Abstract
Background. Placenta previa, occurring when the placenta covers the cervical opening after 28 weeks, can lead to severe postpartum bleeding, especially when coupled with placenta accreta spectrum (PAS), posing risks of organ damage and necessitating hysterectomy. Accurate preoperative diagnosis of PAS in women with placenta previa is crucial to reduce adverse outcomes.
Objectives. This study aimed to develop a risk prediction model for PAS in women with placenta previa.
Materials and methods. A total of 437 patients with placenta previa, delivering babies between January 2012 and December 2018, were included. Data collected encompassed clinical records, neutrophil-to-lymphocyte ratio (NLR) and sonographic findings. Utilizing univariate and multivariate logistic regression analyses, the study identified key factors correlated with PAS in expectant mothers with placenta previa. A risk prediction model was formulated and evaluated through receiver operating characteristic (ROC) analysis. External validation was performed using additional patients diagnosed with placenta previa.
Results. Independent risk factors for PAS in placenta previa included NLR, timing of cesarean section and miscarriage, placenta previa type, presence of placental lacunae, and uterovesical hypervascularity. The predictive model was established using specific coefficients. The ROC curve indicated an area under the curve (AUC) of 0.821, with a sensitivity of 80.6% and specificity of 68.9%. External validation demonstrated a diagnosis coincidence rate of 75%, and the model exhibited good calibration according to the Hosmer–Lemeshow test (p = 0.3742, >0.05).
Conclusions. The developed model showed effective potential in predicting PAS among women with placenta previa. Its application could significantly contribute to the early detection and subsequent management of PAS.
Key words: prenatal diagnosis, NLR, placenta accreta, prediction, placenta previa
Background
Placenta previa, defined as the partial or complete coverage of the cervical opening by the placenta after 28 weeks of pregnancy, affects between 0.24% and 1.57% of pregnancies and is a leading cause of postpartum hemorrhage.1 When placenta previa coexists with placenta accreta spectrum (PAS), the risk of severe postpartum bleeding and damage to adjacent organs significantly increases. In severe cases, hysterectomy may become necessary, posing serious health risks to both mothers and babies.2, 3
Placenta accreta spectrum encompasses a spectrum of disorders characterized by trophoblast tissue invading or adhering to the myometrium, with potential extension into the uterine serosa.1, 4, 5 This spectrum includes placenta accreta, placenta increta and placenta percreta, with the incidence steadily rising due to factors such as maternal age advancement and increasing rates of cesarean deliveries.5, 6 Accurate preoperative diagnosis and risk assessment of PAS in women with placenta previa are crucial to mitigate adverse outcomes and mortality risk.
While ultrasound is the primary method for prenatal PAS diagnosis and risk assessment, its efficacy varies widely, and interpretation depends on operator expertise.7 In cases where specific ultrasound features suggest abnormal placental implantation, magnetic resonance imaging (MRI) is often used for further evaluation.8 However, the efficacy of ultrasound relies on the operator’s skill, and not all sonographers possess the expertise to interpret PAS-related ultrasound characteristics. Additionally, the location of the placenta, especially in cases of posterior placentation associated with placenta previa, can hinder accurate assessment. Although MRI provides a more comprehensive evaluation, it is not universally available, particularly in rural areas.8
In response to the need for reliable predictive biomarkers for PAS, the exploration of serological molecular markers has gained recognition.9 The neutrophil-to-lymphocyte ratio (NLR), commonly used to assess systemic inflammation, has shown independent prognostic significance in various cancers.10 Considering the similarities between excessive trophoblast invasion in PAS and tumor cell invasion and metastasis, NLR emerges as a promising candidate for predicting PAS risk in women with placenta previa.11, 12
By incorporating NLR as a potential biomarker, our study aims to develop and validate a predictive model for preoperatively assessing the risk of PAS in women with placenta previa. This approach may enhance risk stratification and inform clinical decision-making, ultimately improving maternal and neonatal outcomes.
Objectives
Considering the parallels between trophoblasts and tumor cells, and the predictive value of NLR in tumors, this study aimed to explore the effectiveness of NLR as a preoperative assessment tool for PAS in individuals with placenta previa. A 2nd objective was to construct and externally validate a predictive model that integrates clinical, ultrasound and NLR data to identify high-risk individuals.
