Abstract
Background. Coronavirus disease 19 (COVID-19) is a viral infection mediated by coronavirus-2 that causes severe acute respiratory syndrome (SARS-CoV-2). The disease may affect biochemical parameters and electrolytes. C-terminal cross-linking telopeptide (CTX-I) is released during mature bone resorption and is a biomarker for predicting bone resorption.
Objectives. As the pandemic progressed, understanding the effects of COVID-19 disease remained critical. Inflammatory responses triggered by the virus can result in a bone metabolism regulation imbalance. As such, this study aimed to analyze serum levels of CTX-I, calcium (CA), phosphorus (P), magnesium (Mg), C-reactive protein (CRP), and alkaline phosphatase (ALP) in COVID-19 patients to investigate the relationship between bone resorption and the disease.
Materials and methods. The study included 56 individuals with COVID-19 (divided into mild, moderate and severe subgroups depending on disease severity) and 25 healthy adults as a control group. Serum CTX-I concentrations were measured with enzyme-linked immunosorbent assay (ELISA). In addition, CRP, Ca, Mg, P, and ALP levels were measured using an automated clinical chemistry analyzer.
Results. Serum CTX-I levels were significantly higher in COVID-19 patients than in the control group (p < 0.05). Furthermore, a positive weak relationship was detected between CRP and CTX-I (r = 0.303, p < 0.05).
Conclusions. Increased serum CTX-I levels in the patient group caused COVID-19-driven bone degradation, though serum CTX-I levels did not differ according to disease severity.
Key words: bone, COVID-19, CTX-I
Background
Coronavirus disease 19 (COVID-19), caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection, was first detected in December 2019 in Wuhan, China, and spread rapidly over almost the entire globe.1, 2 Coronaviridae is a family of single-stranded ribonucleic acid (RNA) viruses, including SARS-CoV-2, that affect many animals, while other coronaviruses could infect humans.3 Subjective clinical findings are considered more reliable with the concomitant biomarkers, which provide measurable data during the progression of biological processes.4 With the rapid spread of COVID-19 and increased mortality in extreme cases, a better understanding of clinical features, determining its effects on metabolism and finding accurate laboratory markers are required to elucidate the disease process. Indeed, its influence on metabolism is not fully known, and there is a need for studies on the effects of COVID-19 on various human organs and tissues, such as muscle and bone.
The bone organic matrix comprises around 90–98% collagen I, which is proteolytically degraded by osteoclasts during bone turnover, releasing small C-terminal cross-linking telopeptide (CTX-I) peptide fragments.5, 6, 7 Serum CTX-I levels correlate with histomorphometric measurements of bone resorption.8 Although different biomarkers are available for measuring the metabolic products of bone resorption, serum CTX-I stands out for its high correlation with bone turnover rate and its immediate response to changes in bone metabolism, making it a sensitive marker of bone resorption.5, 9
Objectives
Studies show that inflammation and bone metabolism are associated with various biological and clinical mechanisms and suggest measuring bone metabolism markers, such as CTX-I, in COVID-19 patients.10 To investigate the effect of COVID-19 on bone, the current study examined the impact of COVID-19 on serum levels of the bone resorption marker CTX-I.
Materials and methods
Patients
This prospective observational research, conducted in June and July 2020, included 56 patients diagnosed with COVID-19 and 25 individuals with no health problems. The Clinical Research Ethics Committee of Ataturk University (Erzurum, Turkey) approved this research prior to experiments (approval No. B.30.2.ATA 0.01.00/369). All participants provided written informed consent, and the study conformed to the principles outlined in the Declaration of Helsinki.
