Abstract
Background. Chronic soft tissue injury is characterized by sterile inflammation and pain. Gua sha with Masanggoubang oil (GSMO) treatment has been found to possess anti-inflammatory and analgesic effects.
Objectives. To explore the mechanism of GSMO in chronic soft tissue injuries.
Materials and methods. Fifty male rats were randomly divided into 5 groups (n = 10): 1) control group; 2) chronic soft tissue injury model group; 3) GSMO group; 4) inunction with Masanggoubang oil (IMO) group; and 5) ua sha with tea oil (GSTO) group. The control group and model group received no treatment, while the GSTO group and GSMO group received gua sha therapy with tea oil or Masanggoubang oil on the injured sites. The rats in the IMO group were treated with Masanggoubang oil inunction on the injured sites once every other day, 4 times in total. All animals were sacrificed 48 h after the last treatment. Muscle tissue sections from the injured sites of the rats were stained with hematoxylin & eosin (H&E) staining to observe pathological changes. The protein levels of tumor necrosis factor alpha (TNF-α), interleukin 1β (IL-1β), interleukin 6 (IL-6), inducible nitric oxide synthase (iNOS), and β-endorphin (β-EP) in the rats’ skin, serum, and muscle were determined using enzyme-linked immunosorbent assay (ELISA).
Results. Gua sha with Masanggoubang oil treatment alleviated necrosis and the denaturation of muscle fibers at the injured sites, reduced connective tissue proliferation and scar tissue generation, downregulated the levels of TNF-α, IL-6 and iNOS in the skin and TNF-α, IL-1β, IL-6, and iNOS in the muscle and serum, and upregulated β-EP levels in the muscle.
Conclusions. Gua sha with Masanggoubang oil treatment significantly improved the inflammatory response in rats with chronic soft tissue injury, which may be associated with a reduction of M1 macrophage polarization in the peripheral blood and local tissues. Additionally, the combination of gua sha therapy and Masanggoubang oil may have a synergistic effect in treating chronic soft tissue injuries.
Key words: gua sha with Masanggoubang oil, chronic soft tissue injury, M1 macrophage polarization, TNF-α, β-EP
Background
Chronic soft tissue injuries involve microcirculatory disorders and sterile inflammation caused by prolonged chronic stress, characterized by swelling, pain, limb dysfunction, and even disability in severe cases. The incidence reaches approx. 90% in adults,1 severely impacting the patient’s physical and mental health as well as their quality of life.2 Due to its high incidence and difficulty in eradication, it is listed among the top 3 most challenging diseases by the World Health Organization (WHO).3 The mechanisms of pain and limb dysfunction in chronic soft tissue injuries remain unclear, but modern medicine associates it with local microcirculatory disorders, compression of nerve endings and inflammatory responses.4 Recently, there has been a notable surge in attention to M1 macrophage polarization concerning the inflammatory mechanisms of soft tissue injuries.5, 6 M1 macrophages stimulate the inflammatory response through the release of pro-inflammatory cytokines, including interleukin 6 (IL-6), interleukin 1β (IL-1β), tumor necrosis factor alpha (TNF-α), and inducible nitric oxide synthase (iNOS). This process heightens the sensitivity of the peripheral and central nervous systems, ultimately leading to hyperalgesia.7, 8 Tumor necrosis factor alpha plays a pivotal role as an inflammatory mediator, instigating the release of a cascade of other pro-inflammatory cytokines, such as IL-1β and IL-6.9 The expression level of iNOS in skeletal muscles correlates with the magnitude of inflammatory damage.10 Beta-endorphin (β-EP), an endogenous opioid peptide secreted by immune cells and exhibiting characteristic opioid-like effects, acts on opioid receptors in primary afferent neurons, obstructing pain transmission and eliciting an analgesic effect.11 Previous studies have identified potential correlations between levels of TNF-α, IL-6, IL-1β, iNOS, and β-EP and soft tissue injuries.11, 12, 13 Traditional Western medical approaches to chronic soft tissue injuries commonly involve a prescription for nonsteroidal anti-inflammatory drugs. Nevertheless, this therapeutic approach manifests obvious adverse effects, including hepatorenal damage and gastrointestinal reactions, while failing to impede the progression of the disease.
