Abstract
Postoperative urinary retention (POUR) is a common surgical complication that can result in bladder overdistension, urinary tract infection and an extended hospital stay. Although neostigmine is an effective therapy for POUR, its usage remains controversial. The purpose of this study was to investigate the effectiveness of neostigmine in improving POUR after surgery. PubMed, Embase, Web of Science, and the Cochrane Library databases were reviewed. A methodical search approach was used for data extraction, while meta-analysis and bias analysis employed Review Manager 5.2 and MedCalc.
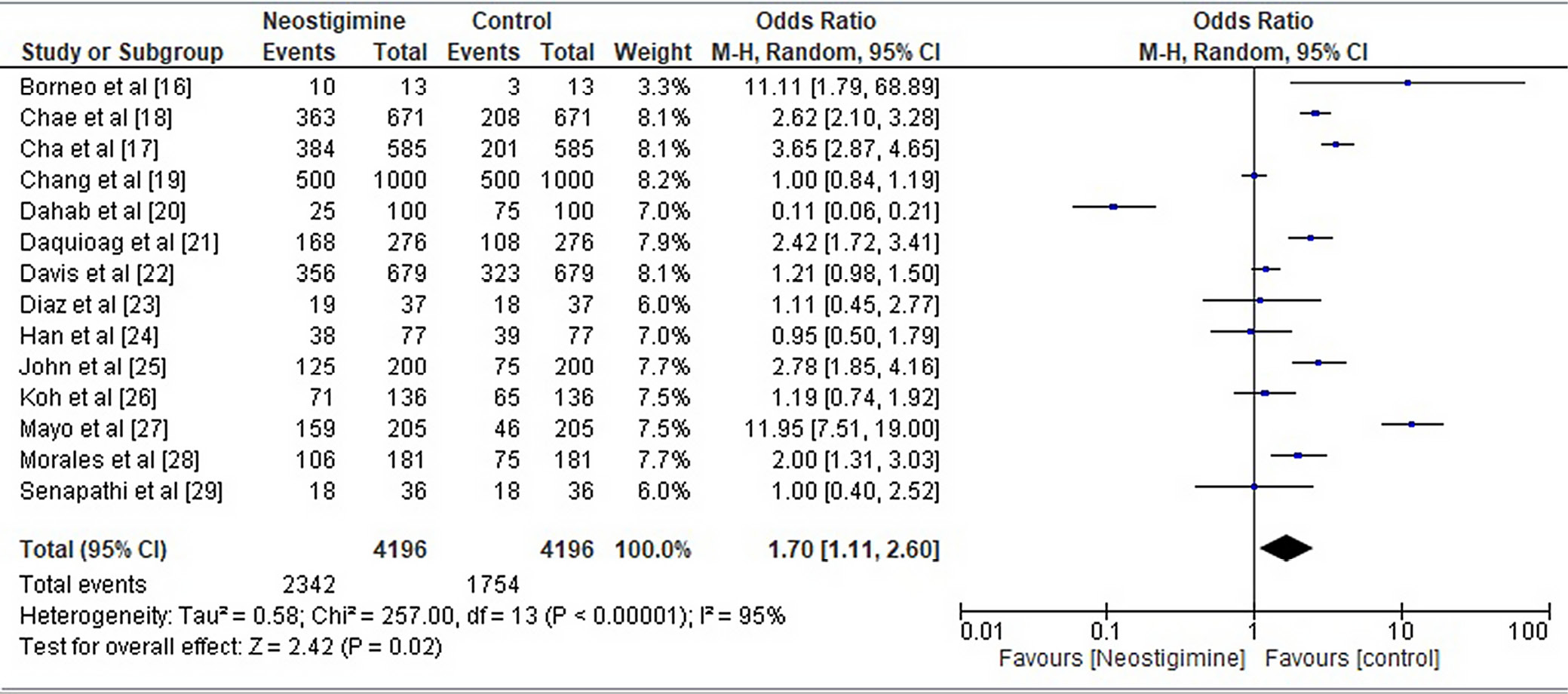
Fourteen studies involving 4196 postoperative patients were included. With an odds ratio (OR) of 1.70, 95% confidence interval (95% CI) of 1.11–2.60 and an overall effect with p < 0.05, our analysis indicated that the patients receiving neostigmine had a greater effective urine retention rate than after other standard therapies. The subgroup analysis showed that neostigmine recipients had reduced residual urine volume (mean difference (MD) = −1.16, 95% CI: −2.05–−0.27, overall p < 0.05, and I2 = 90%) and POUR (standardized MD (SMD) = 3.76, 95% CI: 2.19–5.34, overall p < 0.001, and I2 = 99% using a random effects model) as compared to controls. A random-effects model was utilized due to the substantial heterogeneity between trials. The studies were consistent and had no publication bias. Based on the findings of this meta-analysis, neostigmine can be considered an effective POUR treatment.
