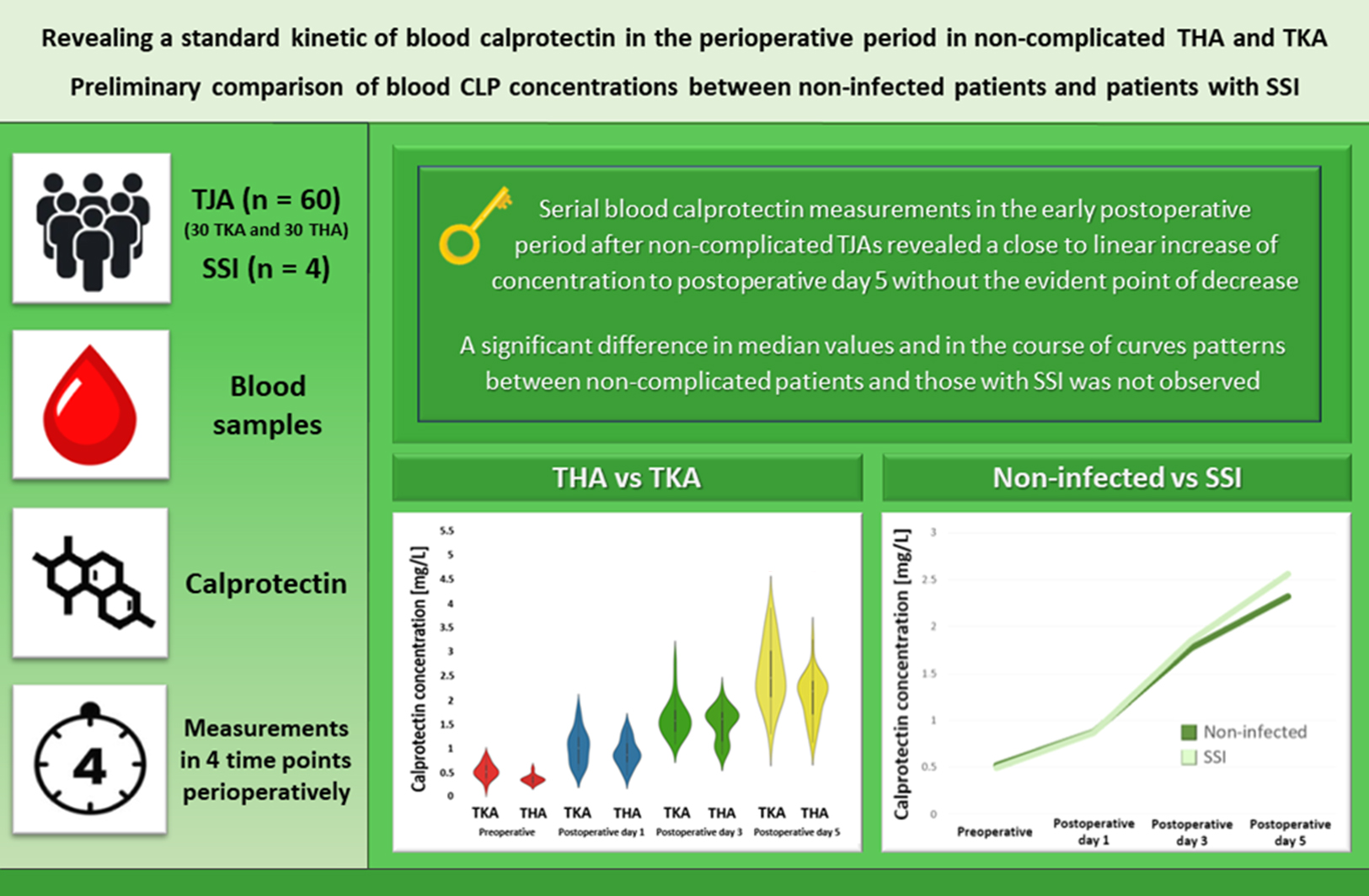
Abstract
Background. Recent studies have revealed the usefulness of synovial calprotectin (CLP) in diagnosing chronic periprosthetic joint infections (PJIs). However, there is still a lack of evidence to support the use of serum CLP in the diagnosis of early PJIs and surgical site infections (SSIs) after total joint arthroplasties (TJAs).
