Abstract
Background. Immunosuppressive therapy in organ transplant ensures proper graft function for many years, but it is burdened with a negative impact on the development of skin cancer in them.
Objectives. To characterize the impact of immunosuppressive therapy in transplant recipients on the development of non-melanoma skin cancers (NMSC).
Materials and methods. A total of 17,207 Polish patients who underwent liver, heart or kidney transplants between 2010 and 2022 and were on immunosuppression were included in the study. Immunosuppression was most commonly achieved using a regimen of tacrolimus (TAC) or cyclosporine A (CsA) combined with mycophenolic acid (MPA) and glucocorticosteroids (GS). Data on NMSC incidence from the National Health Fund in this population were analyzed and compared against incidence of NMSC in general Polish population in the same period.
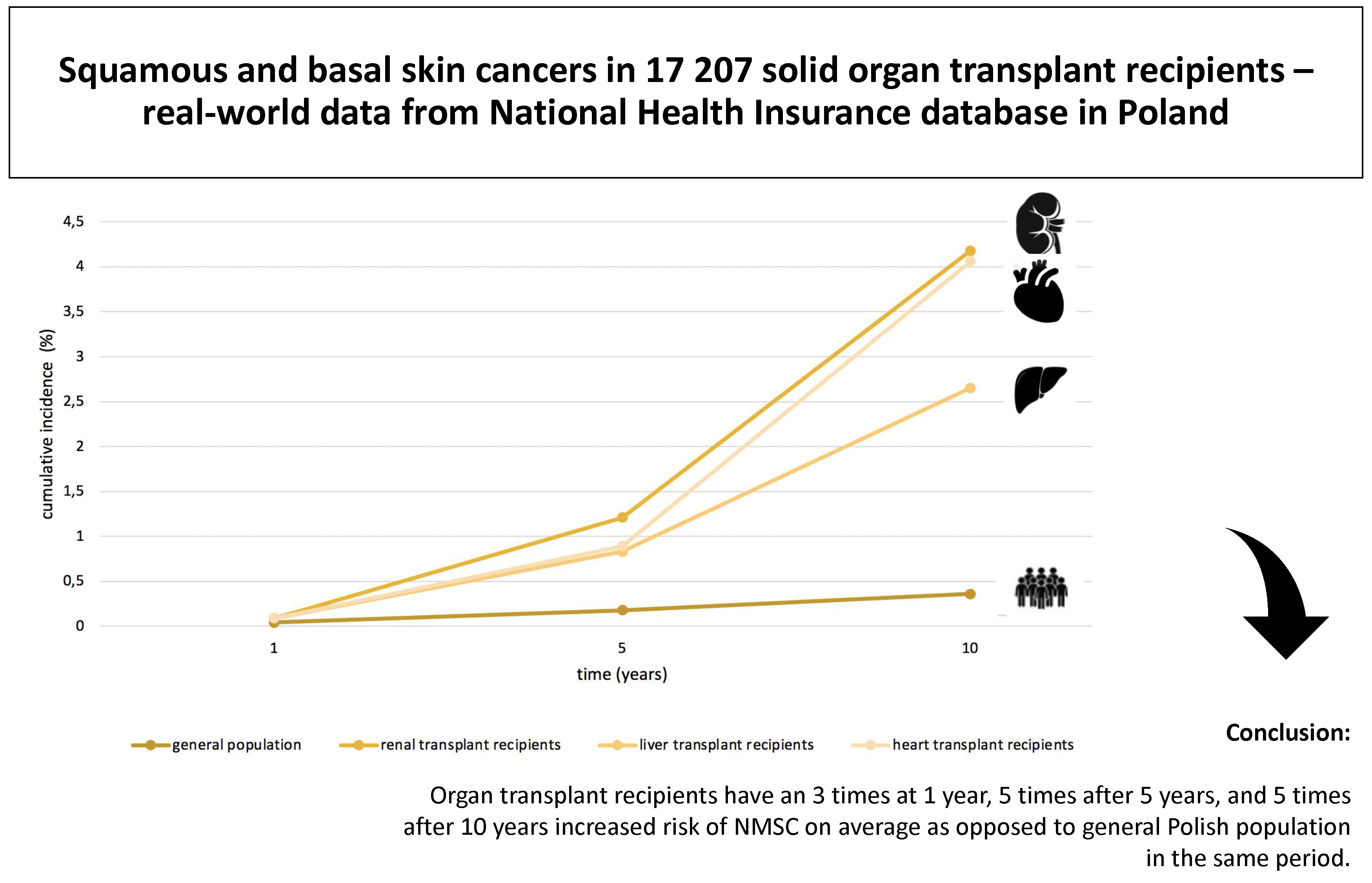
Results. Renal transplant recipients demonstrated a significantly elevated risk of NMSC compared to the general population, with a 1-year cumulative incidence of 0.09% vs 0.04% (p < 0.001), a 5-year incidence of 1.21% vs 0.18% (p < 0.001) and a 10-year incidence of 4.18% vs 0.36% (p < 0.001). Liver transplant recipients exhibited an elevated risk for the development of NMSC, which persisted and increased over time (incidence of 0.09% vs 0.04% at 1 year (p < 0.001), 0.83% vs 0.18% at 5 years (p < 0.001) and 2.65% vs 0.36% at 10 years (p < 0.001)). Heart transplant recipients also showed a significantly higher cumulative incidence of NMSC at 1 year (0.09% vs 0.04%, p < 0.001), 5 years (0.89% vs 0.18%, p < 0.001) and 10 years (4.06% vs. 0.36%, p < 0.001) post-transplantation.
Conclusions. Organ transplant recipients have an 2 times at 1 year, 4,5 times after 5 years and 9 times after 10 years increased risk of NMSC on average as opposed to general Polish population in the same period.
Key words: transplantation, skin cancer, transplant recipients, non-melanoma skin cancer
Background
A total of 1,910 organ transplants, including 1,055 kidney transplants, 550 liver transplants and 178 heart transplants, were carried out in Poland in 2023, according to the National Health Fund.1 This substantially outnumbers earlier figures of transplantation (e.g. 1,608 organ transplants in 2012).2 Currently, organ transplantations are common worldwide, with the numbers of these procedures performed each year globally reaching hundreds of thousands.
