Abstract
Background. Pancreaticoduodenectomy (PD), an abdominal surgery, is known for its complexity, cost and inherent risks. Recently, there has been increasing interest in enhanced recovery after surgery (ERAS) as a therapeutic approach. However, the mechanisms underlying postoperative functional recovery remain uncertain, and there are limited data on the efficacy of ERAS in postoperative physiotherapy following complex PD.
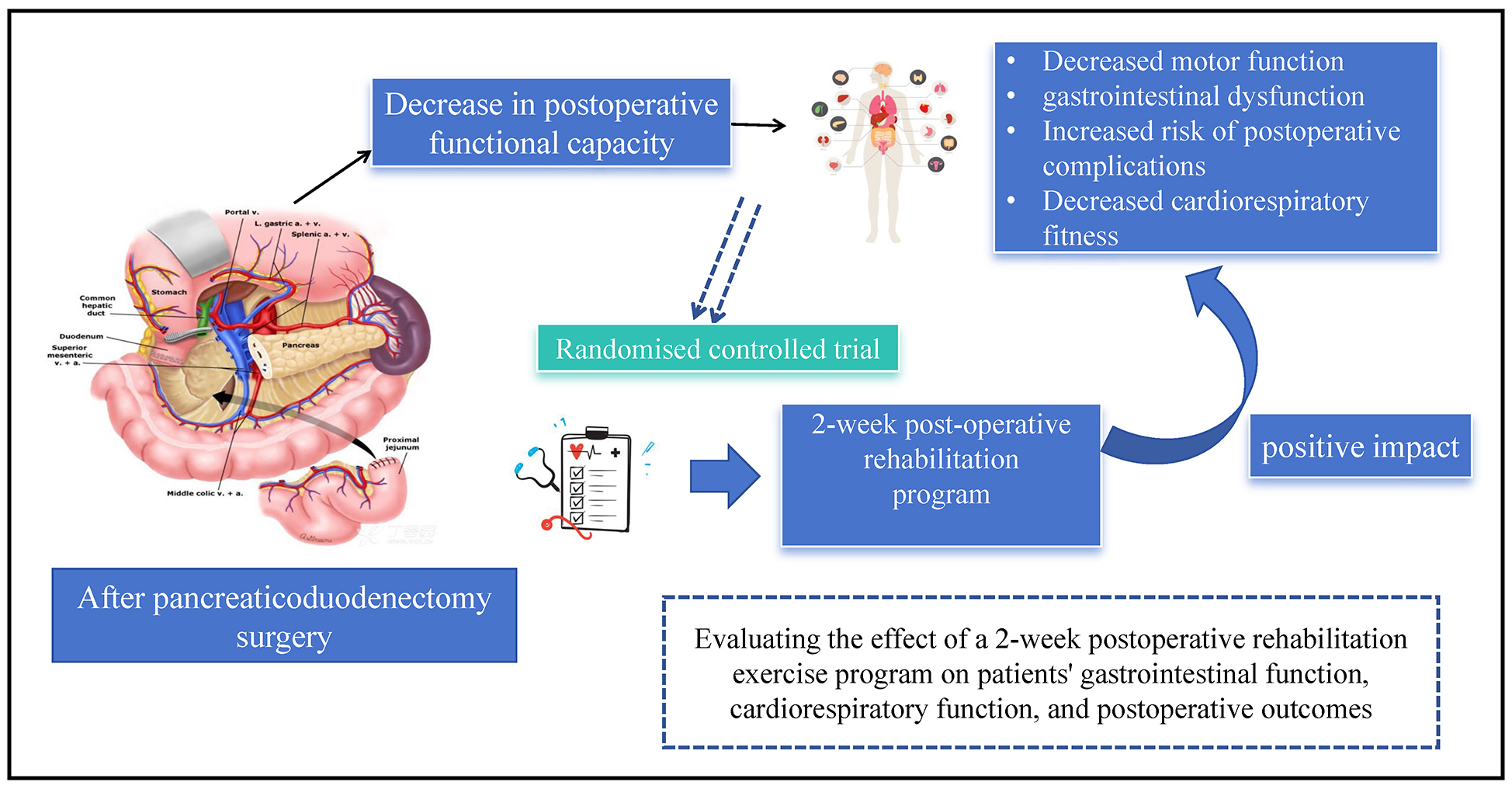
Objectives. This study aims to examine the feasibility and effectiveness of conducting a large powered randomized controlled trial (RCT) to evaluate a 2-week postoperative rehabilitation program based on the ERAS concept for patients undergoing pancreaticoduodenectomy.
