Abstract
Background. Vitamin D supplementation could offer irritable bowel syndrome (IBS) patients significant improvements in terms of symptom severity and overall quality of life (QoL). Yet, the potential benefits and risks associated with vitamin D supplementation still require additional investigation.
Objectives. We aimed to evaluate the impact of vitamin D supplementation on IBS using a systematic review and meta-analysis.
Materials and methods. A comprehensive search was carried out utilizing 4 electronic databases (PubMed, Embase, Scopus, and Cochrane Library) to identify articles published in English-language peer-reviewed journals. The odds ratios (ORs), risk ratios (RRs) and mean differences (MDs), along with their respective 95% confidence intervals (95% CIs), were computed. Heterogeneity was evaluated using the appropriate p-value and Cochrane Q and I2 statistics. For the analysis, RevMan 5.3 was utilized.
Results. Nine randomized controlled trials involving a total of 780 participants were included in this study. Vitamin D supplementation, in adolescents and young adults with IBS, improves the IBS symptoms severity score, QoL and serum 25(OH)D levels compared to controls. We obtained an OR of 2.34 (95% CI: 1.56–3.50) for change in the IBS severity scoring system (IBS-SSS), OR = 2.51 (95% CI: 1.71–3.70) for change in QoL, low risk of any adverse events (RR 0.49 (95% CI: 0.35–0.69)), and substantial changes in serum 25(OH)D level (MD = 11.29 (95% CI: 7.13–15.45)). Results were statistically significant (p < 0.05).
Conclusions. Vitamin D supplementation could lead to better IBS management with a low risk of adverse events.
Key words: irritable bowel syndrome, vitamin-D supplementation, IBS-specific QoL questionnaires, IBS symptom severity, IBS severity score system
Introduction
Irritable bowel syndrome (IBS) is a widespread and intricate gastrointestinal illness characterized by persistent abdominal pain and variations in bowel movements.1 It has a global prevalence of approx. 11%. Approximately 30% of those who exhibit symptoms of IBS suffer from increased levels of stress and a diminished quality of life (QoL).2 Individuals afflicted with IBS may have increased absenteeism, with the degree of work impairment directly correlated to the severity of symptoms.3 Multiple primary and secondary treatments for IBS have been established.4, 5
The majority of current medications target the most prevalent symptoms. Nevertheless, an individualized and meticulous approach to treating IBS should prioritize addressing the fundamental pathophysiological mechanisms rather than the prevailing symptoms. Hence, it is imperative to develop innovative approaches that specifically target the underlying mechanisms of IBS while mitigating any potential adverse effects. Multiple observational studies have indicated that individuals with IBS exhibit a greater occurrence of vitamin D deficiencies compared to control groups.6, 7, 8 Consequently, vitamin D deficiency is believed to have a significant impact on the development of IBS, as well as the severity of symptoms and overall QoL.9, 10 The involvement of vitamin D in the pathogenesis of IBS may be elucidated by its crucial role in the production of antimicrobial peptides, anti-inflammatory agents, immune cell trafficking and differentiation, gut barrier function, and the balance of calcium and phosphate levels.11, 12 Moreover, there is empirical evidence suggesting that vitamin D exerts a substantial beneficial influence on gastrointestinal wellbeing and augments the composition of the gut microbiota. The presence of vitamin D receptors (VDR) in the gut has been found to have an impact on gut function, motility and the symptoms associated with IBS.13
Numerous interventional studies have demonstrated the efficacy of vitamin D administration in mitigating the symptoms associated with depression and anxiety, which are frequently observed as psychological comorbidities in individuals suffering from IBS.14, 15 However, whether vitamin D supplementation helps with IBS is still debatable in randomized controlled trials (RCTs), and its significance in the management of IBS is still an open problem.
Objectives
Given the ongoing debate surrounding the efficacy of vitamin D supplementation in the treatment of IBS, this study performed a systematic review and meta-analysis to determine its impact on the severity of symptoms, change in the irritable bowel syndrome severity score (IBS-SSS) and QoL in individuals with IBS using 9 RCTs selected using pre-specified inclusion-exclusion criteria.16, 17, 18, 19, 20, 21, 22, 23, 24
Materials and methods
This research was carried out in accordance with the recommendations provided by Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA)25 and Assessing the Methodological Quality of Systematic Reviews (AMSTAR).26
Eligibility criteria
The current investigation encompassed articles showing data on the efficacy and safety of the impact of vitamin D supplementation on the severity of symptoms and QoL in individuals with IBS. Studies that satisfied the following inclusion criteria were incorporated: 1) patients with IBS; 2) adolescents and adult individuals ranging in age from 15 to 50 years; 3) evaluating the comparative efficacy and safety of either vitamin D supplementation on the severity of symptoms or QoL in individuals with IBS; 4) implementation of a RCT as the chosen study design. The study selection spanned from 2000 to 2023. We selected studies that contained full text and offered adequate data for the creation of 2×2 table. This meta-analysis included the following quantifiable outcomes as primary measures: improvement in IBS-SSSs, serum 25(OH)D levels and QoL in individuals of both the intervention and control groups. Excluded from consideration were studies that were obsolete, anecdotal or relied exclusively on expert assessments. Additionally, studies based on animal experiments or trials, as well as those where the original data and crucial information from the authors were unavailable, were also excluded. In addition, studies that encompassed patients with IBS alongside HIV, cancer and other systemic diseases, non-research publications, qualitative studies, and articles not published in English were also excluded.
Information sources
The current investigation was carried out after an exhaustive search was conducted in 4 databases: Embase, PubMed, Scopus, and the Cochrane Library. The search encompassed the years 2000–2023, and it made use of particular key words, such as “irritable bowel syndrome”, “IBS”, “vitamin D”, “vitamin D supplementation”, “IBS-specific QoL questionnaires”, “quality of life”, “ QoL,” “gastrointestinal tract disorder”, “symbiotic”, “vitamin D deficiency”, “bloating”, “diarrhea”, “IBS symptom severity”, “IBS severity score system”, “IBS-SSS”, “placebo”, “meta-analysis”, “RCT”, and “randomized controlled trial”. The key words were identified and verified for consistency in both Embase and MEDLINE databases, in accordance with the PICOS framework.27 (Table 116, 17, 18, 19, 20, 21, 22, 23, 24).
Search strategy
The aforementioned key words were entered into the Title (ti)-Abstract (abs)-Keyword (key) field in the Scopus search. Cochrane search terms included “irritable bowel syndrome” and “vitamin D supplementation.” The PICO structure was used for establishing the specific criteria for selection. “P” in this context represented patients diagnosed with irritable bowel syndrome; “I” denoted the supplementation of vitamin D; “C” represented a control; and “O” comprised the enhancement of clinical outcomes, specifically the change in IBS-SSSs, serum 25(OH)D levels, QoL, and risk of adverse events. The research design incorporated in this study was limited to the implementation of RCTs.