Objectives. The primary aim of this study is to investigate the standard kinetics of CLP concentrations in the blood during the very early postoperative period after non-complicated total hip arthroplasty (THA) and total knee arthroplasty (TKA). The secondary aim was to perform a preliminary comparison of CLP concentrations between non-infected patients and patients with recognized SSIs.
Materials and methods. A total of 64 consecutive patients who underwent primary THA and TKA were included in this prospective research. Sixty patients (30 THA and 30 TKA) were scheduled to determine the standard shape of the blood CLP curve and the expected concentrations during the first 5 postoperative days after non-complicated TJAs. In 4 additonal patients, early SSI was confirmed, and they were included in a separate SSI subgroup.
Results. Calprotectin demonstrated a linear increase during the first 5 postoperative days. Statistically significant differences in CLP concentrations between non-infected cases and SSIs were not observed. The preoperative median results with interquartile range (Q1–Q3) were 0.52 (0.39–0.64) mg/dL and 0.5 (0.47–0.52) mg/dL (p = 0.77), while post operation they were as follows: on postoperative day 1: 0.88 (0.53–1.3) mg/dL and 0.86 (0.62–1.1) mg/dL (p = 0.84), on postoperative day 3: 1.77 (1.29–2.08) mg/dL and 1.85 (1.70–1.95) mg/dL (p = 0.72), and on postoperative day 5: 2.32 (1.79–2.67) mg/dL and 2.56 (2.25–2.83) mg/dL (p = 0.55), respectively.
Conclusions. Serial CLP measurements during the early postoperative period revealed a linear (statistically significant) increase in concentration to postoperative day 5 without an evident point of decrease. A significant difference in median values and the course of curve patterns between the non-complicated and SSI groups was not observed.
Key words: calprotectin, periprosthetic joint infection, surgical site infection, total hip arthroplasty, total knee arthroplasty
Background
A surgical site infection (SSI) is a serious early complication of total joint arthroplasty (TJA) with a reported incidence of up to 3.68%.1, 2 This complication is associated with multiple perioperative factors and comorbidities, e.g., type of surgery (primary or revision), diabetes, obesity, rheumatoid arthritis, and smoking.1, 3 Despite the availability of guidelines that can help to recognize SSIs,4 the diagnosis of infection in the early postoperative period after TJA is challenging due to the different expressions of clinical symptoms, their intensification and overlap following surgical trauma, and a wide fluctuation of laboratory marker concentrations in specific patients.5, 6 Current concepts propose the use of clinical symptoms together with single or serial measurements of biochemical blood markers such as C-reactive protein (CRP), white blood cell (WBC) count and interleukin 6 (IL-6).7, 8, 9 However, these biomarkers have shown non-univocal interpretations in the septic scenario. Thus, there is a need to investigate other diagnostic markers with potential value in diagnosing SSI during the early postoperative period.
Calprotectin (CLP) is a zinc and calcium-binding protein that reduces superoxide dismutase and inhibits bacterial defense mechanisms against oxygenation. This heterodimer is released to body fluids from activated granulocytes and monocytes during inflammation and infection.10 Currently, CLP has been helpful in the diagnosis of inflammatory bowel diseases, monitoring the biological treatment of rheumatoid arthritis10, 11 and diagnosing chronic periprosthetic joint infections (PJIs).12, 13, 14 However, there is still a lack of evidence to support the use of serum samples of this marker in the diagnosis of early infection and SSIs after TJA.
Objectives
The primary aim of this preliminary study was to reveal the standard kinetics of CLP concentrations in the blood during the early postoperative period after non-complicated total hip arthroplasty (THA) and total knee arthroplasty (TKA) and to compare this curve shape with curves of CRP and IL-6. The secondary aim was to compare blood CLP concentrations between non-infected patients and patients with recognized early infections/SSIs.
Materials and methods
This study has received Postgraduate Medical Education Center Ethical Committee approval No. 104/PB/2018.