Hailed as the only long-term curative treatment for end-stage liver, heart or kidney disease, organ transplantation involves recipients in extensive, lifelong care. Recipients of the lifelong immunosuppressive therapy required for proper graft function are susceptible to numerous illnesses, and cancer is one of the most common in this group of patients.
Non-melanoma skin cancers (NMSCs), comprising mainly of squamous cell carcinoma (SCC) and basal cell carcinoma (BCC), are the most common tumors developing after organ transplantation and both constitute over 90% of all skin tumors that appear in individuals after organ transplantation.3
The outcomes of various studies have receive transplant recipients experience an increased risk of skin malignancy, ranging from 2 to 250 times higher compared to the general population, with the exact risk depending on specific population under investigation.4, 5, 6, 7, 8, 9 The highest increase in this particular risk was observed in Australia (a 250 times higher risk compared to the general population). In this country, the cumulative incidence of skin cancer progressively rises from 7% after 1 year of immunosuppression to 45% after 11 years, eventually reaching 70% after 20 years of immunosuppression.5 Authors from Scandinavian countries report a 20–100-fold increase in the risk of developing NMSCs.6, 7 In the Netherlands, the risk of skin cancer was observed to be 3% after 5 years, 24% after 15 years and even 40% after 20 years from organ transplantation.8 In one of the Italian studies, the cumulative risk after 5 years was 7.5%, and after 15 years, it was 28.8%.9 The risk of developing skin cancer steadily rises in all the presented data as more time passes since the transplantation. However, the numerical values significantly vary among them, even in the European countries sharing similar remaining NMSC risk factors, like long life expectancy, large percentage of elderly in the demographical structure, UV exposure, and cancer awareness within society.6, 7, 8, 9
Despite sharing a similar geographical latitude, findings from European studies also reveal considerable variation in the risk of neoplasia. This indicates that there may be other risk factors to cancer development, including the effects of administered treatments, genetic predispositions, or the prevalence and influence of HPV infection.10, 11, 12, 13 Regrettably, despite a lot of publications, epidemiological studies with a sizable patient population are lacking; such studies could facilitate an accurate evaluation of the actual risk of skin cancer among transplant recipients in Poland.