Materials and methods. This study is a RCT with a single treatment group. From June 2022 to June 2024, 28 PD patients will participate in the trial. Patients will be randomly assigned to either a control group receiving standard clinical care or an intervention group undergoing a 2-week postoperative rehabilitation program. Cardiopulmonary function will be assessed using the 6-minute walk test (6MWT), and gastrointestinal (GI) recovery will be evaluated using the Intake, Feeling nausea, Emesis, physical Exam, and Duration of symptoms (I-FEED) scoring system.
Results. Secondary outcomes, including changes in recovery quality post-surgery, will be evaluated using the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC-QLQ-C30) and the Quality of Recovery Questionnaire (QOR-40). Additional recorded items will include time to first flatus and feces, daily volume of stomach fluid, time to gastric tube removal, length of hospital stay (LOS), and postoperative complications.
Conclusions. The study will utilize the I-FEED score, a novel tool for assessing GI function, to monitor the impact of a 2-week postoperative rehabilitation exercise program on patients. The primary outcome will focus on improvements in cardiopulmonary capacity following postoperative rehabilitation activities.
Key words: quality of life, pancreaticoduodenectomy, postoperative rehabilitation, gastrointestinal function
Introduction
Pancreaticoduodenectomy (PD) is necessary for treating diseases of the pancreatic head, jugular abdomen, duodenum, and distal bile duct. It is a challenging abdominal surgical procedure with high operational costs and risks.1 The PD involves significant surgical trauma and multiple gastrointestinal (GI) anastomoses, which can cause considerable surgical stress to patients, thereby increasing the risk of postoperative complications in 30–60% of cases. These complications include pancreatic leak, bile leak, postoperative bleeding, and delayed gastric emptying.2 Delayed gastric emptying has an incidence rate as high as 44%; although it is not life-threatening, it can impact the patient’s postoperative nutritional status and lead to postoperative malnutrition.3 This effect prolongs postoperative bed rest and contributes to issues such as pulmonary infection, reduced physical fitness and decreased quality of life (QoL). These factors result in longer hospital stays and increased medical costs.4, 5 Therefore, multidisciplinary management during the perioperative period is crucial.6
Enhanced recovery after surgery (ERAS) has gained increasing attention in clinical settings in recent years. This concept refers to a series of perioperative management measures within a multidisciplinary collaborative treatment model aimed at promoting early patient recovery, reducing traumatic stress responses, shortening hospital stays, and reducing medical costs.7 Although ERAS protocols are mainly surgeon- or technique-based, many optimal clinical management measures adopted during the perioperative period align with the principles of prevention and early rehabilitation advocated by rehabilitation medicine. Surgical interventions impose significant stress on patients, particularly those undergoing PD, who are often elderly and have multiple comorbidities. These factors can significantly reduce their functional ability. Additionally, following the operation, patients may encounter various emergent complications that lead to multisystem dysfunctions such as GI, motor and cardiopulmonary dysfunction.8 Therefore, it is essential to achieve the minimum functional level necessary for discharge and subsequent independent living in the community or at home, which encompasses all biological and cognitive functions. Rehabilitation interventions play a decisive role in attaining this goal.9 Therefore, postoperative rehabilitation for these patients is crucial.
Early postoperative activity is an integral part of ERAS. Scholars advocate that patients should mobilize within 24 h after surgery to accelerate postoperative recovery, in conjunction with other optimal clinical measures. Early postoperative bed activities can help patients maintain normal muscle tone, alleviate postoperative pain, promote GI tract recovery, enhance overall metabolism and blood circulation, and expedite the resumption of daily activities.10, 11 Research by Na et al.12 showed that early moderate exercise positively affects natural killer cell function in vitro following radical surgery in gastric cancer patients. Additionally, a short-term prospective randomized controlled trial (RCT) by Allgayer et al.13 indicated that a short-term moderate exercise program reduces oxidative DNA damage and boosts the immune system. These findings suggest that early rehabilitation programs following abdominal surgery are safe and feasible interventions that can mitigate potential infectious complications. In an RCT involving patients with stage I–III colon cancer, Ahn et al. compared postoperative exercise with standard medical care, revealing that low- to moderate-intensity exercise can shorten hospital stay and improve bowel motility following colectomy.14 This finding shows the importance of postoperative rehabilitation exercise programs in improving functional recovery and reducing hospitalization duration. However, there is limited supporting evidence regarding the application of these interventions in the context of complex PD. Therefore, further investigation based on the ERAS concept is required into the effects of postoperative rehabilitation on GI function, cardiopulmonary function and quality of recovery in PD patients. The results of such studies are expected to inform clinical practice, potentially accelerating postoperative rehabilitation, shortening hospital stay, alleviating the burden of high medical costs, and facilitating an early return to family and community life.
