Abstract
Background. Numerous research studies have explored the impact of micronutrients, including dietary minerals such as iron, zinc, selenium, copper, and vitamins A, B complex, C, D, and E, on periodontitis. However, there is no literature investigating the role of iodine in periodontal health.
Objectives. This study aimed to examine the potential influence of iodine, a trace element, on periodontal health, an area that has not yet been researched.
Materials and methods. The study recruited a total of 73 participants, including 33 periodontally healthy control subjects and 40 patients with stage III periodontitis. Iodine levels in urine samples were measured using a spectrophotometric method, and the results were expressed in µg/L.
Results. Lower iodine levels were observed in patients with periodontitis. Individuals with low iodine levels were found to be 1.04 times more likely to develop periodontitis than those with high iodine levels. The study found that if a person’s urine iodine value is below 76.93 µg/L, the probability of having periodontitis is 72.5%; if it is above this value, the probability of not having periodontitis is 90.9%.
Conclusions. These findings suggest that urinary iodine levels could be a valuable metric for future research, as indicated by the variance in mean urinary iodide levels. Further extensive studies could establish urinary iodine levels as a useful biomarker for the diagnosis, prognosis and treatment plan of periodontitis.
Key words: periodontitis, risk, periodontal health, iodine deficiency, urine iodine level
Background
According to the World Health Organization (WHO), micronutrients are essential in very small quantities (measured in micrograms or milligrams per day) and act as “magical tools” that support the production of enzymes, hormones and other necessary substances for healthy growth and development of the body.1 Micronutrients, including essential vitamins and minerals, are crucial for promoting proper metabolic function, facilitating wound healing, and preventing illnesses and infections.2 In vitro studies have demonstrated that vitamins and trace elements have a significant impact on every aspect of immune function, and deficiencies in these nutrients can compromise immune function.3
Major minerals such as sodium (Na), potassium (K), calcium (Ca), magnesium (Mg), phosphorus (P), and sulfur (S) are required in amounts greater than 100 mg/day, while trace minerals such as iron (Fe), zinc (Zn), selenium (Se), copper (Cu), iodine, fluoride (F), cobalt (Co), chromium (Cr), manganese (Mn), and molybdenum (Mo) are required in amounts less than 100 mg/day.4 Reports indicate that deficiencies in micronutrients have been described as a “silent epidemic” affecting approx. 2 billion people globally, regardless of age or gender.5 The WHO has noted micronutrient deficiencies in several Middle Eastern countries, especially among children and women of childbearing age, involving Ca, Fe, Zn, vitamin A, vitamin D, and folic acid. Iron deficiency is prevalent among a majority of women in these countries, with over 13.2 million preschool children having retinol serum levels below 0.7 μmol/L, leading to approx. 800,000 cases of night blindness.6
