Abstract
Background. Diabetic nephropathy (DN), the most severe microvascular consequence of diabetes mellitus (DM), is the precursor to end-stage renal disease (ESRD). The development of problems linked to DN involves both oxidative damage and inflammation. Natural flavone acacetin (AC) has anti-inflammatory, antioxidant and anti-cancer properties. However, the effect of AC on DN is not clear.
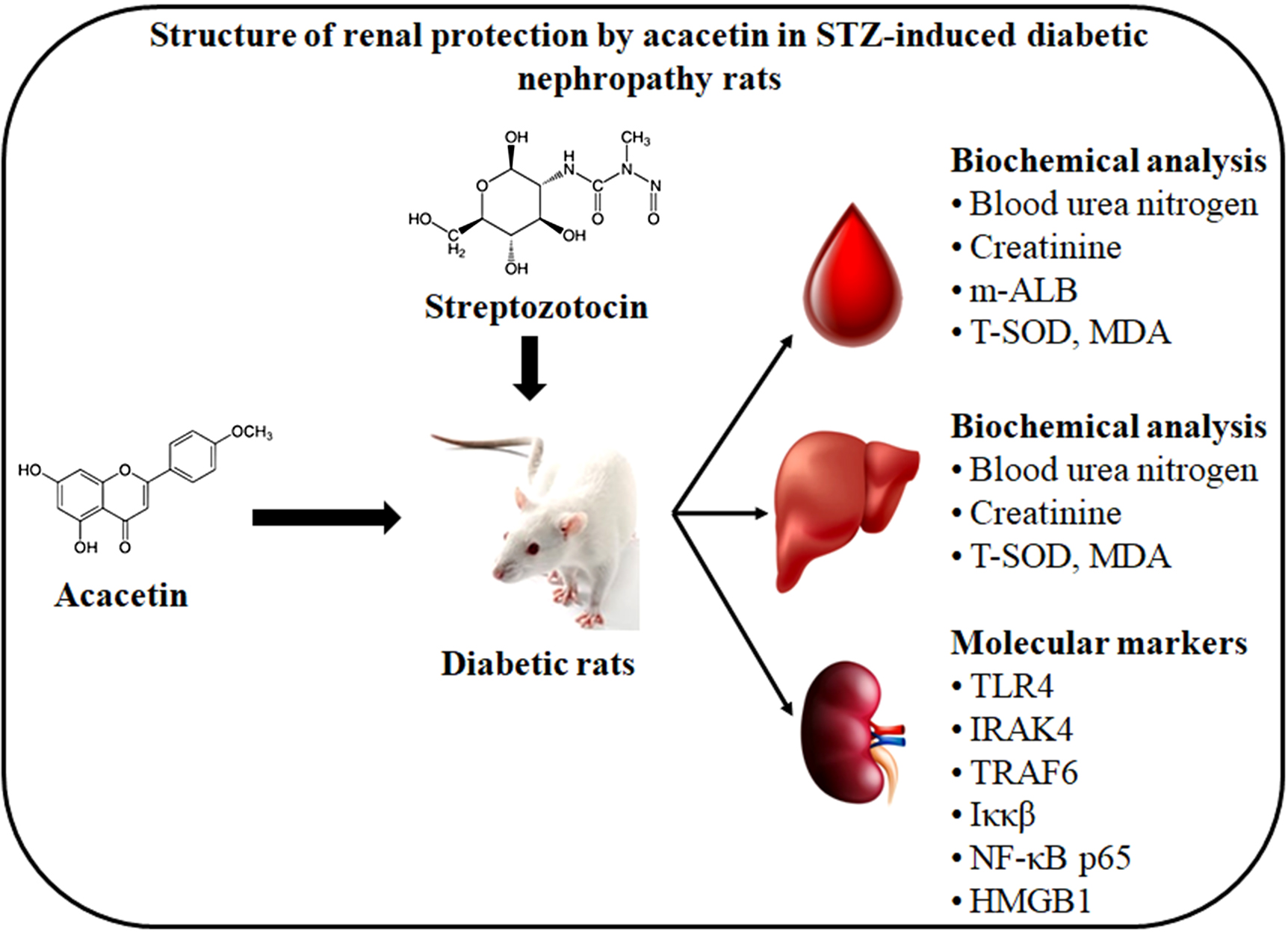
Objectives. To investigate potential nephroprotective effects of AC caused by reducing inflammation and oxidative stress via the TLR4/NF-κB pathway in rats with streptozotocin (STZ)-induced DN.