Materials and methods
Participants
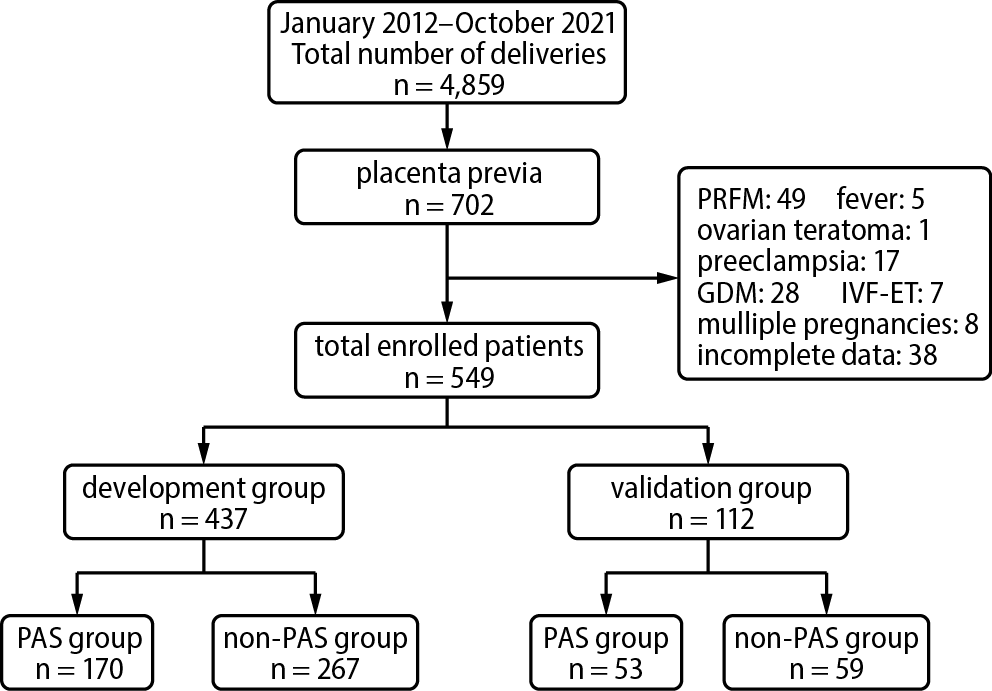
This retrospective analysis included 549 patients with placenta previa who delivered at our hospital from January 2012 to October 2021 (437 patients in the development group, who delivered from January 2012 to December 2018, and 112 patients in the validation group, who delivered from January 2019 to October 2021). Inclusion criteria comprised single pregnancies, live births and gestational weeks ranging from 28 to 42. Exclusion criteria involved neoplastic or infectious diseases, preeclampsia, gestational diabetes mellitus, multiple pregnancies, assisted reproduction, or incomplete data. The research protocol received approval from the Institutional Review Board (approval No. YXLL-KY-2021-018).
The study participants were divided into PAS and non-PAS groups based on intraoperative placental implantation outcomes. The patient inclusion and grouping process are detailed in Figure 1.
Study design and setting
Collection of information
Maternal clinical data encompassed various factors: maternal age, place of residence, gestational age, gravidity, parity, history of previous cesarean sections, number of prior abortions, and any previous uterine surgeries, e.g., laparoscopic or open myomectomy, hysteroscopy, etc. Additionally, perioperative outcomes were examined, including the mode of delivery, post-delivery blood loss within 24 h and infant birth weight.
We collected complete blood counts (CBCs) taken within 1 week before delivery, preceding the administration of dexamethasone for promoting fetal lung maturation. The NLR was derived by dividing the absolute neutrophil count (ANC) by the absolute lymphocyte count (ALC). Similarly, the platelet-to-lymphocyte ratio (PLR) was calculated by dividing the absolute platelet count by the ALC.
Experienced sonographers with over 10 years of practice in ultrasound diagnostics and holding the qualification of Deputy Chief Physician, following guidelines established by the Society for Maternal–Fetal Medicine (SMFM) in 2021, validated the ultrasound diagnosis within 1–2 weeks before delivery.7 The selection of specific ultrasound parameters, including placental lacunae and uterovesical hypervascularity, was based on their documented association with PAS diagnosis and prognosis. Placental lacunae, defined as 3 or more large-sized, irregular, hypoechoic spaces containing vascular flow within the placenta, have been identified as a hallmark feature of PAS on ultrasound imaging. Similarly, uterovesical hypervascularity, characterized by increased flow with the visualization of more than 10 small vessels and/or several main vessels, has been shown to correlate with abnormal placental implantation and increased risk of PAS. By assessing these ultrasound parameters, our study aimed to utilize established criteria to enhance the accuracy and reliability of preoperative PAS risk prediction in women with placenta previa.