In traditional Chinese medicine (TCM), chronic soft tissue injuries are considered tendon and muscle injuries resulting from qi (circulating life force) stagnation, blood stasis, and an obstruction of meridians (paths through which qi flows). The therapeutic strategy is directed towards alleviating pain by promoting blood circulation, resolving blood stasis and improving qi flow. Gua sha, a TCM therapeutic modality extensively applied in Asia, demonstrates notable efficacy in the management of chronic pain. Its mechanism involves the stimulation of acupuncture points (acupoints) and meridians based on the meridians and acupoints theory. In the course of gua sha therapy, a tool such as a buffalo horn or coin is used to scrape the body’s surface, often in conjunction with massage oil or water. This intentional scraping of the skin produces sha (therapeutic petechiae), engendering new biological effects. Gua sha functions to stimulate meridians, promote blood circulation, remove blood stasis, balance yin and yang (complementary and at the same time opposing forces that interact to form a dynamic system), and regulate the intestines.14 From an anatomical perspective, yin and yang are linked to various anatomical regions of the body. Specifically, yin is linked to the inferior portions of the body, including the pelvis and lower extremities, while yang is associated with the superior regions of the body, such as the upper extremities, head and neck, and the back. A previous study proposed that gua sha demonstrates its effects in stimulating cutaneous telangiectasia, activating the serotonergic, noradrenergic and opioid systems,15 elevating 5-hydroxytryptamine, mast cells, CD4+, CD8+, and IL-6 levels in local skin tissues, and reducing IL-1, prostaglandin E2 and phospholipase A2 expression. These mechanisms collectively contribute to pain relief,16, 17, 18, 19, 20 toxin elimination, heightened antioxidant activity, and upregulated immune functions involved in regulating the circulatory, nervous, immune, and other systems, notably the musculoskeletal system.21, 22
Gua sha with Masanggoubang oil (GSMO) is a characteristic external therapeutic technique within Chinese Miao medicine (a branch of TCM practiced by Miao ethnic group in China). This scraping medium is produced by blending Masanggoubang, a Miao herb derived from the root of Astilbe chinensis, with tea oil. Researchers have shown that Masanggoubang contains flavonoids, with Astilbe being the main component. These flavonoids exhibit anti-inflammatory, analgesic, circulatory, and decongestant properties.23, 24 A study has confirmed that Astilbe mediates anti-inflammatory functions by downregulating the expression of proteins such as nitric oxide (NO), prostaglandin E2, iNOS, and cyclooxygenase-2, along with modulating the mRNA expression of IL-6 and TNF-α through its impact on the NF-κB pathway.25 Furthermore, studies published in 2020 have demonstrated that oral administration of Astilbe improved inflammation and slowed down the progression of osteoarthritis in rats by regulating the NF-κB, PI3K/AKT and TLR4/MD-2 pathways.26, 27, 28 Similarly, the external application of Masanggoubang has been found to improve clinical symptoms and enhance the quality of life in patients with knee osteoarthritis and rheumatic arthritis.29, 30 Huang and Xia31 discovered that GSMO mitigated limitations in shoulder joint activity and deltoid muscle atrophy in patients recovering from cervical spine surgery. Another randomized, multicenter, parallel-controlled clinical trial published in 2017 found that GSMO not only alleviated neck and shoulder pain but also improved symptoms of upper limb numbness in patients with cervical spondylotic radiculopathy. Moreover, the efficacy of this intervention was observed to surpass that of gua sha with tea oil (GSTO).32 Furthermore, an independent study in 2018 substantiated the differences in clinical effectiveness in patients with chronic soft tissue injuries when employing scraping with varying concentrations of Masanggoubang oil. This study shed light on the impact of the scraping medium on clinical outcomes.33 Additionally, another study has reported that the external application of Masanggoubang oil does not cause skin irritation or allergic reactions.34 This finding further reinforces the evidence of its efficacy and safety. However, the precise mechanisms underlying its action on chronic soft tissue injuries remain undetermined. Therefore, the objective of this study was to elucidate the effect and mechanism of GSMO therapy on chronic soft tissue injuries, ultimately aiming to advance its clinical application.
Objectives
Generally, chronic soft tissue injuries correlate with elevated levels of IL-1β, IL-6, TNF-α, and iNOS. Both gua sha and Masanggoubang have demonstrated anti-inflammatory and analgesic effects. Consequently, we hypothesized that GSMO therapy holds the potential to mitigate chronic soft tissue injury by regulating the expression of inflammatory and pain-related cytokines. To investigate this, this study aimed to identify the potential mechanism of GSMO in rats with chronic soft tissue injuries by evaluating the expression levels of TNF-α, IL-1β, IL-6, iNOS, and β-EP in skin, muscle and serum samples, along with a meticulous examination of histopathological changes in skin and muscle tissues.
Materials and methods
Animals
Fifty male Sprague Dawley rats, 8 weeks old and weighing 280 ±10 g, were included in this study. The rats were housed under standard laboratory conditions, with a temperature maintained at 22 ±2°C and humidity ranging from 50% to 60%. A 12-h light/dark cycle was followed, and the rats were given unrestricted access to a standard diet (18% protein, 4% fat, 5% crude fiber, 7.6% amino acids, etc.) and water. All animal experiments followed the guidelines of the Ethics Committee for Animal Experiments of the Guizhou University of Traditional Chinese Medicine (Guiyang, China) (approval No. 20160003).
Chemicals, drugs, and instruments
High-dose Masanggoubang oil (Masanggoubang/tea oil: 1.54 g/mL, homemade),33 urethane (Sinopharm Chemical Reagent Co., Ltd., Beijing, China), tea oil (Guizhou Malinghe Plant Oil Co., Ltd., Xingyi, China), a scraping board (Suzhou Medical Products Co., Ltd., Suzhou, China), distilled water, formalin solution (Shenzhen Xigene Biotechnology Co., Ltd., Shenzhen, China), paraffin (Sinopharm Chemical Reagent Co., Ltd., Beijing, China), hematoxylin & eosin (H&E) staining kits, and 8% sodium sulfide hair remover (Guizhou Weiboxin Biotechnology Co., Ltd., Guiyang, China) were used in the experiments. Interleukin 1 beta, IL-6, TNF-α, iNOS, and β-EP enzyme-linked immunosorbent assay (ELISA) kits were purchased from Wuhan Huamei Biospes Co. (Wuhan, China). Scales; a homemade fixator, syringes, latex gloves, a centrifuge, a homogenizer, a slicer, an oven, and a computerized image acquisition and analysis system (Olympus DP70 (Olympus Corp., Tokyo, Japan) photographic system and Olympus BX51 microscope (Olympus Corp.) were also used in the experiments.
Modelling
Following a 1-week adaptation phase, a cohort of 50 rats was selected and divided into 5 different groups using the random number table method. Each group consisted of 10 rats and was designated as the control group, the model group, the GSMO group, the GSTO group, and the inunction with Masanggoubang oil (IMO) group. In the model group, the rats’ right hind thigh underwent initial hair removal, followed by mechanical modeling as per the methodology established by Kami et al.,35 which involves positioning the rats’ right hind limb in knee extension with the ankle maintained at 90° of dorsiflexion. Subsequently, trauma was induced by dropping a weight (335 g) on the gastrocnemius muscle from a height of 80 cm. The weight carried 1.57 J of kinetic energy upon striking the muscle. The impact area was approx. 1 cm2, resulting in discernible swelling following 3 consecutive strikes. While scattered bleeding spots were observed, no lesions formed, and the rats displayed a noticeable limp. The model showed a success rate of 100% based on anatomical and histological verification. In the absence of any intervention, the animals naturally progressed to develop a chronic soft tissue injury model after 2 weeks of normal feeding.
