Abstract
Background. The favorable regenerative effects of some mineral waters on wound healing have long been empirically demonstrated.
Objectives. The aim of this experimental study is to investigate the effects of an Italian salso-bromo-iodic mineral water (Rivanazzano, Italy) on an in vitro human native fibroblast culture model to identify any potential regenerative actions.
Materials and methods. Human native fibroblasts were cultured under different experimental conditions:
− Dulbecco’s modified Eagle’s medium (DMEM) reconstituted with distilled water (control);
− DMEM reconstituted with filtered mineral water collected from the spring;
− DMEM reconstituted with filtered mineral water collected at the balneotherapy facility;
− DMEM reconstituted with filtered, heated mineral water collected at the balneotherapy facility;
− DMEM partially replaced with filtered mineral water collected from the spring at different concentrations (10%, 20%, 30%, 40%, 50%);
− DMEM partially replaced with filtered, heated mineral water collected at the balneotherapy facility at different concentrations (10%, 20%, 30%, 40%, 50%);
− DMEM partially replaced with filtered mineral water collected at the balneotherapy facility at different concentrations (10%, 20%, 30%, 40%, 50%).
Cell proliferation and viability were evaluated using spectrophotometric analysis following staining with the XTT Microculture Tetrazolium Assay. Statistical analyses were performed for each experimental condition at 24, 48 and 72 h.
Results. The best outcomes were observed in fibroblasts cultured with DMEM partially replaced with filtered mineral water collected from the spring, within the range of 20–50%.
Conclusions. Our research results showed that Rivanazzano salso-bromo-iodic mineral water has a stimulating effect on in vitro human native fibroblast cultures. This activity was most pronounced with water collected from the spring, and it decreased with water collected at the balneotherapy facilities. These findings could form the basis for clinical applications in wound healing and balneotherapy.
Key words: human cell culture, fibroblast, microbiota, mineral water, wound healing
Background
Natural mineral water, according to EU Directive 2009/54/EC, is defined as “microbiologically wholesome water, originating in an underground water table or deposit and emerging from a spring at one or more natural or bore exits”.1, 2 The demonstrated effects of natural mineral waters on the skin include anti-inflammatory, antipruritic and anti-oxidative properties.3, 4, 5, 6 Furthermore, they improve skin hydration through the repair of the epidermal–dermal barrier.3, 4, 5, 6
The favorable regenerative effects of some mineral waters on wound healing have long been empirically demonstrated.7, 8, 9, 10, 11 Our research group previously investigated the biological properties of a calcium magnesium bicarbonate-based mineral water (Comano, Italy) and demonstrated the regenerative effect of its native mineral water non-pathogenic microflora on experimental in vitro cell and ex vivo tissue culture models.12, 13, 14, 15, 16, 17
Among the many natural mineral water springs in our region, the Rivanazzano (Italy) salso-bromo-iodic natural mineral water (Table 1) is noted for its anti-inflammatory properties, mucosal antiseptic effects and stimulation of tissue blood circulation, and is traditionally recommended for the treatment of skin and mucosal inflammatory diseases, capillary fragility, water tissue retention, and edematous fibrosclerotic panniculopathy (cellulite). This water is also used for the preparation and topical skin application of therapeutic anti-inflammatory thermal muds.18
We aimed to investigate its biological properties using the same experimental methods as in our previous research on Comano calcium magnesium bicarbonate-based natural mineral water.14
Rivanazzano mineral water is delivered from the natural spring source to 2 different destinations within the thermal baths via a dedicated pipeline. At the balneotherapy facilities, water is kept at the natural spring temperature, whereas at the 2nd location, it is heated to 70°C for the preparation of thermal muds.
Objectives
The general aim of this experimental study is to examine the effects of Rivanazzano salso-bromo-iodic mineral water, collected at different sites of the Rivanazzano spa resort, on an in vitro human native fibroblast culture model.14, 16
Two specific aims were investigated:
1) The effect on native human skin fibroblasts cultured in Dulbecco’s modified Eagle’s medium (DMEM) reconstituted with salso-bromo-iodic water collected at different sites and DMEM reconstituted with distilled water (control) after 24, 48 and 72 h was assessed (Experiment 1).