Placental data collected included placenta type (classified based on the relationship between the placenta and the cervical opening, categorized as marginal placenta, partial placenta or complete placenta) and placental location, referring to the primary area of the placenta. Placenta accreta spectrum diagnosis was confirmed in all patients, either through intraoperative observations or histopathological examination.
Statistical analyses
For statistical analyses, we utilized IBM SPSS v. 25.0 (IBM Corp., Armonk, USA) and R software v. 4.0 (R Foundation for Statistical Computing, Vienna, Austria). Descriptive statistics were performed to summarize both continuous variables (utilizing mean and standard deviation (SD) or median and interquartile range (IQR), depending on distribution normality) and categorical variables (presented as frequency and proportions). The Shapiro–Wilk test, chosen for its suitability with smaller datasets and sensitivity to deviations from normality, was used to assess the normality of distribution. A significance level below 0.05 typically indicates non-normality.
To develop and validate the regression model predicting the outcome, our study followed a rigorous approach. Initially, the dataset was randomly split into a training set (80%) and a test set (20%) to ensure unbiased model evaluation, preventing temporal biases from influencing results.
For predictor selection, least absolute shrinkage and selection operator (LASSO) regression was employed, especially suitable for models with potentially numerous predictors compared to observations. Such regression aids in coefficient shrinkage and enhances model interpretability by performing variable selection within the regularization process. The tuning parameter (lambda) for LASSO was determined through 10-fold cross-validation on the training set to minimize prediction error and avoid overfitting.
Multivariate logistic regression analysis was conducted to identify independent influencing factors and formulate the PAS prediction model. The assumption of linearity between the logit of the outcome and each continuous predictor was assessed with Stata v. 17 (Stata Corp., College Station, USA) using Component Plus Residual (CPR) plots rather than the Box–Tidwell procedure due to the software’s limitations with handling the Box–Tidwell test for models with multiple continuous predictors, which could lead to computational issues or convergence problems.
The CPR plots visually inspected the relationship, ensuring adherence to the linear logit assumption.
To detect multicollinearity among predictors, the variance inflation factor (VIF) was calculated for each variable. The VIF values greater than 10 indicated significant multicollinearity, prompting model review and potential modification for stable estimates.
Influential observations were identified using leverage and Cook’s distance measures, helping to detect points disproportionately affecting model fit. Observations with high leverage or Cook’s distance significantly exceeding typical thresholds were considered potential outliers or possessing high influence.
Model specification correctness was evaluated using Stata linktest, detecting omitted variables and functional form misspecification through the significant _hat squared term (_hatsq), suggesting potential model adjustments.
Overall model performance was assessed via a receiver operating characteristic (ROC) curve and the Hosmer–Lemeshow test, with statistical significance defined as p < 0.05. Additionally, the Brier score and likelihood-based measures such as the Akaike information criterion (AIC) and Bayesian information criterion (BIC) were applied to assess model calibration and refinement.
Results
A total of 547 pregnant women meeting the inclusion criteria were enrolled in our study. Based on intraoperative clinical assessments and pathological findings, 223 women (40.7%) were diagnosed with PAS. The distribution of placenta accreta, increta and percreta cases was 123 (22.5%), 75 (13.8%) and 25 (4.6%), respectively. Notably, only 2 patients in the PAS group had a history of hysteroscopic electrotomy, precluding a comparison regarding previous uterine surgeries between the groups. Hysterectomy was performed in 13 patients (2.4%). No fatalities occurred during the study period. Comprehensive demographic characteristics and perioperative outcomes for both the training and test sets are detailed in Table 1, and all test assumptions are provided in the Supplementary tables.
Variable selection and model development
In the training set, we employed LASSO regression to optimize the selection of predictive variables from clinical characteristics, CBCs and ultrasound findings. This approach was chosen to ensure a rigorous, data-driven selection process, minimizing potential overfitting and enhancing the model’s predictive accuracy. The LASSO regression was particularly effective in handling the high dimensionality of our dataset and identified 6 variables that exhibited the strongest associations with the outcome of interest.
