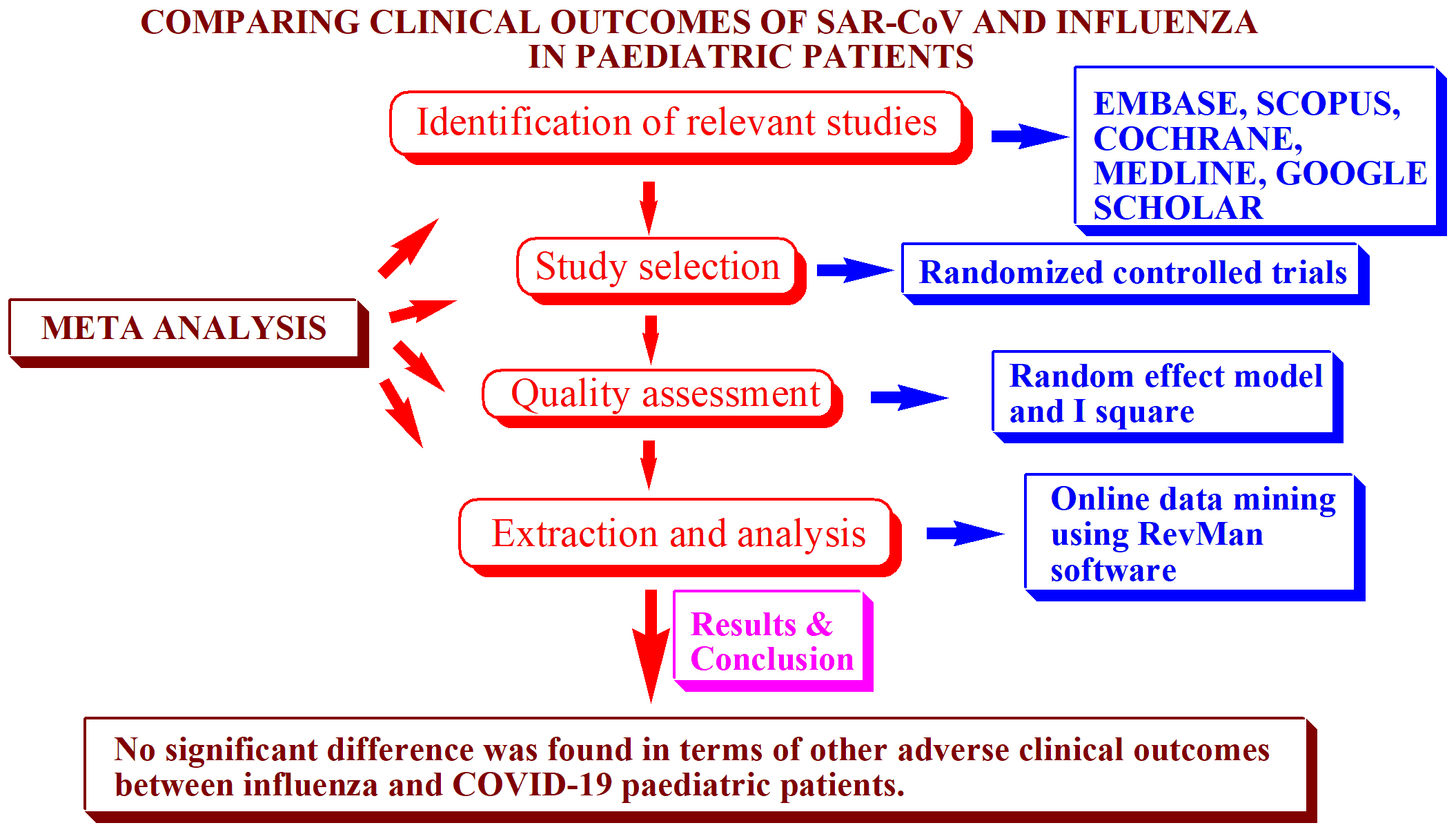
Abstract
Only a few studies have examined the effects of coronavirus disease 2019 (COVID-19) and influenza on clinical outcomes in pediatric patients. Furthermore, no meta-analysis has assessed the impact of these diseases on adverse outcomes. This study aims to compare the clinical outcomes of COVID-19 and influenza in pediatric patients. Searches were conducted from December 2019 to February 2022 in databases including Embase, Scopus, PubMed Central (PMC), MEDLINE, Google Scholar, Cochrane Library, and ScienceDirect. Our meta-analysis used a random-effects model, reporting pooled odds ratios (ORs) or standardized mean differences with 95% confidence intervals (95% CIs). Thirteen studies meeting the inclusion criteria were analyzed. Most studies had poor quality. The pooled OR was 0.13 for oxygen requirement (95% CI: 0.04–0.45; I2 = 74%) and 0.03 for steroid requirement (95% CI: 0.01–0.19; I2 = 60.8%). No significant differences were found in outcomes such as intensive care unit (ICU) admission, duration of inpatient stay, invasive/non-invasive ventilation, death, acute respiratory distress syndrome (ARDS), and acute kidney injury (AKI). SARS-CoV-2 infection was comparable to influenza regarding mortality, pediatric intensive care unit (PICU) admissions, mechanical ventilation, and AKI incidence, but with notable differences in oxygen supplementation.