Objectives
In this study, we performed a large-scale investigation regarding the specific risk faced by transplant recipients by focusing on the real-world incidence of NMSC in Poland over the last 13 years. The data on skin cancer episodes involving all Polish residents were sourced from the National Health Fund dataset, a governmental public health institution. This is the largest analysis conducted on this topic in Poland to date, aiming to shed light on the frequency and risk factors associated with NMSC among organ transplant recipients.
Materials and methods
The National Health Fund provided information on skin cancer incidence in patients who had received a kidney, heart or liver transplant. The International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) code for skin cancer (C44.0–C44.9) was used to identify a cohort of patients with a diagnosis of skin cancer based on any inpatient or outpatient claim associated with the dataset. Code C44.0–C44.9 is used by hospitals for reporting to the National Health Fund the diagnosis of NMSC regardless of histopathology (SCC and BCC are reported using the same code). Patients who underwent renal, heart or liver transplantation between 2010 and 2022 are included in the dataset and were identified by specific codes used for transplantation reimbursements by the National Health Fund. A history of prior organ transplantation as well as the transplantation of more than 1 of the specified organs were considered exclusion criteria. Basic demographic characteristics of the patients included in the analysis is presented in the Table 1.
Statistical analyses
The primary objective of this analysis was to compare the cumulative incidence rates of skin malignancies, excluding melanoma, in patients who have undergone organ transplantation (kidney, heart or liver) with those in the general population. An additional aim was to determine whether organ transplantation is a significant risk factor for the development of skin cancers when compared to the general population. The studied hypotheses were formulated as follows:
H₀: The cumulative incidence rate of skin cancer is the same in both the specific post-transplant (Tx) cohort and the general population at each time point (1, 5 and 10 years).
H₁: The cumulative incidence rate of skin cancer differs between the specific Tx cohort and the general population at each time point.
The cumulative incidence rate at specific time point t is calculated as the sum of the incidence rates for each year up to that point using Equation 1:
(1)
We compared cumulative incidence rates of skin cancer between the Tx cohort and the general population at 1 year, 5 years and 10 years using a Z-test for 2 proportions.
The proportion of patients who developed skin cancer by the specified time point (1, 5 or 10 years) was calculated for both the Tx cohort and the general population.
The pooled incidence rate was calculated with Equation 2:
(2)
where nTxn and nPop represent the number of patients at risk in the Tx cohort and general population, respectively; P̂Tx– cumulative incidence rate in the Tx cohort; and P̂Pop– cumulative incidence rate in general population.
The Z-statistic for comparing the cumulative incidence rates between the cohort of patients after transplantation and overall population was calculated using Equation 3:
(3)
This Z-statistic was compared to the standard normal distribution to obtain the p-value.
When comparing the Tx cohort to the general population, the Tx cohort is not excluded from the population dataset. Due to the very small proportion of Tx patients, their inclusion does not impact the observed differences.
An alpha level α = 0.05 was set as the criterion for determining statistical significance. A p < 0.05 was considered statistically significant, indicating a difference in cumulative incidence rates between the 2 groups at the respective follow-up periods.
Moreover, to establish a transplantation as a risk factor, we estimated risk ratio (RR) comparing the risk of skin cancer in the transplantation cohort to the risk in the general population using Equation 4:
(4)
To compute the 95% confidence interval (95% CI) for RR, we applied an approximation for the standard error (SE) of the logarithm of the RR using Equation 5:
(5)
where eTx, ePo is the number of skin cancer events in the Tx cohort and overall population respectively.
The 95% CI for RR was calculated using Equations 6,7.
(6)
(7)
The RR is significantly greater than 1.0, which suggested that the transplantation is a risk factor for skin cancer.
Analyses were conducted using the R statistical language (v. 4.3.3; R Core Team, 2024; R Foundation for Statistical Computing, Vienna, Austria) on Windows 11 Pro 64 (build 22631).
We computed the cumulative incidence rate of NMSC, coded as C44, among recipients of organ transplants (Tx) in comparison to a control group over a 1-, 5- and 10-year follow-up period.