The variables selected using LASSO regression included:
• time of caesarean section;
• time of miscarriage;
• NLR;
• type of placenta previa;
• presence of placental lacunae;
• uterovesical hypervascularity.
These variables were then further analysed using multivariate logistic regression to determine their independent effects on the risk of PAS.
A risk model to predict PAS
Following the variable selection, multivariate logistic regression was conducted, which confirmed that the 6 variables identified by LASSO regression (time of cesarean section, time of miscarriage, NLR, type of placenta previa, presence of placental lacunae, and uterovesical hypervascularity) were significant predictors of PAS (Table 2). The logistic risk prediction model was formulated as follows: P = exp(w)/ [1 + exp(w)], where W = −3.942 + 0.393 × time of miscarriage + 0.482 × time of cesarean section + 0.296 × NLR + 1.546 × partial placenta (1 for yes, 0 for no) + 1.755 × complete placenta (1 for yes, 0 for no) + 0.871 × uterovesical hypervascularity (1 for yes, 0 for no) + 1.488 × placental lacunae (1 for yes, 0 for no).
Our evaluation of linearity between predictors and the log odds of the outcome confirmed that the relationship is appropriately linear. The plots showed no systematic deviations from a straight line, suggesting that the linear assumption necessary for logistic regression holds true across all continuous variables.
The VIF analysis revealed that all predictors had VIF values well below the threshold of 10, with the highest being 3.2. This indicates that our model does not suffer from multicollinearity, affirming that the coefficient estimates are stable and reliable. Investigation into influential outliers through leverage and Cook’s distance identified no observations, with leverage exceeding twice the average or Cook’s distance greater than the commonly used cutoff of 4/(N-k-1). This outcome suggests that our model is not unduly influenced by outliers, and the results are robust across the sample.
The linktest used for assessing model specification indicated no signs of misspecification. The significance of the _hat term (p < 0.001) and the nonsignificance of the _hatsq term (p = 0.995) confirm that the functional form of the model is correct and that there are no omitted variable biases influencing our results.
Effectiveness of the logistic risk prediction model
The performance of our predictive model in the training set, with an AUC of 0.821 (Figure 2), indicates a strong ability to distinguish between the outcomes of interest, suggesting that the model is highly effective in capturing the essential predictors. The Youden index optimal cutoff value of 0.344, which maximizes the sum of sensitivity and specificity, highlights a well-balanced trade-off between both measures, leading to an overall accuracy of 73.5%. These metrics underscore the robustness of the model in the training phase.
Upon external validation in the test set, the model maintained a robust performance with an AUC of 0.775. This slight decrease compared to the training set is typical, as models often perform slightly better on the data on which they were trained. However, the high specificity of 84.7% in the test set indicates that the model is particularly effective at identifying true negatives, which is crucial for clinical settings where false positives can lead to unnecessary interventions.
The sensitivity in the test set was 64.2%, which, while lower than in the training set, still represents substantial predictive power, especially when considered alongside the high specificity. The accuracy of 75.0% in the test set further validates the model’s utility in external populations and reinforces its generalizability (Figure 3, Table 3).
Additionally, the Hosmer–Lemeshow test result (p = 0.3742) confirms good calibration of the model across different subgroups within the dataset, indicating that the predicted probabilities of outcomes are consistent with the observed probabilities. This aspect of model performance is crucial for clinical applicability, ensuring that the model’s predictions are not only accurate but also calibrated to the realities of clinical practice. The Brier score, which measures the mean squared difference between the predicted probabilities and the actual outcomes, was 0.18, indicating a good level of predictive accuracy. Furthermore, the AIC and BIC were employed to evaluate model complexity and fit, ensuring that the final model avoids overfitting while maintaining predictive robustness.
Discussion
This study aimed to create a model to identify individuals with placenta previa at high risk for developing PAS by integrating clinical risk factors, NLR and ultrasound findings. The developed risk prediction model was externally validated based on these clinical variables, exhibiting superior predictive accuracy compared to clinical diagnosis alone. This study marks the initial exploration of the potential role of NLR as a predictive biomarker for PAS.