Selection process
Relevant studies were identified through an independent, comprehensive evaluation of the pertinent literature by 2 researchers who independently collected the demographic profiles of the patients and event data containing relevant components from the studies that were included.
Assessment of risk of bias
To assess the possibility of bias in the research examined, a well-established and standardized questionnaire was utilized. The evaluation of bias was performed utilizing a method from the Cochrane Collaboration as detailed in the Cochrane Handbook (version 5.3).28 Randomization-induced bias, bias arising from variations from intended interventions, bias due to missing outcome data, bias in outcome assessments, and bias in the selection of reported outcomes were the 5 components taken into consideration in this instrument.
Data collection process and data items
The evaluation of potential bias was carried out independently by 2 reviewers. An additional reviewer acted as an arbitrator to settle any outstanding disputes. In the end, the potential bias was evaluated and classified as either “high risk”, “low risk” or “unclear risk.” The existence of publication bias was evaluated by the utilization of a funnel plot,29 while the statistical significance of this bias was determined by conducting Begg’s test30 using MedCalc (MedCalc Software, Ostend, Belgium).31
Statistical analyses
The effect of multiple dichotomous and continuous outcomes was assessed and analyzed using the software application Review Manager (RevMan) 5.3 (The Nordic Cochrane Centre, The Cochrane Collaboration, Copenhagen, Denmark).32 The integration of RevMan software has facilitated the optimization of the procedure for arranging, retrieving and removing superfluous references. The researchers used forest plots33 to assess the impact of outcome factors across all the investigations. The DerSimonian and Laird approach was utilized to determine the odds ratio (OR), risk ratio (RR) and mean difference (MD). This was accomplished by adopting a 2×2 table34 that was constructed using event data. The binary outcomes were evaluated using the OR35 along with a 95% confidence interval (95% CI). Statistical approaches, including the γ2 test with a corresponding p-value and the I2 test,36 were employed to evaluate heterogeneity. Given the varying settings under which the investigations were performed, a random effect model37 was used.
Results
Literature search results
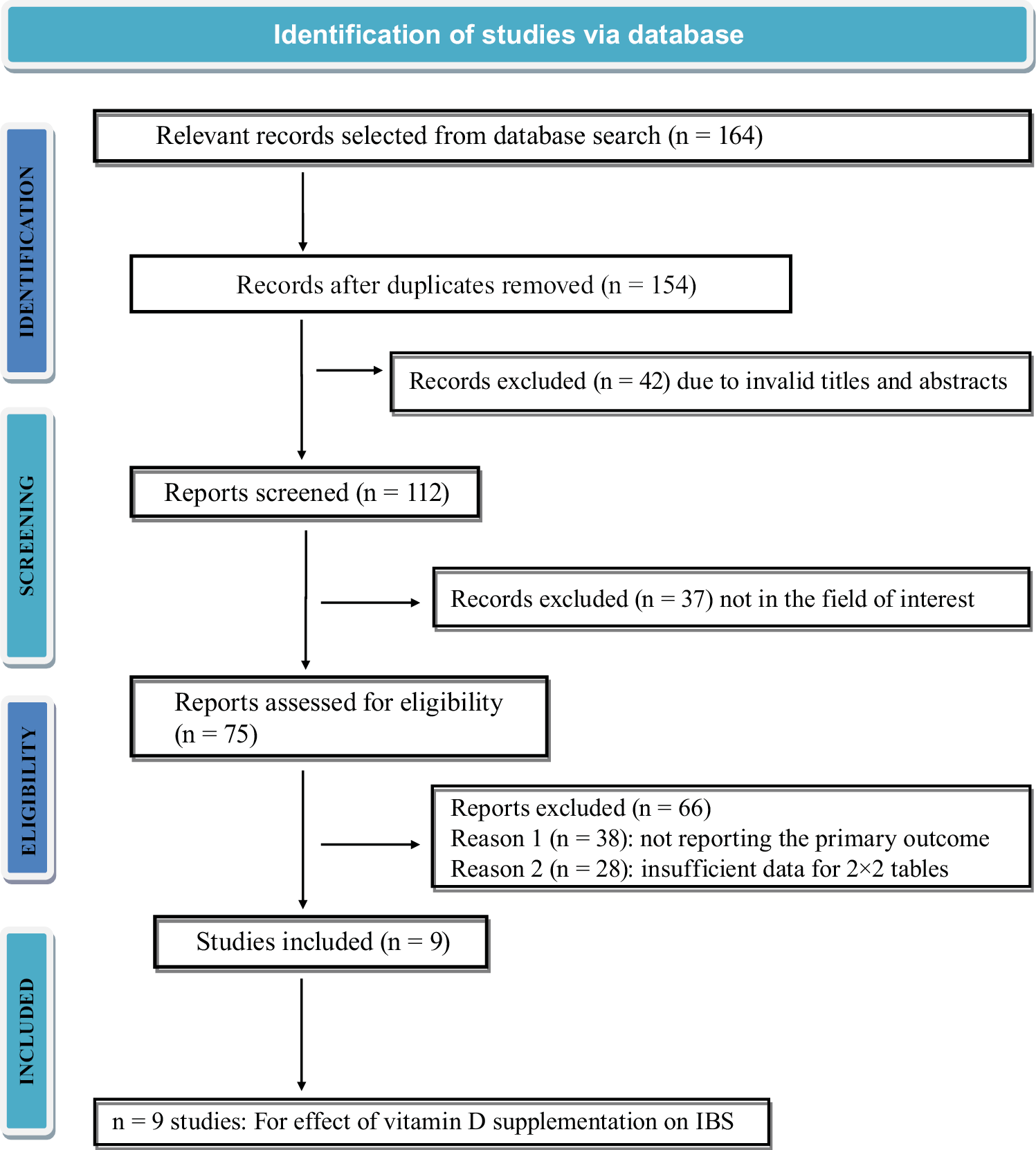
Electronic scanning techniques were employed to conduct an extensive search in Embase, PubMed, Scopus, and the Cochrane Library, yielding a total of 164 papers that satisfied the inclusion criteria specified by the PICOS framework. However, 154 papers were eliminated owing to being duplicates, and 42 papers were omitted because their titles and abstracts were erroneous. Subsequently, a total of 112 records were subjected to further screening, resulting in the exclusion of 37 records owing to unrelated fields. The remaining 75 records were then evaluated to determine their eligibility. Nevertheless, following the application of the inclusion-exclusion criteria, cumulative counts of 66 studies were determined to be ineligible and thus excluded. The main issues that led to the exclusion of studies were a lack of enough data for the construction of 2×2 tables and the absence of essential outcome measures. This meta-analysis employed a total of 9 RCTs that matched the planned inclusion criteria and covered the time period from 2000 to 2023, as seen in Figure 1.