Treatment
The untreated control and model groups received no intervention. In the case of the remaining 3 groups, a designated individual immobilized the rats while a second person applied tea oil or Masanggoubang oil to the injured sites using a buffalo horn scraper. This procedure lasted 5 min, performed at an approx. rate of 70 repetitions per min until the appearance of sha on the skin. This treatment regimen was administered once every other day until sha disappeared, totaling 4 sessions. In the IMO group, 1 person immobilized the rats while another applied Masanggoubang oil to the injured soft tissue once every other day, amounting to a total of 4 applications.
Sample preparation
Tissue and serum cytokine sample preparation
Serum cytokine samples were prepared 48 h after the last treatment (until sha disappeared). To ensure consistency, all animals underwent a 12-h fasting period prior to anesthesia induction. For anesthesia, the rats were intraperitoneally (ip.) injected with 20% urethane at a dosage of 0.8 mL per 100 g of body weight. Subsequently, 5 mL of blood was collected from the abdominal aorta while the rats were under anesthesia. The collected samples were allowed to stand at room temperature for 1 h before being centrifuged at 3,500 rpm/min for 15 min. The supernatant was then collected and stored at –20°C for later use.
Muscle tissue samples were prepared 48 h after the last treatment (until sha disappeared). The rats were euthanized by cervical dislocation, and local muscle tissues were isolated. Blood was rinsed with pre-cooled normal saline at 4°C, and the samples were blotted dry with filter paper. The tissues were weighed and homogenized to a concentration of 10%. Subsequently, the homogenized samples were subjected to centrifugation at 5,000 rpm/min for 10 min at 4°C. The resulting supernatant was collected and stored at –20°C for later use.
Histopathological section sample preparation
Forty-eight hours after the last treatment, all the rats were euthanized by the cervical dislocation method after the sha had faded. The local muscle tissues were then separated, fixed and individually preserved in a formalin solution.
Sample testing
Tumor necrosis factor alpha, IL-1β, IL-6, iNOS, and β-EP levels in the skin, muscle and serum samples were determined with ELISA according to the kits’ instructions.36, 37
Paraffin-embedded tissue sections were prepared as follows. The muscle tissues were cut separately along the injury site. They were then rinsed in saline solutions, dehydrated, embedded in paraffin, and sectioned. Subsequently, the sections were stained with H&E to generate pathological sections for observing the pathological changes.
Statistical analyses
All data were analyzed using IBM SPSS Statistics for Windows v. 24 (IBM Corp., Armonk, USA). Due to the small number of observations in the group (n ≤ 10), the assumption of normality of distribution for parametric tests was not checked, and a nonparametric test was used. Results were expressed as median and quartiles. The Kruskal–Wallis test was used. The Mann–Whitney–Wilcoxon U test for rank sums for independent samples was employed for post hoc comparisons of each group with the control group, and a post hoc Dunn’s test was used for comparisons of the individual groups. A significance level of α = 0.05 was adopted. A Bonferroni correction of multiple comparisons with a significance level of α = 0.01 was applied to the Kruskal–Wallis test and Man–Whitney–Wilcoxon test for the rank sums of independent samples. No correction for multiple comparisons of significance level was applied to the post hoc Dunn’s test.
Results
All 50 rats were included in the analysis.
Histopathological findings
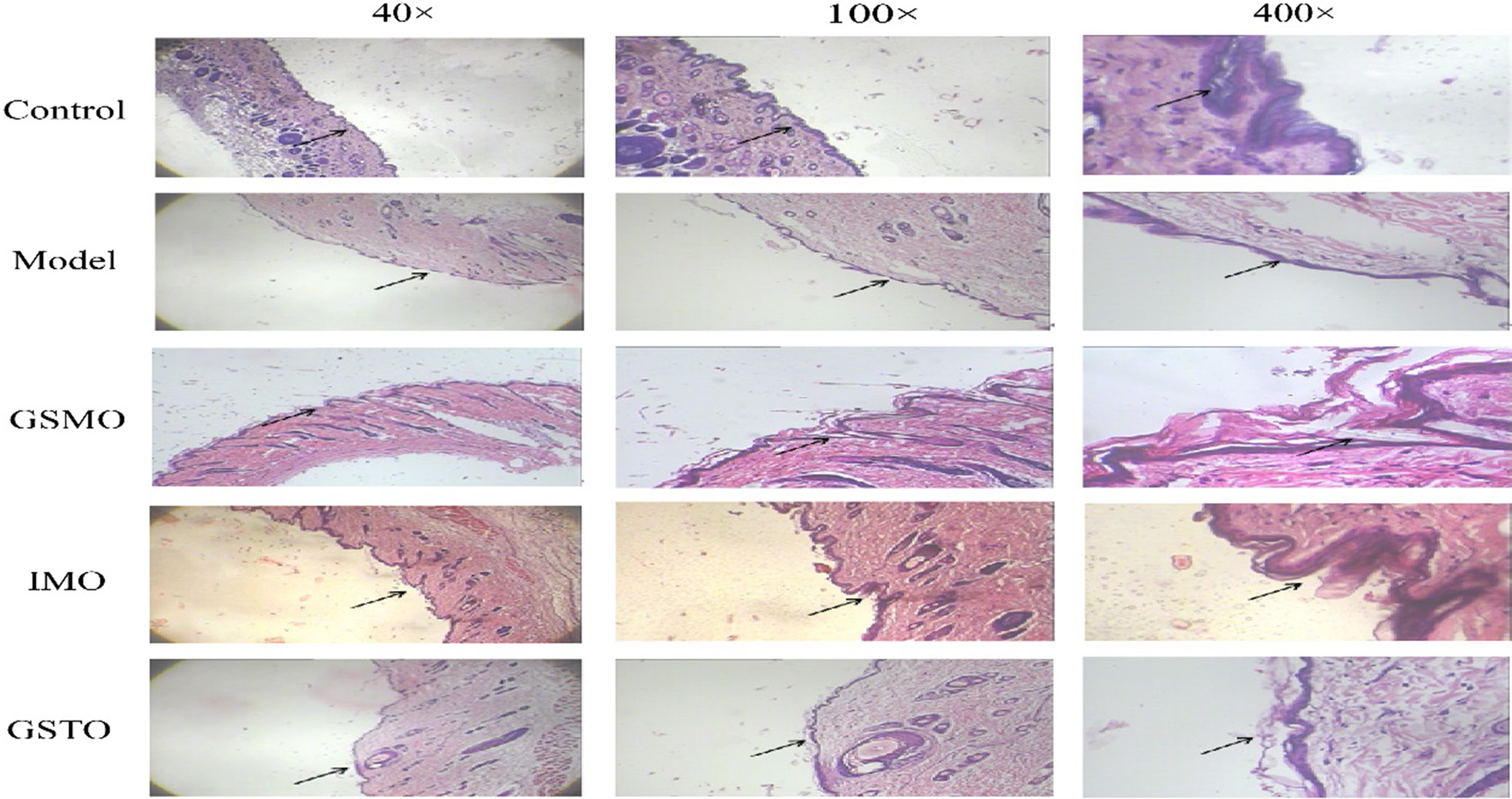
Histopathological findings in skin samples
The results of H&E staining showed that in the model group, the stratum corneum exhibited a strip-like and uniform distribution, with tightly arranged cells involved in the formation of the stratum corneum. In comparison to the model group, both the GSTO group and GSMO group showed a loosely arranged stratum corneum with increased intercellular spaces, indicating that the physical stimulation of gua sha enlarges the interstitial spaces in the skin tissue of the applied area (Figure 1).