Key words: COVID-19, SARS-CoV, meta-analysis, pediatrics, influenza
Introduction
In recent years, public health experts have been anticipating the potential emergence of a highly contagious respiratory virus with the capacity to cause a pandemic.1 Upon the emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in late 2019, a prompt comparison was made between this virus and both seasonal and pandemic influenza viruses due to the notable similarities exhibited by these viral entities. The disease conditions induced by both of these viruses exhibit comparable clinical manifestations, including symptoms such as fever and respiratory distress. These symptoms range from milder forms, such as cough and sore throat, to more severe manifestations, including lung infections.2, 3 Both SARS-CoV-2 and influenza viruses have comparable modes of transmission, as they can be spread through respiratory droplets, facilitating efficient human-to-human transmission.3, 4 SARS-CoV-2 and influenza are preventable through vaccination.2 Although the vaccine may be less effective in older individuals, it can make the illness less severe and reduce the chance of complications and death.
The clinical manifestations of hospitalized patients with SARS-CoV-2 and seasonal influenza exhibit significant differences.5 There is a high risk of SARS-CoV-2 infection in children of all ages, but the disease typically manifests in a mild manner and usually does not cause lasting consequences. Critical illness and death from SARS-CoV-2 in children are extremely rare.6 Among SARS-CoV-2 patients, the primary mode of treatment is mostly supportive, although several experimental antiviral medications are currently being evaluated.7 Prevention, timely judgment and adequate supervision of pediatric patients infected with influenza are crucial. In contrast, there is a lack of pediatric data comparing influenza and SARS-CoV-2. Newer and more severe forms of clinical expressions related to SARS-CoV-2 are continually developing in the pediatric age group. However, to the best of our knowledge, earlier published meta-analyses did not compare the clinical outcomes between SARS-CoV-2 and influenza, specifically in pediatric patients.
Objectives
The main objective of the current study was to compare the clinical outcomes between influenza and SARS-CoV-2 patients in the pediatric age group.
Materials and methods
Study participants
Studies containing both influenza and SARS-CoV-2 patients in the pediatric age group (<18 years) as a separate group were included.
Outcomes
The included outcomes were as follows: mortality,, intensive care unit (ICU) admission, necessity for mechanical ventilation (invasive and non-invasive), sepsis, oxygen requirement, acute kidney injury (AKI), steroid requirement, acute respiratory distress syndrome (ARDS), and duration of inpatient stay.
Study design
Prospective and retrospective observational study designs were eligible. Full-text studies were included. Research questions were framed in the PICO (P – patients, I – intervention; C – comparator; O – outcome) format as follows: P: influenza and SARS-CoV-2 patients in the pediatric age group; I: none; C: non-influenza and SARS-CoV-2 patients in the pediatric age group; O: mortality, ICU admission, necessity for mechanical ventilation (invasive and non-invasive), sepsis, oxygen requirement, AKI, steroid requirement, ARDS, and duration of inpatient stay.
Information sources and search strategy
A thorough and rigorous literature review was conducted by systematically searching multiple databases, including Embase, Scopus, PubMed Central, Cochrane Library, MEDLINE, Google Scholar, and ScienceDirect. Our search strategy included Medical Subject Headings and free-text terms with Boolean operators (“AND” & “OR”). An additional English-language filter and a time point restriction from December 2019 to February 2022 were applied. Terms referring to children and SARS-CoV-2 or influenza were used, with the complete list available in the Supplementary data.