Blood samples from 64 consecutive patients (25 men and 39 women) who underwent primary THA and TKA between January 2019 and December 2019 were tested. Initially, this study aimed to analyze data collected from 60 patients (30 THA and 30 TKA) to determine the standard shape for the concentration curve of blood CLP during the first 5 days after the surgery. Despite the fact that in this study, patients after TKA and THA were analyzed, in terms of the examined markers these groups were homogenous and were combined as 1. Patients followed the standard protocol of observation and postoperative rehabilitation used routinely in the authors’ department and were discharged on the 5th postoperative day. All patients in the non-infected group were observed for a minimum of 2 years postoperatively, and there were no misdiagnosed cases. During the collection of the samples on the appointed days, in 4 patients (1 after THA and 3 after TKA), SSIs following Centers for Disease Control and Prevention (CDC) criteria were recognized.4 These patients were enrolled in a separate subgroup to conduct the additional analysis. Patients with chronic inflammatory diseases (e.g., rheumatoid arthritis, ankylosing spondylitis), with active or previous oncological treatment, patients with preoperative CRP concentration in blood >10 mg/L, those who have undergone surgical procedures on the knee joint in the past or were treated due to septic arthritis or PJIs, and those who underwent THA after femoral neck fractures were excluded from this study.
The CLP concentrations in blood were measured with an immunoturbidimetric Calprotectin Immunoassay (GCal; Gentian, Moss, Norway). Blood samples were collected at 7 AM from patients at 4 time points: the day before surgery, during admission to the hospital, and on postoperative days 1, 3 and 5. All samples were collected in test tubes with lithium heparin. Afterward, the quantitative analysis was done with the use of a Mindray BS-400 analyzer (Diamond Diagnostics Inc., Holliston, USA). Additionally, serum CRP (CRP Ultra; Cormay, Łomianki, Poland) and serum IL-6 (Interleukin 6 ELISA kit; Roche, Basel, Switzerland) concentrations were measured from the same blood samples.
Statistical analysis was performed with Statistica v. 13.1 (TIBCO Software Inc., Palo Alto, USA). The Shapiro–Wilk test was utilized to check the normality of the data. For dichotomous data, the Fisher’s exact test was performed. For continuous variables, Mann–Whitney U tests were employed, and results were presented as medians with interquartile ranges (Q1–Q3). For repeated variables, non-parametric Friedman’s analysis of variance (ANOVA) was performed. To assess a trend for analyzed markers in subsequent days, the Page test was used. A p-value < 0.05 was considered significant.