Materials and methods. In this study, we investigated the nephroprotective effect of AC compared to that of a positive control therapy of irbesartan (IRB) in DN induced with STZ. In this model, rats were given an intraperitoneal injection of STZ (180 mg/kg body weight (BW)), along with daily doses of AC (10 mg/kg BW) or IRB (180 mg/kg BW) to induce DN. Histopathology, albumin, blood glucose (Bg), BW, oxidative stress indicators, and western blot of inflammatory signaling pathways in the kidney were examined.
Results. Reduction of blood glucose, proteinuria, serum malondialdehyde (MDA), serum creatinine, and blood urea nitrogen (BUN), as well as the inhibition of toll-like receptor 4 (TLR4), high mobility group box 1 (HMGB1) and nuclear factor kappa B (NF-κB) protein expression were observed. These data demonstrated that AC could improve BW, antioxidant enzyme and renal histopathology in rats with STZ-induced DN.
Conclusions. Results from the rat model highlight how AC-suppressed inflammation and oxidative stress can attenuate STZ-induced DN by downregulating the TLR4/NF-κB pathway in rats.
Key words: inflammation, diabetic nephropathy, oxidative stress, acacetin, TLR4/NF-κB signaling
Background
Diabetes mellitus (DM) is an endocrine illness caused by an acquired or inherited deficit of insulin formation.1 Nearly 537 million adults suffered from DM in 2021, and it is predicted to affect 783 million by 2045.2 Diabetes nephropathy (DN) is one of the severe enduring complications of DM resulting in hyperglycemia, proteinuria and reduced glomerular filtration rate (GFR). Diabetes nephropathy has an average occurrence of almost 3% per year throughout the initial 10–20 years after the onset of DM.3, 4 Around 30–40% of DM patients develop DN that advances to renal injury,5 which can lead to chronic kidney failure and end-stage renal disease (ESRD). Characteristic pathogenesis related to DN resulting in kidney failure and ESRD comprises hypertrophy, glomerular hyperfiltration, basement membrane hardening, nodular glomerulosclerosis, mesangial matrix accretion, and proteinuria.6 Advanced kidney damage can result in lipid disorders, inflammation, hemodynamic abnormalities, oxidative stress, apoptosis, and the release of inflammatory mediators and cytokines,7 all of which can trigger inflammation, hyperproduction of reactive oxygen species (ROS) and mitochondrial impairment.8 The pathogenesis of DN is not fully understood owing to its complex nature,9 and existing therapeutic approaches cannot efficiently prevent the progression of DN, warranting further in-depth research.
Generally, DN has been regarded as an endocrine and metabolic disorder that, through hyperglycemia, worsens inflammation and triggers the stimulation and discharge of numerous chemokines, cytokines, growth factors, and cell adhesion molecules.10, 11 It has been established that toll-like receptor 4 (TLR4)/nuclear factor kappa B (NF-κB) is a central signaling pathway that plays a critical role in DN by facilitating immune-inflammatory reactions.12, 13 High mobility group box 1 protein (HMGB1) is a vastly non-histone conserved protein that belongs to a highly migratory protein family.14 The HMGB1 pathway is mostly intermediated by glycation end-product receptors and toll-like receptors (TLRs). The HMGB1 extracellular domain and its receptors trigger monocytes/macrophages, ultimately leading to NF-κB stimulation and production of pro-inflammatory mediators. The NF-κB is a ubiquitous and significant nuclear transcription element that facilitates numerous inflammatory pathways.15 Stimulation of NF-κB may induce the expression of HMGB1 and its receptors, liberating cytokines that promote monocyte and macrophage stimulation. In the innate immune system, pattern recognition receptors of conserved families of TLRs stimulate inflammatory downstream signaling in reaction to microbial exogenous agents.16 Triggering of TLRs signal is connected with the stimulation of NF-κB activity and results in elevated chemokine and cytokine release, including monocyte chemoattractant protein 1 (MCP1), interleukin (IL)-1β and IL-6, which sequentially induce native inflammation and accretion of the leukocyte.17, 18 Specifically, TLR4 has been shown to be involved in the pathogenesis of severe kidney damage, the incidence of DN and prolonged kidney illnesses.19 The downstream effector of the TLR4 signaling is NF-κB, and it is widely thought that activation of NF-κB has a fundamental role in inflammation in the kidney during DN progression.16 Hence, we hypothesize that TLR4/NF-κB signaling pathway suppression is capable of reducing the renal inflammatory responses associated with DN.