The Polish National Cancer Registry provided the information used to build the dataset for this time period (2010–2022).14
Results
Population of patients with non-melanoma skin cancer (C44)
The population of Poland during the period from 2013 to 2022, according to data from the World Bank,15 ranged from 38.04 million at the beginning of the analyzed timeframe to 36.1 million by the final year of assessment. Incidence data for the respective years within the study period were derived based on the findings of the reports from the Polish National Cancer Registry.16 The search criteria were focused on identifying newly reported cases of incidence associated with NMSC (C44) across the entirety of the Polish region, encompassing both genders and all age groups.
The cumulative incidence rates were calculated in accordance with established epidemiological methodologies, taking into account the annual variations in population size as well as the yearly incidence of new cancer cases reported by the Polish National Cancer Registry. These results, as detailed in Table 2, provide insight into the burden of skin cancer in the Polish population over a 10-year follow-up period. The results demonstrate a steady increase in the cumulative incidence rates for NMSC (C44) over the 10-year follow-up period in the Polish population.
The incidence of NMSC (C44) is significantly higher from the outset, with a cumulative incidence rate starting at 0.0355% in 2013 and increasing to 0.3597% by 2022. The consistently higher annual incidence, ranging from 10,810 to 15,716 cases, suggests that NMSC represent a more substantial and increasing risk to the population.
Cohort of patients with kidney transplantation
The number of patients at risk for developing skin malignancies following kidney transplantation, over the period from 2010 to 2022, was approx. 12,205, as detailed in Supplementary Table 1. This cohort represents individuals who underwent kidney transplantation and were subsequently exposed to long-term immunosuppressive therapy, which is well-documented to increase the risk of certain malignancies, particularly skin cancers.5, 6, 7, 8, 9
Table 3 provides a detailed summary of the cumulative incidence of non-melanoma skin cancer (NMCS) (C44) over a 10-year follow-up period post-transplantation.
Notably, the cumulative incidence rate for other skin malignancies is significantly higher, rising from 0.0857% at 1 year to 4.1835% after 10 years.
Results of comparing incidents rate between the cohort of patients after kidney transplantation and overall population are reported in Table 4.
The results in Table 4 indicate that kidney transplant recipients have a significantly higher risk of developing NMSC (C44) compared to the general population. The risk ratio for NMSC (C44) at 1 year, the cumulative incidence rate for transplant recipients is 0.0857%, with an RR = 2.41, indicating more than double the risk compared to the general population. By 5 years, the incidence rate rises dramatically to 1.2145%, with an RR = 6.84, and by 10 years it reaches 4.1835%, with an RR = 11.63. These findings demonstrate that kidney transplant recipients are at a significantly elevated risk of developing NMSC over time, with the risk increasing substantially as the follow-up period lengthens.
The Z-test results for NMSC is highly significant (p < 0.001) at all time points, showing that the observed differences between the transplant cohort and the general population are not due to random chance. This strongly supports the conclusion that organ transplantation, specifically kidney transplantation, is a major risk factor for the development of NMSC.
Liver transplant recipients vs general population
The data from Table 5 indicate that patients who undergo liver transplantation are at an elevated risk for NMSC (C44) over a 10-year period. By the end of the first year, 3 new cases of NMSC are observed, resulting in a cumulative incidence rate of 0.0931%. This rate continues to rise. By year 5, the cumulative incidence of NMSC reaches 0.8311%, and by year 10, it escalates to 2.6462%. The consistent appearance of new cases throughout the follow-up period, especially between years 5 and 10, highlights the prolonged and progressive risk of NMSC in liver transplant recipients.
The results from Table 6 show that liver transplant recipients are at a significantly higher risk of developing NMSC (C44) compared to the general population. The risk for other skin malignancies (C44) is pronounced. At 1 year, the cumulative incidence rate in liver transplant recipients is 0.0931%, with an RR = 2.61 (95% CI: 0.84–8.12), suggesting a moderately increased risk, though with some uncertainty due to the wide confidence interval. By 5 years, the cumulative incidence rises sharply to 0.8311%, with an RR = 4.68 (95% CI: 2.99–7.35), reflecting a significantly elevated and more precisely estimated risk. At 10 years, the cumulative incidence for NMSC reaches 2.6462%, with an RR = 7.35 (95% CI: 6.77–7.99), indicating that liver transplant recipients are more than 7 times as likely to develop NMSC compared to the general population. This long-term risk is highly significant and consistent.