Nutrients are crucial for maintaining the health of the human body and all metabolically active tissues. They also play critical roles in the regeneration process, managing oxidative stress and maintaining adequate immunity.7 While substantial data support the significance of vitamins and major minerals in periodontal and overall health, there is limited evidence supporting the functions of trace minerals. Inadequate intake of trace minerals could increase the risk of periodontal disease, either by exacerbating infections caused by periodontopathogens or by influencing biological gradients and host hemostasis.8
Periodontitis is a multifactorial disease resulting from complex interactions between the dental plaque biofilm and the host’s immunological inflammatory response. Furthermore, several environmental and host-related risk factors significantly contribute to the disease development, altering the host’s response and disease progression.9 Several approaches have been employed to slow the progression of periodontitis. Alongside conventional periodontal therapies, host-modulating agents such as dietary components (e.g., omega-3 fatty acids), dietary minerals (e.g., Fe, Zn, Se, Cu) and vitamins (e.g., A, B complex, C, D, and E) with immunomodulatory properties have been utilized to elicit favorable host responses. Furthermore, trace minerals impact periodontal health by exerting local effects on hard and soft tissues and systemic effects on immune-inflammatory mechanisms.10
Iodine, with an atomic weight of 126.9 g/mol, is a crucial component of thyroid gland hormones, which are essential in mammals. Despite its widespread presence in the environment, iodine is unevenly distributed, with oceans containing the highest iodide levels (approx. 50 μg/L). Iodide ions in seawater oxidize into elemental iodine, which is volatile and evaporates into the atmosphere, returning to the soil through rainfall, thus completing the cycle. However, in many regions, the iodine cycle is slow and incomplete, resulting in iodine-deficient soils and groundwater. Crops grown in these soils have low iodine concentrations, contributing to iodine deficiency in humans and animals that consume these crops.11
Studies have shown that iodine functions as an antioxidant in various tissues, including the breasts.12, 13, 14 When exposed to hydrogen peroxide, iodide acts as an electron donor, which may reduce the damage caused by oxygen free radicals.15 Iodide efficiently eliminates reactive oxygen species (ROS) within human blood cells.14 In addition, iodine has been found to interact with acute-phase proteins and is utilized by immune system cells and liver cells.16, 17 Besides its antioxidant properties, iodine has anti-inflammatory effects. Although levels of C-reactive protein (CRP) and interleukin 6 (IL-6) remain stable after administering different doses of iodide, negative correlations are observed between CRP and free tetraiodothyronine (FT4), as well as between urinary iodine and free triiodothyronine (FT3) and IL-6. This suggests a connection between iodide, thyroid hormone production and inflammatory status assessed with CRP and IL-6.18
Objectives
While several studies have explored the impact of trace elements, such as Fe, Zn, Se, and Cu, on periodontal health, the current study is unique in its emphasis on the significance of iodine, a trace element, in relation to periodontal health. The hypothesis of the study and the primary outcome suggest that the urinary iodine content differs between patients with periodontitis and periodontally healthy individuals.
