Abstract
Background. Pulmonary metastasectomy (PM) is an important procedure for the treatment of metastatic nodules in the lung. The choice of surgical approach, whether thoracotomy or video-assisted thoracoscopic surgery (VATS), remains controversial in terms of the impact on patient prognosis.
Objectives. This study aimed to evaluate the outcomes and impact on survival of patients undergoing PM with VATS compared to thoracotomy.
Materials and methods. A retrospective evaluation of 136 patients who underwent PM between September 2012 and July 2020 was performed. Data on the demographics, primary tumor histopathology, metastatic features, surgical approach, surgical outcomes, and survival status were analyzed. Statistical analyses included descriptive statistics, survival analysis and Cox regression models.
Results. Of the participants, 84 underwent thoracotomy and 52 underwent VATS. The median survival time of thoracotomized patients was 86.6 months, while it was 99.6 months for VATS patients. A gender-specific analysis revealed a significantly longer survival time for female VATS patients compared to thoracotomy. Multivariate analysis showed significant independent effects of specific tumor types and the number of nodes removed on survival. Overall, no significant difference in survival was found between the 2 surgical methods.
Conclusions. Both VATS and thoracotomy are effective and safe options for PM. Video-assisted thoracoscopic surgery may offer advantages, particularly in certain patient groups and tumor types, potentially prolonging survival. Gender-specific analyses suggest a survival benefit of VATS, particularly in women. Further studies are needed to validate these results and optimize surgical decision-making in PM.
Key words: survival, pulmonary metastases, surgical approach, metastasectomy
Background
Pulmonary metastasectomy (PM) is a procedure to surgically remove metastatic nodules in the lungs. Before this procedure can be performed, several important criteria must be met. A candidate for PM must first have the primary tumor under control. This may mean surgical removal of the primary tumor or its control through other treatments. A patient who is eligible for PM should have no extrathoracic metastases. This means that the metastases should be confined to the lungs. The patient must be a suitable candidate for surgery. For this purpose, the general state of health, lung function and ability to tolerate surgery are assessed. The decision is made after weighing the surgical risks and benefits.1, 2
There are several surgical approaches for metastasectomy in PM: Video-assisted thoracoscopic surgery (VATS) is a minimally invasive approach that reduces surgical trauma and postoperative pain while preserving lung function. This results in advantages such as a shorter hospital stay, fewer complications and a reduced need for intensive care. Thoracotomy involves making a larger incision in the chest to allow manual examination and exploration of the lung tissue.3
The surgical approach used depends on the patient’s specific disease, the size and location of the metastases, the patient’s overall health, and the experience of the surgical team. Each approach has its advantages and disadvantages and should be chosen based on the patient’s individual needs.4
Objectives
In the present study, the treatment outcomes and effects on survival of patients undergoing PM with VATS and thoracotomy were evaluated.
Materials and methods
A total of 136 patients who underwent PM between September 2012 and July 2020 were retrospectively evaluated at the Department of Thoracic Surgery, Istanbul Training and Research Hospital, University of Health Sciences (Istanbul, Turkey). A total of 84 patients underwent thoracotomy and 52 patients underwent VATS.
All patients were evaluated according to age, sex, histopathology of the primary tumor, location and number of radiologically and surgically detected metastatic nodules, type of surgery, type of resection, number of operations, disease-free survival, surgical morbidity and mortality, survival status, and time after the first metastasectomy. Patients for whom follow-up was no longer possible were contacted by telephone. The death data of patients who died were recorded.
In all patients, the primary tumor was surgically controlled, and it was confirmed that there were no metastases in organs other than the lungs. Preoperative investigations included a physical examination, a chest X-ray and an electrocardiogram (ECG). Respiratory function tests were performed to determine whether patients had sufficient lung capacity for possible anatomical resections. Most patients also underwent positron emission tomography (PET-CT) and magnetic resonance imaging (MRI) of the brain.
Video-assisted thoracoscopic surgery, or thoracotomy, was used as the surgical approach in the patients. Surgical reports were evaluated according to the resection procedure used, and pathology reports were assessed for tumor histology, resection status (R0, R1, R2, and RX) and distance between the staple line and the tumor in patients undergoing VATS.
This study was conducted in accordance with the Declaration of Helsinki. It was reviewed and approved by the Ethics Committee of the Istanbul Training and Research Hospital (approval No. 2020/1488).