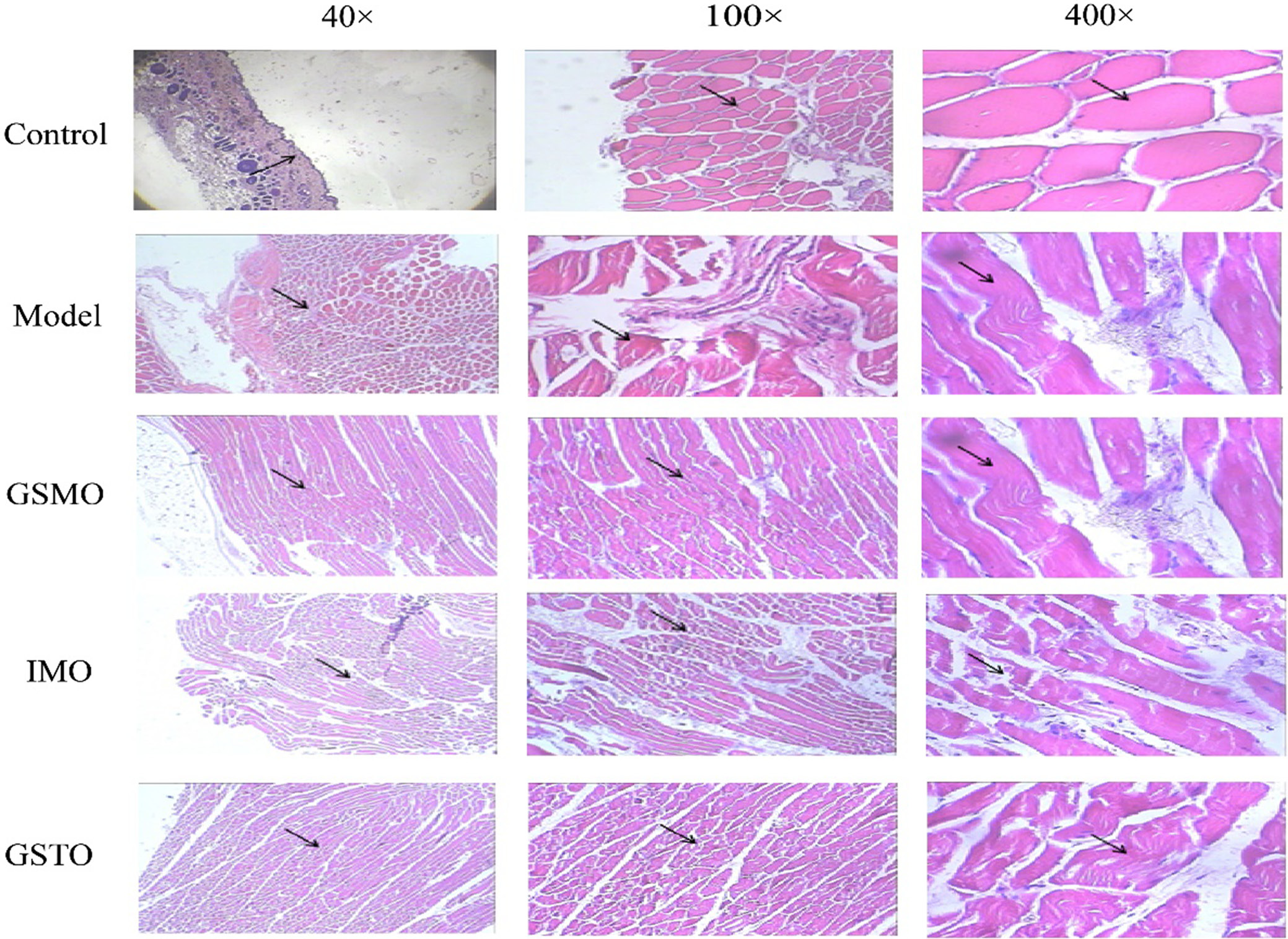
Histopathological findings of muscle samples
Hematoxylin and eosin staining also revealed that in the model group, the local muscle tissues in the injured area exhibited irregular cell arrangement, muscle fiber degeneration, necrosis, and dead muscle fibers, along with the proliferation of connective tissue. In comparison to the model, IMO and GSTO groups, the pathological changes in muscle tissues were less pronounced in the GSMO group, suggesting that GSMO significantly improves the degree of soft tissue damage (Figure 2).