Literature on risk assessment and prediction models for PAS in placenta previa remains limited. Prior studies13, 14, 15 have explored scoring systems diagnosing PAS using clinical characteristics and ultrasound findings, while Delli Pizzi et al.14 employed an MRI-based predictive model. These models achieved AUC values ranging from 0.833 to 0.925, sensitivities between 83.3% and 100%, and specificities ranging from 77% to 85.7%. In our study, the prediction model assessing PAS yielded an AUC of 0.821, a sensitivity of 80.6% and a specificity of 68.9%. While our diagnostic efficacy may not surpass that of other studies, the factors included in our prediction model are simple, cost-effective and easily applicable in clinical settings.
The NLR is recognized as a potential biomarker for predicting survival outcomes in various diseases, including cancer and coronary artery disease (CAD).11 Elevated NLR in association with PAS is believed to involve multifactorial mechanisms.
Studies indicate that women with prior cesarean sections exhibit increased uterine artery resistance and a reduced proportion of uterine blood flow in comparison to those with a history of vaginal delivery.16 This discrepancy in oxygen supply to placental tissue may trigger a local inflammatory response, resulting in the production of cytokines, particularly vascular endothelial growth factor (VEGF). Research has demonstrated common occurrences of local fibrous tissue increase and inflammatory cell infiltration around the scar site.4, 17 Immunohistochemistry findings have revealed heightened VEGF and phosphotyrosine in extravillous trophoblast cells, potentially promoting trophoblast infiltration and vascular remodeling among PAS cases compared to normal controls.4, 18
Lymphocytes play essential roles in cytotoxic cell death and cytokine production, impacting immune response inhibition. Pregnant women with placental implantation complications have notably decreased natural killer cells (NK cells), which release cytokines to regulate invasion.19 Recent work by Shainker et al.20 employing a novel proteomics platform unveiled differential plasma protein expression between PAS and control groups. Pathway analyses implicated these differentially expressed proteins in closely regulating inflammation, coagulation, angiogenesis, and invasion in PAS cases. Furthermore, secondary disruptions in the endometrium–myometrial interface might contribute to microenvironmental changes within the uterine scar, leading to immune balance disruptions.
The escalating prevalence of PAS disorders is closely linked to the rising rates of cesarean sections, substantiated by robust epidemiological evidence.5, 21, 22, 23, 24, 25 Notably, a study conducted in Hong Kong observed a surge in PAS incidence among women with prior cesarean deliveries compared to those with unscarred uteruses.24 Correspondingly, as cesarean section rates increased, so did the incidence of PAS disorders. A meta-analysis highlighted an escalating odds ratio (OR) for PAS, rising from 8.6 to 17.4 with 1–2 previous cesarean sections, and soaring to 55.9 following 3 or more prior cesarean deliveries.5, 25 Another comprehensive systematic review revealed an increase in PAS rates from 0.3% after a single prior cesarean section to 6.7% after 6 previous cesareans.26
The underlying mechanism contributing to the surge in PAS may involve uterine scarring, impairing the decidua interface at the implantation site. This scarring potentially diminishes the integrity of the decidua, fostering increased trophoblast adhesion or infiltration. Consequently, placental implantation into the myometrium may occur in subsequent pregnancies. Surgeries leading to endometrial integrity compromise, such as uterine curettage, postpartum endometritis, hysteroscopic surgery, endometrial ablation, and uterine artery embolization, have also been associated with PAS disorders in subsequent pregnancies. Additionally, our study established abortion as an independent risk factor for PAS.
The primary method for diagnosing PAS is transvaginal ultrasound (TVS). Classical ultrasound signs indicative of PAS encompass features such as placental lacunae, loss of the clear zone, interruption of the bladder wall, uterovesical hypervascularity, placental bulge, and myometrial thinning.27 These ultrasound observations are correlated with the pathophysiology of PAS.1, 4 Among these signs, placental lacunae have notably been linked to PAS, with studies emphasizing its association with cesarean hysterectomy and maternal complications.7, 28, 30 Conversely, in patients with placenta previa and prior cesarean sections, the absence of lacunae demonstrated a high negative predictive value (NPV) ranging from 88% to 100% for PAS.7, 29, 30
Alterations in the uterovesical interface present compelling specificity (97.5–99.8%), albeit with lower sensitivity (49.6%).1, 31, 32 Markers observed at the uterus and bladder interface include bridging vessels, hypervascularity between these organs, and interruption of the bladder wall. Color Doppler imaging has proven effective in detecting neovascularity in most PAS cases7; however, some of the ultrasound signs mentioned are not exclusive to PAS. For instance, myometrial thinning may result from a prior cesarean scar.1, 7 Thus, our study specifically selected 2 easily identifiable signs – placental lacunae and uterovesical hypervascularity – which were found to be significantly valuable in distinguishing between the PAS and non-PAS groups.