Materials and methods
Study design
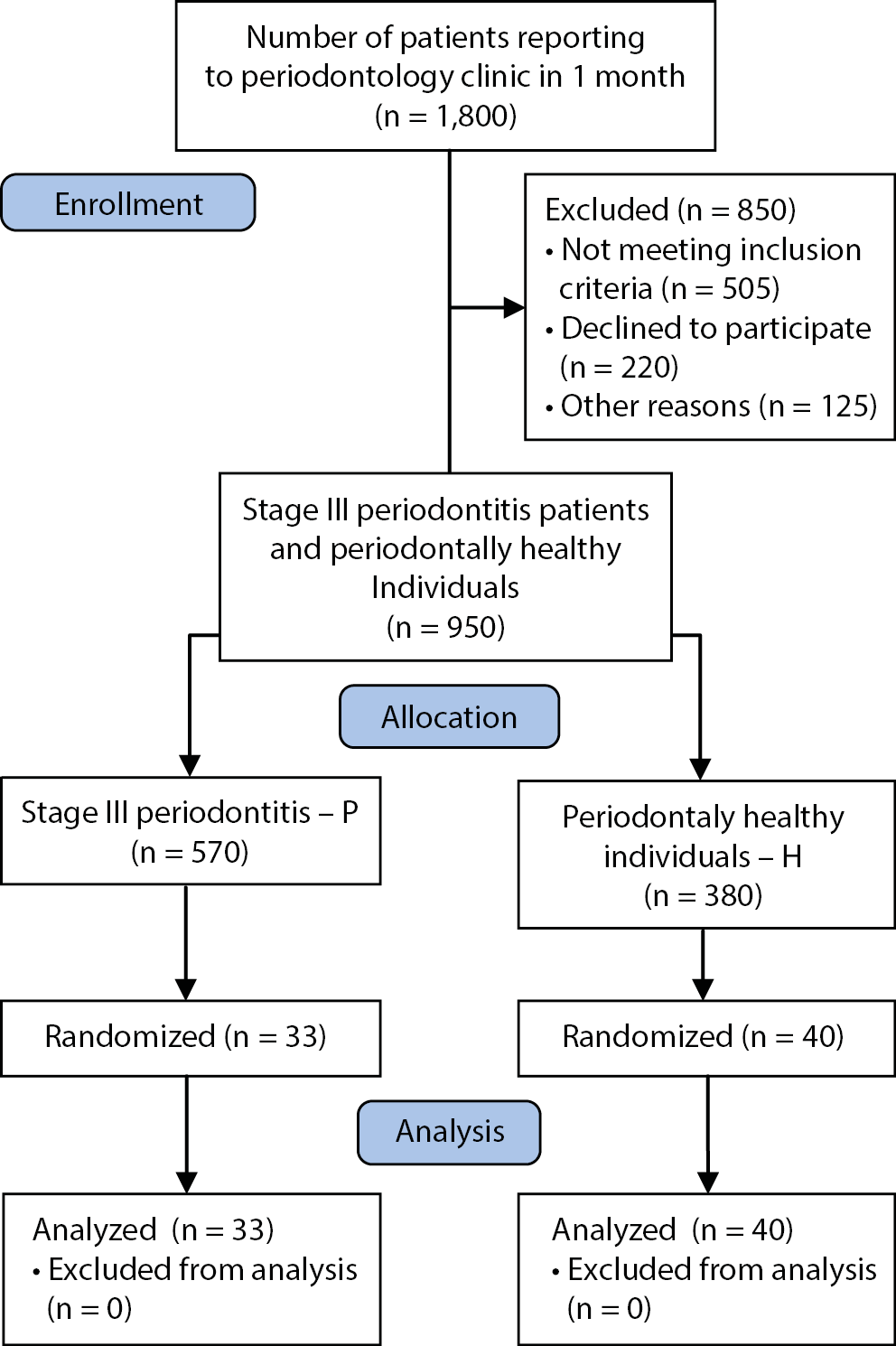
The present cross-sectional study included a total of 73 participants referred by the Department of Periodontology, Faculty of Dentistry, Atatürk University (Erzurum, Turkey), in 2022. Participants were systemically healthy, non-smoking individuals comprising 33 periodontally healthy control subjects (H) and 40 patients with stage III periodontitis (P). All participants were provided with information about the study and signed an informed consent form. The study protocol was approved by the Clinical Research Ethics Committee of the Faculty of Medicine, Atatürk University (approval No. 2021/11), and registered under Clinical Trials ID NCT05724251. The study was conducted in accordance with the Declaration of Helsinki.
Settings and division of patients into groups
To be eligible, individuals had to be free of systemic illnesses or conditions such as hepatic or renal dysfunction, diabetes, organ transplantation or cancer therapy, cardiovascular disease, pregnancy or lactation, goiter, hyper- or hypothyroidism, or unbalanced thyroid-stimulating hormone levels, and had to be in good systemic health. Those previously treated for periodontal disease requiring antibiotics for infective endocarditis prophylaxis during dental procedures were excluded. Additionally, subjects who had taken antibiotics, immunosuppressive drugs or nutritional supplements (especially iodine) in the past 6 months were excluded. Participants were asked about their dietary habits regarding the use of iodine-containing salt in daily nutrition or avoiding salt at meals, and only those with similar dietary patterns were enrolled in the study. Vegetarians or participants with dietary habits potentially affecting iodine intake were excluded (Figure 1).
Clinical periodontal status was assessed through intraoral examination and by referring to panoramic X-ray records. The control group (H) was defined as having a probing depth (PD) ≤3 mm, no clinical signs of gingival inflammation (<10% of sites with bleeding upon probing and absence of gingival redness/edema), no radiographic evidence of alveolar crestal bone loss, and no tooth loss due to periodontitis (each subject had at least 5 teeth per jaw section and 20–22 teeth in total).19
Patients classified with stage III periodontitis met criteria including interdental clinical attachment level (CAL) ≥5 mm, tooth loss ≤4 teeth due to periodontitis (at least 5 teeth per jaw section and 22 teeth per patient), PD ≥ 6 mm, vertical bone loss ≥3 mm, and Class II or III furcation involvement, as per the 2017 World Workshop criteria for periodontitis classification.20
Blinding
Physicians at the periodontology clinic categorized patients into periodontally healthy and periodontitis groups without disclosing group assignments to the patients before urine sample collection. Urine samples were collected by physicians unaware of group assignments. Biochemical analyses were performed by physicians blinded to participant groupings. Data results were recorded and forwarded to the author for statistical analysis without mentioning the group identifiers.