Objectives
This study aims to examine the feasibility and effectiveness of conducting a large, powered RCT to determine the role of a 2-week postoperative rehabilitation program based on the ERAS concept for patients undergoing PD.
Materials and methods
Design
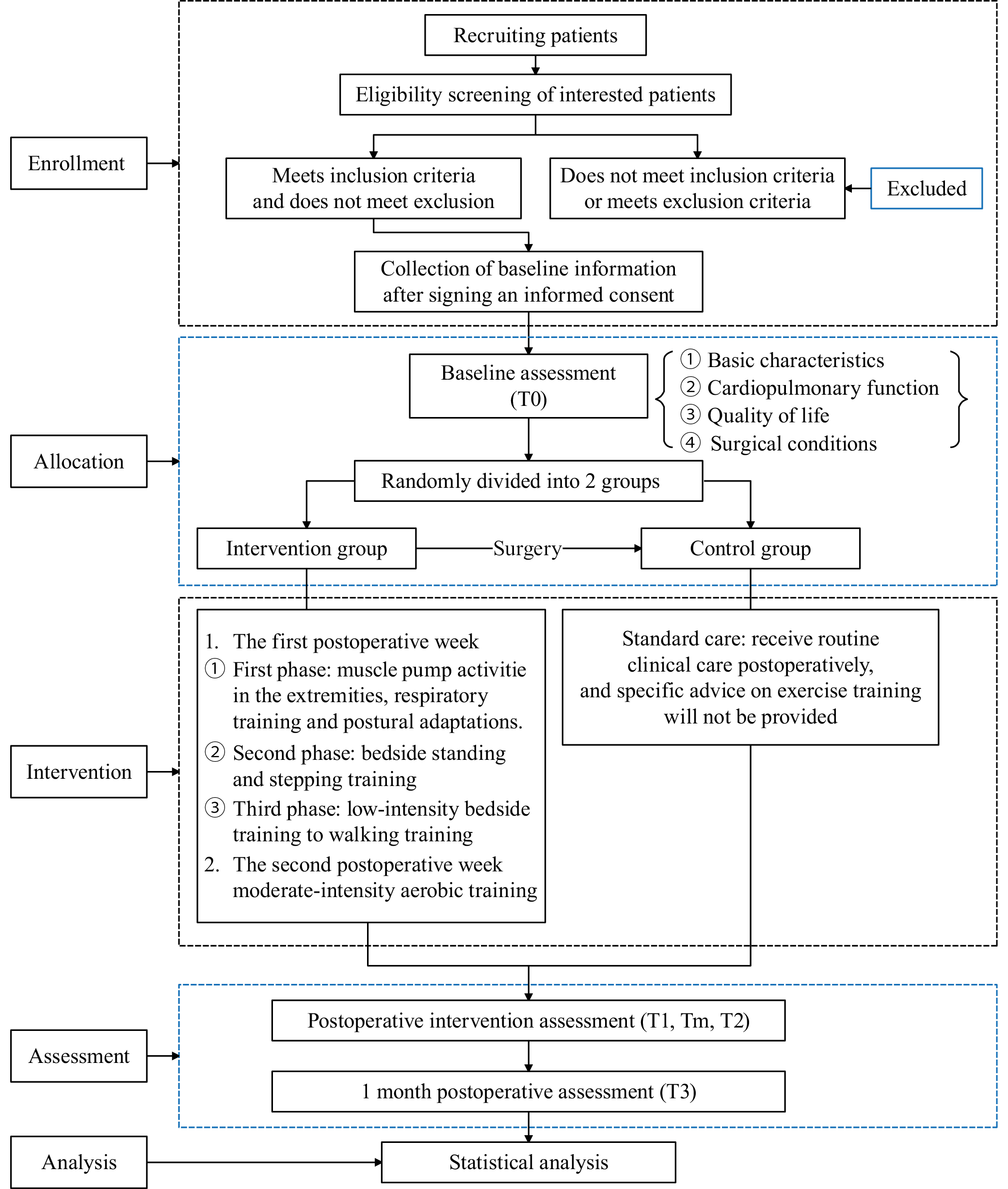
This study is a RCT with a single treatment group, which will enroll a total of 28 PD patients between June 2022 and June 2024. Participants will be randomly assigned to either a control group receiving standard clinical care or a trial group participating in a 2-week postoperative rehabilitation program. Cardiopulmonary function will be assessed using the 6-minute walk test (6MWT) at preoperative day 0 (T0), postoperative day 7 (T1), postoperative day 14 (T2), and postoperative month 1 (T3) in both groups. The primary outcome measure for both groups will be GI recovery at 2 weeks postoperative (T0–T2), measured using the Intake, Feeling nausea, Emesis, physical Exam, and Duration of symptoms (I-FEED) symptom score. Quality of recovery from T0 to T3 will be compared between the 2 groups using secondary outcomes measured with QOR-40 and EORTC-QLQ-C30. Secondary outcomes, including time to first flatus and defecation, daily volume of stomach fluid, time to gastric tube removal, length of hospital stay (LOS), and postoperative complications, will be measured 1 and 14 days post-surgery (Tm). The conceptual flowchart of the study is shown in Figure 1. The study design conforms to the The SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) checklist and SPIRIT diagram (Figure 2).