The analysis includes a total of 780 adolescents and young adults from different age groups. The demographic features of the studies included in this meta-analysis are presented in Table 2. The text presents a comprehensive account of the authors’ identification, publication year, journal of publication, country of publication, intervention, study design, total participant count, participant age, number of individuals in the intervention and control groups, supplement dosages, study duration, and primary outcomes assessed. Afterwards, the previously indicated event data was used to carry out the meta-analysis.
Assessment of the risk of bias
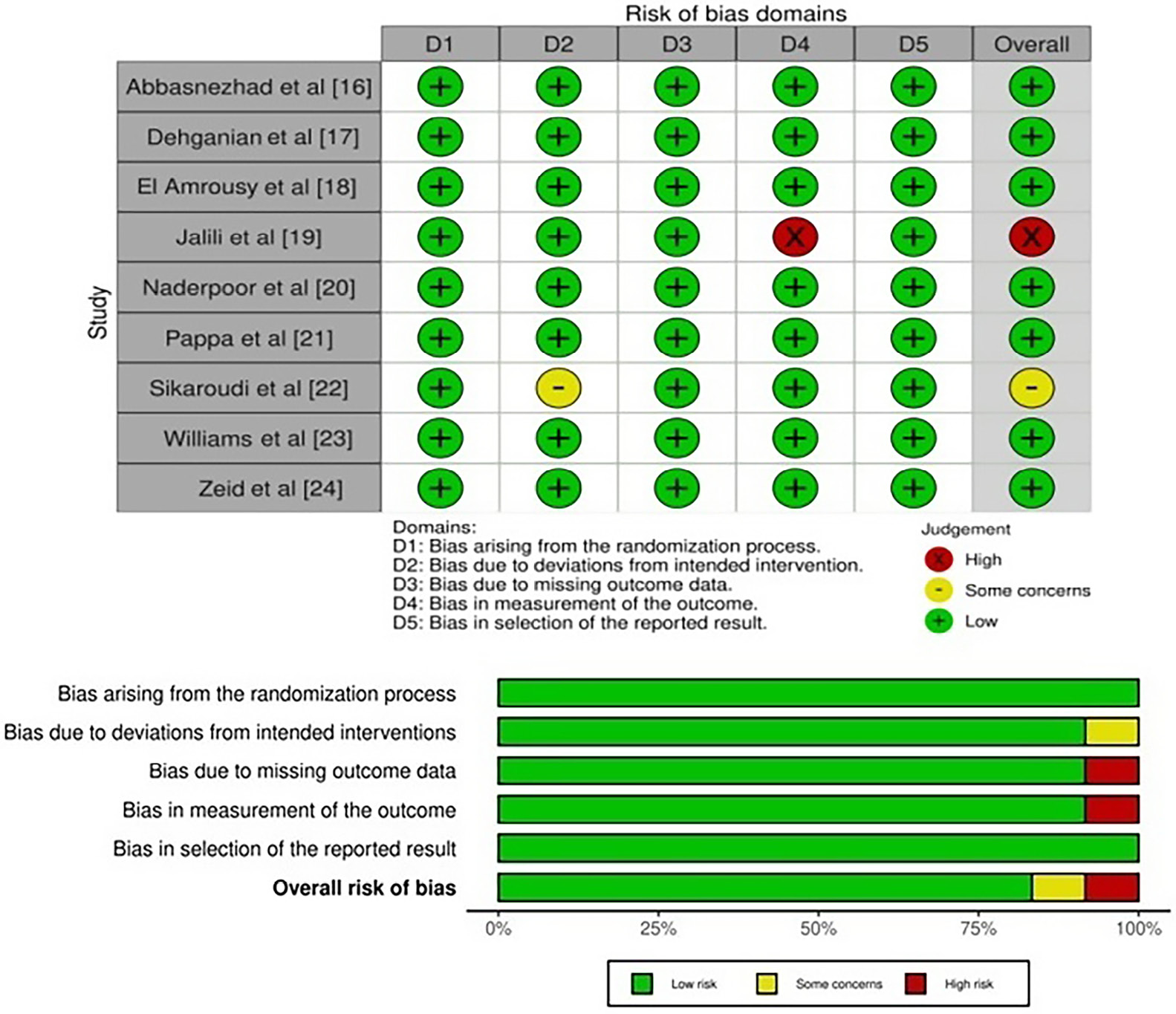
Risk of bias evaluation results for the included studies, as evaluated with the preset questionnaire, are presented in Table 3. The graph depicting the risk of bias and the description of the risk of bias in Figure 2 indicates that the present meta-analysis exhibits a minimum risk of bias. Among the 9 RCTs used in the analysis, it was observed that 7 studies had a low risk of bias, whilst 1 paper showed a moderate risk of bias. The bias resulting from the divergence from the targeted intervention was identified as the cause of the moderate risk. Nevertheless, our research demonstrated a notably elevated risk as a result of bias in the assessment of the outcome.