Randomization
Randomization was performed using computer-generated random number allocation. After diagnosis, patients with periodontitis and periodontally healthy individuals were randomly assigned to groups (Figure 1).
Collection and storage of urine samples
Study participants provided spot urine samples via normal micturition at the Department of Periodontology, Faculty of Dentistry, Atatürk University, before any surgical intervention or drug treatment. Samples were centrifuged at 2,000 rpm for 5 min at 4°C. The resulting supernatant was collected using an automatic pipette and divided into aliquots. Samples were then stored at –80°C until the day of analysis. There were no periods of recruitment or follow-up sessions for urine sample collection.
Analysis of urine iodine levels
Urine iodine levels were measured using a spectrophotometric method with the LTA IODIDE IN URINE (LTA s.r.l., Milan, Italy) kit in accordance with the manufacturer’s instructions. Results were reported in µg/L within the following categories: severe deficiency: 0–19 µg/L; moderate deficiency: 20–49 µg/L; mild deficiency: 50–99 µg/L; optimal nutritional value: 100–199 µg/L; adequate nutritional value: 200–299 µg/L; excessive nutritional value: >300 µg/L.
According to the WHO, most iodine absorbed by the body eventually appears in the urine. Therefore, measuring urinary iodine concentration (UIC) in casual urine specimens is recommended for monitoring iodine status. The UIC is highly sensitive to recent changes in iodine intake, as up to 90% of iodine is absorbed and excreted in the urine. Renal clearance of iodine, in the range 30−50 mL plasma/min, primarily depends on glomerular filtration in humans, with no reported tubular secretion or active transport mechanisms.21, 22
Statistical analyses
For the difference (effect size: 0.71) between the 2 groups to be significant, the G*Power (https://www.psychologie.hhu.de/arbeitsgruppen/allgemeine-psychologie-und-arbeitspsychologie/gpower) program calculated that there should be at least 33 subjects in each group at 80% power and 95% confidence level. A total of 73 subjects were included in the study, with 33 having periodontitis and 40 being healthy.
Statistical analyses were conducted using IBM SPSS v. 20 (IBM Corp., Armonk, USA). Descriptive statistics such as the mean, standard deviation (SD; for normally distributed variables) or median, 1st quartile and 3rd quartile (Q1 and Q3; for non-normally distributed variables or insufficient sample size to determine normality), percentage, and number were used to summarize the data. The normal distribution of continuous variables was assessed using various methods, including the Shapiro–Wilk test, Kolmogorov–Smirnov test, Q–Q plot, skewness, and kurtosis. The Mann–Whitney U test was employed to compare 2 independent groups when normality assumptions were not met. Iodine concentration and age variables were compared using the Mann–Whitney U test due to their non-normal distribution in the periodontitis and healthy groups. Gender and periodontitis/healthy status were analyzed using a 2×2 contingency table, with the appropriate χ2 test selected based on expected values: Pearson’s χ2 test for values greater than 5. The Fisher–Freeman–Halton test was used to compare iodine levels and periodontitis/healthy status, given the expected value was less than 5. Logistic regression analysis was conducted to model the relationship between age, gender, iodine concentration, and periodontitis/healthy status. Results from the logistic regression model were expressed as odds ratios (ORs) with a 95% confidence interval (95% CI). Analysis of covariance (ANCOVA) was employed to determine the effect of age on the comparison of periodontitis/healthy status and iodine concentration. The statistical analysis in this study was exploratory and did not incorporate correction for multiple comparisons. Therefore, caution is advised in interpreting the results due to the potential for uncontrolled type I errors, which could affect the validity of future meta-analyses. This exploratory analysis provides a foundation for future research in the field. Receiver operating characteristic (ROC) analysis was used to determine whether iodine concentration could be used in the diagnosis of periodontitis. Sensitivity and specificity were calculated to assess the validity of the diagnostic test results. In addition, the Youden index was used to determine the cutoff point. Statistical significance was set at p < 0.05.