The Z-test results are significant (p < 0.001) for NMSC at all time points, confirming that the differences in cumulative incidence rates between liver transplant recipients and the general population are statistically significant. The data clearly established liver transplantation as a major risk factor for NMSC, where the risk increases markedly over time.
These findings highlight the critical need for regular dermatologic screening and preventive strategies in liver transplant recipients, with a specific focus on NMSC, which pose a substantial and growing risk in this population.
Heart transplant (Tx) recipients vs general population
The following Table 7 presents the risk of developing NMSC over a 10-year period in a cohort of heart transplant recipients. The data highlight both the cumulative incidence of these cancers and the number of new events at each follow-up interval, providing valuable insights into the timing and progression of skin cancer risk in this vulnerable population.
The data in Table 7 highlight the increasing cumulative incidence of non-melanoma skin cancer (NMSC) (C44) over a 10-year follow-up period in heart transplant recipients. The incidence of NMSC (C44) shows a steady increase over time, with new events occurring consistently throughout the follow-up period. By the 10th year, the cumulative incidence rate of NMSC reaches 4.0609%, indicating a high risk. The data suggest that heart transplant recipients are at significantly greater risk for NMSC, with the risk progressively increasing as follow-up extends.
The data in Table 8 show a distinct pattern of non-melanoma skin cancer (NMSC) risk in heart transplant recipients compared to the general population. The risk of NMSC (C44) is high and clearly defined. At 1 year, the cumulative incidence rate in heart transplant recipients is 0.0909%, with an RR = 2.56 compared to the general population. Although this indicates more than double the risk, the confidence interval (95% CI: 0.37–18.18) suggests a lack of precision, likely due to the smaller number of cases early on. By 5 years, the cumulative incidence rate increases sharply to 0.8871%, with an RR = 5.00 and a much narrower confidence interval (95% CI: 2.24–11.13), demonstrating a significantly elevated risk. At 10 years, the cumulative incidence reaches 4.0609%, with an RR = 11.29, and the confidence interval (95% CI: 9.64–13.22) indicates a highly significant and reliable increase in the risk of NMSC in heart transplant recipients.
The Z-test results across all time points for NMSC is significant (p < 0.001), confirming that the differences in cumulative incidence rates between heart transplant recipients and the general population are not due to chance.
Discussion
Following organ donation, NMSC are frequent and significantly increase mortality.13, 17 Numerous research studies have looked at the risk of NMSC following transplantation, showing a wide range of the reported rise in incidence. This raises many questions regarding the scale of this phenomenon in Poland. The studies that are available so far for the Polish population either focus on a small number of participants, describe individual cases, or relate to earlier periods when immunosuppressive medication was frequently administered differently than it is today.16, 17, 18, 19 The purpose of our research was to reexamine the relationship between Poland’s organ transplant recipients and the incidence of SCC or BCC in comparison with the corresponding incidence observed over the same time span across the general Polish population. After analyzing data from the National Health Fund, we discovered that abundant NMSC cases were present in the 3 most numerous post-transplant patient groups: those who had received a kidney, liver or heart transplant.
Upon comparing data from the National Health Found to population data published by the National Cancer Registry,14 the risk of NMSC occurrence in renal transplant recipients was nearly 12 times higher after 10 years of transplantation compared to the population risk at the same time (cumulative risk 4.18% vs 0.36%). The escalating incidence in the renal transplant recipient cohort over time is probably indicative of a persistent and compounding risk factor for NMSC attributable to long-term immunosuppressive treatment, which may not only enhance the survival of malignant cells but also promote the accumulation of oncogenic mutations and inhibit the body’s natural antitumor immune defenses. This impact of immunosuppression seems to be independent from other well-known risk factors, like UV exposure, as other risk factors are evenly affecting general population in Poland as equally as they affect post-transplant patients.
The 10-year data also show an increase in the cumulative risk to 2.65% among liver transplant patients, compared to 0.36% in the general population (2010–2022). This enduring elevation in NMSC incidence underscores the chronic risk associated with long-term immunosuppression and possible additional hepatic comorbidities that may further compromise immune surveillance against skin cancer.