Statistical findings
Change in irritable bowel syndrome symptoms severity score
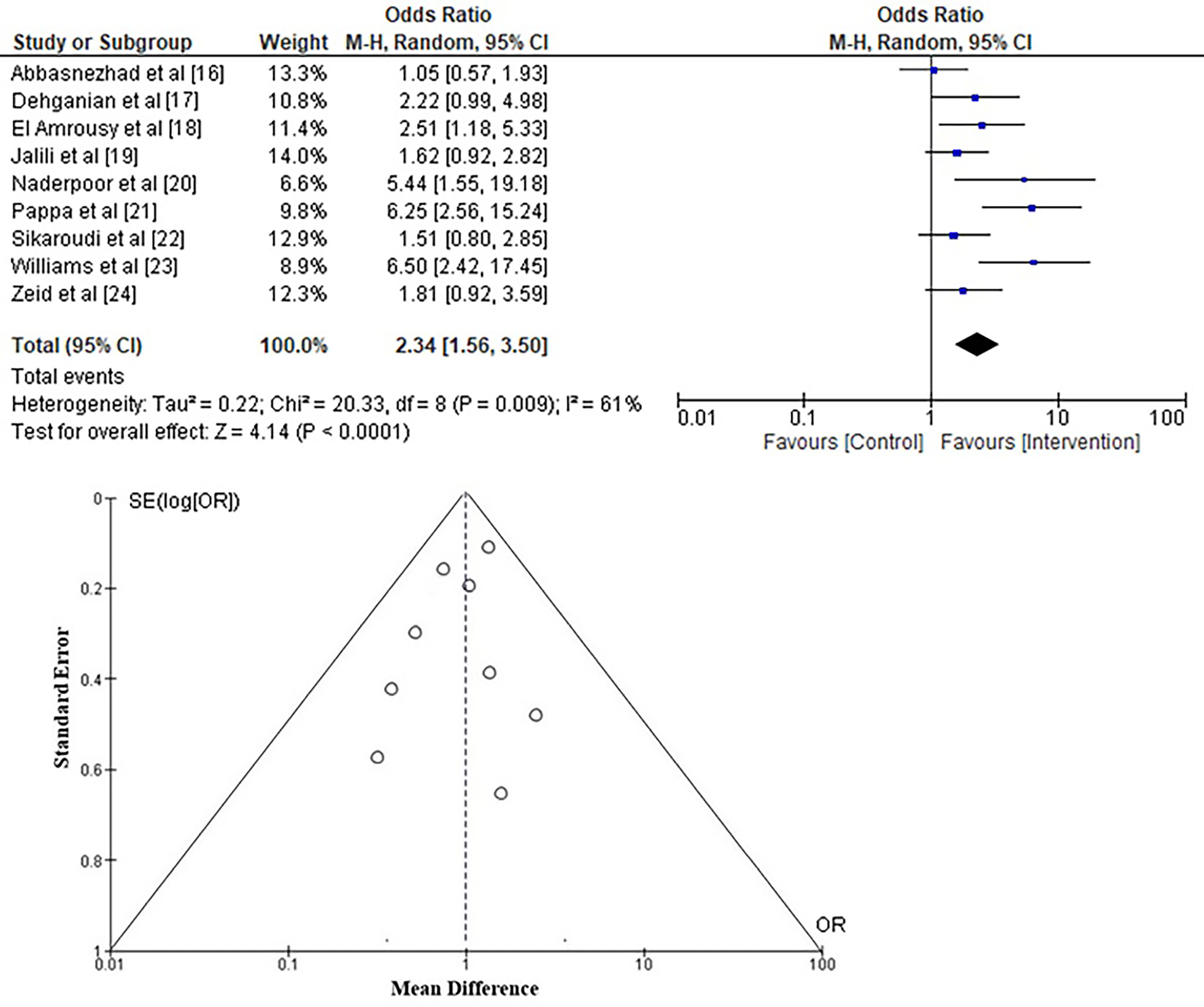
To investigate the likelihood of a change in IBS-SSSs in adolescents and young adults with IBS using vitamin D supplementation, the OR was calculated from the event data extracted from the included studies, as depicted in Figure 3. It was found that vitamin D supplementation has a higher likelihood of improving IBS-SSS scores than control, with an OR of 2.34 (95% CI: 1.56–3.50) and a Tau2 value of 0.220, χ2 = 20.331, degrees of freedom (df) = 8, Z = 4.14, I2 = 61%, and p < 0.001. Furthermore, the funnel plots shown in Figure 3 exhibit a symmetrical structure, suggesting a low likelihood of publication bias. Additionally, the p-value of Begg’s test (p = 0.182) was statistically insignificant, above the specified significance level of 0.05.
Change in quality-of-life in individuals with IBS
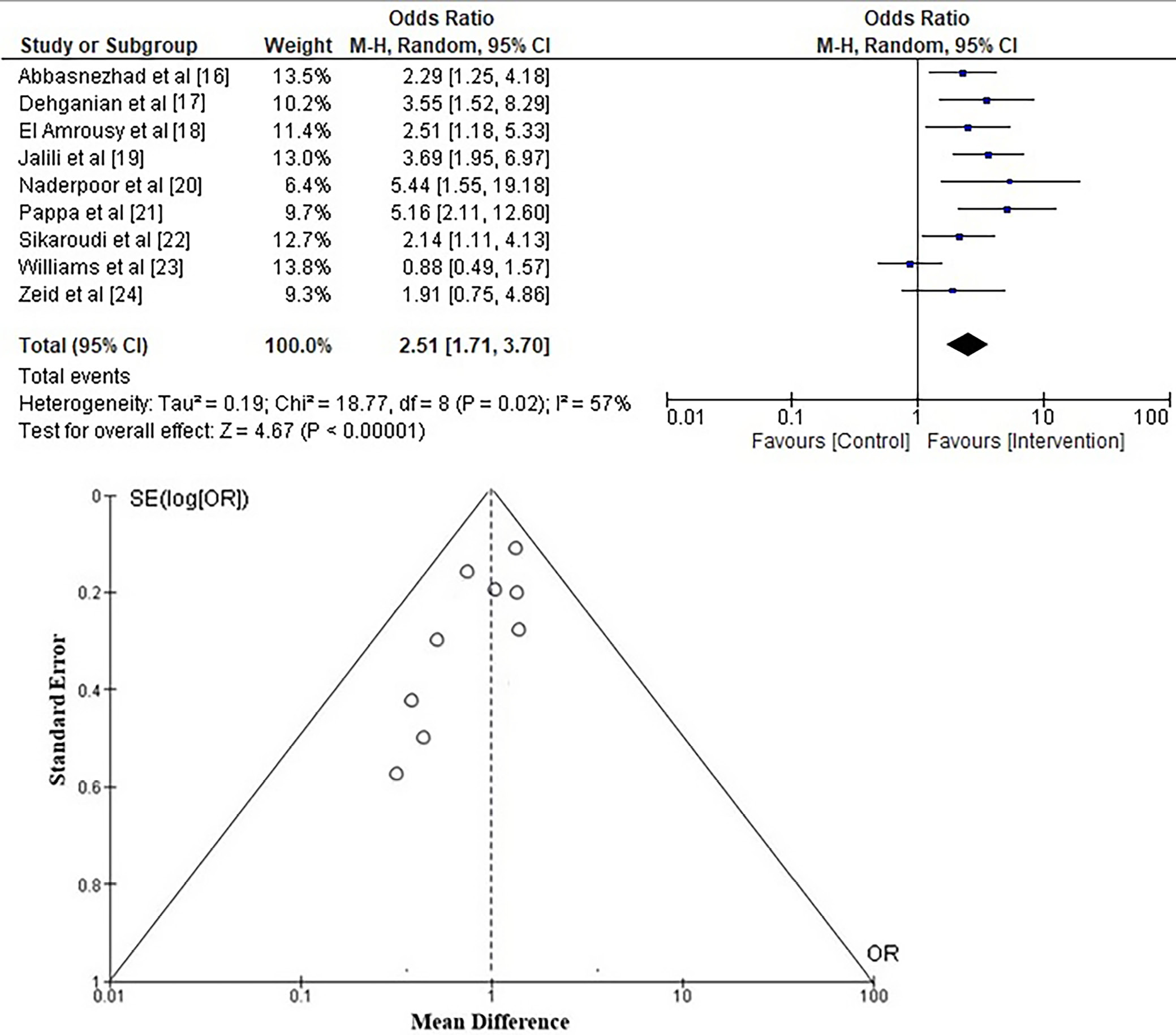
To determine the possible impact of vitamin D supplementation on the QoL of adolescents and young adults with IBS, we calculated the OR using event data from the included trials, as shown in Figure 4. The findings suggest that vitamin D supplementation is more likely to improve QoL compared to the control group. The OR was 2.51 (95% CI: 1.71–3.70; Tau2 = 0.191, χ2 = 18.77, df = 8, Z = 4.67, I2 = 57%, p < 0.001]. Furthermore, the symmetrical configuration of the funnel diagrams depicted in Figure 4, along with a statistically negligible p-value on Begg’s test (p = 0.314), above the specified significance level of 0.05, clearly suggest a minimal likelihood of publication bias.