2) The effect on native human skin fibroblasts cultured in DMEM reconstituted with distilled water and partially replaced with different percentages of filtered salso-bromo-iodic water collected at different sites compared to DMEM reconstituted with distilled water (control) after 24, 48 and 72 h was described (Experiment 2).
Materials and methods
The experimental in vitro study was conducted in cooperation with the Plastic and Reconstructive Surgery Unit of the Department of Clinical-Surgical, Diagnostic, and Pediatric Sciences; the Advanced Technologies for Regenerative Medicine and Inductive Surgery Research Center; the Immunology and General Pathology Laboratory of the Department of Molecular Medicine of the University of Pavia (Italy); the Unit of Biostatistics and Clinical Epidemiology of the Department of Public Health, Experimental, and Forensic Medicine of the University of Pavia; and the Plastic and Reconstructive Surgery Unit, Department of Surgery of the Istituti Clinici Scientifici (ICS) Maugeri SB SpA IRCCS in Pavia. The experiments were carried out from September 2018 to February 2020. The study was approved by the Ethics Committee of the ICS Maugeri SB SpA IRCCS in Pavia (project identification code 2064) on September 26, 2016. The study conformed to the 1975 Declaration of Helsinki, and informed written and signed consent was obtained from all the patients.
Human skin specimen collection and processing
Human skin samples were collected in the operating rooms of the ICS Maugeri. The samples were obtained from anatomical specimens harvested during reduction mammoplasty or abdominoplasty sessions performed on 4 healthy female patients aged 43–60 years. The specimens were processed in the Immunology and General Pathology Laboratory of the Department of Molecular Medicine of the University of Pavia using the same methods as in previous studies conducted by our research group on other natural mineral waters.14, 15, 16, 17, 19, 20
Mineral water collection
The natural source of Rivanazzano salso-bromo-iodic water is connected to the thermal baths by a dedicated piping system. The mineral water samples were collected from December 2019 to February 2020 by a single individual using an aseptic procedure. The sample gatherer, wearing sterile surgical gloves, collected 250 mL of the water in sterile bottles from 3 sites: the natural spring source; the balneotherapy facility in the thermal baths at the end of the dedicated pipeline where the water had been heated to 70°C (HRWB); and the balneotherapy facility in the thermal baths at the end of the dedicated pipeline, which preserved the natural spring temperature (RWB). The samples were then transported under isothermal conditions (mean temperature 13.2°C) to the Immunology and General Pathology Laboratory of the Department of Molecular Medicine of the University of Pavia.
Fibroblast cell cultures
The human native skin fibroblasts were extracted and processed using the same methods as in previous studies.14, 16
To reduce potential sources of bias, a cell suspension of human native skin fibroblasts was seeded in triplicate using a multichannel pipette into 198 wells within a 96-multiwell plate at a concentration of 2 × 103 cells per well. The cells were maintained in DMEM with 4,500 mg/L glucose, 0.584 g/L L-glutamine and 0.11 g/L sodium pyruvate (Sigma-Aldrich, St. Louis, USA; Merck KGaA, Darmstadt, Germany), reconstituted with distilled water (Milli-Q, Merck-Millipore, Darmstadt, Germany) and enriched with 3.7 g/L sodium bicarbonate, 10% fetal bovine serum (FBS) and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA). The cultures were incubated in an atmosphere of 95% humidified air with 5% CO2 at 37°C for 24 h to allow cell adhesion within the wells. After this period, the culture media were removed from all wells, and a gentle rinse was performed with phosphate-buffered saline (PBS; Sigma-Aldrich; Merck KGaA). Each well was then refilled with 100 µL of different culture media according to the experimental conditions. The culture media were changed on the 2nd day. All samples were incubated in an atmosphere of 95% humidified air with 5% CO2 at 37°C for 24, 48 and 72 h.