Results
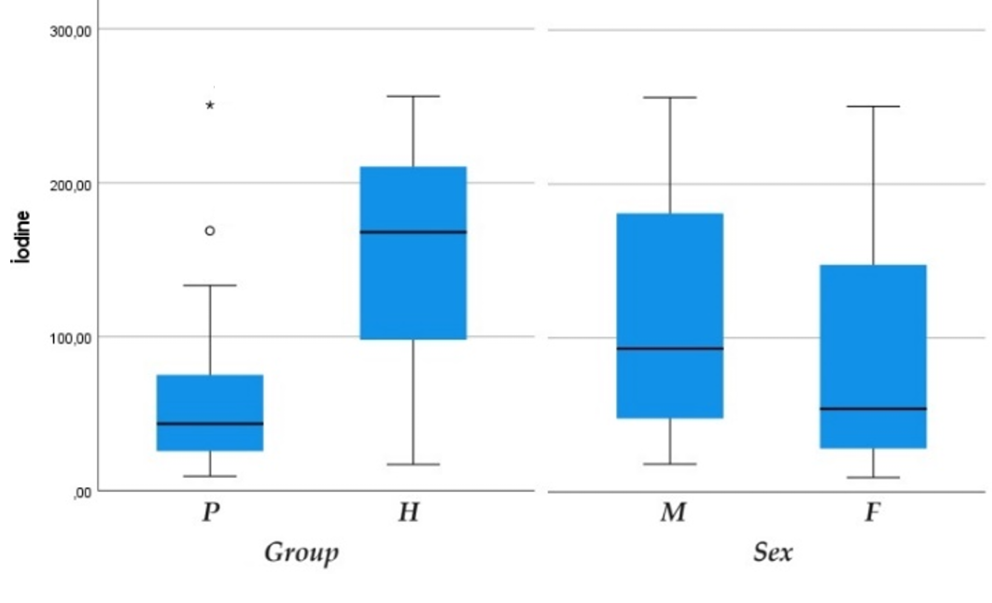
Table 1, Table 2 shows that there were significant differences in sex, age, and iodine levels between patients and controls (p = 0.029, p < 0.001 and p < 0.001, respectively; Figure 2). After adjusting for sex and age, the difference in iodine levels remained statistically significant (p < 0.001). Iodine levels were markedly lower in the patient group compared to the control group, and this difference was unaffected by sex or age. Table 3 shows no significant difference in sex and iodine levels among the study subjects (p = 0.073; Figure 2).
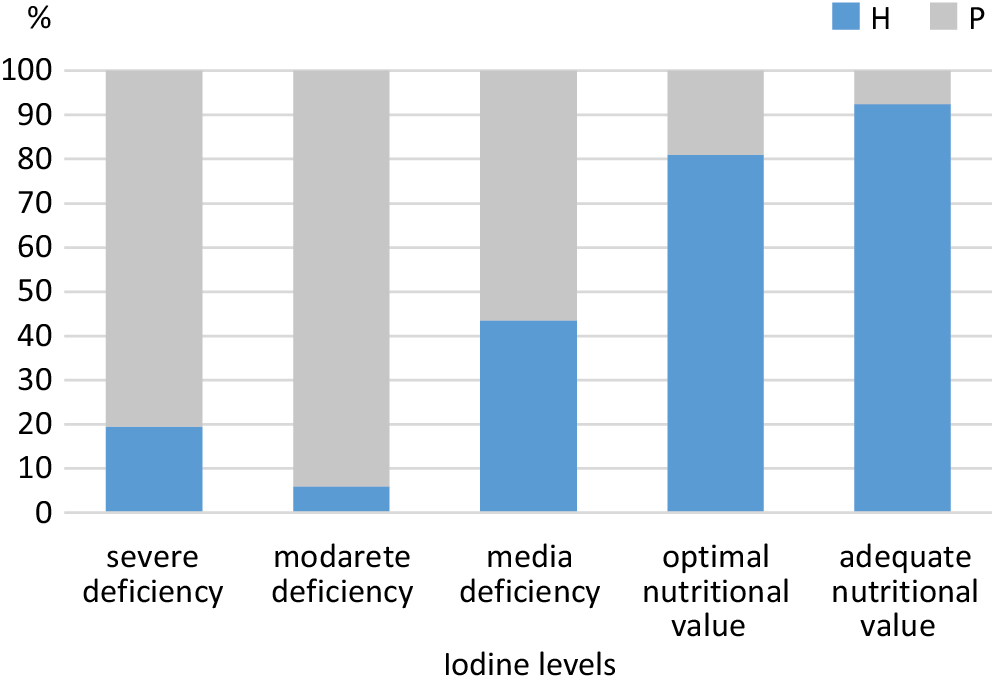
We found a significant difference in urinary iodine levels between the patient and control groups (p < 0.001). Urinary iodine levels in the control group were within the optimal and adequate range, whereas those in the patient group were classified as severely to moderately deficient (Table 4, Figure 3). Logistic regression analysis further indicated that age, sex and iodine levels were statistically significant between the patient and control groups (p < 0.001). The analysis revealed that the patient group had lower iodine levels than the control group. Individuals with low iodine levels were 1.04 times more likely to develop periodontitis than those with higher iodine levels (Table 5).