Participants provided nasopharyngeal and oropharyngeal samples for real-time polymerase chain reaction (PCR) COVID-19 detection, with those testing positive stratified to the COVID-19 group and those testing negative to the control group. Those diagnosed with COVID-19 (n = 56) were divided into mild, moderate and severe subgroups based on disease severity, with 25 healthy individuals used as a control group. Power analysis was performed before determining sample size. Subgroups included a non-complicated group (NCG), with mild fever and cough present/no and no radiological pulmonary findings, a moderate group (MG) with fever-cough, radiological uni/bilateral lung involvement and oxygen saturation SpO2 > 93%, and a severe group (SG) displaying lung radiological imaging with uni/bilateral involvement and SpO2 < 93%. Exclusion criteria encompassed being aged less than 18 years, the presence of diseases affecting bone metabolism, undergoing chemotherapy or immunosuppressive drug treatment, and using calcium, magnesium or calcitriol supplements. The MG and SG included some patients with hypertension, diabetes mellitus and chronic obstructive pulmonary disease (COPD).
Blood sampling
Participants provided fasting blood on the first day of hospital admission, with venous blood drawn using a 10 mL syringe and transferred to gel-containing vacutainers. After 30 min, samples were centrifuged at 1,500 × g for 10 min, and serum was stored at –80°C until the day of measurement.
Measurement and data collection
Serum CTX-I concentrations were analyzed using a human-specific CTX-I sandwich enzyme-linked immunosorbent assay (ELISA) (cat. No. CSB-E11224h; Cusabio Technology, Wuhan, USA). The kit detection range was 0.625–40 ng/mL, with an intra-assay coefficient of variance (CV%) <8%, inter-assay precision CV% of <10% and sensitivity of 0.156 ng/mL. The ELISA measurements used serum samples (100 µL) according to the manufacturer’s instructions, with samples analyzed using an XS PowerWave multi-plate spectrophotometer (Agilent Technologies, Santa Clara, USA). Other biochemical parameters were measured using compatible kits and detected on a cobas 8000 modular analyzer (Roche Diagnostics, Basel, Switzerland), with C-reactive protein (CRP) immunoturbidimetrically detected in the e801 module, and calcium (Ca), magnesium (Mg), phosphorus (P), and alkaline phosphatase (ALP) in the c502 module. These results were obtained from existing patient files. Analysis was carried out at the Faculty of Medicine of Ataturk University.
Statistical analyses
Data analysis employed IBM SPSS 20.0 (IBM Corp., Armonk, USA). Shapiro–Wilk tests evaluated numerical data distribution, and F test/Brown–Forsythe tests confirmed the homogeneity of variances. Normally distributed data were calculated as mean ± standard deviation (SD). Student’s t-test was used to compare 2 groups and analysis of variance (ANOVA) 3 groups. Data with a non-normal distribution were calculated as median and interquartile range (Me (IQR)), with Mann–Whitney U tests used for paired group comparisons and Kruskal–Wallis post hoc with Dunn’s test used for 3 group comparisons. Spearman’s correlation coefficient was employed to evaluate the monotonic component in the relationship between the data. A p < 0.05 was considered significant, and p < 0.001 very significant. Supplementary tables (https://doi.org/10.5281/zenodo.10719329) provide confirmation of parametric test assumptions and related data for the control group, COVID-19 patient group and COVID-19 subgroups (NCG, MG and SG).
Results
Table 1 shows the clinical and demographic characteristics of COVID-19 patients. While 30 of the 56 COVID-19 patients were female and 26 were male, the control group contained 11 women and 14 men. There was no statistical difference between group mean age (p > 0.05).