Results
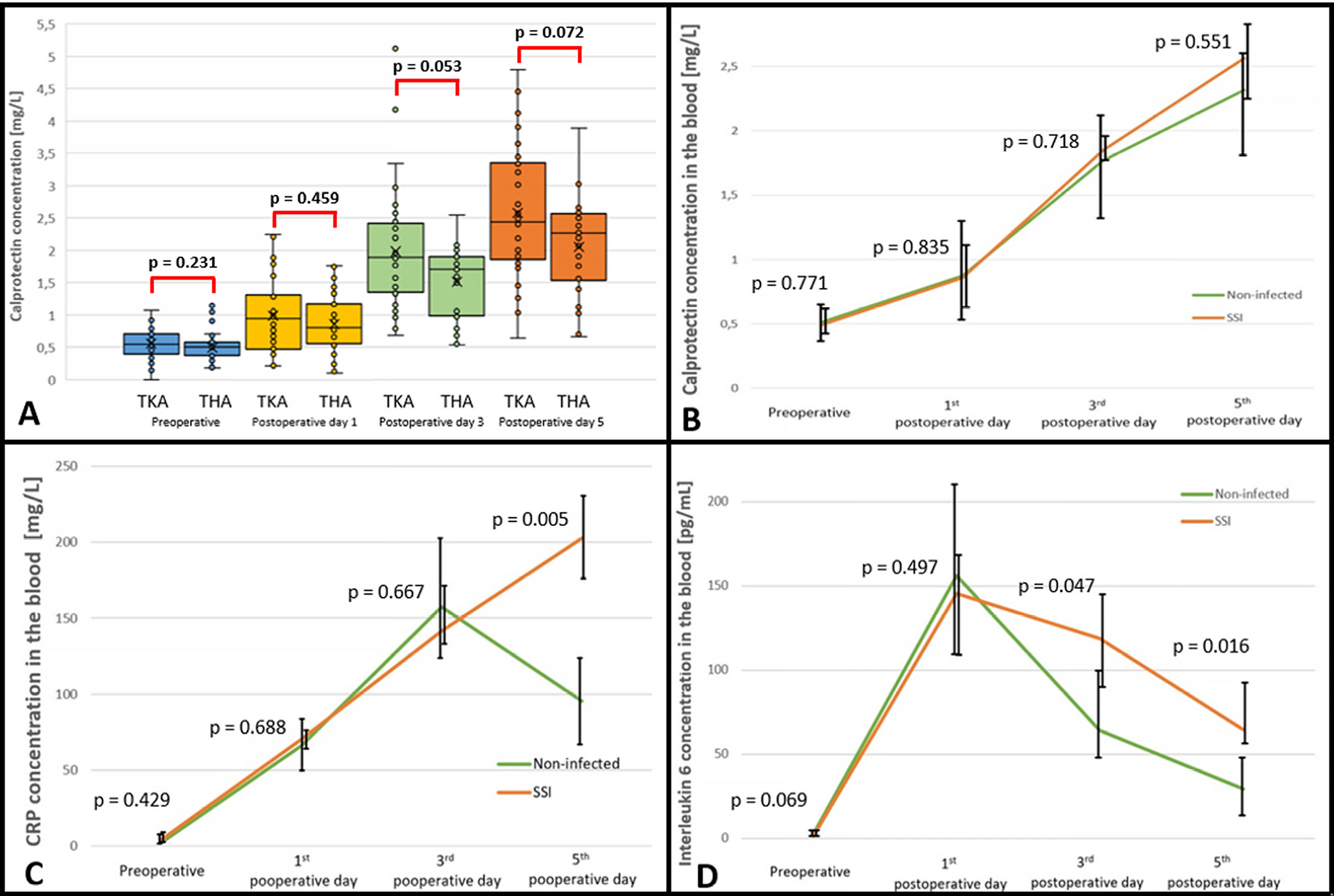
Demographic and clinical data of the study groups are presented in Table 1. Statistically significant differences between the medians (Q1–Q3) for CLP concentrations in TKA and THA groups were observed between: 1) preoperative measurements and postoperative day 3; 2) preoperative measurements and postoperative day 5; 3) postoperative day 1 and postoperative day 3; and 4) postoperative day 1 and postoperative day 5 (Mann–Whitney U test: p < 0.001 for all comparisons). A statistically significant increasing trend of CLP concentrations after TKA (Page test: p < 0.001) and THA (Page test: p < 0.001) was observed. Statistically significant differences in median (Q1–Q3) CLP concentrations between THA and TKA from preoperative and postoperative measurements were not observed (Figure 1A).
In consecutively operated patients after primary THA and TKA, a SSI was confirmed in 4/64 patients (6.25%). In this group, 3 cases (1 THA and 2 TKA) presented with superficial infection secondary to wound healing issues without joint capsule perforation, for which surgical debridement and irrigation were performed. In 1 patient (after TKA), according to CDC criteria, deep space/organ SSI (early PJI) was recognized, and debridement, antibiotics and implant retention with a mobile elements exchange (DAIR) procedure were administered. Statistically significant differences in CLP concentrations between non-infected cases and SSI were not observed (Figure 1B).
For CRP, statistically significant differences between non-infected and SSI groups were noted only on postoperative day 5 (95.55 (70.95–125.40) mg/L vs 202.50 (174.63–232.13) mg/L; Mann–Whitney U test: p = 0.005). However, for preoperative measurements, on postoperative day 1 and postoperative day 3, significant differences were not observed (Figure 1C).
For IL-6 concentrations, significant differences were noted on postoperative day 3 (64.10 (47.05–100.40) pg/mL vs 118.75 (91.43–146.53) pg/mL; Mann–Whitney U test: p = 0.047) and postoperative day 5 (29.10 (19.28–48.55) pg/mL vs 64.40 (60.48–91.50) pg/mL; Mann–Whitney U test: p = 0.016). Values of IL-6 concentrations measured preoperatively and on postoperative day 1 did not differ significantly (Figure 1D). Median values with Q1–Q3 are presented in the Table 2.