Acacetin (AC) is an O-methylated flavone sequestered from many herbal plants, including Betula pendula, Robinia pseudoacacia, Carthamus tinctorius, and Turneradiffusa.20, 21 Acacetin has displayed numerous neuroprotective, antioxidant, anti-peroxidative, and anti-inflammatory effects, and has shown activity against prostate, lung, skin, and breast carcinomas.22, 23, 24, 25, 26 Acacetin exerts a protective influence in assorted cardiovascular diseases, including atherosclerosis, atrial fibrillation, doxorubicin-induced cardiomyopathy, and ischemia/reperfusion (I/R) heart injury.27, 28, 29, 30, 31 Recently, it has been documented that AC mitigates the PPAR-α/AMPK pathway in streptozotocin (STZ)-induced diabetic cardiomyopathy by constraining energy metabolism and oxidative stress.32 Acacetin repressed E-selectin manifestation by the MAPK signaling and NF-κB stimulation in human umbilical vein endothelial cells (HUVECs).33 Acacetin has also demonstrated the ability to avert cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) inflammatory mRNA levels in RAW 264.7 cells by suppressing NF-κB triggering via involving with the MAPK and PI3K/Akt/IKK signaling.34 Furthermore, a prior study reported that AMPK-facilitated Nrf2 triggering by AC is included in cardiomyocyte defense counter to re-oxygenation and hypoxic harm by its anti-inflammatory, anti-oxidative and anti-apoptotic actions.29 Together, these results illustrate the contribution of AC in inflammation-related disorders and oxidative stress. However, the effect of AC on DN has not been completely explored.
Streptozotocin is an effective experimental model for inducing DN, as it selectively abolishes pancreatic β-cells by producing ROS, which affects insulin secretion.35 Streptozotocin produces an inflammatory reaction by discerningly abolishing islets of β-cells through manifold consecutive injections, which cause additional impairment of β-cell function that leads to decreased insulin and high glucose levels.36 Consequently, multiple consecutive injections of the STZ are generally employed for creating experimental DM animal models.35, 36
Objectives
Our research aims to examine the possible nephroprotective mechanisms of AC using an STZ-induced DN model in rats and confirm the hypothesis that AC ameliorates oxidative stress and inflammation through the regulation of the TLR4/NF-κB pathway.
Materials and methods
Chemicals
Acacetin and biochemicals were purchased from Ruicong Ltd (Shanghai, China). Streptozotocin was obtained from Novartis (Beijing, China). Irbesartan (IRB) was delivered by Sun Pharmaceutical Industries Ltd (Shanghai, China). Antibodies for western blot analysis were bought from Ese-Bio (Shanghai, China). Analytical-grade biochemicals and solvents were used.
Experimental animals
A total of 40 adult male Sprague Dawley rats aged 8–10 weeks and weighing between 180–200 g were selected 1 week prior to trials. Animals were preserved in aseptic polypropylene cages under specified laboratory environment, and nourished with a regular pellet diet ad libitum. All treatment protocols were piloted according to the animal ethical committee of the ChunanCounty Traditional Chinese Medicine Hospital, Hangzhou, China (approval No. 024).