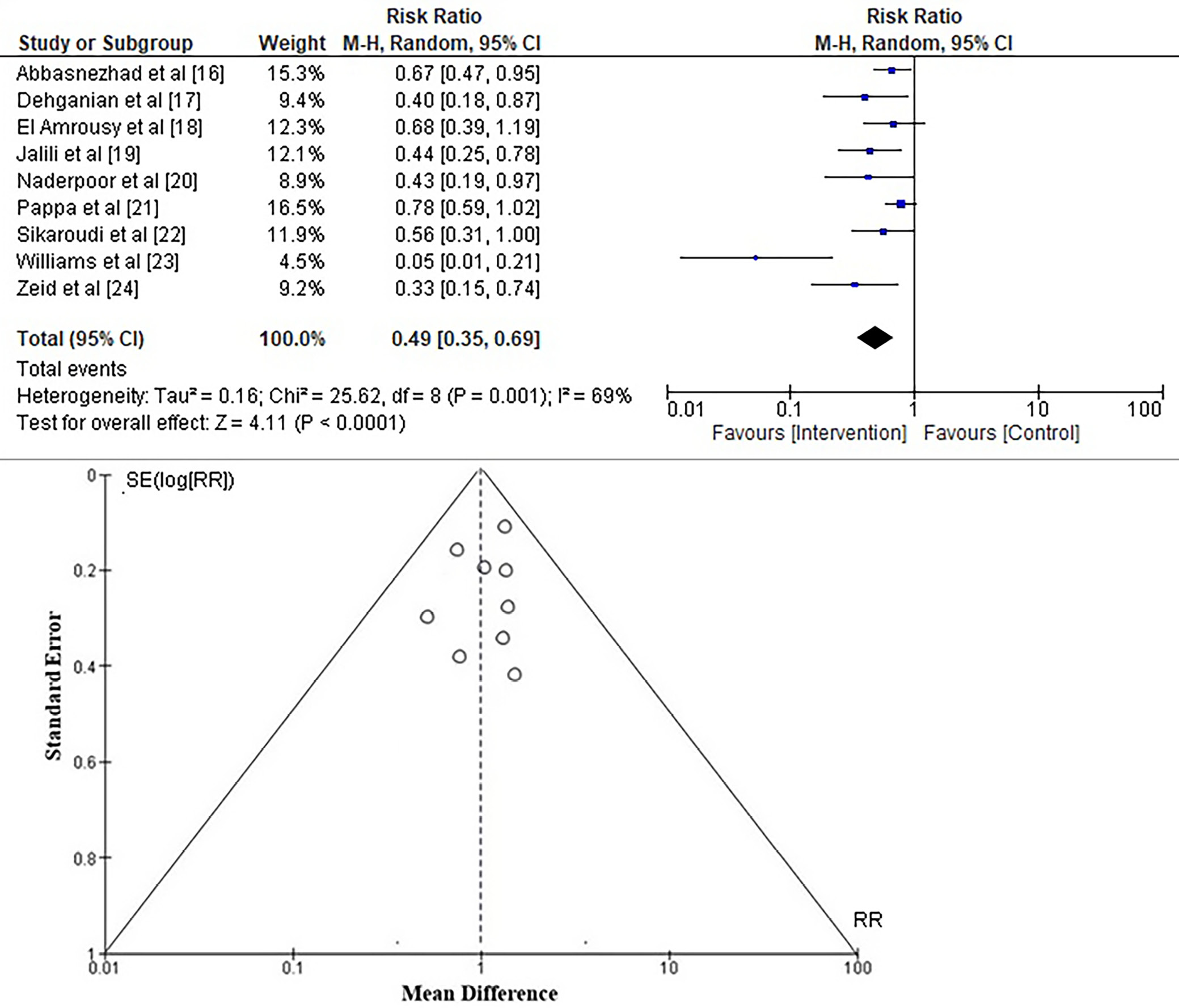
Risk of adverse events
To assess the risk of any adverse events in adolescents and young adults with IBS using a vitamin D supplement, the RR was calculated from the event data extracted from the included studies, as depicted in Figure 5. It was found that vitamin D supplementation has a low risk of adverse events, with an RR of 0.49 (95% CI: 0.35–0.69) and a Tau2 value of 0.16, χ2 = 25.62, df = 8, Z = 4.11, I2 = 69%, and p < 0.001. The symmetrical structure of the funnel plots in Figure 5 and the insignificant statistical p-value of 0.284 for the Begg’s test – which is higher than the preset significance level of 0.05 – also suggest that there is little chance of publication bias.