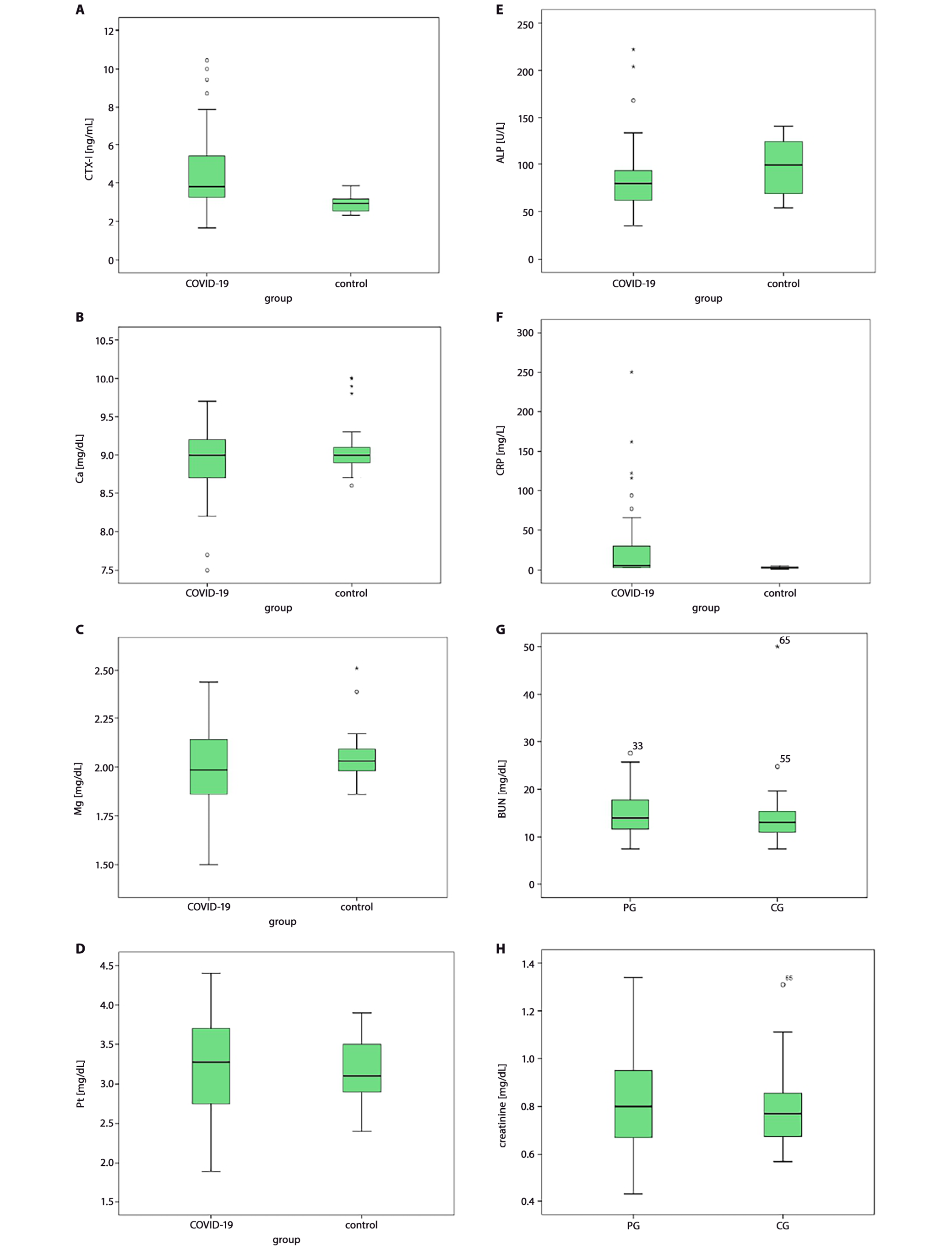
Serum CTX-I levels were higher in COVID-19 patients than in the control group (p < 0.001), as were CRP levels (p < 0.001). On the other hand, serum Ca, Mg, P, ALP, creatinine, and blood urea nitrogen (BUN) levels were similar in both groups (p > 0.05). Table 2, Table 3 show the biochemical tests measured in both groups, while Figure 1A–H provides box-whisker graphics of the group comparison.