Surgical approach
In the thoracotomy group, patients generally underwent a lateral thoracotomy through the 5th intercostal space. For pulmonary nodules <3 cm in diameter and in peripheral locations, wedge resection was preferred to preserve lung tissue, especially in those with respiratory distress or comorbidities, as well as in the elderly. Lobectomy was reserved for nodules >3 cm, for nodes in the hilar region that were unsuitable for wedge resection, or for multiple nodules >3 cm in 1 lobe. Additional nodules that were not detected using CT were palpated manually after resection. Lymph node dissection was performed for nodules with suspected lymph node metastases (cN1/2), especially those of gastrointestinal origin.
In VATS metastectomy, which was usually performed with 2–3 ports using a uniportal or biportal-triportal wedge resection, systematic manual palpation of the lung was not possible.
Statistical analyses
The collected data were summarized using descriptive statistics. To this end, continuous variables were tabulated using means ± standard deviations (±SD) or medians with minimum and maximum values depending on their normal distribution characteristics, whereas categorical variables were expressed as counts and percentages. The normality of numerical variables was assessed using the Shapiro–Wilk, Kolmogorov–Smirnov and Anderson–Darling tests.
In comparing the differences in categorical variables between the groups, Pearson’s χ2 test was used for 2×2 tables with expected cells of 5 or more, Fisher’s exact test for 2×2 tables with expected cells of less than 5, and a Fisher–Freeman–Halton test for R×C tables. Additionally, in comparing the differences in numerical variables regarding surgical and survival outcomes between 2 independent groups, the independent sample t-tests were used for numerical variables determined to conform to normal distribution, and the Mann–Whitney U test was used for numerical variables determined not to conform to normal distribution. After the assumption of homogeneity of variances was confirmed in all t-test analyses, the results were reported.
Given the number of comparisons across various subgroups, we used the Benjamini–Hochberg correction procedure to control the false discovery rate. We analyzed the survival times and influencing factors with Cox proportional hazards regression models using the R-project 4.3.3 (R Foundation for Statistical Computing, Vienna, Austria; https://www.R-project.org) software package and its associated packages, namely “survival,” “survminer,” “haven,” “gridExtra,” and “readxl.” The assumptions underlying the Cox regression models were evaluated using Schoenfeld’s global test featuring a variable-based and global examination of whether the proportionality of hazards assumption was violated, with all p-values found to exceed 0.05.
Based on these results, we proceeded to the model selection phase. To this end, we developed 5 distinct Cox proportional hazard models, using different sets of independent variables or data subgroups in each model. Notably, the 4th and 5th models were specifically tailored based on gender to thoroughly examine the effects of various malignancies on mortality risks within the sample. Gender-specific differences were observed among the malignancies studied. Accordingly, while some cancers, i.e., breast and endometrial cancers, were exclusive to women, others, i.e., testicular cancer, were exclusive to men. To more accurately assess the impact of these gender-specific malignancies, we stratified the sample by gender and constructed separate Cox regression models for each group.
We assessed the appropriateness and complexity of the models using Akaike’s Information Criterion (AIC) and Bayesian Information Criterion (BIC). Additionally, we tested the validity of the proportionality of the hazards assumption for each model using the “cox.zph” function and Schoenfeld residuals, which allowed us to determine the most viable models. We evaluated the impact of surgical methods on survival by gender using the Breslow (generalized Wilcoxon) and Tarone–Ware tests. The log-rank test, which assumes that hazard ratios (HRs) between compared groups remained constant over time, was deemed inappropriate if survival curves crossed, indicating that HRs changed over time and were disproportionate.
In contrast, the Breslow test, which gives more weight to early events, provided more consistent results with crossing curves, better detecting the differences observed in the early period. On the other hand, the Tarone–Ware test, which provides a balanced weighting of early and late events, evaluates HRs over a broader time spectrum and thus offers a more flexible alternative for general use. Because of the foregoing, we used both tests.5
We conducted the statistical analyses using the Jamovi project 2.3.28 (Jamovi, v. 2.3.28.0, https://www.jamovi.org) and JASP 0.17.3 (Jeffreys’ Amazing Statistics Program, v. 0.17.3, https://jasp-stats.org) software packages, setting the significance level (p-value) at 0.05.