Key words: meta-analysis, urinary retention, neostigmine, postoperative urinary retention (POUR), volume of urine excreted
Introduction
Postoperative urinary retention, commonly referred to as POUR, is a condition characterized by the inability of patients to effectively void their bladders after surgical interventions despite having a full bladder. The condition results in an elevated postvoid residual volume. Untreated POUR can cause adverse outcomes, such as acute renal injury, detrusor injury and excessive bladder dilatation. These events may lead to extended hospital stay and necessitate supplementary care after discharge.1
After a surgical procedure or anesthesia, POUR can present in various forms, including tenderness or uneasiness in the suprapubic region, bladder contractions, urinary incontinence, and an inability to void.2 Urinary retention is a prevalent medical condition impacting a significant proportion of the population, with a reported incidence rate of 5–70%.3 Several factors can contribute to increased urinary retention susceptibility after a surgical procedure, including anesthesia administration, the type of operation performed, the presence of postoperative inflammation, and limited mobility.4 If left untreated, POUR may result in significant bladder distension, acute renal dysfunction and detrusor muscle damage. Consequently, a patient’s discharge from the hospital and their subsequent treatment may be delayed.5, 6
Catheterization is a frequently recommended therapeutic intervention for POUR due to its potential to facilitate the management of the condition. Notwithstanding its benefits, this method is associated with an increased likelihood of urinary tract infection and other potential complications.7 However, it is possible to reduce the incidence of complications and postoperative morbidity by employing diverse methods that improve patients’ physical and emotional well-being while also avoiding POUR, as suggested by previous research.8 Hence, plausible preventive measures encompass implementing anesthetic and analgesic interventions in conjunction with acupuncture, heated compresses and massage therapy.9 Pharmacological intervention for POUR management can involve drugs that impede β-adrenergic and cholinergic activity.10
Neostigmine is classified as a parasympathomimetic drug due to its ability to mimic the effects of the parasympathetic nervous system. It functions as a reversible acetylcholinesterase inhibitor via indirect activation of nicotinic and muscarinic receptors by inhibiting acetylcholine breakdown, specifically targeting step 5 of the process.11 Neostigmine has demonstrated effectiveness, safety and success in various POUR patient management trials.12, 13 Nonetheless, certain studies have reported unsatisfactory outcomes due to the development of tension in the smooth muscle of the bladder.14, 15 The data suggest that further research is required to explore the utilization of neostigmine for POUR. Consequently, pertinent publications16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29 were procured and reviewed to conduct a thorough investigation into the efficacy of neostigmine in POUR.
Objectives
This investigation aimed to assess the efficacy of neostigmine in ameliorating POUR.
Materials and methods
Eligibility criteria
All possibly relevant papers were examined in their entirety to assess whether or not they fulfilled the inclusion criteria listed below: 1) studies that compared patients receiving neostigmine to conventional therapy; 2) studies including patients diagnosed with POUR; 3) studies that contained indicators assessing efficacy or additional pertinent variables comparing neostigmine treatment to standard treatment; and 4) studies that were readily available in their entirety. The exclusion criteria were as follows: 1) studies regarding other disorders; 2) studies including comparisons with other therapies; 3) studies with insufficient data; and 4) reviews, abstracts or duplicate publications.
Information sources and literature search strategy
We conducted a search for randomized controlled trials (RCTs) published between January 1, 2000, and January 1, 2023, in the PubMed, Web of Science, Embase, and Cochrane Library databases using the following search terms: 1) neostigmine; 2) postoperative urinary retention OR POUR; 3) clinical effects; 4) volume of urine excreted; and 5) urinary retention. Within the context of the search strategy, the Boolean operator “AND” was used to combine the Medical Subject Headings (MeSH) with the text keywords. We carried out a comprehensive search across various databases and did not impose any limitations on the language used or the publication status of the studies. Two researchers, NL and YW, independently searched the literature and analyzed the bibliographies to find additional publications related to the topic.