Levels of TNF-α, IL-1β, IL-6, iNOS,
and β-EP in skin tissue
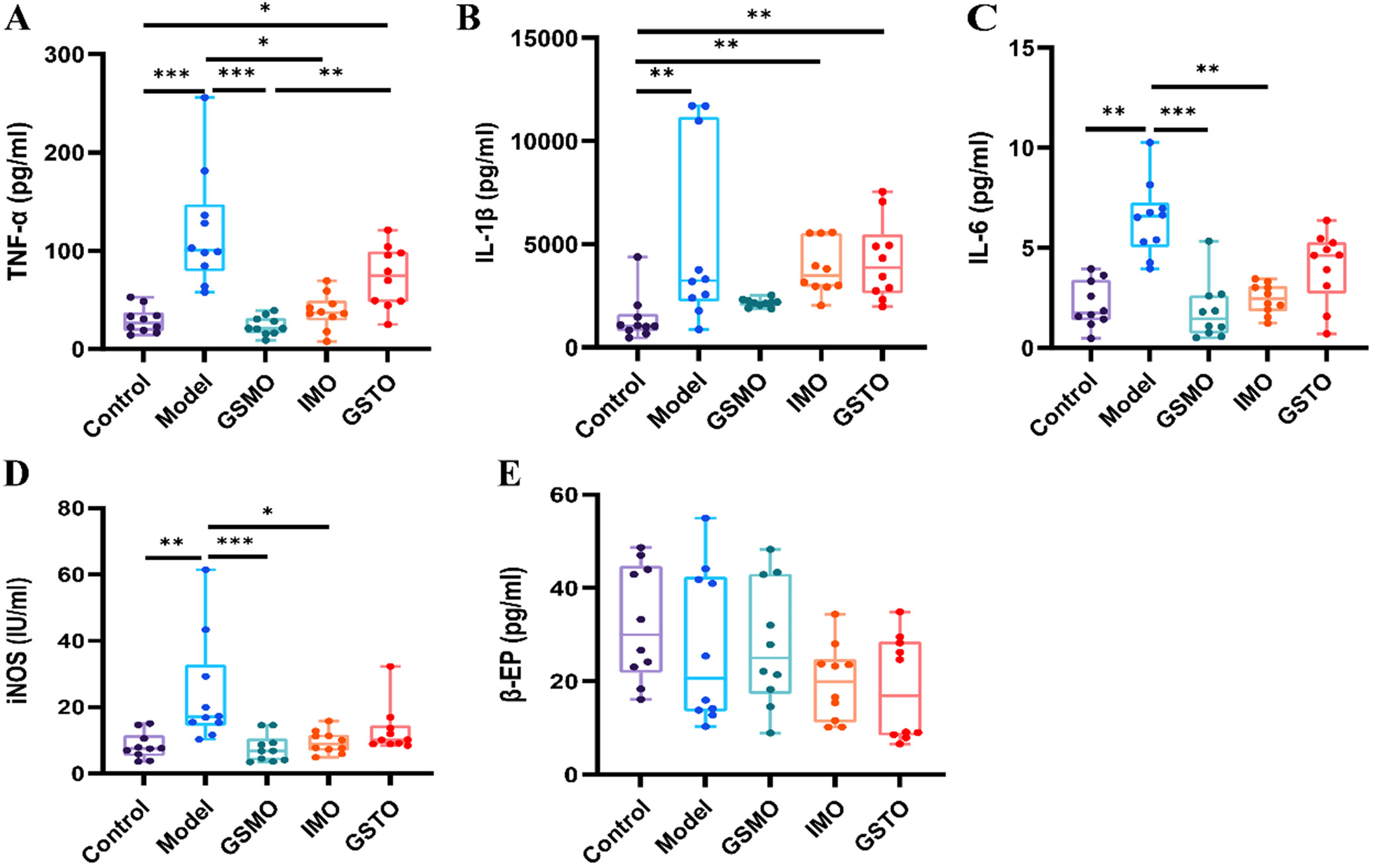
The ELISA results indicated a noteworthy elevation in skin levels of TNF-α (post hoc Dunn’s test: p < 0.001), IL-1β (post hoc Dunn’s test: p = 0.008), IL-6 (post hoc Dunn’s test: p = 0.001), and iNOS (post hoc Dunn’s test: p = 0.002) in the model group compared to the control group, but no statistically significant difference was observed in the level of β-EP.
After treatment, a notable reduction in the levels of TNF-α (post hoc Dunn’s test: p < 0.001, p = 0.012, respectively), IL-6 (post hoc Dunn’s test: p < 0.001, p = 0.005, respectively) and iNOS (post hoc Dunn’s test: p < 0.001, p = 0.024, respectively) were observed in the skin tissues of the GSMO and IMO groups compared to the model group. Conversely, no significant disparity was evident in the GSTO group. Additionally, the levels of TNF-α (post hoc Dunn’s test: p = 0.004) significantly decreased in the skin tissues of the GSMO group in comparison to the GSTO group, while no significant variance was noted in the IMO group. Furthermore, these cytokine levels displayed no discernible differences between the GSMO and IMO groups. Moreover, in comparison to the control group, the GSMO group exhibited no significant differences in skin levels of TNF-α, IL-1β, IL-6, and iNOS. Similarly, the IMO group showed no significant differences in skin levels of TNF-α, IL-6 and iNOS when compared to the control group. Likewise, the GSTO group exhibited no significant differences in skin levels of IL-6 and iNOS in comparison to the control group (Table 1, Figure 3).