Results
Of the patients, 80 were male (58.8%) and 56 female (41.2%). The mean age was 57.42 ±12.93 years (min: 25, max: 76). The origin of the primary tumor was as follows: 36 patients (26.5%) had colorectal carcinoma, 17 (12.5%) had renal cell carcinoma, 26 (19.1%) had breast carcinoma, 21 (15.4%) had osteosarcoma, 10 (7.4%) had soft tissue sarcomas, 12 (8.8%) had endometrial carcinoma, and 14 (10.3%) had testicular carcinoma.
The thoracotomy group exhibited significantly higher values than the VATS group in age, length of hospitalization, number of metastatic nodules removed during surgery, and mortality rate (p < 0.05 for each case). Regarding the types of malignancy, colorectal carcinoma and endometrial carcinoma were found to be significantly more prevalent in the thoracotomy group (p = 0.035 and p = 0.029, respectively), whereas osteosarcoma and testicular carcinoma were found to be significantly more prevalent in the VATS group (p = 0.002 and p < 0.001, respectively). There was no significant difference between the thoracotomy and VATS groups in gender, follow-up duration, number of metastatic nodules detected with thoracic CT, recurrence of metastasis, and the presence of renal cell carcinoma, breast carcinoma and soft tissue sarcoma (p > 0.05 for each case, Table 1).
Survivors had significantly longer follow-up periods (p < 0.001), a significantly lower age (p = 0.034) and pulmonary nodule size (p = 0.019), and shorter hospitalization (p = 0.007) compared to the deceased. Survival rates of patients with breast carcinoma were significantly higher than those with other types of malignancies (p = 0.005). Conversely, mortality rates of patients with endometrial and soft tissue sarcomas were significantly higher than those with other malignancies (p = 0.004 and p = 0.001, respectively). There was no significant difference between the survivors and the deceased regarding gender, number of metastatic nodules removed during surgery, number of metastatic nodules detected with thoracic CT, recurrence of metastasis during postoperative follow-up, and the presence of colorectal carcinoma, renal cell carcinoma, osteosarcoma, and testicular carcinoma (p > 0.05 for each case, Table 2).
The univariate analysis, including only female patients, revealed that breast carcinoma was associated with a 93% decrease in the mortality risk (95% confidence interval (95% CI): 0.01–0.57, p = 0.012), whereas soft tissue sarcomas had a 68.28-fold increase (95% CI: 6.01–775.58, p = 0.001) and each 1-year increase in age had a 10% increase in mortality risks (95% CI: 1.02–1.19, p = 0.017). There was no significant correlation between mortality risk and the surgical method, presence of metastasis, renal cell carcinoma or osteosarcoma, pulmonary nodule size, and number of metastatic nodules removed during surgery (p > 0.05 for each case).
Multivariate analysis revealed that breast carcinoma was associated with a 91% decrease in the mortality risk (95% CI: 0.01–0.66, p = 0.018), whereas osteosarcoma with a 9.12-fold increase (95% CI: 1.31–63.50, p = 0.026), soft tissue sarcomas had a 520.48-fold increase (95% CI: 41.06–6597.76, p < 0.001), and each 1-year increase in age was related to a 17% increase in the mortality risk (95% CI: 1.09–1.26, p < 0.001). There was no significant correlation between the mortality risk and surgical method, presence of metastasis or renal cell carcinoma, pulmonary nodule size, or number of metastatic nodules removed (p > 0.05 for each case).
The univariate analysis, including only male patients, revealed that recurrent metastases during the post-surgical period were associated with a 2.95-fold increase in mortality risk (95% CI: 1.26–6.92, p = 0.013), osteosarcoma had a 3.11-fold increase (95% CI: 1.12–8.63, p = 0.029) and soft tissue sarcomas had a 8.04-fold increase (95% CI: 2.98–21.68, p < 0.001). Each additional metastatic nodule removed during surgery was associated with a 32% increase in the mortality risk (95% CI: 1.10–1.59, p = 0.003). There was no significant correlation between the mortality risk and surgical approach, presence of colorectal cancer or renal cell carcinoma, age, and number of pulmonary nodules (p > 0.05 for each case).