Induction of diabetes mellitus
Animals were starved overnight and then STZ (180 mg/kg body weight (BW))37 dissolved in 0.1 M citrate buffer (pH 4.5) was injected intraperitoneally (i.p.). After the 3rd day of STZ administration, sample blood was collected by tail puncture, and blood glucose (BG) levels were tested using a glucometer (LifeScan Inc., Malvern, USA).. Animals with BG levels above 300 mg/dL were identified as the diabetic controls.38
Investigational protocol
Rats were separated into a normal control group and 3 diabetic groups, with 10 rats in each. They were divided into 4 sets: normal control (NC); diabetic control (DC), DC+IRB (180 mg/kg/day); and DC+AC (10 mg/kg/day) for 8 weeks. On completion of the experimental period, animals were starved for 12 h and sedated with pentobarbital sodium (i.p., 45 mg/kg).39 Blood samples were collected through cardiac puncturing of the retro-orbital and various biochemical parameters were estimated after sacrificing the animals. Centrifugation was performed at 10 × g rpm for 15 min for the separation of serum, and the separated serum sample was kept at –80°C for future use. The kidney was weighed, sliced and fixed in a 10% formalin-neutral buffer for histopathological examination.
Estimation of body weight and blood glucose level
Body weight and BG were determined at 2-week intervals using blood samples taken by tail puncture in the lateral vein. Blood glucose levels were tested using a glucometer (LifeScan Inc.)
Biochemical analysis
The levels of blood urea nitrogen (BUN) and creatinine (Cr) in urine and serum were estimated spectrophotometrically using the commercial assay kits cat. No. ab83362 (Jiancheng Bioengineering Institute, Nanjing, China) in an Olympus AU 600 Autoanalyzer (Olympus Corp., Tokyo, Japan). Malondialdehyde (MDA) and total superoxide dismutase activity (T-SOD) in serum and kidney were measured with commercial assay kits cat. No. ab118907 provided by Biomed (Badr City, Egypt). The quantity of microalbuminuria (m-ALB) was assayed by commercial kit cat. No. E-TSEL-H0005 (Elabscience Biotechnology, Wuhan, China) as per the manufacturer’s protocol.
Histopathological analysis of kidney
For histopathological examination, the kidney was separated, tissues fixed with formalin (10%) and paraffin-embedded. To evaluate histopathological variations in the kidneys, 5-µm sections were cut using rotary microtome (HV-HSMA-1090A; Hoverlabs, Ambala, India) stained with hematoxylin and eosin (H&E), and observed through a microscope (BS-2074T; Beijing BestScope Technology Co., Ltd., Beijing, China).
Analysis of western blot
Total proteins were isolated from the rat’s kidney tissue homogenate for the assessment of the role of AC and IRB on the signaling of TLR4/NF-κB. Kidney tissues were prepared by adding lysis buffer in ice-cold conditions for western blot analysis. Briefly, total protein was estimated using the Protein Assay Kit (Pierce Chemical Co, Rockford, USA), quantified, electrophoretically dispersed, and transferred to a polyvinylidene difluoride (PVDF) film. It was blocked by using a probe at room temperature for 1 h with treated primary antibodies TLR4, interleukin 1 receptor-associated kinase 4 (IRAK4), tumor necrosis factor receptor-associated factor 6 (TRAF6), inhibitory kappa B kinase beta (IKKβ), nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κBp65), and HMGB1. in 1:1,000 dilutions, and kept at 4°C overnight. Then, horseradish peroxidase (HRP)-conjugated secondary antibodies were administered. The protein bands were stained and imaged for protein recognition. Protein bands were quantified through densitometry with ImageJ (National Institutes of Health (NIH), Bethesda, USA) and normalized to glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression.
Statistical analyses
The data from each group were analyzed statistically using GraphPad Prism v. 8.0.2 (GraphPad Software, San Diego, USA) and IBM SPSS v. 25 (IBM Corp., Armonk, USA). The measurement data were reported as the median (min–max). The normality of the distribution was tested using the Kolmogorov–Smirnov test. Since all the distributions were normal, the Brown–Forsythe test was used to establish the equality of variances. Significant differences between multiple groups were analyzed using the Kruskal–Wallis test, and Dunn’s post hoc test was used for multiple comparisons. If p < 0.05, the data divergence was considered statistically notable. All tests in this study were bilateral.