The endpoint
Quantification of cell proliferation and viability (endpoint) was evaluated using spectrophotometric analysis to measure absorbance at 475 nm after cell staining with the XTT Microculture Tetrazolium Assay, using the SPECTROstar® Omega microplate reader (BMG Labtech-Euroclone; Pero, Milano, Italy).16, 21
Experimental design
The experimental design was based on a previous study on human native skin fibroblast in vitro cultures.14 Two different experimental plans were implemented to achieve the specific aims.
In both experimental plans, 3 wells within a 96-multiwell plate without cells were filled with 100 µL of DMEM to provide a baseline “white” absorbance assessment. The XTT solution was prepared by mixing 5 mL of XTT labeling reagent with 0.1 mL of electron coupling reagent. In each well with cells, the medium was removed, and a gentle rinse was performed with PBS. After removing the PBS, each well was refilled with 100 µL of DMEM. A volume of 50 µL of XTT solution was then added to each well and incubated in a humidified atmosphere of 95% air with 5% CO2 at 37°C for 4 h.
Spectrophotometric analyses were conducted at 24 and 48 h after the addition of different culture media, along with the white absorbance assessment. Subsequently, at 48 h, the culture media were removed from the 3rd multiwell. A gentle rinse was performed with PBS, and each well was refilled with 100 µL of the specific culture medium according to the experimental conditions. The XTT staining procedure was then repeated at 72 h.
Experiment 1: Human native skin fibroblasts cultured with DMEM reconstituted with distilled water (control) vs DMEM reconstituted with filtered salso-bromo-iodic water collected at different sites at 24, 48 and 72 h
Four different experimental conditions were compared:
A. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with distilled water and enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA) (control).
B. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with filtered RWS enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA).
C. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with filtered HRWB enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA).
D. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with filtered RWB enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA).
Experiment 2: Human native skin fibroblasts cultured with DMEM reconstituted with distilled water (control) vs DMEM reconstituted with distilled water and partially replaced with different amounts of filtered salso-bromo-iodic water collected at different sites at 24, 48 and 72 h
A total of 16 different experimental conditions were compared:
A. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with distilled water and enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA) (control).
B. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with distilled water and enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA), partially replaced with filtered RWS at 5 different percentages (10%, 20%, 30%, 40%, and 50%).
C. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with distilled water and enriched with 3.7 g/L sodium bicarbonate, 10% FBS and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA), partially replaced with filtered HRWB at 5 different percentages (10%, 20%, 30%, 40%, and 50%).
D. DMEM without NaHCO3 with 1.0 g/L D-glucose reconstituted with distilled water and enriched with 3.7 g/L sodium bicarbonate, 10% FBS, and 1% (10,000 U/mL) penicillin and streptomycin (10 mg/mL) (all from Sigma-Aldrich; Merck KGaA), partially replaced with filtered RWB at 5 different percentages (10%, 20%, 30%, 40%, and 50%).
Statistical analyses
The quantitative variables were expressed as the mean with standard deviation (±SD) in the different experimental conditions at different times. To examine the effect of time and sites of water collection, a 2-way analysis of variance (ANOVA) for repeated measures (RM-ANOVA) was used after checking the assumptions. According to Verma, if the epsilon of Greenhouse and Geisser (ε-GG) was more than 0.750, the p-value with the correction of Huynh–Feldt (ε-HF) was reported.22
Finally, if the ANOVA proved significant, all comparisons among the sites of water collection per time were tested using a post hoc test with the Bonferroni approach. To describe the effect of different water partial replacement of the cell culture media at different times per site of water collection, the average level of absorbance with SD was used. The level of statistical significance was set at 0.05. All the analyses were made with STATA® v14 (https://doi.org/10.5281/zenodo.12762630).