Selection process
The initial phase of study selection was conducted by 2 independent investigators (C.L. and F.H.), who screened titles, keywords and abstracts. Duplicates (same study available in different databases) were identified and excluded. Both investigators collected the full-text studies and subsequently narrowed down the selection for the next round of screening, considering the predetermined eligibility criteria. Any inconsistencies during the initial phase were resolved through mutual agreement between these 2 investigators. During the 2nd phase, the same 2 investigators (C.L. and F.H.) reviewed the retrieved full texts. Only eligible studies were selected for further analysis. The review employed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist 2020 for guidance.8 Conflicting assessments were resolved after discussion among authors.
Data collection process and data items
Following the completion of the selection process for relevant full-text articles to be included and analyzed in the review, the 2 above authors actively participated in the manual extraction of data to obtain information such as authors’ details, study title, publication year, study period and duration, design, setting, country, sample size, outcome details, and mean age of the participants.
Risk of bias assessment
Two authors (C.L. and F.H.) assessed the risk of bias using the Newcastle-Ottawa scale for observational studies, which includes selection, comparability and outcome domains. Studies with scores between 7–8 stars were considered of “good” quality, 5–6 stars indicated “satisfactory” quality and 0–4 stars indicated “unsatisfactory” quality.9
Statistical analyses (synthesis methods, effect measures, reporting bias assessment)
This meta-analysis was conducted using the Metafor R package (R Foundation for Statistical Computing, Vienna, Austria). The binary outcomes were assessed by inputting the number of events and sample size for each group, and the combined effect was presented as the pooled odds ratio (OR) with a 95% confidence interval (95% CI).
The model selection was based on the variation among the studies in terms of study design, sample size, type of participants, methodology, etc. The random effects model was applied. Heterogeneity was evaluated using a χ2 test, and the level of inconsistency was quantified using the I2 statistic. I2 < 25% indicated mild heterogeneity, I2 between 25% and 75% indicated moderate heterogeneity, and I2 > 75% indicated substantial heterogeneity.10 Forest plots showed the study-specific and pooled estimates. Sensitivity analysis was performed to determine whether the pooled estimate varied after removing studies one by one in the analysis. This ensured the robustness of the estimates and assessed the possibility of any small study effects. Publication bias was assessed for outcomes with at least 10 studies.
Results
Study selection
A total of 1,120 items were found. Of these, 801 studies were excluded for various reasons, such as not involving either SARS-CoV-2 or influenza patients, review articles, case reports, case series, systematic literature reviews, meta-analyses, formulation development for SARS-CoV-2 and influenza patients, as well as analytical studies. Finally, 99 studies were deemed relevant, and full texts were obtained. A final consensus was reached to include 13 studies with a total of 18,516 participants (Figure 1 and Table 1) after screening against the eligibility criteria.11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23
Characteristics of the included studies
Almost all analyzed studies (except Pokorska-Śpiewak et al.)18 were retrospective. Most studies were conducted in China (3 studies) and Turkey (3 studies). Sample sizes varied from 47 to 10,169. The average age in the SARS-CoV-2 group ranged from 12 to 128 months, and in the influenza group from 16 to 112 months. Most studies involved both influenza A and B patients. All studies were found to be of fair and good quality, as indicated in Table 1.
Admission to Intensive Care Unit
Eight studies compared ICU admission rates between SARS-CoV-2 and influenza patients in the pediatric age group. The pooled OR was found to be 1.26 [0.86, 1.86], indicating no significant difference between pediatric patients with SARS-CoV-2 and those with influenza (Figure 2A). However, heterogeneity among studies was high, as indicated by the I2 statistic. Cochran’s Q test (p < 0.01) and χ2 test (p = 0.161) also indicated significant heterogeneity among the studies. Therefore, a sensitivity analysis was conducted.
The sensitivity analysis results showed the impact of high sample size studies on the outcome (Figure 3A). The heterogeneity among studies was found to decrease. Furthermore, no significant difference between pediatric patients with SARS-CoV-2 and influenza was observed even after the removal of both high sample size studies,17, 21 as shown in Figure 3. A symmetrical funnel plot and Begg’s test (p = 0.322) (Supplementary Fig. 1) both indicated that there was no substantial publication bias.