Study population and recruitment
Commencing in June 2022, subjects will be recruited from the Department of Hepatobiliary and Pancreatic Surgery at the Second Affiliated Hospital of Hainan Medical College (Haikou, China). Initially, the project manager will brief the team on the study’s goals and eligibility requirements. Then, patients scheduled for PD due to hepatobiliary and pancreatic tumors will receive information about the study’s goals, methods, potential benefits, and risks. Individuals interested in participating in the current research will undergo an interview with recruiting investigators to verify their eligibility. Finally, eligible participants or their guardians will sign an informed consent form to ensure confidentiality.
No patients or members of the public have been involved in the design, conduct, reporting, or dissemination plans of this research. Ethical approval has been obtained from the Ethics Committee of the Second Affiliated Hospital of Hainan Medical College (approval No. LW2022040) and is registered on ClinicalTrials.gov (No. ChiCTR2200060468). Written informed consent will be obtained from each participant before performing any procedures. The results of the study will be published in a peer-reviewed journal. This trial is currently in the recruitment phase.
Randomization, allocation and blinding
Eligible subjects will be randomly assigned to either the postoperative quantitative rehabilitation protocol or the control group at a 1:1 ratio based on their inclusion numbers using the PROC PLAN function of SAS v. 9.1 statistical software (SAS Institute, Cary, USA). The principal investigator will maintain confidentiality by placing random numbers inside opaque envelopes to determine the sequence of allocations. The allocation will remain undisclosed to the data analyst and appraiser; if they do become aware, they will be replaced. Participants will be instructed to keep their assigned interventions confidential from the evaluator.
Sample size
According to literature and previous studies such as “Changes in motor function and quality of life after surgery in patients with pancreatic cancer”, the mean 6MWT distance after treatment is 498 m for the rehabilitation program group and 403 m for the control group, with a standard deviation (SD) of 80 m.15 A two-sided test with equal sample sizes (κ = 1), a type I error probability (α) of 0.05 and a type II error probability (β) of 0.2 will be used for both groups. The sample size was calculated using the following equation (Equation 1):
 . (1)
. (1)
This calculation showed that 12 participants were required for the rehabilitation program group and 14 participants for the control group, totaling 28 participants and accounting for a 10% missed visit rate.
Participants
Inclusion criteria
Participants must meet the following criteria: 1) age 18–75 years; 2) absence of distant organ metastases; 3) no history of abdominal surgery; 4) absence of pancreatic, bile duct or jugular occupancy; and 5) signed informed consent for PD diagnosed through preoperative imaging.
Exclusion criteria
Patients will be excluded if they: 1) do not meet the indications for surgery; 2) have other serious complications incompatible with general anesthesia; 3) require immediate surgery; 4) have severe heart, lung, liver, kidney, or other organ insufficiencies; 5) have had recent cerebrovascular accidents resulting in limb or cognitive dysfunction; or 6) have a diagnosis of PD combined with serious infections, endocrine dysfunction or other factors limiting participation in physical activities.
Intervention
Patients in both groups will receive preoperative education, including explaining surgical precautions and coordination points and assisting patients in completing surgery-related examinations or preparations. The advantage of the intervention group lies in its ability to promptly engage in the postoperative rehabilitation program, with the aim of facilitating swift recovery of postoperative function.