Multivariate analysis revealed that undergoing VATS was associated with a 7.49-fold increase in the mortality risk (95% CI: 1.08–51.98, p = 0.042), osteosarcoma had a 29.24-fold increase (95% CI: 2.38–358.96, p = 0.008) and soft tissue sarcomas had a 88.53-fold increase (95% CI: 4.88–1606.17, p = 0.002). Each unit increase in pulmonary nodule size was associated with a 2.47-fold increase in the mortality risk (95% CI: 1.24–4.89, p = 0.010) and each additional metastatic nodule removed with a 1.40-fold increase (95% CI: 1.08–1.82, p = 0.010). There was no significant correlation between mortality risk and colorectal cancer, renal cell carcinoma and age (p > 0.05 for each case, Table 3).
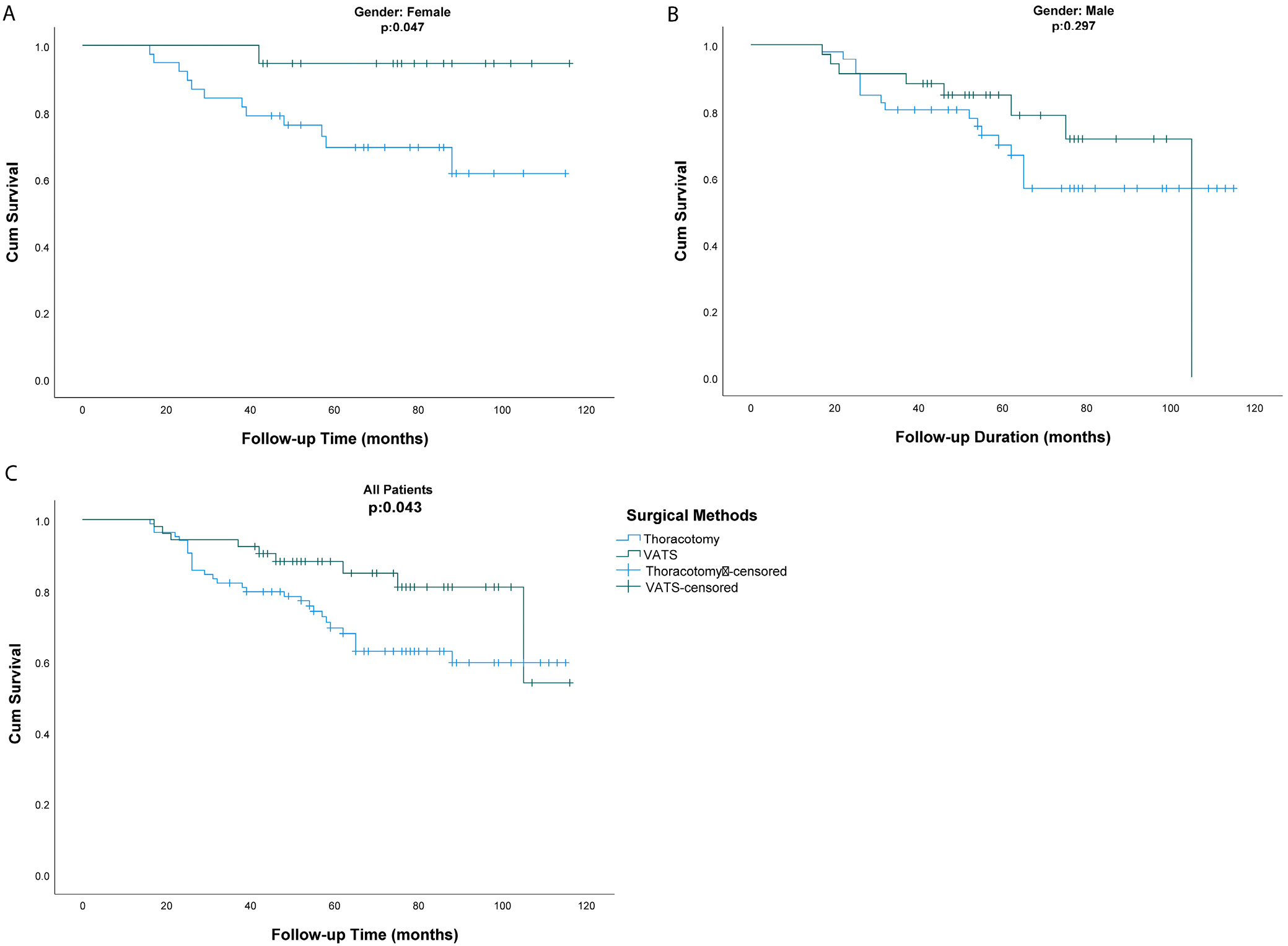
The mean survival time of female patients was 96.45 months, and the survival time of female patients who underwent VATS was significantly longer than those who underwent thoracotomy (p = 0.047). The mean survival time of male patients was 87.5 months. There was no significant difference between the male patients who underwent VATS and those who underwent thoracotomy in survival time (p = 0.297). The survival analysis of the overall sample revealed a mean survival time of 91.80 months, 86.60 months for those who underwent thoracotomy, and 99.57 months for those who underwent VATS. There was a significant difference between the survival time of patients who underwent VATS and those who underwent thoracotomy (p = 0.043, Table 4, Figure 1).