Results
Experiment 1: Human native skin fibroblast cultures with DMEM reconstituted with distilled water (control) vs DMEM reconstituted with filtered salso-bromo-iodic water collected at different sites
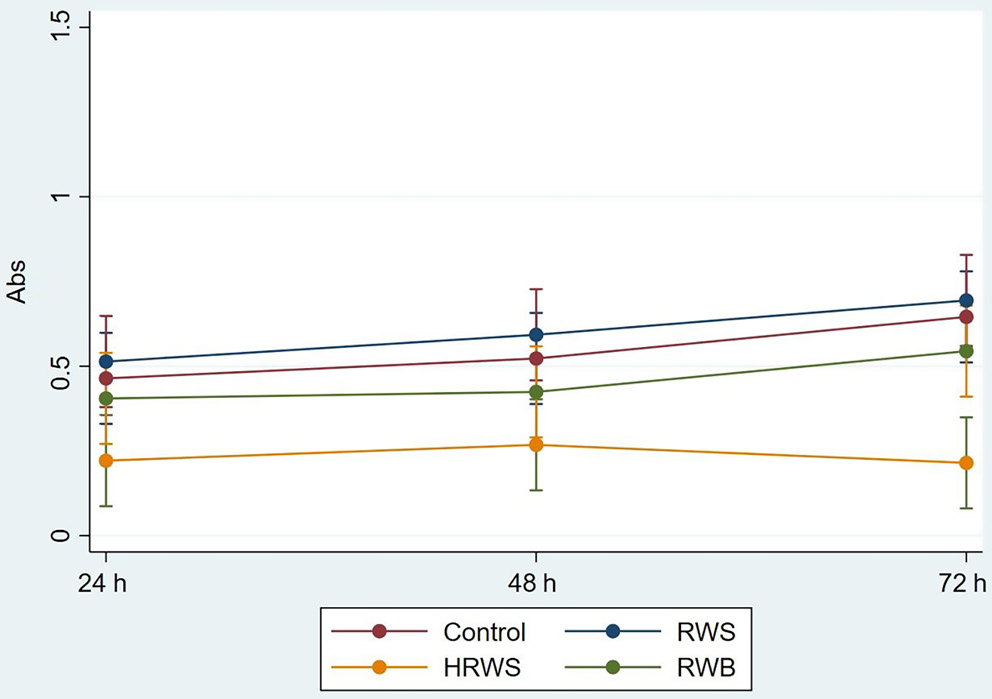
The evaluation indicated that the normality assumption was not met (p < 0.05); however, with ε-HF = 1 it was possible to consider RM-ANOVA robust to non-normality, as reported by Blanca et al.23 The cultures with DMEM reconstituted with filtered RWS displayed a slightly higher mean absorbance value compared to the controls at all observation times (Figure 1, Table 2). The cultures with DMEM reconstituted with filtered RWB, in contrast, displayed lower absorbance values than the controls at all times. The cultures with DMEM reconstituted with filtered HRWB displayed even lower values, with the lowest values observed at 24, 48 and 72 h.
The interaction between time and site of water collection was not statistically significant (F = 0.56, degrees of freedom (df) = 6, p = 0.758). However, both the main effects of time and site of water collection were statistically significant (F = 3.34, df = 2, p with Huynh–Feldt correction = 0.038 and F = 16.72, df = 3, p < 0.001, respectively).
More precisely, there was a growing trend in vitality over time for DMEM, RWS and RWB. The trend for HRWB reached a plateau at 48 h and then decreased (Figure 1). The multiple comparisons revealed that, for the main effect of time, there was a statistical difference only between 24 and 72 h (t = 2.57, df = 118, p = 0.033), but not for comparisons at 48 h (24 h vs 48 h t = 1.06, df = 118, p = 0.874; and 48 h vs 72 h t = 1.51, df = 118, p = 0.397) (Table 2). For the main effect of having 2 separate collection sites, there was a difference only with HRWB compared to the other sites of water collection (control vs HRWB t = –5.57, df = 88, p < 0.001; RWS vs HRWB t = –6.57, df = 88, p < 0.001; RWB vs HRWB t = 4.02, df = 88, p = 0.001) (Table 2).