Control group
Subjects in the control group will receive standard postoperative care, which involves advising patients to rest in bed after surgery and gradually mobilize as their condition stabilizes. No personalized exercise programs will be recommended.
Interventional study of postoperative rehabilitation
According to the following guidelines, a trained physical therapist will assess all patients starting on the day of surgery. Patients must meet the following criteria: 1) they must be awake and responsive to calls; 2) their blood pressure, heart rate and oxygen saturation were stable during surgery; 3) they did not experience any postoperative bleeding, with abdominal drainage of 600 mL or less over 24 h; 4) their muscle strength was assessed at level 3 or above; and 5) their wound pain score (on a numerical scale) was less than 4. During the first week after surgery, patients can participate in a 30–40-min early activity rehabilitation program with a physical therapist. The rehabilitation regimen during the 1st week after surgery (postoperative days 1–7 (POD1–7)) consists of 3 stages. First, patients will engage in a series of “muscle pump” activities designed to stimulate blood flow to the extremities. Second, they will practice standing and stepping on the bed. Third, they will transition gradually from low-intensity bedside training to walking training within the hospital setting. Table 1 shows the postoperative exercise schedule for the 1st week (T1).
The results of the 6MWT at T1 will inform the development of moderate-intensity aerobic training to be implemented during the 2nd postoperative week (T2). Table 2 displays specific recommendations for aerobic activities. Vital signs and patient complaints will be monitored during one-on-one exercises; however, if patients are unable to remain upright or report feeling fatigued, the activity will be terminated immediately. Based on the results of the 6MWT at T1, moderate-intensity aerobic training will be continued into T2. The time interval from illness to intermittent participation will be noted on the case report form (CRF) and illustrated during data interpretation, allowing participants to resume exercise intermittently if they have to stop for 1 or 2 days due to illness.
Outcome measurements
Primary outcomes
To assess the functionality of a patient’s digestive system after surgery, a novel objective evaluation tool called the I-FEED scale will be implemented.16 This scale focuses on 5 areas: postoperative intake, nausea, vomiting, physical examination findings, and symptom duration. Postoperative GI intolerance was defined as a score of 3, and GI dysfunction as a score of 6 on this 5-factor scale.17
Condition of the heart and lungs
The 6MWT is a walking speed and distance test conducted over 6 min. It is an easy-to-administer and reliable test that can be repeated with consistent results. This test predicts the likelihood of postoperative complications with high accuracy and correlates strongly with peak oxygen intake.18 The risk of significant postoperative complications due to hepatopancreatobiliary malignancy can be determined using the 6WMT.19
Secondary consequences
To compare the postoperative recovery quality between the 2 groups, we will administer the 40-item Quality of Recovery Questionnaire (QoR-40). The QoR-40 is considered the best QoL evaluation questionnaire for Chinese patients undergoing surgery due to its excellent reliability, validity and responsiveness.20 It covers 5 domains: emotional state (9 items), physical comfort (12 items), psychological support (7 items), self-care ability (5 items), and pain (7 items). Higher total scores indicate successful rehabilitation, with a possible range of 40–200 points.
Quality of life
The Quality-of-Life Questionnaire (QLQ-C30), developed by the European Organization for Research and Treatment of Cancer (EORTC), will be used to evaluate postoperative improvements in patients’ QoL.21 It includes the Global Health Status and Quality of Life scales, as well as the eight symptom scales (fatigue, pain, nausea/vomiting, dyspnea, sleep problems, loss of appetite, constipation, and diarrhea) among its 30 items. Responses for items 29 and 30 will be rated on a scale from 1 to 7, whereas the rest will be categorized into 4 levels (none, a little, more, and a lot) and rated from 1 to 4. Higher scores in symptom categories indicate worse health and functioning, whereas higher scores in functioning and overall health suggest better QoL.22
Clinical evaluation indices
Additional clinical evaluation indices will include time to first flatus and feces, daily volume of stomach fluid and time of gastric tube removal. Anaerobic digestion and elimination are signs that intestinal peristalsis is beginning to function again post-surgery. While they are closely associated with the recovery of intestinal function, they cannot independently determine it. Therefore, time to exhaustion and defecation remain crucial indicators for assessing digestive tract health post-surgery.23 Nurses or researchers will record patients’ exhaustion and defecation times, daily volume of gastric juices, and gastric tube removal times from postoperative day 1 to day 14 to calculate the time to first flatus and defecation.