Levels of TNF-α, IL-1β, IL-6, iNOS,
and β-EP in muscle tissue
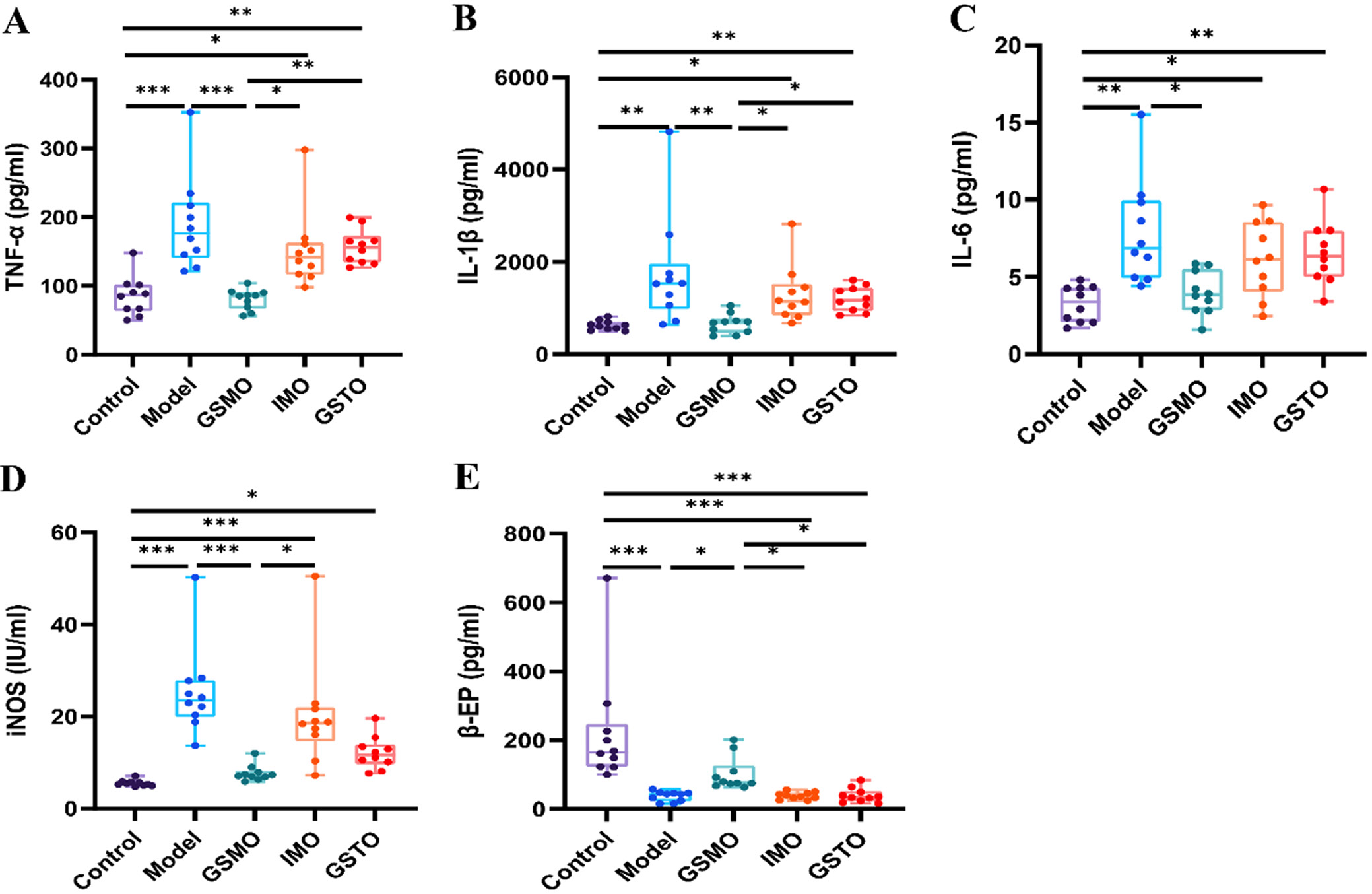
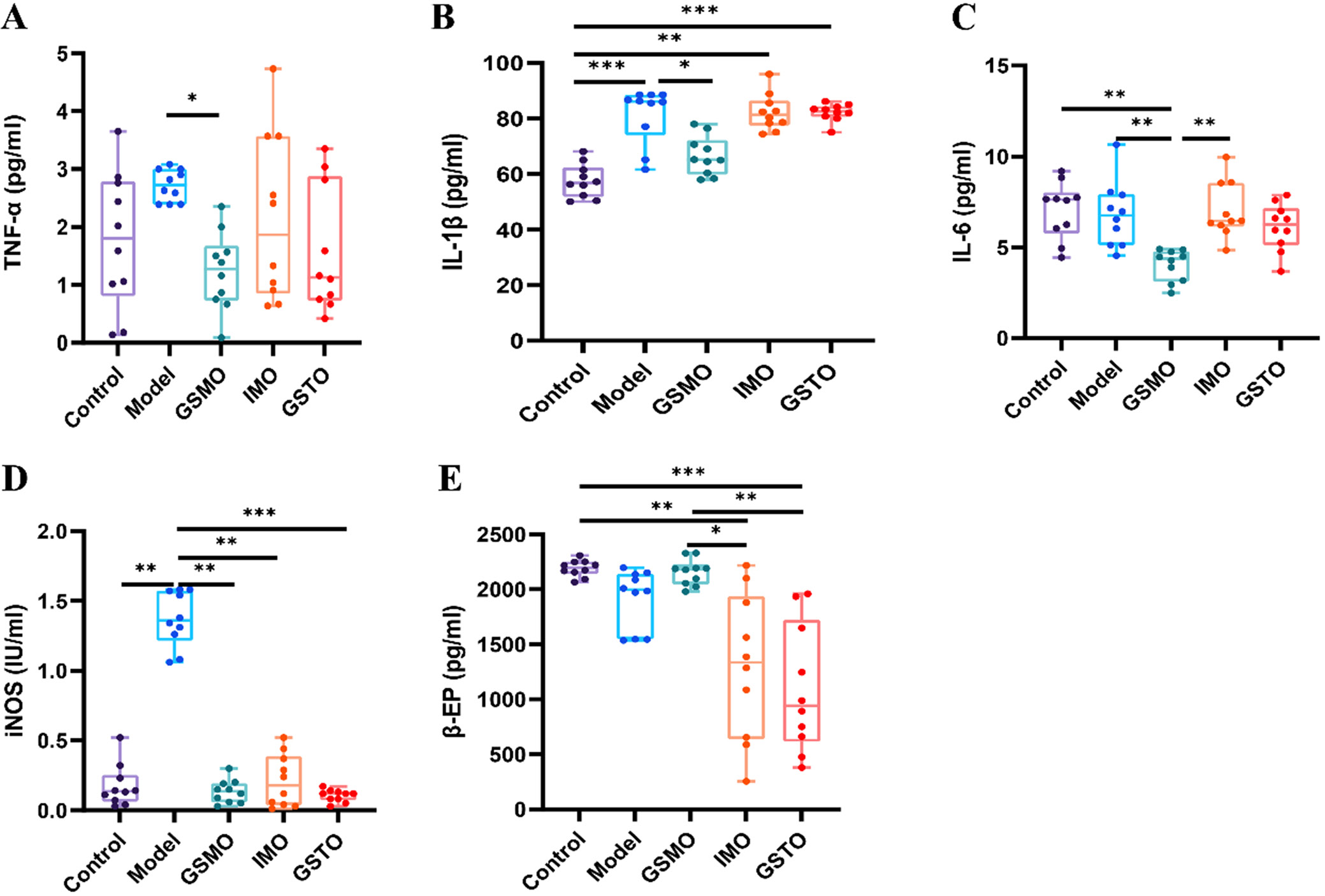
The ELISA results revealed markedly higher levels of TNF-α (post hoc Dunn’s test: p < 0.001), IL-1β (post hoc Dunn’s test: p = 0.001), IL-6 (post hoc Dunn’s test: p = 0.001), and iNOS (post hoc Dunn’s test: p < 0.001) in the muscle tissues of the model group compared to the control group. Conversely, β-EP (post hoc Dunn’s test: p < 0.001) exhibited a significant decrease.
Following the intervention, the GSMO group demonstrated a notable reduction in muscle levels of TNF-α (post hoc Dunn’s test: p < 0.001), IL-1β (post hoc Dunn’s test: p = 0.003), IL-6 (post hoc Dunn’s test: p = 0.013), and iNOS (post hoc Dunn’s test: p < 0.001) compared to the model group. Additionally, a significant increase in β-EP (post hoc Dunn’s test: p = 0.014) was observed. Conversely, the IMO group and GSTO groups did not exhibit significant differences in muscle levels of TNF-α, IL-1β, IL-6, iNOS, and β-EP. Furthermore, comparing the GSMO group to the GSTO group, significant differences were observed in TNF-α (post hoc Dunn’s test: p = 0.002), IL-1β (post hoc Dunn’s test: p = 0.018) and β-EP (post hoc Dunn’s test: p = 0.018) levels. Similarly, the GSMO group revealed significant variances in TNF-α (post hoc Dunn’s test: p = 0.017), IL-1β (post hoc Dunn’s test: p = 0.034), iNOS (post hoc Dunn’s test: p = 0.040), and β-EP (post hoc Dunn’s test: p = 0.019) levels compared to the IMO group. Nevertheless, no significant differences were found in these cytokine levels between the GSTO and IMO groups. Furthermore, the GSMO group exhibited no significant differences in these cytokine levels compared to the control group (Table 2, Figure 4A–E)
Levels of TNF-α, IL-1β, IL-6, iNOS, and β-EP in serum
The ELISA results unveiled notably elevated levels of IL-1β (post hoc Dunn’s test: p < 0.001) and iNOS (post hoc Dunn’s test: p = 0.003) in the serum of the model group compared to the control group.