Mechanical ventilation
(invasive/non-invasive)
Four investigations revealed differences in the need for mechanical ventilation between children with SARS-CoV-2 and those with influenza. No significant difference was observed between pediatric patients with SARS-CoV-2 and those with influenza, as indicated by the pooled OR of 1.41 (95% CI: 0.74–2.67; I2 = 84%) (Figure 3A). The heterogeneity among studies decreased from 84% to 67%, as shown in Figure 3A,B. The Cochran’s Q and χ2 tests (p = 0.303) also indicated significant heterogeneity among the studies. Sensitivity analysis results showed no impact on the conclusion (Figure 3B). A symmetrical funnel plot and Begg’s test (p = 0.497) (Supplementary Fig. 2) both indicated no substantial publication bias.
Invasive mechanical ventilation
Three studies compared the requirement for invasive mechanical ventilation between pediatric influenza and SARS-CoV-2 cases. The pooled OR was 1.34 (95% CI: 0.51–3.48), indicating no significant difference in the need for invasive mechanical ventilation between pediatric influenza and SARS-CoV-2 patients (Figure 4). Cochran’s Q test (p < 0.01) and χ2 tests (p = 0.447) also indicated significant heterogeneity among the studies. Sensitivity analysis was not performed due to the availability of only 2 studies. The funnel plot indicated minimal publication bias (Supplementary Fig. 3).
Non-invasive mechanical ventilation
Two studies compared the requirement for noninvasive mechanical ventilation between SARS-CoV-2 and influenza patients in the pediatric age group. The pooled OR was 1.34 (95% CI: 0.51–3.48; I2 = 93%), indicating no significant difference between pediatric patients with SARS-CoV-2 and those with influenza (Figure 4). Cochran’s Q test (p < 0.01) and χ2 tests (p = 0.447) also indicated significant heterogeneity among the studies. Due to the limited number of studies, sensitivity analysis could not be performed. The funnel plot indicated minimal publication bias (Supplementary Fig. 4).
Oxygen requirement
Four investigations compared oxygen requirements between the 2 groups. The pooled OR was 0.34 (95% CI: 0.11–1.06; I2 = 0%), showing no significant differences (Figure 5A). Cochran’s Q test (p = 0.61) and χ2 test also indicated nonsignificant heterogeneity among studies. Sensitivity analysis results showed no impact of outliers on the study outcome, as shown in Figure 5B. The funnel plot indicated minimal publication bias (Supplementary Fig. 5).
Steroid requirement
Two investigations examined the difference in steroid requirements between SARS-CoV-2 and influenza patients in the pediatric age group. The pooled OR was 0.56 (95% CI: 0.06–5.31; I2 = 92%), indicating no significant differences (Figure 6A). Cochran’s Q test (p < 0.01) and χ2 test (2.451) also indicated significant heterogeneity among studies. Sensitivity analysis was not performed due to the limited number of studies.
Acute kidney injury
Four studies compared AKI between SARS-CoV-2 and influenza patients in the pediatric age group. The pooled OR was 1.31 (95% CI: 0.84–2.04; I2 = 94%), indicating no significant difference between influenza and SARS-CoV-2 pediatric patients (Figure 6B). Cochran’s Q test (p < 0.01) and χ2 test (0.168) also indicated significant heterogeneity among studies. Sensitivity analysis results showed no significant impact on the outcome, as shown in Figure 7A–C. The symmetrical funnel plot and Begg’s test (p = 0.497) (Supplementary Fig. 7) both indicated no substantial publication bias.
Mortality
Seven studies reported on the difference in mortality between influenza and SARS-CoV-2 pediatric patients. The pooled OR was 1.16 (95% CI: 0.71–1.90; I2 = 72%), indicating no difference between pediatric patients with SARS-CoV-2 and those with influenza (Figure 8A). Cochran’s Q test (p < 0.01) and χ2 test (p = 0.212) also indicated significant heterogeneity among studies. The symmetrical funnel plot (Supplementary Fig. 8) suggested significant publication bias (Begg’s p = 0.652). Sensitivity analysis results showed no impact of outliers on the outcome, as shown in Figure 8B.