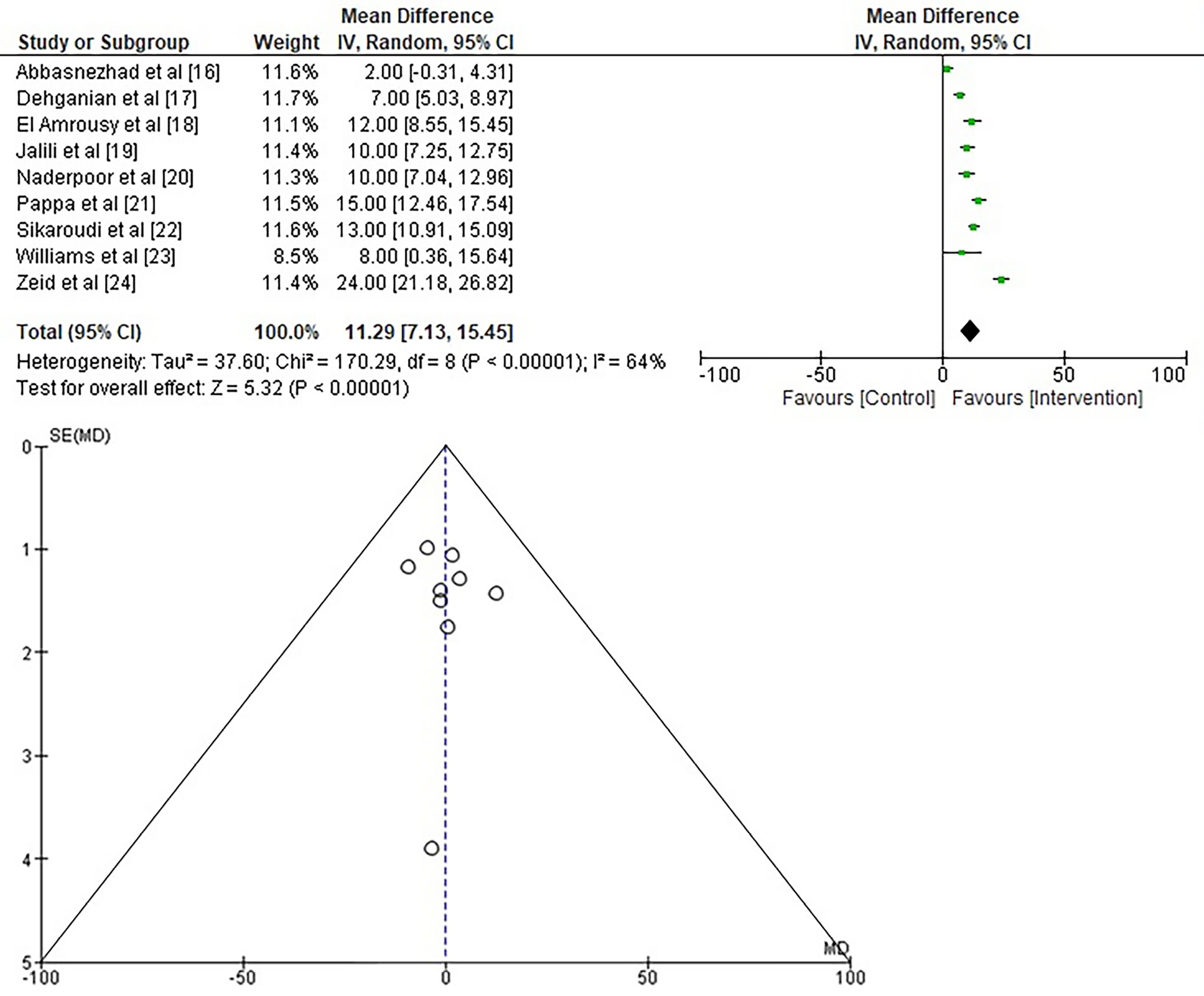
Change in serum 25-hydroxy vitamin D levels
To assess the effect of vitamin D supplementation on the fluctuation of serum 25(OH)D levels in adolescents and young adults with IBS, the MD was calculated using the data collected from the studies included, as shown in Figure 6. The study found a significant increase in serum 25(OH)D levels with vitamin D supplementation. The statistical analysis showed a MD = 11.29 (95% CI: 7.13–15.45; Tau2 = 37.60, χ2 = 170.29, df = 8, Z = 5.32, I2 = 64%, and p < 0.001). Similarly, the symmetrical configuration of the funnel plots depicted in Figure 6 and the statistically insignificant p-value of Begg’s test (p = 0.244), surpassing the predetermined significance level of 0.05, provide more evidence for the unlikelihood of publication bias.
Discussion
Irritable bowel syndrome is a prevalent and persistent illness that impacts the gastrointestinal tract. Common symptoms include cramping, stomach pain, bloating, excessive gas, and alternating episodes of diarrhea and constipation.38 Irritable bowel syndrome can manifest at any age and may arise from either a bacterial or parasitic infection of the intestines or as a result of stress.39, 40 Several effective treatments for IBS, targeting the most common symptoms, have been developed. They include dietary changes, medication supplementation and psychological therapies.41, 42 These emergent treatments have demonstrated favorable outcomes in many studies with regard to enhanced QoL and IBS-SSS scores. The IBS-SSS scores were determined using 5 visual analogue scales (VAS): pain severity, pain duration, stomach bloating, bowel satisfaction, and interruption with daily activities. The severity scores of IBS were measured on a scale ranging from 0 (indicating the least severe) to 500 (indicating the most severe).43, 44
Several studies found that people with IBS experienced less severe symptoms and an overall better QoL after supplementing with fiber, microbiota and vitamin D since deficiency of these factors is often linked to IBS.45, 46, 47 For example, Huang et al.48 reported in their study that vitamin D supplementation was superior to placebo for IBS treatment with greater improvement in IBS-SSS scores (weighted mean difference (WMD): –84.21, 95% CI: –111.38 to –57.05).
Similarly, in a study conducted by Chong et al.,49 the impact of the addition of vitamin D on IBS was examined. Their study included a total of 685 patients from 8 different studies. The findings of this analysis revealed a significant improvement in IBS-SSS scores as a result of vitamin D addition. They reported a significant effect size (standardized mean difference (SMD)) of –0.77 (95% CI: –1.47 to –0.07, p = 0.04, I2 = 91%). Nevertheless, it was noticed that there were improvements in IBS-QoL ratings, but these improvements were not statistically significant (SMD: 0.54; 95% CI: –0.34 to 1.41, p = 0.15, I2 = 87%). There are, however, a lot of studies on different IBS treatment plans. In the same vein, Abuelzam et al.50 concluded that vitamin D was ineffectual in alleviating gastrointestinal symptoms in IBS, whereas Yan et al.51 found that vitamin D supplementation may be linked to a decrease in IBS-SSS scores but not an increase in IBS-QoL scores. However, there is still debate about the number of patients who may benefit from vitamin D supplementation and whether or not there are any adverse effects.
Consequently, the purpose of this study was to assess the efficacy of vitamin D supplementation regimens for IBS in terms of their influence on QoL and the degree to which they improved the IBS-SSS score in patients who suffer from IBS. The study included 10 RCTs with a total of 831 participants. The primary findings of the included RCTs are as follows:
The study conducted by Abbasnezhad et al.16 employed a randomized, double-blind clinical design to investigate the effects of vitamin D on gastric and intestinal symptoms and health-related QoL in a sample of 90 persons diagnosed with IBS. The research entailed the random allocation of individuals who were designated to acquire either 50,000 IU of vitamin D3 or a placebo at biweekly intervals over 6 months. The IBS-SSS and IBS-specific QoL questionnaires were used for baseline and after-intervention assessments. During the 6-month intervention period, the patients who were administered vitamin D demonstrated a notable enhancement in symptoms associated with IBS, such as pain in the abdomen and distention, bloating, trembling, and overall symptoms of the gastrointestinal tract (excluding discontentment with bowel habits), in comparison to the placebo group. Following the intervention, the vitamin D group demonstrated notable enhancements in both the IBS-SSS and IBS-QoL scores in comparison to the placebo group. The average alterations in IBS-SSS scores were –53.82 ±23.3 and –16.85 ±25.01, respectively (p < 0.001). In a similar vein, the average change in IBS-QoL scores was 14.26 ±3 compared to 11 ±2.34, respectively (p < 0.001). The researchers have concluded that vitamin D is a feasible and reliable option for improving the QoL and mitigating symptoms associated with IBS.