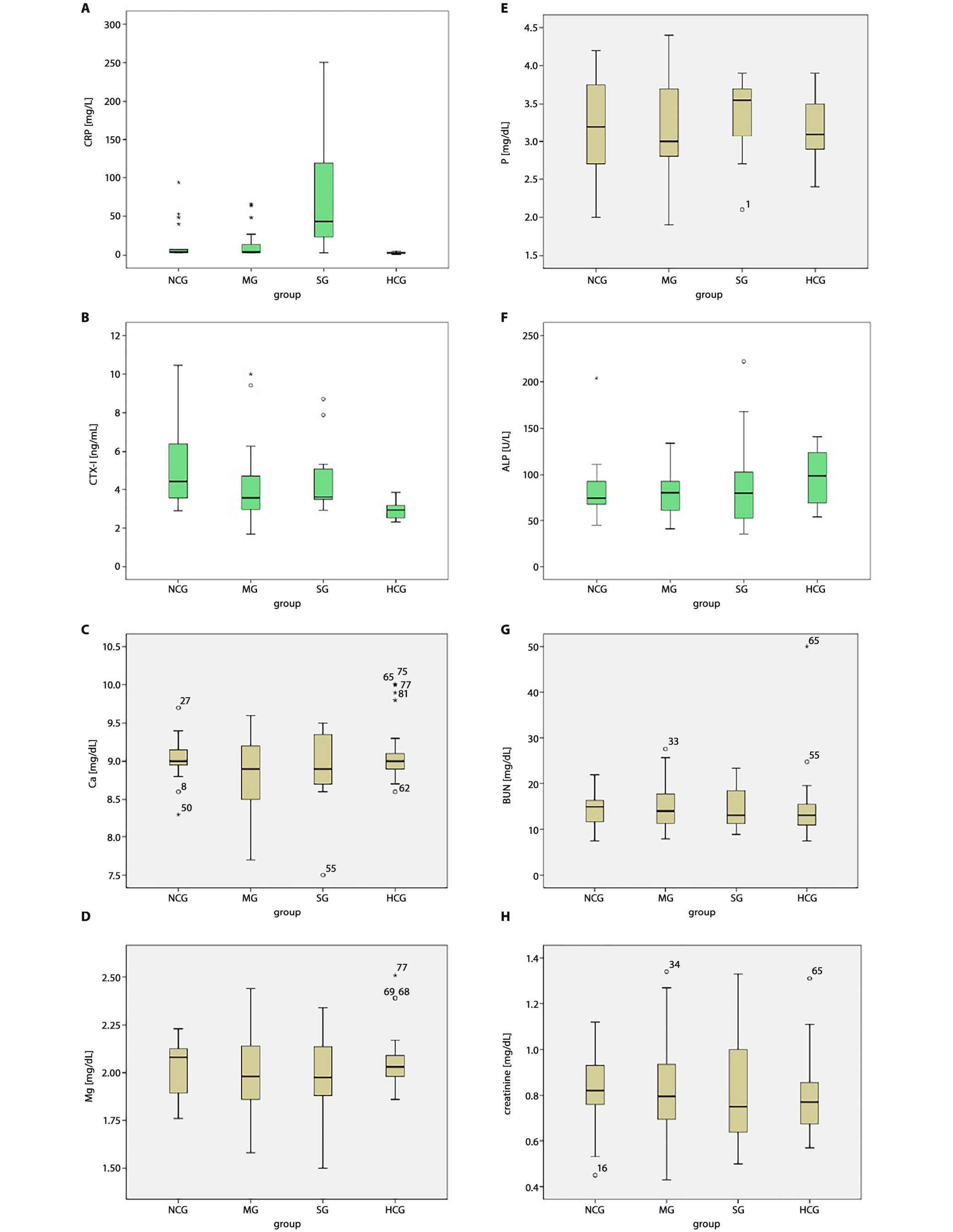
When the COVID-19 subgroups were compared and evaluated, the highest serum CTX-I levels were found in the NCG, but this difference was not significant (p > 0.05). While CRP was significantly higher in the SG compared to the other groups (p < 0.05), no difference was observed between other parameters (p > 0.05). Table 4, Table 5 show the COVID-19 subgroup’s serum biochemical tests, and Figure 2A–H presents box-whisker graphics of the group comparisons.