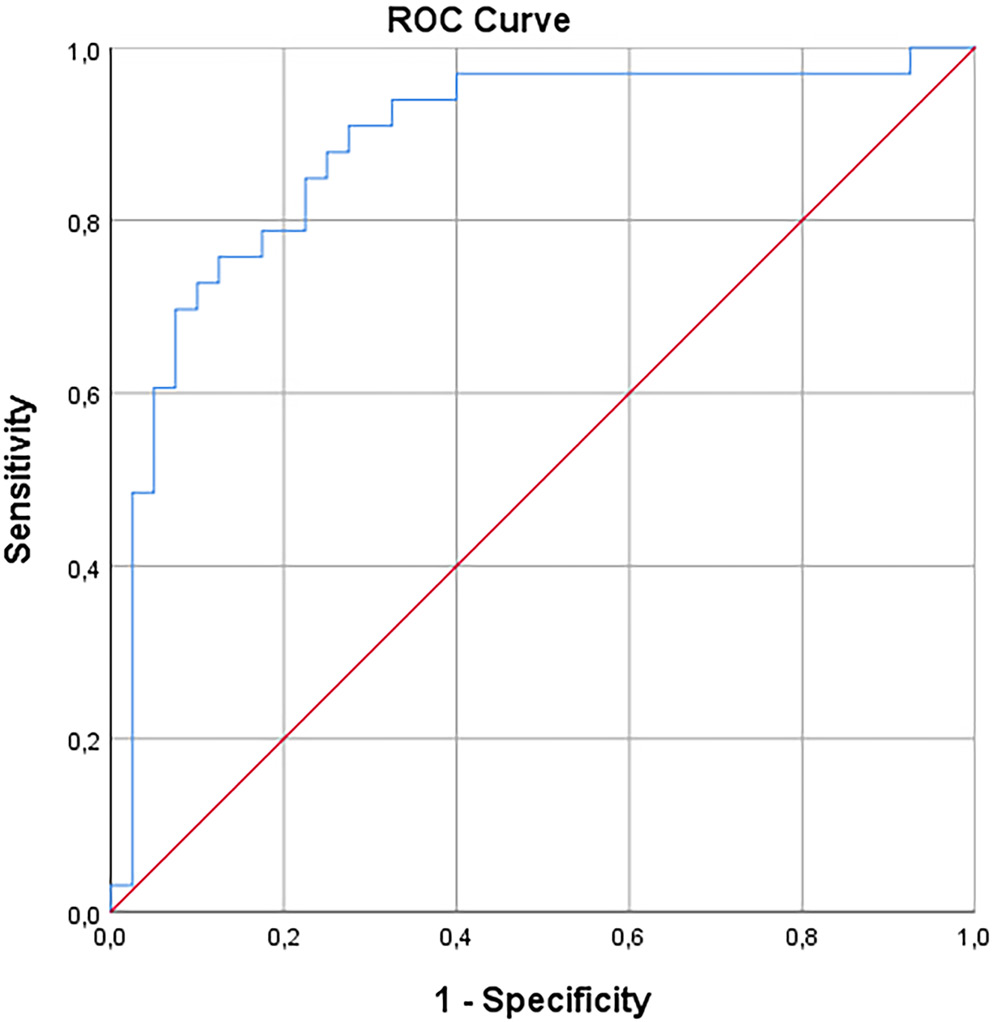
According to the results of the ROC analysis, the presence or absence of periodontitis can be predicted by examining the iodine values of the patients (0.884 ±0.042; p < 0.001). When individuals’ iodine levels were below 76.93, the likelihood of periodontitis was 72.5%; when above this value, the probability of not having periodontitis was 90.9% (Table 6, Figure 4).