Discussion
Coronavirus disease 2019 (COVID-19) is a novel disease caused by a newly identified virus, SARS-CoV-2. It is a positive-sense single-stranded RNA virus that mainly affects the respiratory system.24, 25 Influenza and SARS-CoV-2 are both preventable via vaccination.26, 27, 28 While numerous experimental antiviral medications are presently undergoing assessment, supportive care remains the predominant approach for managing SARS-CoV-2 patients.29 In this evaluation, we compared SARS-CoV-2 patients with influenza patients of all ages to assess the relative risk of several adverse clinical outcomes in terms of severity and complications. We have compiled 13 studies in total. China and Turkey topped the list of countries where these studies were performed, followed by Sweden, France and Poland. Except for the study by Pokorska-Śpiewak et al.,18 all of the other research used a retrospective design, and the majority of them were highly at risk of bias.
A sensitivity analysis showed no significant change in effect magnitude. While no earlier pediatric case evaluations were available to compare with the present study, the reported results are consistent with previous pandemic infections.29, 30 The severity of the SARS-CoV-2 infection among pediatric patients was evidently different from that in adult patients.31 This suggests that the pathogenicity of SARS-CoV-2 infection in children was similar to that of other severe respiratory illness-associated coronaviruses, such as Middle East Respiratory Syndrome Coronavirus (MERS-CoV) and SARS-CoV. Children with SARS/MERS coped better than adults with these illnesses.32, 33
Other adverse clinical outcomes (death, ICU admission, mechanical ventilation, AKI, ARDS, and length of hospital stay) did not differ substantially between SARS-CoV-2 and influenza pediatric patients. However, previous studies on adult patients have found greater mortality and more frequent hospitalization and requirement for mechanical ventilation with late hospital discharge, particularly among individuals over the age of 50.34, 35, 36 This could be due to related comorbidities such as diabetes, hypertension or heart disease among middle-aged and older adults, skewing the adverse outcome rates higher for SARS-CoV-2 patients. More longitudinal research into the outcomes of influenza and SARS-CoV-2 in pediatric patients is needed.
The review’s major strengths are rigorous methodology and extensive investigation of the relevant literature. The evidence comparing the clinical outcomes of children with SARS-CoV-2 and children with influenza is scant, and this study fills a gap in that area. For any of the outcomes under evaluation, there was no discernible publication bias. The reliability of the findings may be enhanced as a result.
Limitations
The majority of the findings revealed a substantial amount of variation from one study to the next. This might influence the external validity (generalizability) of the study results. Virtually all of the studies were retrospective, making it challenging to establish a causal relationship. Therefore, longitudinal research is necessary for determining credible effect estimates and contributing to evidence-based recommendations for creating interventions in the hospital context. Most of the included studies lacked information about comorbidities, which play a substantial role in the outcomes of both infectious agents. Therefore, further studies are required to confirm the effects of comorbidities on clinical outcomes. We have considered only outcomes related to the respiratory system. Neurological outcomes, coagulation issues and other infection-related complications such as myocarditis, Guillain–Barré syndrome (GBS) or multisystem inflammatory syndrome in children (MIS-C) were not considered. There were differences in the results of studies including very young patients compared to older ones, which can produce predefined bias.
Conclusions
Based on our findings and experiences, SARS-CoV-2 infection is comparable to influenza in terms of mortality, pediatric intensive care unit (PICU) admissions, mechanical ventilation, and AKI incidence, with differences mainly observed in oxygen supplementation. Furthermore, the impact of influenza on specific age groups is influenced by the strain. The use of glucocorticoids also plays a significant role in the outcomes of both strains. Therefore, more large-scale and longitudinal studies are required to make a precise judgment on the severity of both of these diseases. The results of this investigation may provide preliminary evidence that SARS-CoV-2 infection may become permanently embedded in everyday life for many years to come (similar to influenza), particularly concerning the differences between these 2 conditions in terms of adverse clinical outcomes.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.13340768. The package includes the following files:
Supplementary Fig. 1. Symmetrical funnel plots and Begg’s p = 0.322 show no publication bias.
Supplementary Fig. 2. Symmetrical funnel plots and Begg’s p = 0.497 show no publication bias.
Supplementary Fig. 3. A funnel plot shows less publication bias for invasive mechanical ventilation.
Supplementary Fig. 4. A funnel plot shows less publication bias for noninvasive mechanical ventilation.
Supplementary Fig. 5. A funnel plot shows less publication bias for oxygen requirement.
Supplementary Fig. 6. A funnel plot shows less publication bias for steroid requirement.
Supplementary Fig. 7. A symmetrical funnel plot and Begg’s p = 0.497 show no notable publication bias.
Supplementary Fig. 8. The symmetrical funnel plot indicated that there was significant publication bias (Begg’s p = 0.652).