Experiment 2: Human native skin fibroblast cultures with DMEM reconstituted with distilled water (control) compared to DMEM reconstituted with distilled water partially replaced with different amounts of filtered salso-bromo-iodic water collected at different sites
The results of the cultures with DMEM compared to DMEM partially replaced with filtered salso-bromo-iodic water collected at different sites are summarized in Table 3.
A. Human native skin fibroblast cultures with DMEM reconstituted with distilled water (control) compared to DMEM reconstituted with distilled water partially replaced with different amounts of filtered salso-bromo-iodic water collected at the spring
At 24 h, all experimental conditions with Rivanazzano mineral water showed a mean absorbance value higher than the controls. The absolute highest value was observed in the cultures with DMEM partially replaced with 20% filtered mineral water collected from the spring.
At 48 h, all experimental conditions with Rivanazzano mineral water showed a mean absorbance value higher than the controls. The cultures with DMEM partially replaced with 20%, 30% and 40% filtered mineral water collected from the spring showed the highest absorbance values. At 72 h, the cultures with DMEM partially replaced with 20%, 30%, 40%, and 50% filtered salso-bromo-iodic water collected from the spring showed higher values than the controls. However, the mean absorbance value in the cultures with DMEM partially replaced with 10% filtered salso-bromo-iodic water was lower than in the controls.
B. Human native skin fibroblast cultures with DMEM reconstituted with distilled water (control) vs DMEM reconstituted with distilled water partially replaced with different amounts of filtered HRWB
At 24 h, all experimental conditions with filtered HRWB showed a mean absorbance value higher than the controls. Specifically, the highest absorbance values were found in the cultures with DMEM partially replaced with 20% and 10% filtered HRWB.
At 48 h, only the cultures with DMEM partially replaced with 10% filtered HRWB showed a mean absorbance value higher than the controls. All other replacement percentages had absorbance values lower than in the controls.
At 72 h, all experimental conditions with filtered HRWB showed a mean absorbance value lower than the controls.
C. Human native skin fibroblast cultures with DMEM reconstituted with distilled water (control) vs DMEM reconstituted with distilled water partially replaced with different amounts of filtered RWB
At 24 h, all experimental conditions with filtered RWB showed a mean absorbance value higher than the controls. The highest absorbance values were observed in the cultures with DMEM partially replaced with 20% and 10% filtered RWB.
At 48 h, all experimental conditions with filtered RWB also showed a mean absorbance value higher than the controls. Within this group, the highest values were observed in the cultures with DMEM partially replaced with 10% filtered RWB.
At 72 h, the absorbance values in the cultures with DMEM partially replaced with 10%, 20%, 30%, and 40% filtered RWB were higher than in the controls. Under these experimental conditions, the highest values were observed in the cultures with DMEM partially replaced with 40% filtered RWB. In the cultures with DMEM partially replaced with 50% filtered RWB, the values at 72 h were almost equal to those in the controls.