Study selection and data collection process
The parameters for this review were based on the most recent edition of the Cochrane Handbook for Systematic Reviews of Interventions.30 A predesigned data collection form was used to extract data from the main research. Two investigators (NL and YW) independently screened titles, abstracts and full texts of potentially eligible studies and used the 27-item Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist criteria to grade each study as “yes” (1 point), “partially” (0.5 points) or “no” (0 points). The points assigned to each study comply with PRISMA guidelines. Both investigators extracted the data independently, and the 3rd investigator (QC) was involved if the data extracted from the same study differed between the 2 investigators. Information, including the name of the first author, publication year, journal, country, patient population, number of participants, age, sex (male/female), intervention dosage (neostigmine), and primary outcome measures, was extracted for each arm. The primary endpoints were the amount of urine excreted and the extent of POUR decrease. The likelihood of bias across studies was assessed using a visual examination of a funnel plot31 and Egger’s test.32
Risk of bias evaluation
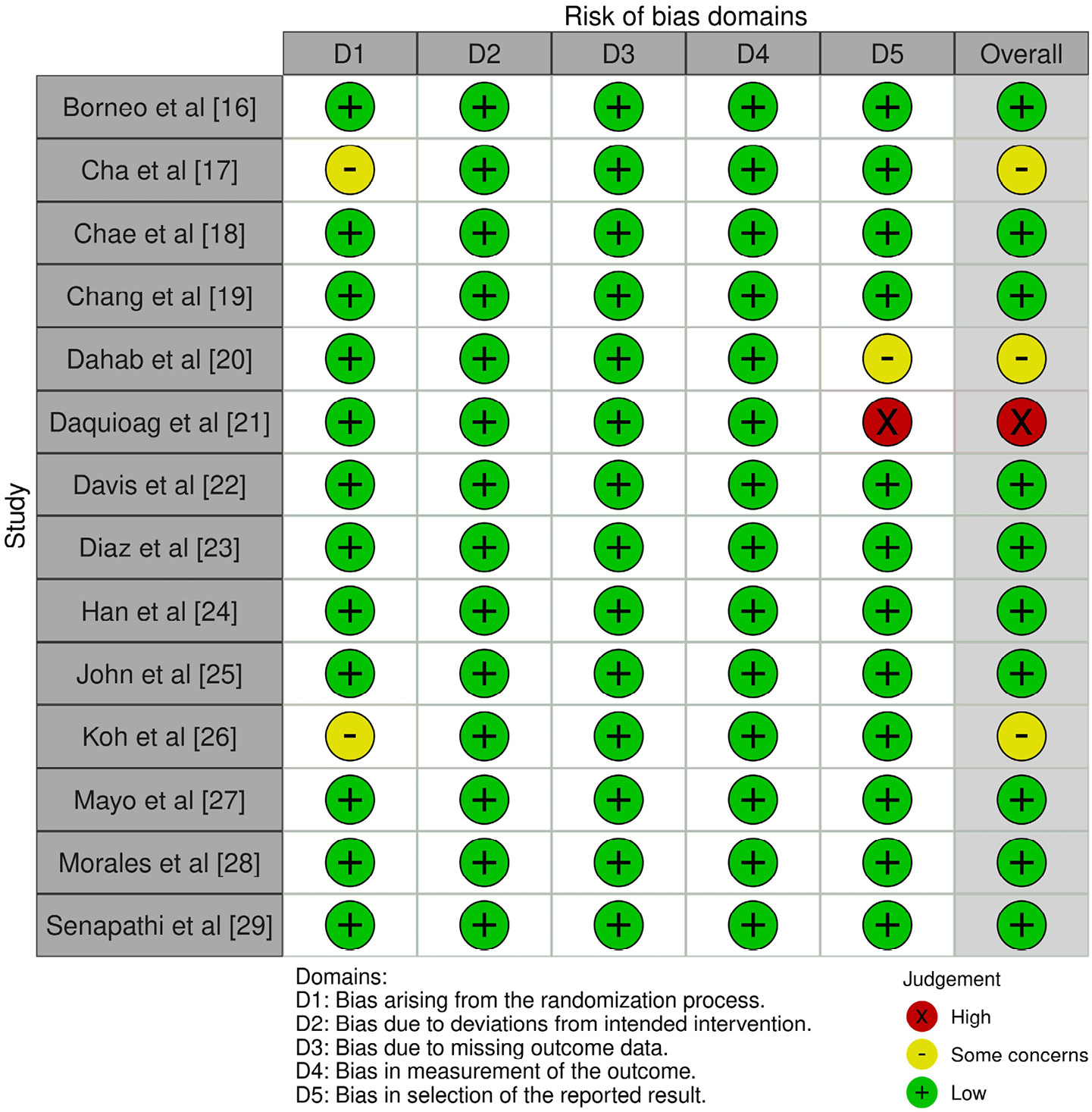
The “risk of bias” table prepared in the Review Manager (RevMan) software (v. 5.3; The Nordic Cochrane Centre, Copenhagen, Denmark) was used to assess study quality.33 The table documented random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessments, insufficient outcome data, selective reporting, and other forms of bias. Based on the retrieved data, we assigned a score of “low”, “high”, or “some concerns” to each parameter for each study. The inquiry was independently conducted by 2 investigators (NL and YW). Any disagreements were addressed by the 3rd investigator (QC).