El Amrousy et al.17 performed a RCT to examine the effectiveness of vitamin D supplementation in a group of 112 adolescents diagnosed with IBS and vitamin D deficiency. The individuals were allocated randomly to 2 groups that were properly matched based on age and gender. One cohort received oral vitamin D3 at a daily dosage of 2,000 IU for 6 months, whilst the 2nd cohort received a placebo for the same period. Prior to and 6 months after the intervention, the study evaluated the levels of vitamin D and results of assessment using different IBS scoring systems (IBS-SSS, IBS-QoL and total scores). The administration of vitamin D supplements to individuals diagnosed with IBS over 6 months resulted in significant improvements in IBS-SSS (p < 0.001), IBS-QoL (p < 0.001) and overall scores (p = 0.02) compared to the control group receiving a placebo for IBS. A notable elevation in serum vitamin D levels was observed in persons with IBS following the injection of vitamin D, with a twofold increase from 17.2 ±1.3 to 39 ±3.3 ng/mL (p < 0.001). No significant change in serum vitamin D levels was seen in the placebo group (p = 0.66). No adverse effects were observed over the trial period, suggesting that vitamin D was tolerated well. The efficacy of vitamin D supplementation in controlling IBS and vitamin D insufficiency in teenagers was determined by those researchers.17
Dehghanian et al.18 conducted a clinical experiment with 66 patients to investigate the effects of vitamin D3 supplementation on the clinical signs in individuals with IBS and lack of vitamin D. The study revealed that the administration of vitamin D supplements to patients with IBS led to a considerable elevation in serum 25(OH)D levels (with a MD of less than 14 at the conclusion of the study: –23/24 ±14). Additionally, there was a noteworthy reduction in the intensity of clinical manifestations (p < 0.05). The efficacy of the vitamin D dosage used in this trial (50,000 IU per week) in alleviating symptoms of IBS was determined by those researchers.18
Jalili et al.19 conducted a study to examine the impact of vitamin D supplementation on individuals diagnosed with IBS. The study employed a randomized, double-blind, placebo-controlled clinical trial design to examine the impact of weekly administration of 50,000 IU of vitamin D or a placebo comprising medium chain triglycerides on a cohort of 116 patients diagnosed with IBS for 6 weeks. The research findings indicated a significant increase in the serum levels of 25-hydroxy vitamin D, with values ranging from 21.10 ±5.23 to 36.43 ±12.34 in the group receiving vitamin D (p < 0.001). Nevertheless, the placebo group did not exhibit any notable disparity in serum levels prior to and following the experiment. At weeks 0 and 6, the severity scores of symptoms related to IBS, the disease-specific QoL and the total score were evaluated. The group administered vitamin D showed significantly larger improvements in IBS-SSS, IBS-QoL and overall ratings compared to the group receiving a placebo (p < 0.05). The results of this study indicate that the administration of vitamin D can potentially improve the severity of symptoms and overall QoL in patients diagnosed with IBS.
Naderpoor et al.20 conducted a randomized controlled study to investigate the effects of vitamin D supplementation on intestinal microbiota. This research was comprised of a sample of 26 individuals who exhibited either vitamin D deficiency (together with a 25-hydroxyvitamin D (25(OH)D) level below 50 nmol/L), overweight or obesity (with a body mass index (BMI) above 25 kg/m2), and were otherwise in a state of physical wellbeing. The participants provided fecal samples initially and after being administered a loading dosage of 100,000 IU of cholecalciferol, which was followed by a daily dose of 4,000 IU or a placebo for 16 weeks. The analysis of the fecal microbiota was conducted using 16S rRNA sequencing, with a specific focus on the V6–8 region. There were no significant differences observed in the microbiome diversity between the groups treated with vitamin D or placebo at both the initial and final stages of the research (all p > 0.05). Additionally, no clustering was seen concerning the administration of vitamin D throughout the time of follow-up (p = 0.3). However, a significant association was seen between the composition of the community and the genus-level with the supplementation of vitamin D (p = 0.04). According to a linear discriminatory analysis with a threshold of 3.0, the vitamin D group showed a higher occurrence of the genera Lachnospira and a lower occurrence of the genus Blautia. Additionally, it was noted that individuals with a 25(OH)D level exceeding 75 nmol/L exhibited a higher percentage of the Coprococcus genus and a lower percentage of the Ruminococcus species, as compared to those with a 25(OH)D level below 50 nmol/L. The findings of their research demonstrate that the intake of vitamin D supplements has significant effects on the type of fecal bacteria.
Pappa et al.21 undertook a randomized clinical trial to compare 2 regimens designed to maintain sufficient levels of vitamin D in children and adolescents with a diagnosis of IBS. The study comprised a cohort of 63 participants, ranging in age from 8 to 18 years, who were diagnosed with IBS and had a baseline 25(OH)D level over 20 ng/mL. Among the participants, 48 individuals successfully concluded the trial, but 1 participant withdrew from the study due to experiencing unfavorable effects. A daily dosage of 400 IU of oral vitamin D2 was supplied to the intervention Group A, consisting of 32 participants. A daily dosage of 1000 IU was delivered to Group B during the summer/fall period and 2,000 IU throughout the winter/spring season (n = 31). The key outcome was the probability of consistently achieving a 25(OH)D level of 32 ng/mL or higher during all regular checkups every 3 months over 12 months. Three participants in Group A (9.4%) and 3 individuals in Group B (9.7%) achieved the main outcome (p = 0.97). No significant disparities were seen in the incidence of adverse reactions, all of which were of a minor magnitude. Participants in Group A exhibited a greater percentage of individuals with a C-reactive protein (CRP)level of 1 mg/dL or above (31% vs 10%, p = 0.04) and interleukin 6 (IL-6) levels exceeding 3 pg/mL (54% vs 27%, p = 0.05). The researchers reached the determination that daily oral administrations of vitamin D2 up to 2,000 IU were inadequate for the maintenance of normal 25 (OH) D levels, notwithstanding their demonstrated tolerability.