Patients who have undergone heart transplants in Poland also statistically more frequently received a NMSC diagnosis over the course of follow-up. Cumulative incidence climbs to 4.06%, compared to 0.36% within the general population during the same time. This trend suggests a continued increased risk of NMSC associated with long-term immunosuppression, and possibly other factors unique to heart transplant recipients, such as the increased incidence of viral infections that can contribute to skin cancer risk. In conclusion, the cumulative risk of NMSC compared to the general population is also significantly higher in all 3 mentioned groups of patients.
Limitations
It should be noted that our investigation is subject to various limitations. Firstly, information from the National Health Fund database on the matter is limited to the period after 2010. Furthermore, we tried to reduce the possibility of overdiagnosis by only taking into account hospital and clinical data related to the diagnoses (i.e., we eliminated ICD codes entered into the national online reporting system at primary care facilities without prior histopathological confirmation). Additionally, since SCC and BCC were not given distinct ICD-10 codes, it is not possible to conduct a separate analyses on the incidence of these 2 skin malignancies. Fortunately, since 2022, all Polish pathology reports have been entered into nationwide online reporting system, which in future will enable pathology diagnosis-directed, rather than code-based, analysis.
Additionally, another weakness of this study is the well-known phenomenon of underreporting of skin cancers like SCC and BCC.20 On the other hand, the underreporting of the C44 code applies to the same extent both to transplant recipients and the general population, thus allowing for valid comparison.
There are many well-established factors influencing the increased incidence of skin cancers, such as the patient’s age, UV exposure or HPV infection. However exposure to these factors is no different in general population and its subpopulation of transplant recipients in Poland. It might be even speculated, that transplant recipients are younger as opposed to general population at the time of NMSC diagnosis – we are currently exploring that issue in the ongoing further epidemiological study. Therefore, we believe that they do not have a significant impact on the final result.
Additionally, the type of immunosuppression used and the duration of its application also affect the incidence of cancers. There are many drugs and their combinations used in organ recipients to prevent the rejection of the transplanted organ. However the most commonly used immunosuppressive treatment regimen for solid organ transplant recipients in our dataset was: tacrolimus (TAC) or cyclosporine A (CsA) combined with mycophenolic acid (MPA) and glucocorticosteroids (GS). We are intending to perform subgroups analyses on strictly and narrowly defined groups receiving specific immunosuppression.
Nevertheless, the strength of the report derives from a rigorous examination of the available data, stringent inclusion criteria, and their integration to produce a clinically meaningful consensus. Utilizing data from one of the most trustworthy medical information repositories for transplant recipients in Poland, this study represents, to the best of our knowledge, the first extensive analysis of such a large group of organ transplant recipients in Poland and presumably in Europe as well. According to our research, organ transplantation and subsequent immunosuppression exposes transplant recipients to a much higher risk of NMSC. This underlines the need to comprehensibly inform the patients on the risk and educate them how to perform self-examinations.21 It was acknowledged in recently published Polish Oncological Society guidelines for melanoma and NMSC post-treatment surveillance – 5 years following transplantation, guidelines advise that patients who have undergone transplantation should have their skin evaluated for cancer at least twice a year.22, 23, 24
Conclusions
The risk of NMSC in patients who have long-term survived renal, heart and liver transplants has been steadily rising over time since transplantation in Poland. Based on >17,000 transplant recipients, we concluded that the risk of developing NMSC after transplantation was higher than the general population risk. The 10-year cumulative incidence of NMSC increased in renal (4.18% vs 0.36, p < 0.001), liver (2.65% vs 0.36, p < 0.001) and heart recipients (4.06% vs 0.36, p < 0.001) vs general Polish population. The overall NMSC risk was continuously rising since transplantation staring from on average 2 times higher risk at 1 year to 12 times higher risk after 10 years compared to the risk of NMSC in general Polish population.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.14606095. The package includes the following files:
Patient population at risk following kidney, heart, and liver transplantation in 2010–2022.
Supplementary Table 1. Number of patients at risk following kidney transplantation by year of transplantation with 10-year follow-up.
Supplementary Table 2. Number of patients at risk following heart transplantation by year of transplantation with 10-year follow-up.
Supplementary Table 3. Number of patients at risk following liver transplantation by year of transplantation with 10-year follow-up.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.