Discussion
Around 30% of cancer patients develop lung metastases. The primary tumors that are most likely to spread to the lungs include colorectal carcinomas, bone and soft tissue sarcomas, malignant melanomas, head and neck tumors, germ cell tumors, breast cancer, and renal cell carcinomas.4, 6
Patient selection, imaging, characteristics of the nodule, and surgeon experience are critical to PM. Thoracotomy offers a comprehensive view, while VATS is less invasive and better suited for remetastasectomy.7, 8, 9
With VATS, some lesions can only be detected to a limited extent, while CT can reveal small nodules and those that were not palpated during surgery. However, thoracotomy is more effective for certain procedures.10, 11, 12 In our study, some patients underwent multiportal (2 or 3) VATS surgery with incisions for manual palpation of nodules. However, more additional PMs were performed by thoracotomy than by VATS, suggesting that thoracotomy is still more effective for PMs.
Morbidity depends on the patient’s condition, the surgical method and the extent of the resection. Postoperative complications such as atelectasis, pneumonia, cardiac arrhythmias, and bronchopleural fistulas occur in 10–15% of patients after PM.13, 14 In our study, postoperative morbidity was observed in 11.7% of patients, with the most common complications being dyspnea, pneumonia, cardiac arrhythmias, and persistent air leaks.
In the literature, recurrence is reported in more than 50% of patients after PM. Recurrence after PM can occur particularly in primary sarcomas and melanomas.15 In our study, however, recurrence was observed in only 18.4% of patients (carcinomas: 15, sarcomas: 10). These results indicate that the recurrence rate in our study was lower compared to the overall recurrence rates reported in the literature.
Markowiak et al.16 recommend VATS for metastasectomy, especially in patients with a single metastasis. They achieved an R0 resection rate of 90.5 with VATS, similar to thoracotomy, and found no advantage in terms of survival or surgical outcomes over open surgery. In our study, similar results were obtained, with only 3 patients undergoing an R1 resection, while the remaining patients underwent an R0 resection. These results show that VATS is an effective option for metastasectomy and provides similar results compared to open surgical procedures.
Nakajima et al.17 and Nakas et al.18 reported no significant differences in survival between VATS and thoracotomy patients. However, Gossot et al.19 found that VATS resulted in better overall survival rates at 1, 3 and 5 years postoperatively compared with thoracotomy (VATS: 87.4%, 70.9%, and 52.5%, respectively; thoracotomy: 82.3%, 63.6% and 34%, respectively). Similarly, Nakajima et al.20 and Carballo et al.21 found that the 5-year survival was better in VATS patients compared with thoracotomy patients (Nakajima et al.: 49.3% vs 39.5%; Carballo et al.: 69.6% vs 58.8%). Chao et al.22 also reported that VATS had no additional negative impact on patient survival compared with thoracotomy (5-year survival of 51% vs 43%).
In our study, the median survival was 86.6 months for thoracotomy patients and 99.6 months for VATS patients. Female patients who underwent VATS had a significantly longer survival compared to those who underwent thoracotomy, suggesting that VATS provides a better survival benefit in PM, especially for women. Further studies are needed to confirm these results, particularly concerning gender differences.
Rusidanmu et al.23 found that VATS in PM has similar survival rates to thoracotomy but offers better perioperative outcomes, suggesting that VATS is a viable surgical alternative. Survival studies comparing thoracotomy and VATS provide conflicting results, with some favoring thoracotomy and others showing no significant difference. Overall survival and disease-free survival appear to be comparable for the different types of primary tumors.24
The complete removal of all lung metastases is associated with a longer survival. Over 62% of patients who undergo surgical resection survive for more than 5 years. The number of nodules removed during surgery influences the prognosis, with better results observed when only 1 nodule is removed.25, 26
In our study, we found that survival was longer in the VATS group than in the thoracotomy group. Video-assisted thoracoscopic surgery prolonged survival and reduced the risk of death, especially in female patients with breast cancer, but tumors such as soft tissue sarcomas and osteosarcomas were associated with a higher risk of death. This suggests that VATS has the potential to improve the efficacy of breast cancer treatment and the quality of life of patients.
