cell development, and unrestrained cell growth is the main hallmark of tumors.33 A known proliferative marker, PCNA, shows a significant function in DNA replication, repair, cell cycle regulation, chromatin remodeling, and epigenetic conservation.34 Proliferating cell nuclear antigen overexpression was observed in the genetic outcome of numerous tumors.35 We showed that EUP can reduce the PCNA protein expression in human PTC cells BCPAP in a dose-dependent manner. The p53 wild type is efficient in initiating cell apoptosis under stress conditions.17 Conversely, inactivation, mutation or depletion of the p53 gene leads to the failure to respond to radiotherapy and chemotherapy in several tumors.17, 18 There is evidence to support the idea that cancer cells bear the responsibility for apoptotic cell death when p53 function is restored.21 In the current study, p53 and caspase-3 were upregulated by EUP in BCPAP human PTC cells. Anti-tumor medication-facilitated apoptosis in tumor cells is mostly regulated by intrinsic (mitochondrial) or extrinsic (death receptor) pathways through the activation of caspases. Caspase-3 triggers the mitochondrial-mediated intrinsic apoptosis, as observed in this work, which was enabled by the morphological examination of fluorescence microscopy images using DAPI, Rh-123 and AO/EB staining. These results indicate that EUP might be effective for the suppression and remedy for TC.
Chemotherapeutic agents targeting a reduction in pro-inflammatory mediators to explore novel strategies to avert the inflammatory disorders involved with malignant tumors.19 The hyperactivation of NF-κB has a critical role in cancer pathogenesis, letting malignant cells persist and offering a favorable target for the improvement of chemotherapeutic drugs.19, 20 During inflammation, several cytokines such as iNOS, IL-6, TNF-α, and COX-2, which are hyper-expressed in tumor cells, contribute to the malignant pathogenesis.20, 21 However, a stimulated NF-κB signaling pathway creates high levels of nitric oxide (NO) and prostaglandin E2 (PGE2) for an extended period, which influences tumor progression and inflammation. In this study, EUP administered to the BCPAP cells effectively reduced pro-inflammatory mediators, including iNOS, IL-6, TNF-α, NF-κB, and COX-2 protein expression measured using western blot examination, which specifies that EUP may deactivate NF-κB signaling in BCPAP cells.
Adhesion of cancer cells to the ECM has a crucial function in metastasis development.36 Inhibiting and reducing the quantity of adhering BCPAP cells, the EUP modulates cell adhesion and represses metastasis, which is in line with our results from the apoptosis analysis. The number of adherent cells was noticeably reduced after BCPAP cell suppression of cell metastasis. The PI3K/Akt signaling pathway has been demonstrated to exert a vital function in cellular proliferation, cell adhesion, migration, invasion, metastasis, and apoptosis in several human cancers.23 The PI3K-Akt pathway is directly linked to tumor cell proliferation.37 The overexpression of the PI3K/AKT pathway figures to be linked to malignancies and a poor prognosis in numerous human carcinomas.23 Furthermore, targeting the PI3K/AKT pathway has been confirmed to be effective for treating both human solid tumors and TC.25, 26 For instance, the antioxidant rutin has an antiproliferative effect on hepatic carcinoma by regulating the PI3K/Akt signaling pathway.38 Another bioactive compound, quercetin, can modulate the PI3K/Akt pathway, thus showing anti-tumor efficacy on liver, gastric and oral carcinomas.39, 40 Likewise, phloretin can induce apoptosis in prostate cancer and mammary carcinoma cells through the PI3K/Akt signaling pathway.41, 42 In this study, EUP treatment reduces the PI3K/Akt and NF-κB pathways related to PTC cells that interrupt their communication, favoring BCPAP cell proliferation. Hence, EUP exerts its antiproliferative, anti-metastatic and apoptotic activity through the NF-κB and PI3K/Akt signaling pathways.
Limitations
The current study investigated EUP on human PTC cell proliferation, apoptotic and inflammatory pathways in a cancer cell line model. An in vivo experimental study in a mouse or rat model is preferred to analyze the molecular mechanism and correct route signaling.
Conclusions
Our report explores the efficacy of EUP against BCPAP human PTC cells and has elucidated its molecular actions involving cell proliferation, inflammation, cell adhesion, and apoptosis. It was noticed that EUP stimulated mitochondrial-facilitated apoptosis and a loss of MMP. The suppression of BCPAP cell proliferation and metastasis occurred in a concentration-dependent mode. Furthermore, it inhibited proliferation, inflammation and metastasis that triggered apoptosis through the NF-κB/COX-2 and PI3k/Akt signaling pathways. Our data show that EUP can inhibit the BCPAP human PTC cells. However, more studies are required on EUP for its clinical use in the therapy for TC patients.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.12683722. The package includes the following files:
Supplementary Fig. 1. Results of Kruskal–Wallis test as presented in Figure 1.
Supplementary Fig. 2. Results of Kruskal–Wallis test as presented in Figure 5.
Supplementary Fig. 3. Results of Kruskal–Wallis test as presented in Figure 6.
Supplementary Fig. 4. Results of Kruskal–Wallis test as presented in Figure 7.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.