After treatment, in comparison to the model group, the GSMO group demonstrated a significant decrease in serum levels of TNF-α (post hoc Dunn’s test: p = 0.019), IL-1β (post hoc Dunn’s test: p = 0.015), IL-6 (post hoc Dunn’s test: p = 0.004), and iNOS (post hoc Dunn’s test: p = 0.001). Additionally, both the IMO and GSTO groups exhibited a significant reduction in serum levels of iNOS (post hoc Dunn’s test: p = 0.003, p < 0.001, respectively) when compared to the model group. When compared with the GSTO group, the GSMO group demonstrated an increased level of β-EP (post hoc Dunn’s test: p = 0.001). However, no significant differences were observed in these cytokine levels between the GSTO and IMO groups. Notably, the GSMO group displayed remarkable differences in IL-6 (post hoc Dunn’s test: p = 0.002) and β-EP (post hoc Dunn’s test: p = 0.015) levels compared to the IMO group. Furthermore, compared to the control group, a notable reduction in the levels of IL-6 (post hoc Dunn’s test: p = 0.001) was observed in the serum of the GSMO group, while no significant differences were observed in TNF-α, IL-1β, iNOS, and β-EP levels in the GSMO group when compared to the control group. Similarly, no significant differences in the levels of TNF-α, IL-6 and iNOS were observed in the serum of the IMO and GSTO groups compared to the control group (Table 3, Figure 5A–E).
Discussion
Chronic soft tissue injuries refer to a traumatic syndrome in which soft tissues or skeletal muscles are subjected to a direct or indirect force or prolonged chronic strain, characterized by aseptic inflammation and resulting in soft tissue spasms and scarring.2 Gua sha with Masanggoubang oil therapy is known as one of the distinctive external therapies in the traditional Miao medicine for its anti-inflammatory and analgesic effects.38 Its efficacy and safety in the treatment of chronic soft tissue injuries have been extensively confirmed.23, 32, 34 However, its underlying mechanism of action remains unclear. Therefore, this study explores the therapeutic effects of GSMO on rats with chronic soft tissue injuries and identifies its potential mechanisms of action.