Discussion
Spring water’s connection to life has been relevant in human history, health, work, culture, and religion since ancient times. In Roman culture, the use of mineral water was part of everyday life, both for hygienic and cosmetic purposes. The therapeutic properties of some mineral waters on wound healing have long been empirically demonstrated.7, 8, 9, 10, 11 These favorable effects are traditionally attributed to their chemical-physical composition, unique to every spring. Recently, increased evidence has suggested that the therapeutic properties of natural mineral waters might also be related to their biological components, complementing the chemical-physical ones.24, 25 The role of some natural mineral waters’ native non-pathogenic bacterial microflora and bacterial extracts in actively enhancing both cell function and a complex skin regeneration process has already been reported.16, 26, 27, 28, 29, 30, 31 Within this context, the mineral waters’ native microbial population and chemical-physical constitution are likely to mutually interact in promoting the water-specific therapeutic effects.32, 33, 34, 35
The medically relevant properties of natural mineral waters are anti-inflammatory, immune-modulating and regenerative. Within the wound healing process, an inversely proportional relationship between the magnitude of the inflammatory reaction and the effectiveness of the proliferative phase has been presented.36 Topical treatments with natural mineral waters have demonstrated favorable effects both on skin irritations37 and cutaneous re-structuring after aggressive cosmetic skin procedures.38, 39
The regenerative properties of calcium magnesium bicarbonate-based mineral water have been investigated and showed over the last decade by our research group using both animal in vivo experimental models and in vitro and ex vivo human models.12, 13, 14, 15, 16, 17
Our study focused on salso-bromo-iodic natural mineral water, whose therapeutic effects have long been empirically related to anti-inflammatory, antiseptic and angio-trophic properties.18
The mineral water replacement of the cell culture medium, at any percentage, with filtered mineral water allowed for a significant enhancement of fibroblast proliferation in the cultures with water collected from the spring. The strongest stimulation of human native skin fibroblast proliferation was found in the cultures with DMEM partially replaced with 40% Rivanazzano salso-bromo-iodic natural mineral water collected from the spring. The same result was observed with the unheated mineral water collected at the balneotherapy facility, although to a lesser degree. The heated mineral water, however, demonstrated poor results compared to both the replacements with the mineral water collected at other sites and the controls, except at 24 h with the lower replacement percentages. These outcomes might suggest that the favorable effects of Rivanazzano mineral water could be related to active substances influenced by environmental factors, such as the delivery in the pipeline system and thermal exposure.
The DMEM reconstitution with filtered mineral water collected from the spring did not demonstrate a substantial advantage in fibroblast proliferation compared to the controls, thus showing only a non-toxic effect.
All these data suggest that the partial replacement of the culture medium with filtered mineral water collected from the spring represents a potentially favorable combination for fibroblast in vitro proliferation, although the mechanism remains unexplained. Conversely, the reconstitution of DMEM with the same mineral water collected at the end of the dedicated pipeline in the balneotherapy facility showed substantial inhibition of in vitro cell proliferation. This effect was even worse with the DMEM reconstitution with heated mineral water. These effects might be related to cell-inhibiting substances produced during water delivery through the pipeline and, more significantly, after thermal conditioning at 70°C. It is unknown whether the fibroblast proliferation inhibiting effect was due to catabolites from the biological component of the mineral water or to irreversible damage within the water’s inorganic composition. However, the general picture from our study suggests that in the Rivanazzano mineral water collected from the spring, an active role in fibroblast proliferation and viability might be played by thermolabile, non-pathogenic microbial flora in harmonious combination with the water’s inorganic substrate.
Limitations
In this study, we deliberately used native skin-derived human fibroblasts. This option has both limitations and advantages. Only a limited number of replication cycles are allowed with these cells. They are far more delicate than immortalized cells because they have higher trophic requirements and display greater sensitivity to the contact inhibition process. Furthermore, they exhibit some inconsistency in their functional features according to different donors.40 However, native skin-derived human fibroblasts can be considered the closest approximation to in vivo ones because they share the same morphology, physiology and genetic features. Therefore, results from studies on native skin-derived human fibroblasts might best approximate the condition of fibroblasts in the in vivo context.
The results from this study should be considered just a preliminary step in investigating any stimulating effects of Rivanazzano salso-bromo-iodic water on human skin because fibroblasts represent only a single cell component within the complex integrated human skin environment. Therefore, we aim to further investigate the cell proliferative stimulating effect of Rivanazzano natural mineral water by repeating the same experimental sequence previously performed on Comano mineral water:
1) Identification of Rivanazzano mineral water’s native non-pathogenic bacterial microflora;
2) Assessment of the water’s effects on a human ex vivo skin experimental wound healing model.
Conclusions
The results from our research demonstrate some favorable effects of Rivanazzano salso-bromo-iodic mineral water on in vitro human native skin fibroblast cultures. This study could be the first step in exploring the potential regenerative properties of Rivanazzano salso-bromo-iodic natural mineral water on human skin, potentially expanding its therapeutic applications to the field of wound healing.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.12762630. The package includes the following file:
Supplementary File 1. STATA® v14 output.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.