Despite the pivotal role of ultrasound in the prenatal diagnosis of PAS, the American College of Obstetricians and Gynecologists/The Society for Maternal-Fetal Medicine (ACOG/SMFM) underscores that a negative ultrasound cannot entirely exclude the possibility of PAS. They emphasize the significance of clinical risk factors, highlighting their equal importance to ultrasound findings in predicting PAS (grade 1A).33, 34 Moreover, the Society of Obstetricians and Gynecologists of Canada (SOGC) highlights that the efficacy of ultrasound in diagnosing PAS is contingent on various factors, including knowledge of clinical risk factors, imaging quality, operator expertise, gestational age, imaging techniques, and adequate bladder filling.35 These considerations further underscore the relevance and importance of our composite scoring model.
As anticipated, the type of placenta played a significant role in the risk of placental implantation disorders. Specifically, complete placenta previa demonstrated a higher likelihood of association with PAS. The absence of endometrial re-epithelialization in the scar region, coupled with inadequate blood supply, potentially facilitated wider and deeper invasion by trophoblast and villous tissue.
While the location of placental attachment showed minimal significance, prior studies have indicated that maternal age above 35 years could elevate the odds of PAS disorders.36, 37 However, according to the 2018 International Federation of Gynecology and Obstetrics (FIGO) guidelines on the prenatal diagnosis and screening of PAS,5 this association might be influenced by confounding factors such as multiparity and previous uterine surgeries, rather than solely by advanced maternal age itself. Our present study did not establish a significant association between age and PAS.
The notable strength of our study lies in its pioneering approach incorporating serum markers into the construction of a predictive model. This strategy facilitated a quantifiable prenatal assessment, specifically aimed at identifying cases of placenta previa at high risk for PAS. We rigorously validated this predictive model and confirmed its accuracy using independent samples. By integrating NLR into our predictive model, we not only improved predictive accuracy but also gained valuable insights into the pathophysiological mechanisms driving PAS development. This holistic approach, encompassing both clinical and inflammatory markers, enhances the robustness of our predictive model and provides clinicians with a more comprehensive tool for risk assessment in placenta previa patients.
Limitations
This study was conducted at a single center, and both model development and validation were retrospective. Notably, the enrolled participants had placenta previa, raising uncertainties about the model’s applicability to PAS patients without placenta previa. In future investigations, we intend to prospectively verify our findings and extend the prediction model to encompass all PAS patients, particularly focusing on the 1st and 2nd trimester. This approach is crucial, considering that current studies predominantly concentrate on high-risk pregnancies in the 3rd trimester. Ultimately, such efforts aim to enhance pregnancy outcomes through improved predictive strategies.
Conclusions
The integration of clinical characteristics, NLR and ultrasound findings in a preoperative prediction model holds significant promise for improving patient care and maternal outcomes in cases of PAS among women with placenta previa.
By leveraging our prediction model, clinicians can effectively stratify patients into high-risk categories, enabling the formulation of tailored delivery plans and timings. This approach facilitates early identification of high-risk cases, allowing for closer monitoring and timely interventions when necessary. As a result, unnecessary interventions can be minimized, while optimal management strategies can be implemented to mitigate the risk of maternal morbidity and mortality associated with PAS.
The implementation of our prediction model is anticipated to not only enhance patient care by ensuring appropriate management strategies but also improve maternal outcomes by reducing the incidence of complications associated with PAS.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.12694596. The package includes the following files:
Supplementary Table 1. Variance inflation factor for all the variables in the final prediction model.
Supplementary Table 2. Cook’s distance for all the variables in the final prediction model.
Supplementary Table 3. Verification of test assumptions in the final prediction model.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.


















.jpg)
.jpg)