Discussion
Periodontitis is a condition characterized by immune-inflammatory responses that result from the accumulation of dental plaque biofilm, leading to inflammation of the periodontal ligament and alveolar bone. Some experts suggest that a periodontal lesion can be considered a type of “wound” that requires an environment conducive to healing.23
The consensus across all studies is that maintaining optimal levels of micronutrients is crucial for periodontal health. Nutritional deficiencies can negatively affect the immune response and contribute to the progression of periodontal disease. Maintaining balanced levels of trace elements, including Fe, Zn, Se, and Cu, is vital for preventing the advancement of chronic diseases such as periodontitis.8 Therefore, this study aimed to investigate the potential effects of iodine deficiency on periodontitis.
The findings of this study revealed a significant reduction in urinary iodine levels among individuals with periodontitis (p < 0.001). No prior studies have reported similar results. Previous research has predominantly relied on serum or saliva samples to measure micronutrient levels.8
Iodine is an essential micronutrient that depends mainly on the level of dietary iodine intake. Adequate food and micronutrient supply is crucial for an effective and rapid immune response in inflammatory processes.24 The main dietary sources of iodine in the human population are iodized salt, seafood containing iodine from seawater, and cereals grown in iodine-rich soils. However, recurrent floods and soil erosion in mountainous regions cause iodine deficiency in the soil, resulting in inadequate iodine content in water and food. As a result, humans and animals consuming food grown in these lands may also experience iodine deficiency.25 Nutritional deficiencies in certain food components can lead to dental defects, oral mucosal problems and periodontal issues affecting oral health.26 In this study, participants in the periodontitis group had an iodine level of 51.63 ±4.98, which was below the normal range (with a median deficiency of 50–99 µg/L), whereas the iodine level in the healthy group (160.30 ±11.90) was within the normal range (with an optimal nutritional value of 100–199 µg/L).
Iodine deficiency has a geographical distribution affecting 40% of the world’s population.27 It is characterized by iodine-deficient soil washed away by glaciers, heavy rains or floods, often seen in mountainous regions such as the Himalayas, the Andes and large mountain ranges in China. Iodine deficiency also occurs in Central Asia, Central Africa and Europe. Literature indicates that iodine deficiency is prevalent among adults in the geographical region where the participants of the study reside.28, 29 A study in the same region found that the extent of iodine deficiency in subjects was classified as severe (15.6%), moderate (22%) and mild (26.8%).28 Another study in the same region investigating the relationship between iodine deficiency and dental caries found lower urinary iodine levels in patients with dental caries compared to healthy persons.30 Our study identified iodine deficiency levels in periodontitis patients as severe (12.5%), moderate (47.5%) and mild (27.5%). This finding among participants may be attributed to their geographical region and corresponding dietary habits that lack sufficient iodine.