Discussion
In our study, we assessed the usefulness of the serum CLP marker, which recently showed a high usefulness in the diagnosis of chronic PJIs when it is determined in synovial fluid.12, 13, 15 Unfortunately, the potential value of CLP in the diagnosis of early PJIs and SSIs after primary TJAs was not investigated before, and to our knowledge, this is the first report that revealed a standard pattern for the kinetic curve of CLP after non-complicated THA and TKA. We have revealed a constant, linear increase during the first 5 postoperative days. In this pilot study, we did not reveal significant differences between non-infected and SSI cases in terms of the CLP concentration values on corresponding postoperative days.
In relation to blood CRP, after major orthopedic operations such as THA and TKA, in non-complicated cases, concentrations increase with a peak after 2 or 3 days, followed by a rapid decrease.8, 16 Larsson et al. reported the highest CRP values are seen 48–72 h after THA and TKA. Moreover, they revealed that normal or close to normal CRP levels returned by 14–21 days after surgery.8 This was also confirmed by Waśko et al., who also observed a wide range of blood CRP concentrations from 100 mg/L to 260 mg/L on the 3rd day after THA and TKA procedures.7 Based on the standard kinetics of CRP in the postoperative period, different studies were conducted to assess whether the increase in CRP correlates with the risk of developing PJIs. Despite this fact, there is still no consensus about the usefulness of serial CRP measurements after TJA in diagnosing early PJIs.17 Niskanen et al. concluded that the rise in CRP levels at 1 or 2 weeks after TJA may be an indicator of a serious complication.16 Similarly, Kim et al. concluded that elevated CRP levels may indicate postoperative infection; however, they emphasized that there is a need to differentiate between non-infection causes and other site infections.18 On the other hand, Rohe et al. and Windish et al. revealed that serial measurements of CRP have no predictive value in diagnosing early PJIs after THA and TKA.19, 20 We are aware that this requires further investigation. However, we believe that serial measurements and the observation of the curve pattern are currently the best and widely available laboratory methods to diagnose an early postoperative infection. Due to the wide range of the received values of CRP, there is a need to compare the obtained results with those measured on the day of the concentration peak.
In the case of IL-6, our results also agree with those reported by the other authors. Compared to the curve trajectory of CRP, higher values of IL-6 are expected earlier. In non-complicated TJAs, the IL-6 concentration curve presents different kinetic patterns than CLP and CRP, with a maximum, rapid increase on the 1st postoperative day, and a more dynamic decrease in the following days. Maniar et al. reported that the IL-6 concentration increased rapidly, with a peak at 12 h after TKA, and returned to a level in the range of 3–14 pg/mL within 2 weeks.21 Waśko et al., in the study mentioned above, observed that IL-6 reaches the highest value during the first 24 h after TJA and decreases over the following postoperative days.7 Despite this fact, we observed significantly higher concentrations of IL-6 on days 3 and 5 between SSI and non-complicated cases and agreed with Aichmair et al. that the increased serum values of this marker cannot predict an early onset PJI.22
Limitations
This study has some limitations which should be considered before data analysis. First, although this is a prospective study, the SSI group (4 patients) was significantly smaller than the non-infected group (60 patients). Second, the analysis covered only a short postoperative period (the 5th postoperative day to the day of the patient’s discharge). A standard point of concentration decrease for CLP was not established in contrast to CRP and IL-6. Finally, in this study, we did not analyze the influence of additional demographical and clinical factors that may have influenced the final results.
Conclusions
1. Serial serum CLP measurements during the early postoperative period revealed a close to linear increase of concentration up to postoperative day 5 without the evident point of decrease in non-complicated cases.
2. A significant difference in median values and the course of the curve patterns between the non-complicated and SSI groups was not observed. There is a need to verify our findings and the potential value of CLP in the diagnosis of PJI/SSI on a larger group of patients to determine when the peak, the start of concentration decrease and its normalization occurs after elective TJAs.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.