This study found that GSMO can alleviate soft tissue necrosis, degeneration and scar tissue proliferation at the injury site, mitigate inflammatory responses, downregulate TNF-α, IL-6 and iNOS levels in the skin tissues, as well as TNF-α, IL-1β, IL-6, and iNOS levels in muscle and serum, and upregulate β-EP levels in the muscles of rats with chronic soft tissue injuries.
Chronic soft tissue injuries primarily manifest as inflammatory responses and pain. In recent years, there has been increasing attention on the role of M1 macrophage polarization in inflammatory diseases.5, 6 M1 macrophages promote inflammation by densely releasing pro-inflammatory factors such as IL-6, IL-1β and TNF-α. High expression of iNOS catalyzes the production of the neurotransmitter NO, thereby increasing the sensitivity of the peripheral and central nervous systems and inducing pain hypersensitivity.7, 8 Among these, TNF-α is an important mediator in the inflammatory response, inducing a cascade of IL-6 and IL-1β release, thereby increasing inflammation.9 Simultaneously, a decrease in TNF-α is closely associated with the improvement of limb disabilities.20 Previous studies have found macrophage polarization to play a crucial role in suppressing soft tissue inflammation and promoting tissue repair and regeneration.39, 40 Yang et al.12 discovered that gua sha therapy improves pain perception and the inflammatory response in chronic soft tissue injuries by downregulating the expression of TNF-α, IL-1β and IL-6 in the serum, which is consistent with our findings. Bai found that electroacupuncture alleviates inflammation in rats with myofascial injuries by downregulating iNOS levels.41 We observed that the levels of serum and muscle iNOS were downregulated in the GSMO group, which is consistent with the literature. Beta-EP is an important endogenous opioid neuropeptide that exerts powerful analgesic effects by inhibiting pain transmission pathways.42 Peripheral and central opioid receptors participate in the analgesic effects during the early stages of inflammation.43 He and Zhou13 found that tuina (a hands-on body treatment that uses Chinese Daoist principles to bring the 8 principles of TCM into balance) massage alleviates pain symptoms by upregulating serum β-EP levels in patients with lumbar disc herniations. Our research shows that the GSMO group exhibited upregulated muscle β-EP levels, which may promote blood circulation in the body to facilitate the exchange of β-EP from serum to tissues and enhance local analgesic effects.44 This finding is consistent with the literature.
Similarly, this study found that IMO and GSTO therapies also improved the inflammatory response to varying degrees in rats with chronic soft tissue injuries. Our experimental results revealed decreased TNF-α, IL-6 and iNOS levels in the skin tissues at the injury site, as well as serum iNOS levels in the IMO group. In the GSTO group, the serum iNOS levels also decreased. The observed improvements in the IMO group can be attributed to the active ingredients in Masanggoubang oil penetrating the dermis of the damaged area and exerting anti-inflammatory effects to some extent.38 Gua sha with tea oil partially alleviated the inflammatory response in the injured soft tissues by improving local blood circulation and facilitating the metabolism of inflammatory factors.44
Furthermore, this study found that GSMO, IMO and GSTO therapies all improved the inflammatory response in rats with chronic soft tissue injuries to varying degrees. Among them, GSMO therapy was superior to IMO and GSTO therapies alone, revealing a synergistic effect of combining gua sha therapy and Masanggoubang oil. The scraping medium was identified as an important factor influencing the efficacy of gua sha therapy. Compared to IMO, GSMO significantly improved muscle TNF-α, IL-1β, iNOS, and β-EP levels. This may be attributed to gua sha therapy’s ability to expand the interstitial space, facilitating the deep absorption of active ingredients from Masanggoubang oil into the muscle layer. Compared to GSTO, GSMO significantly improved skin TNF-α levels, muscle TNF-α, IL-1β and β-EP levels, as well as serum β-EP levels, possibly due to the combined effect of gua sha therapy and Masanggoubang oil, which may enhance the anti-inflammatory action and significantly improve soft tissue inflammation.
Limitations
We found that GSMO can upregulate muscle β-EP levels in rats with chronic soft tissue injuries. Therefore, we speculate that GSMO may also alleviate pain in rats with chronic soft tissue injuries. However, we only tested 1 pain factor, so further evaluation of its analgesic effectiveness should include pain threshold measurement scores and other serum pain substances such as substance P and neuropeptide Y.45, 46 Additionally, the underlying mechanisms of GSMO for chronic soft tissue injuries remain unclear. It would be beneficial to explore the relevant signaling pathways of scraping therapy using gene chip technology and further clarify its mechanism of action by combining immunological techniques and proteomics. Moreover, future studies should consider extending the observation period to investigate the long-term effects of GSMO. Furthermore, conducting comparative efficacy studies between GSMO and prescription drugs would provide a comprehensive evaluation of its therapeutic effects on chronic soft tissue injuries.
Conclusions
Our results indicate that GSMO can alleviate the inflammatory response in rats with chronic soft tissue injuries, which may be related to the inhibition of M1 macrophage polarization. Additionally, we found that the combination of gua sha therapy and Masanggoubang oil has a synergistic effect in treating chronic soft tissue injuries. Therefore, this study may provide valuable insights into the clinical treatment of chronic soft tissue injuries. In summary, GSMO demonstrates a favorable therapeutic effect of chronic soft tissue injuries, significantly improving the inflammatory response in rats with chronic soft tissue injuries. The mechanism behind this improvement may be associated with the reduction of M1 macrophage polarization in the peripheral blood and local tissues. Gua sha with Masanggoubang oil shows promise as an effective method for the treatment of chronic soft tissue injuries, and our research provides scientific evidence supporting its use in this context.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.10889055. The package includes the following files:
Supplementary Table 1. Comparative analysis of TNF-α, IL-1β, IL-6, iNOS, and β-EP skin levels compared control group to the other 4 groups.
Supplementary Table 2. Comparative analysis of TNF-α, IL-1β, IL-6, iNOS, and β-EP muscle levels compared control group to the other 4 groups.
Supplementary Table 3. Comparative analysis of TNF-α, IL-1β, IL-6, iNOS, and β-EP serum levels compared control group to the other 4 groups.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.