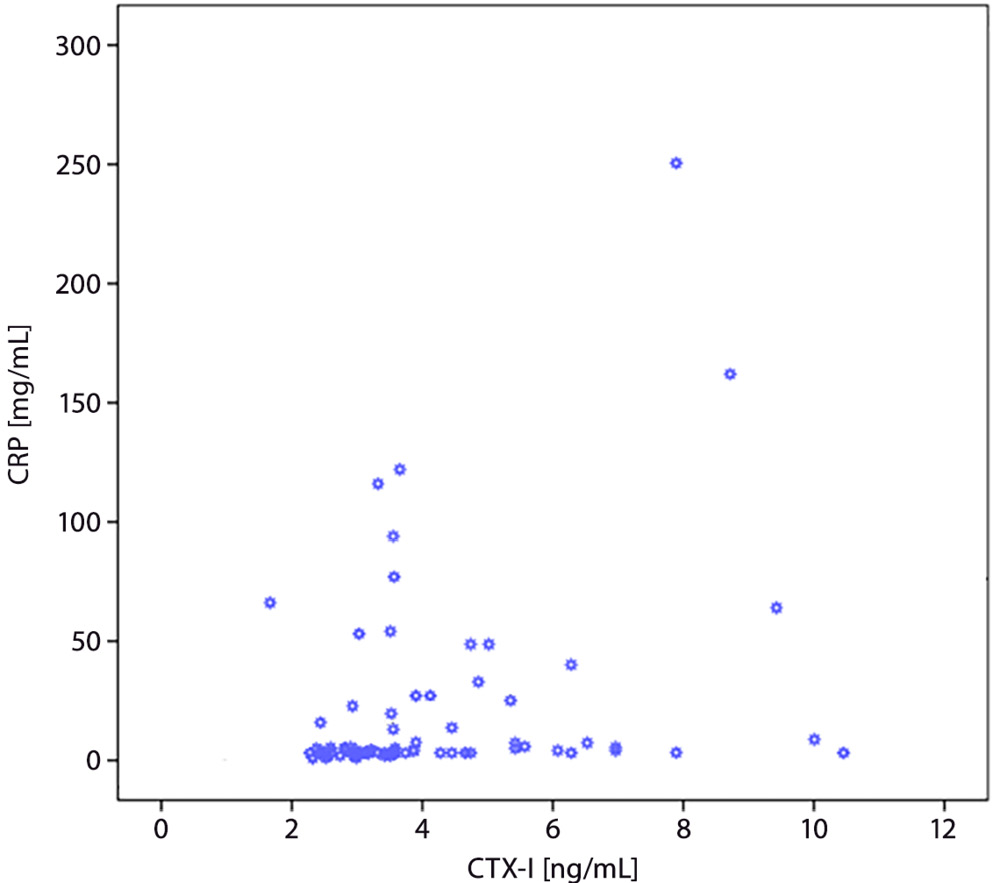
Spearman’s correlation analysis indicated a positive weak monotonic correlation between CTX-I and CRP in all participants (r = 0.303, p < 0.05). Table 6 shows the analysis of the monotonic component of the relationship between CTX-I and other tests, and Figure 3 shows the relationship between CTX-I and CRP
Discussion
To the best of our knowledge, this is the first study reporting on serum CTX-I levels in patients with COVID-19. The findings demonstrated that serum CTX-I levels were statistically higher in the COVID-19 patient group than in the healthy controls. However, there were no differences between COVID-19 patients divided into subgroups according to disease severity (mild, moderate and severe).