Results
The results of the Kruskal–Wallis test and Dunn’s post hoc test are presented in Table 1, Table 2.
Influence of acacetin on body weight and blood glucose levels
The body weight (BW) gain was considerably reduced (p < 0.05) in DC rats, whereas the BG level was substantially elevated (p < 0.01) against NC (Table 1, Figure 1, Figure 2). Administration of IRB (180 mg/kg) and AC (10 mg/kg) considerably (p < 0.05) enhanced BW gain; however, they reduced the level of BG during the experiment period. The effect was higher in IRB-treated DC rats than in AC-treated rats.
Influence of acacetin on microalbuminuria and diabetic nephropathy factors
As shown in Table 1, and Figure 3A, STZ administration expressively (p < 0.01) increased the level of microalbuminuria (m-ALB), BUN and Cr in serum, whereas the levels of Cr and BUN were significantly (p < 0.05) lower in urine, confirming that STZ-administration could cause DN in rats (Table 1, Figure 3B). Irbesartan and AC may inverse this inclination significantly (p < 0.05), as revealed by a reduced level of BUN, m-ALB, and Cr in serum, while elevated BUN and Cr were significant in urine. On the other hand, IRB and AC considerably (p < 0.05) reduced the m-ALB contained in the 24-h urine sample. These results established that IRB and AC could relieve STZ-induced renal impairments.
Histopathological investigation of kidney
Histopathological analysis of the kidney of DC rats depicted apparent glomerulus hypertrophy, mesangial cells proliferation, expanded Bowman’s capsule, hyperplasia, basement tissue thickening, infiltration of inflammatory cells, and a few vacuolar tubular cells collapse with glycogen accumulation, in contrast to NC rats (Figure 4). Conversely, the rats administered with IRB and AC demonstrated less Bowman’s capsule expansion, slight thickening of mesangial cells, absence of palpable extension of the glomerulus, and mild glycogen accumulation due to their nephroprotective effects.
Effects of acacetin on oxidative markers in serum and kidney
Oxidative markers in DC rats revealed that T-SOD status in serum was reduced substantially, while there was no apparent alteration in the contents of T-SOD in renal cells (Table 1, Figure 5). In DC model rats, the T-SOD content was expressively (p < 0.01) less in serum than that in the NC, whereas the IRB and AC group had significantly (p < 0.05) elevated T-SOD level. The concentration of serum MDA was considerably elevated (p < 0.01) in DC rats, whereas the MDA dropped to near ordinary levels in STZ-induced rats administered IRB and AC. No statistical difference (p < 0.05) was noted in the MDA level of the kidneys between groups. These results suggest that IRB and AC could attenuate oxidative stress and improve the antioxidant status in the STZ-induced DN rat model.
Analysis of TLR4/NF-κB pathway
Streptozotocin-induced DC rats were seen to expressively upregulate HMGB1 (p < 0.01) and TLR4/NF-κB compared to the NC group, signifying that the TLR4/NF-κB pathway was triggered in the STZ-induced DN rats (Table 1, Figure 6). These proteins were considerably (p < 0.05) downregulated after treatment with IRB and AC, suggesting that AC can avert the pathway of TLR4/NF-κB in DN rats by exhibiting nephroprotective action against renal inflammation.