Iodine is considered the most electron-rich essential element for animal and human nutrition and plays a crucial role in early human development. Iodine deficiency disorders encompass a range of consequences, from endemic goiter (thyroid gland enlargement in areas with low iodine content in soils or water) to growth retardation, central nervous system development impairment in children (cretinism), intellectual impairment, neonatal hypothyroidism (insufficient newborn thyroid function), pregnancy loss, and infant mortality.31
Its inorganic form, iodide, acts as an antioxidant by counteracting the damaging effects of hydrogen peroxide (H202) on human cells.32 Reactive oxygen species cause oxidative damage and can be categorized into free oxygen radicals and non-radical molecules. Non-radical ROS, such as H2O2, can react with cellular macromolecules, including DNA, leading to damage. Reactive oxygen species can cause severe DNA damage in periodontal tissue cells, contributing to periodontal tissue destruction.33 The significant reduction in iodine levels observed in patients with periodontitis underscores the potential importance of iodine in maintaining periodontal health. The antioxidant properties of iodide suggest it may act as an antimicrobial agent in saliva. The presence of an H2O2/peroxidase system in salivary glands supports the idea that iodide may have a bactericidal or bacteriostatic effect.34
Soriguer et al.35 conducted a study in which healthy volunteers were administered a diet program with a specific daily amount of iodine for 6 months. They observed a negative correlation between urinary iodine levels and CRP, an inflammation marker, and a positive correlation with glutathione peroxidase enzymes, which have antioxidant effects. These findings support the hypothesis that iodine possesses anti-inflammatory and antioxidant properties. The precise mechanism of iodine action remains unclear; however, it is known to penetrate microorganisms and target specific molecules such as amino acids (including cysteine and methionine), nucleotides and fatty acids, resulting in cell death. Iodine also has antiviral properties, but its effectiveness varies among different virus types. It is less effective against viruses lacking lipid envelopes and parvoviruses. Iodine may target the surface proteins of enveloped viruses and destabilize membrane fatty acids by reacting with unsaturated carbon bonds.36
Daily intake of trace elements, including Fe, Zn, Se, Cu, iodine, and Mn, ranging from 50 µg to 18 mg, is recommended for normal body functions. In patients with chronic renal failure, trace element levels may decrease due to inadequate food intake, increased dialysis and catabolic reactions. Maintaining optimal trace element levels has been shown to enhance immune function – reducing infection susceptibility and improving quality of life in these patients. Studies indicate that imbalances in trace element homeostasis correlate with mortality in end-stage renal disease patients.37 A healthy diet is essential for maintaining good health and a strong immune system, and it is justifiable to correct any micronutrient deficiencies. This can be achieved through dietary modifications or dietary supplements. Salt iodization is considered the most effective method to combat iodine deficiency, and it is a cost-effective way to contribute to economic and social development in almost all countries. Because only a small portion of the diet consists of micronutrients, maintaining balance sometimes requires the administration of dietary supplements.38 Fortified foods and supplements may offer a practical solution, especially when dietary modification is difficult. In cases where salt iodization is not feasible, vulnerable groups may benefit from iodine supplements or iodophor antiseptics.39, 40 However, there is still controversy surrounding the cost–benefit and widespread use of multimicronutrient supplements, and this issue has yet to be resolved.8 Further investigation is needed to assess the efficacy of supplement administration in periodontitis patients through randomized controlled clinical trials (RCTs).
Limitations
The study only examined a limited number of potential risk factors and did not consider other factors that might contribute to the development of periodontitis, such as smoking or genetics. Another limitation is that the study only included patients and controls from a specific geographic region, which could restrict the generalizability of the findings to other populations. The study population consisted of patients with stage III periodontitis. From a clinical perspective, investigating the levels of iodine trace elements in patients with early-stage periodontitis could be more interesting. Lastly, the study only assessed urinary iodine levels as a proxy for iodine status, which may not accurately reflect iodine levels in the body.
Conclusions
The findings of this study suggest that iodine levels in the body can indicate the ongoing destruction process in periodontitis. Differences in average urine iodide levels could be a useful measure for future investigations. As more extensive studies are conducted, iodine levels may become a useful biomarker for the diagnosis, prognosis and treatment plan of periodontitis. However, the research results presented in this paper are preliminary and should be treated as such. For generalizable results that can be applied broadly, they should undergo more extensive analysis.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.11609037. The package includes the following files:
Supplementary Table 1. ANCOVA age analysis.
Supplementary Table 2. ANCOVA sex analysis.
Supplementary Table 3. Logistic regression analysis.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.