Length of hospital stay
The LOS for patients undergoing PD will be evaluated during routine clinical checks. It was defined as the time from surgical resection completion until the patient’s discharge. Data will be retrieved from patients’ medical records post-discharge.
Postoperative complications
Routine clinical assessments of PD patients will be used to assess the frequency and severity of postoperative complications. Surgical complications were defined and will be assessed for severity using the Clavien–Dindo classification.
Safety
Methods for tracking and reporting medical mishaps are essential. An adverse event was defined as any unexpected medical issue or damage that occurs during the trial. On the observation form, the patient’s condition, noting symptom severity, onset, duration, and interventions taken are recorded. Rapid notification of adverse events during rehabilitative exercises will allow the investigator to pause the intervention, adjust the training cycle and provide focused treatment, while also informing the research unit and ethics committee as needed. Event occurrence will be analyzed.
Data collection and management
Data in the CRF should be promptly, accurately and truthfully recorded by the researcher based on participants’ original records. The monitoring person will verify that all CRFs are correctly filled out and are consistent with the original data; any discrepancies will prompt immediate correction by the investigator. The investigator’s signature and correction date are required to maintain the integrity of the original records.
Statistical analyses
Participants were blindly randomized, and data will be analyzed using the full sample. A complete case analysis will be performed if the data missing rate is less than 10%. For data missing more than 10%, extensive interpolation will be used to fill gaps. Data processing will utilize IBM SPSS v. 25.0 statistical software (IBM Corp., Armonk, USA). Mean and SD will be used to represent normally distributed data; Student’s t-tests will be used to compare data between groups; analysis of variance (ANOVA) hybrid design, using general linear model (GLM) tests, will be used to compare data across multiple time points; non-normally distributed data will be represented by median and interquartile range (M(IQR)) and compared using the χ² test. The threshold for statistical significance was set at p < 0.05.
Discussion
The 5-year survival rate for patients treated with PD, the principal treatment for pancreatic and Vater ampulla carcinoma (VPC), ranges only from 8% to 17%.24 The PD is a highly invasive surgical procedure. Despite continuous advancements in surgical techniques that have increased the overall survival rate,25 patients still face challenges in recovering after surgery.26 A major obstacle to full recovery post-surgery is the increased likelihood of postoperative complications, including reduced physical function, lung infections, GI dysfunction, and other systemic dysfunctions arising from prolonged bed rest due to factors such as postoperative discomfort and the presence of tubes. Surgeons’ recognition of the impact of their patients’ functional status on postoperative QoL and their pursuit of improved clinical outcomes have led to a surge in interest and demand for post-surgery rehabilitation.
The ERAS concept heavily emphasizes early postoperative mobilization, aligning with functional recovery post-surgery. Exercise regimens designed for postoperative rehabilitation following abdominal surgeries such as colonoscopy, thoracotomy and hernia repair have been shown to be successful.27 A single-blind, parallel-arm, RCT comparing early postoperative mobility programs with standard care among patients undergoing major abdominal tumor surgery demonstrated the safety and feasibility of early mobilization programs, incorporating exercises such as core stability, gait training, upright training, and aerobic and resistance training, twice daily, which also improves patient functions.28 Furthermore, in a meta-analysis and retrospective analysis of the ERAS program for PD, it has been universally concluded that postoperative ERAS programs are feasible and safe and can improve postoperative outcomes.29, 30 These studies highlight the importance of early mobilization for patients undergoing this procedure.31 Patients participating in postoperative rehabilitation programs experience lower risks of major complications, shorter hospital stays and reduced out-of-pocket medical expenses. Early, intensive physical therapy after surgery has been shown to significantly enhance QoL by restoring or improving muscle strength, reducing fatigue and enhancing physical functioning among cancer patients.32 Physical therapists can more effectively monitor patient progress and ensure adherence to daily exercise regimens during hospital-based postoperative rehabilitation.
Conclusions
In this study, the I-FEED score, a novel tool for assessing GI function, will be used to monitor the impact of a 2-week postoperative rehabilitation exercise program on patients’ GI function. The primary outcome measure will focus on improvements in cardiopulmonary capacity following postoperative rehabilitation activities. Secondary outcomes include examining the effects of postoperative rehabilitation on recovery quality, complications and LOS. This experimental evidence will contribute to larger hypotheses guiding future multicenter RCTs.