Many studies on diverse viruses have sought to characterize their roles in bone metabolism,10 with those on bone metabolism in SARS-CoV-2 indicating that much more research is required on the risk of fracture in patients recovering from SARS-CoV-2 infection.10 Although not all features of COVID-19 are fully understood, pathological studies on the lungs, liver and heart have shown the potential negative effects of COVID-19 on certain tissues.11 In this regard, bone-forming capacity is prone to worsen due to the progression of underlying diseases resulting from SARS-CoV-2 infection.12
Serum CTX-I levels increase following proteolysis of type I collagen, the main component of the bone extracellular organic matrix, during mature bone resorption. Therefore, CTX-I levels decrease in patients with suppressed bone resorption.9, 13 Most research on viruses and CTX-I shows that virus infection causes increased CTX-I levels. Huang et al.14 showed that the dengue virus caused a temporary inflammatory reaction in bone tissue and increased osteolytic activity to release CTX-I. Research on human immunodeficiency virus (HIV) and hepatitis C virus (HCV) infections showed that HIV was related to increased bone resorption and formation, and increases in CTX-I, whereas HCV infection was not associated with CTX-I.15 Increased bone resorption has been shown in the pathogenesis of bone loss, which was evaluated by measuring CTX-I in hemophiliacs and correlations with HIV infection.16
A study on biochemical markers of bone metabolism in SARS conducted in Hong Kong showed significant bone resorption, as indicated by a significant increase in serum CTX-I from certain days after fever onset.17 Patients with chronic hepatitis C (CHC) and osteoporosis or osteopenia had higher serum CTX-I levels, which may mean that bone loss in the early stages of CHC is not due to reduced bone synthesis but secondary to increased bone resorption.18 Furthermore, animal studies on SARS-CoV-2 demonstrate a significant reduction in several bone parameters.19
Tao et al. hypothesized that angiotensin-converting enzyme 2 (ACE2) deficiency caused by SARS-CoV2 invasion may result in bone matrix reduction and that COVID-19 can cause anaphase bone loss and earlier muscle disorder by targeting ACE2 in the bone marrow microenvironment.12 The current study supports this hypothesis by showing higher CTX-I levels in patients with COVID-19 than in the healthy controls.
Magnesium has many biological functions, especially in bone, with 60% of the total body content found in bone.20 A recent meta-analysis showed an association between unstable Mg homeostasis and COVID-19, and suggested incorporating Mg into the ionogram and supplementing it externally.21 In parallel, a lower Mg level was observed in the COVID-19 group in our study, but this decrease was not significant. In addition, moderate and severe patients had lower Mg than non-complicated patients, though this difference was not significant. A retrospective study by Pal et al.22 found widespread hypocalcemia in mildly severe COVID-19 patients and attributed it to the disease, though they did not clarify its pathophysiological mechanism.
In a study of 420 COVID-19 patients and 165 healthy controls, serum Ca was lower in the COVID-19 patients. Although the current study found a slight discrepancy in Ca levels, it was not considered significant.
Alkaline phosphatase is a bone-building enzyme with several isoforms. Although studies on bone-specific ALP (B-ALP) in COVID-19 are limited to only a few case reports,23, 24 a study showing elevated total ALP in COVID-19 patients suggested it was primarily due to liver damage. In the current study, serum ALP values were similar in both groups and although it increased with COVID-19 severity, this difference was not significant.
Since CTX-I excretion takes place in the kidneys, the CTX-I level may be high in the plasma due to impaired renal function. However, kidney function tests of serum creatinine and BUN showed no differences between the healthy group and COVID-19 patients, indicating that the high CTX-I level was of COVID-19 origin. The CRP serum level, which is a positive acute phase reactant, increases during infection,25 with many reports showing increased CRP in COVID-19 patients.26, 27 Our findings showed an increase in CRP levels in COVID-19 patients, which is compatible with current research results. In addition, a positive weak relationship was found between CTX-I and CRP.
Limitations
Study limitations include the absence of B-ALP and radiological images to support bone resorption. While this study includes critical clinical laboratory parameter results, the small sample size could be a limitation, particularly within the COVID-19 subgroups. Another limitation is the lack of parathyroid hormone (PTH) and calcitriol measurement since they play a role in bone metabolism.
Conclusions
The study showed that COVID-19 patients had increased serum CTX-I levels, a reference marker for bone resorption, though there were no differences based on disease severity. Our data may be foundational to clinical studies investigating bone metabolism in COVID-19.
Supplementary data
The Supplementary materials are available at https://doi.
org/10.5281/zenodo.10719329. The package includes the following files:
Supplementary Table 1. Verification of parametric test assumption for control.
Supplementary Table 2. Verification of parametric test assumption for COVID-19 patient group.
Supplementary Table 3. Verification of parametric test assumption for NCG COVID-19 subgroup.
Supplementary Table 4. Verification of parametric test assumption for MG COVID-19 subgroup.
Supplementary Table 5. Verification of parametric test assumption for SG COVID-19 subgroup.
Supplementary Table 6. Tests of homogenity.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.