We can say that more aggressive tumors, such as sarcomas, require more careful follow-up and treatment planning after surgical treatment. It is possible to increase the reliability and generalizability of the results by conducting such studies with larger sample groups and prospective designs.
In Figure 1B, the sharp decline in the Kaplan–Meier survival curve for the male VATS group does not mean that all patients died. This decline is largely due to censored cases and the end of the observation period. Censored cases include patients who withdrew from the study or were still alive at the end of the observation period and were not counted as events (such as death) in the survival calculations.27 In the group of male VATS patients, the rapid decline in the survival curve reflects the lack of follow-up data after the last recorded death, which marks the end of the observation period and the censoring of surviving patients. Our analysis shows that 8 male patients in the VATS group died, while 26 are still alive. Therefore, it would be wrong to conclude from the observed decline in the curve that all patients died; rather, it illustrates the impact of the censored cases and the end of the observation period.
The intersection of the survival curves observed in Figure 1C indicates that the risk of events fluctuates over time. Consequently, at different time points, 1 group may exhibit higher risk, while at another point, the same group may show lower risk. This phenomenon has been described as a “delayed treatment effect”.28
The early intersection of survival curves in thoracotomy and VATS patients indicates that the impact of these techniques on mortality in the initial postoperative period may be comparable. However, in the long term, VATS shows a reduction in the risk of mortality. Literature indicates that VATS is associated with a lower mortality risk compared to thoracotomy in non-small cell lung cancers (NSCLC). Video-assisted thoracoscopic surgery, a minimally invasive procedure, is linked to reduced complications, faster recovery and lower surgical trauma in the early postoperative period, which aids in quicker immune system recovery and lower infection risk. Studies report lower rates of postoperative pneumonia and arrhythmia, shorter hospital stays and higher 5-year overall survival rates for VATS (e.g., 75.5% vs 56.1%). Lymph nodule dissection performed employing VATS may positively impact cancer recurrence rates.29, 30, 31
However, the early intersection of survival curves may influence the power of statistical tests sensitive to early differences, such as the Gehan–Breslow–Wilcoxon method. This potential limitation in our analysis must be considered when interpreting our results.
Limitations
The limited sample size of our study, heterogeneous distribution of the patient’s primary tumor, the presence of other treatment factors such as re-metastasectomies, and biases in the choice of surgical method may influence the results.
Conclusions
It can be assumed that VATS is a more effective option for PM compared to thoracotomy and can prolong patient survival. Gender-specific analyses show that VATS significantly prolongs survival in women compared to thoracotomy. In contrast, these differences were not found to be significant in men. These results illustrate the influence of the surgical method and the type of disease on patient survival. Our results support the choice of VATS for PM and an improvement in patient survival.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.12548526. The package includes the following files:
Supplementary Table 1. Assessment of proportional hazards assumption using global Schoenfeld tests across different models.
Supplementary Table 2. Comparative analysis of surgical outcomes by type and associated variables in PM.
Supplementary Table 3. Comparative analysis of surgical outcomes by type and associated variables in PM.
Supplementary Table 4. Analysis of survival status relative to post-surgical recurrent metastasis and primary cancer focus.
Supplementary Table 5. Distribution of survival outcomes by cancer type and presence.
Supplementary Fig. 1. Graphical representation of Schoenfeld residuals for individual variables and global model assessment in Cox proportional hazards analysis – Model 1.
Supplementary Fig. 2. Graphical representation of Schoenfeld residuals for individual variables and global model assessment in Cox proportional hazards analysis – Model 2.
Supplementary Fig. 3. Graphical representation of Schoenfeld residuals for individual variables and global model assessment in Cox proportional hazards analysis – Model 3.
Supplementary Fig. 4. Graphical representation of Schoenfeld residuals for individual variables and global model assessment in Cox proportional hazards analysis – Model 4.
Supplementary Fig. 5. Graphical representation of Schoenfeld residuals for individual variables and global model assessment in Cox proportional hazards analysis – Model 5.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.