Discussion
Streptozotocin-induced DM affects the β-cells damage in the islets of Langerhans due to depleted insulin secretion, which further induces diabetes-associated complications, particularly DN.35, 36 In the present research, STZ-induced rats exhibited characteristic features of DM, such as loss of BW and high glucose levels, along with enhanced renal tissue impairment. Diabetic nephropathy, as a severe microvascular complication, shows histologically significant renal injury, including glomerulosclerosis, mesangial dilatation, renal tubule interstitial fibrosis, and hardening of the glomerular basement membrane, progressively diminishing the normal function of the kidneys.5, 6 This persistent disorder is mostly related to a constant elevation of proteinuria and a severe decline in the GFR. Throughout the course of DM, BW reduction is a main complication of DN, and our results also showed reduced BW in STZ-prompted DN in rats. It has been previously reported that dehydration and catabolism of protein, as well as fats, lead to the loss of BW after STZ administration.40 In DN progression, proteinuria is a vital index, as it directly reveals kidney function through GFR and renal tubular re-absorption.3, 4 This study demonstrated that AC not only effectively attenuated renal histopathological damage and BG levels, but also increased serum Cr, BUN and m-ALB 24-h urinary protein. Acacetin treatment considerably increases BW owing to its potential effect in preventing the wasting of muscle. Streptozotocin can affect the function of the β-cells of the islets of Langerhans, subsequently restraining insulin discharge, which in turn forms hyperglycemia and DM-allied complications, including DN. Our research aimed to assess the protective mechanism of AC against oxidative stress and inflammation and its ability to exert nephron protection from STZ-incited DN in rats. To the best of our knowledge, this study offers the first evidence for the potential of AC against kidney impairment in the STZ-prompted DN model.
Hyperglycemia is the key mark of DM, which generates extreme ROS within the body and leads to elevated levels of cytokines formation, hastening renal cell destruction. The joint action of inflammation and oxidative stress has a precise, crucial role in the pathogenesis and progression of renal impairment in DM. In DM, extreme ROS generation can lessen the activity of the antioxidant defense mechanism that causes the oxidation of DNA, proteins and lipids after DN.7, 8 Furthermore, high BG is susceptible to oxidative stress-prompted cell injury caused by attenuation of antioxidant capability in glycation of scavenging enzymatic antioxidants comprising glutathione peroxide (GPx) and superoxide dismutase activity (SOD). This may be attributed to the connection of glucose with proteins to form advanced glycation end products (AGEs), which deactivate enzymatic antioxidants by blocking receptors.15 In STZ-induced DM in rat models, inflammation and oxidative stress have a crucial role in the progression of DN. In our research, STZ-induced DN exhibited elevated levels of MDA; conversely, T-SOD levels were considerably lower (p < 0.05) than in the control. The administration of AC (10 mg/kg BW) lowered the MDA levels while elevating the T-SOD level significantly (p < 0.05) in contrast to STZ-induced DN control rats. These findings support the theory that the ROS foraging by AC may be moderately connected to the enhanced action of enzymatic antioxidants and anti-inflammatory effects.22, 23, 24, 25, 26 Lipid peroxidation products, such as MDA, are used as a marker of tissue impairment due to increased ROS formation. Streptozotocin is recognized to enhance MDA levels in tissues of several organs, particularly kidneys.22, 23 Acacetin was reported to attenuate kidney damage by suppressing oxidative stress and apoptosis in mice models.41 An earlier study has recognized that AC mitigates STZ-induced diabetic cardiomyopathy by averting oxidative stress and energy metabolism via PPAR-α/AMPK signaling.32 Another study concluded that AC could diminish the inflammatory reaction of myocardial tissue by constraining the levels of IL-6 and TLR4 signaling. Myocardial cell apoptosis results from multiple elements, including myocardial I/R inflammation and oxidative stress.42 The above data suggest that the addition of AC lessens oxidative stress and inflammation, inhibiting nephropathy development in STZ-induced DM rats.