The efficacy of vitamin D3 supplementation on symptom improvement, corticotrophin-releasing hormone levels in serum and IL-6 levels in a cohort of 88 patients with diarrhea-predominant IBS (IBS-D) who exhibited vitamin D deficiencies was investigated in a RCT conducted by Khalighi Sikaroudi et al.22 Participants were randomly allocated to 2 groups. In the experimental group, a weekly dosage of 50,000 IU of vitamin D3 was administered, whereas the control group received a placebo for 9 weeks. Furthermore, all patients received mebeverine at a dosage of 135 mg twice a day, in addition to receiving supplementation. The IBS-SSS, blood 25(OH)D3, serum corticotrophin-releasing hormone (CRH) levels and IL-6 levels were assessed before and during treatments. A total of 74 patients participated in the completion of this study. The experimental group demonstrated a statistically significant reduction in the severity of symptoms associated with IBS (p < 0.01) and IL-6 levels (p = 0.02) compared to the control group. Nevertheless, no statistically significant disparities were detected in the serum concentration of CRH. In addition, it was observed that the therapy group demonstrated a significant reduction in IBS-SSS scores and IL-6 levels at the end of the study compared to initial measurements (p < 0.01 and p < 0.03, respectively). The findings of their research indicate that the ingestion of vitamin D3 has the ability to modulate the concentrations of CRH and IL-6 in the circulatory system, hence potentially ameliorating symptoms in individuals diagnosed with IBS-D. Patients with IBS-D who have deficiencies in vitamin D should consider taking vitamin D3 supplements.
The study by Tazzyman et al.28 employed a randomized, double-blind, three-arm parallel strategy to examine the effects of vitamin D on the QoL of persons diagnosed with IBS. The study conducted a comparative analysis of the impacts of vitamin D, placebo and a combination with probiotics. Furthermore, the subjects were classified according to their level of vitamin D, namely, whether levels were deficient or sufficient. The assessment of vitamin D levels was performed using blood tests administered at both the initial and final stages. The evaluation of IBS symptoms was conducted utilizing a validated questionnaire. Furthermore, the assessment of dietary intake was conducted by utilizing a food frequency questionnaire. Insufficient recruitment of individuals from the replete stratum was seen among a significant portion of the population affected by IBS due to a deficit in vitamin D. A significant association was identified in the initial dataset between the concentration of vitamin D in the circulatory system and general wellbeing. The prevalence of vitamin D deficiency among individuals diagnosed with IBS was significant, indicating the need for screening and possibly supplementation to address this issue.
In a study conducted by Williams et al.,23 a randomized clinical trial was carried out. The trial included 135 volunteers who were randomly assigned to receive either vitamin D (3,000 IU per day) or a placebo for 12 weeks. The study revealed that the administration of vitamin D did not exert any influence on the severity of symptoms and the overall wellbeing of persons suffering from IBS. The primary variable of interest was the change in the severity of symptoms related to IBS, whereas the secondary variables comprised alterations in QoL associated with IBS. An intent-to-treat strategy was employed to analyze individuals. In the initial stages of the investigation, it was observed that 60% of the participants exhibited a shortage or inadequacy of vitamin D. In comparison to the placebo group, the intervention group exhibited a statistically significant elevation in vitamin D levels (45.1 ± 32.88 nmol/L vs 3.1 ± 26.15 nmol/L; p < 0.001). There was no significant difference in IBS severity of symptoms over time between the active and placebo trial arms (–62.5 ± 91.57 vs –75.2 ± 84.35, p = 0.426). The trial arms did not exhibit a statistically significant distinction in QoL changes (–7.7 ±25.36 vs –11.31 ±25.02, p = 0.427). There was a lack of evidence to substantiate the efficacy of vitamin D in the management of symptoms associated with IBS. Nevertheless, the prevalence of vitamin D deficiency suggests that it is necessary to implement frequent screening and supplementation in this population to achieve comprehensive health advantages.
The efficacy of vitamin D3 (cholecalciferol) supplementation on gastrointestinal symptoms in adults with IBS was investigated by Zeid et al.24 through a randomized clinical trial including 80 patients at El-Mahsama Family Practice Center in Ismailia, Egypt. The study participants were randomly allocated to 2 separate groups: An intervention group and a control group. An oral dose of 4,000 IU of vitamin D3 was administered to the experimental group, whereas the control group received a placebo made up of edible paraffin. The approved treatment was delivered to both groups daily for 12 weeks. The IBS-SSSs were assessed at the start of the trial and again after 12 weeks. After 12 weeks, the intervention group showed a notable decrease in IBS-SSS scores (114.36 ±67.36) compared to the control group (292.13 ±74.77, p < 0.001). The study found that there was a significant difference in IBS-SSS ratings between persons receiving vitamin D (–164.72 ±67.77) and those receiving the placebo (–12.13 ±50.78, p < 0.001). The researchers concluded that individuals suffering from IBS reported improved overall health and relief of their gastrointestinal symptoms after receiving vitamin D3 therapy.
According to the findings of our meta-analysis, the administration of vitamin D supplements resulted in a significant improvement in QoL and IBS-SSS scores in comparison to the utilization of a placebo. The results were supported by ORs more than 1 (OR = 2.34; 95% CI: 1.56–3.50) for changes in IBS-SSS scores, (OR = 2.51, 95% CI: 1.71–3.70) for changes in QoL, and statistically significant p-values below 0.05. Furthermore, results have indicated that the administration of vitamin D supplements is linked to a decreased occurrence of negative outcomes (RR: 0.49; 95% CI: 0.35–0.69) and a notable alteration in serum 25(OH)D concentrations (MD = 11.29; 95% CI: 7.13–15.45).
Limitations
One notable component of this research is the utilization of extensive search phrases that encompass the investigation of “irritable bowel syndrome” and “vitamin D supplementation” across many databases. Nevertheless, it is important to elucidate specific constraints. This study excluded research in languages other than English. Second, it is essential to recognize the potential presence of selection bias in this research, as a significant number of the studies were purposefully omitted. Third, it was not possible to establish a correlation between the outcomes and factors such as age, ethnic background and gender. Fourth, the small sample size of participants in each subgroup and the limited number of studies resulted in substantial levels of heterogeneity and variation. Fifth, it is unfeasible to conduct a sensitivity analysis to identify the source of this variation. Lastly, since the follow-up period of the trials was short, the long-term effects of vitamin D on IBS remain unclear.
Conclusions
This is a comprehensive review of RCTs evaluating both the safety and efficacy of vitamin D supplementation within adolescents and young adults diagnosed with IBS. This study revealed that the administration of vitamin D supplements yields notable improvements in both IBS-SSS scores and QoL assessments among IBS patients, compared to the control group. Given their cost-effectiveness and safety, vitamin D supplementation is a favorable and practical approach to addressing IBS. However, because there is a limited number of investigations included, along with a significant variation and a wide range of ages, further studies are necessary to obtain a more thorough knowledge of the effects of supplementation with vitamin D.