TLR4/NF-κB signaling is a typical inner inflammatory signal transduction pathway and the NF-κB pathway can be triggered by TLR4.12, 13 The TLR4 initiation successively activates the NF-κB allied pathway via the MyD88-reliant pathway, which mediates the discharge of inflammatory chemokines and cytokines responsible for leukocyte accumulation and innate inflammation (MCP-1, IL-6, IL-1β, and tumor necrosis factor alpha (TNF-α)).10, 11 The NF-κB controls the expression level of cellular genes responsible for inflammation as a significant transcription factor in DN pathogenesis. Under standard physiological conditions, NF-κB and IκB associate to form an inactive complex, which is confined in the cytoplasm. Then, IκB is degraded once phosphorylation occurs, and this is followed by ubiquitylation while the cells are in reaction to the peripheral stimuli. Successively, NF-κB arrives at the nucleus to affix to target genes that facilitate inflammation. The NF-κB activation is frequently stimulated by oxygen free radicals, cytokines, hyperglycemia, and even viruses. Moreover, proteinuria connected with DN also plays a significant role in its stimulation.3, 4, 7 There is strong evidence that NF-κB activation is integral in the progression of nephritis and fibrosis resulting in DN.16, 17 Several studies have established that DN in rats was reduced by constraining the NF-κB stimulation and subsequently reducing other inflammatory mediators.18, 19 The elevated levels of inflammatory mediators indicate inflammation in the body. These results established that AC expressively ameliorated kidney dysfunction and oxidative stress in DN rats. Furthermore, a recent study has substantiated that AC inhibited phosphorylation of NF-κB, and IκB degradation, ultimately leading to the inhibition of NF-κB nuclear translocation,43 consistent with our findings. In the current experiments, AC attenuated the HMGB1 and TLR4 in the DN rat’s kidney tissue simultaneously and downregulated the TLR4 downstream proteins, including IRAK4, TRAF6 and Iκκβ, to facilitate further reduction of NF-κB stimulation.
High mobility group box 1 is a highly conserved non-histone DNA binding protein, which is extensively dispersed between numerous body parts, including the brain, lung, heart, kidney, and liver, and it can be discharged from necrotic cells through active discharge or passive release, causing inflammation.14 It is discharged from inflammatory or necrotic cells, comprising dendritic cells, macrophages and monocytes, as a strong pro-inflammatory cytokine, and interacts with several cell-surface receptors such as TLR2, TLR4 and receptor for advanced glycation end products (RAGE).15 Hence, HMGB1 is known as the endogenous ligand of TLR4, and the communication of HMGB1 with TLR4 marks an auxiliary translocation of NF-κB from the cytoplasm into the nucleus, prompting an inflammatory reaction.14, 15 In agreement with these results, HMGB1 plays a vital action in the pathogenesis of renal illness and DN. Previously, hyperglycemia-prompted release of HMGB1 was tested as a cause of kidney damage in DM rats. The pathogenic action of this HMGB1 is dependent on TLR4, with further stimulation of NF-κB. Thus, HMGB1/TLR4/NF-κB is a significant inflammatory pathway in kidney syndromes and recent literature showed an analogous enhancement in TLR4, HMGB1 and NF-κB in DN.15, 16, 17, 18 Similarly, our research suggests that AC administration could increase amounts of HMGB1 and TLR4 in DM rats, emphasizing the possible nephron-protective influence of AC through reducing inflammation in DN. The present study demonstrates that the nephroprotective effect of AC in STZ-activated DN in rats encompasses cell signaling pathways, including HMGB1, TLR4 and NF-κB.
Limitations
Altogether, the nephroprotective effect of acacetin in streptozotocin-induced diabetic nephropathy was revealed in rats. TLR4, IRAK4, TRAF6, IkkB, NF-KB p65, and HMGB1 were studied only in proteins levels; the function of the nuclear level needs to be further verified.
Conclusions
This study revealed that AC mitigated kidney damage in STZ-stimulated DN rats by suppressing oxidative stress and inflammation through suppression of the TLR4/NF-κB pathway. These findings propose that AC might be a potential nephroprotective agent for the treatment of DN. However, further molecular mechanisms require investigation to fully understand the renoprotective efficacy of AC.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.13148016. The package includes the following files:
Supplementary Fig. 1. Results of Kruskal–Wallis test as presented in Figure 1.
Supplementary Fig. 2. Results of Kruskal–Wallis test as presented in Figure 2.
Supplementary Fig. 3. Results of Kruskal–Wallis test as presented in Figure 3.
Supplementary Fig. 4. Results of Kruskal–Wallis test as presented in Figure 5.
Supplementary Fig. 5. Results of Kruskal–Wallis test as presented in Figure 6.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.

















.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)