Statistical analyses
Statistical analysis of the findings from the selected studies was performed using RevMan software (v. 5.3; The Nordic Cochrane Center). Pairwise meta-analyses were performed with a DerSimonian and Laird random-effects model34 to calculate the pooled estimates of odds ratio (OR) and mean difference (MD), with 95% confidence intervals (95% CIs) of direct comparisons between the experimental and control groups. A random-effects model was used due to the substantial heterogeneity among the studies. The purpose of these analyses was to determine the degree to which the effect size (OR and MD) remained consistent. It was determined that heterogeneity ranging from 0% to 40% “might not be important”, heterogeneity ranging from 30% to 60% was considered “moderate heterogeneity”, heterogeneity ranging from 50% to 90% was treated as “substantial heterogeneity”, and heterogeneity ranging from 75% to 100% was “considerable heterogeneity”. The random-effects model was implemented due to the high heterogeneity in the data.35
Results
Literature search results
The preliminary search yielded 657 articles in the PubMed, Embase, Web of Science, and Cochrane Library databases. Following the initial screening, 316 records remained, and after screening titles and abstracts, additional 211 studies were removed due to their type (review articles, letters, case reports, comments, or editorials). Then, 105 studies were evaluated, of which 38 were selected for final screening. Out of these, 24 publications were deemed ineligible for further consideration for various reasons, such as not reporting the required outcome or having insufficient data. Ultimately, 14 studies involving 4196 patients fulfilled the inclusion criteria and were eligible for meta-analysis. Figure 1 depicts the selection procedure, which followed PRISMA guidelines,36 and includes an explanation of the factors that led to the exclusion of certain studies.
Study characteristics
The primary features of the 14 clinical studies16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29 are outlined in Table 1. The publication dates ranged from 2000 to 2023. During the interventions, experimental groups received neostigmine, and control groups were given different types of medicine. A total of 4196 patients participated in these investigations, with 2342 patients in the intervention groups and 1854 in the control groups. The number of people in the study samples ranged from 13 to 1000.
Risk of bias and publication bias assessment
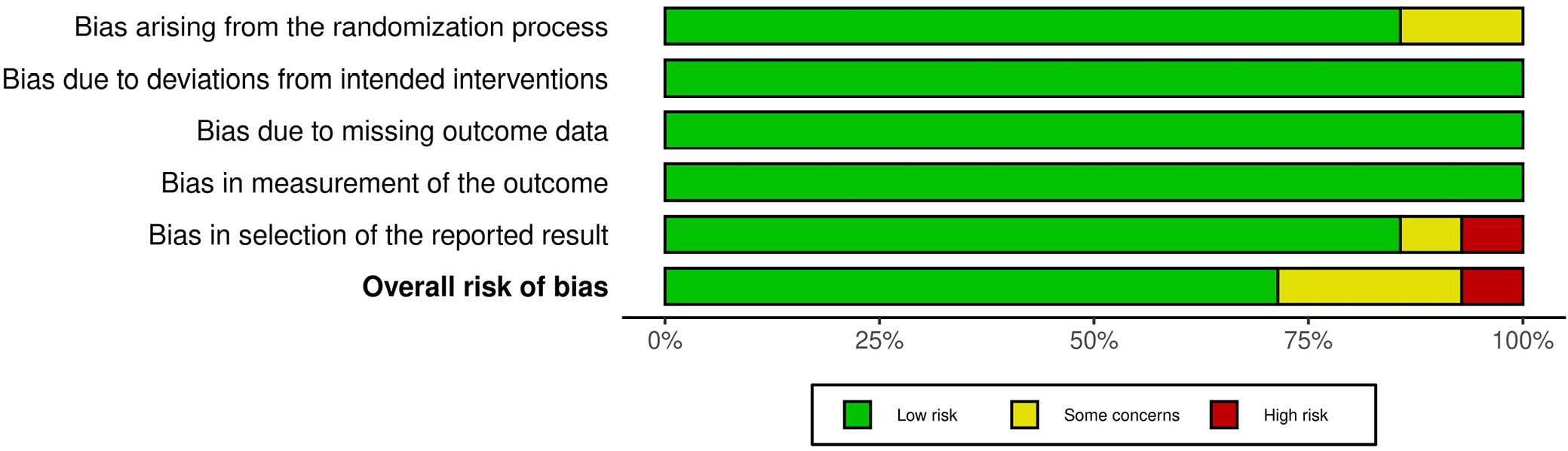
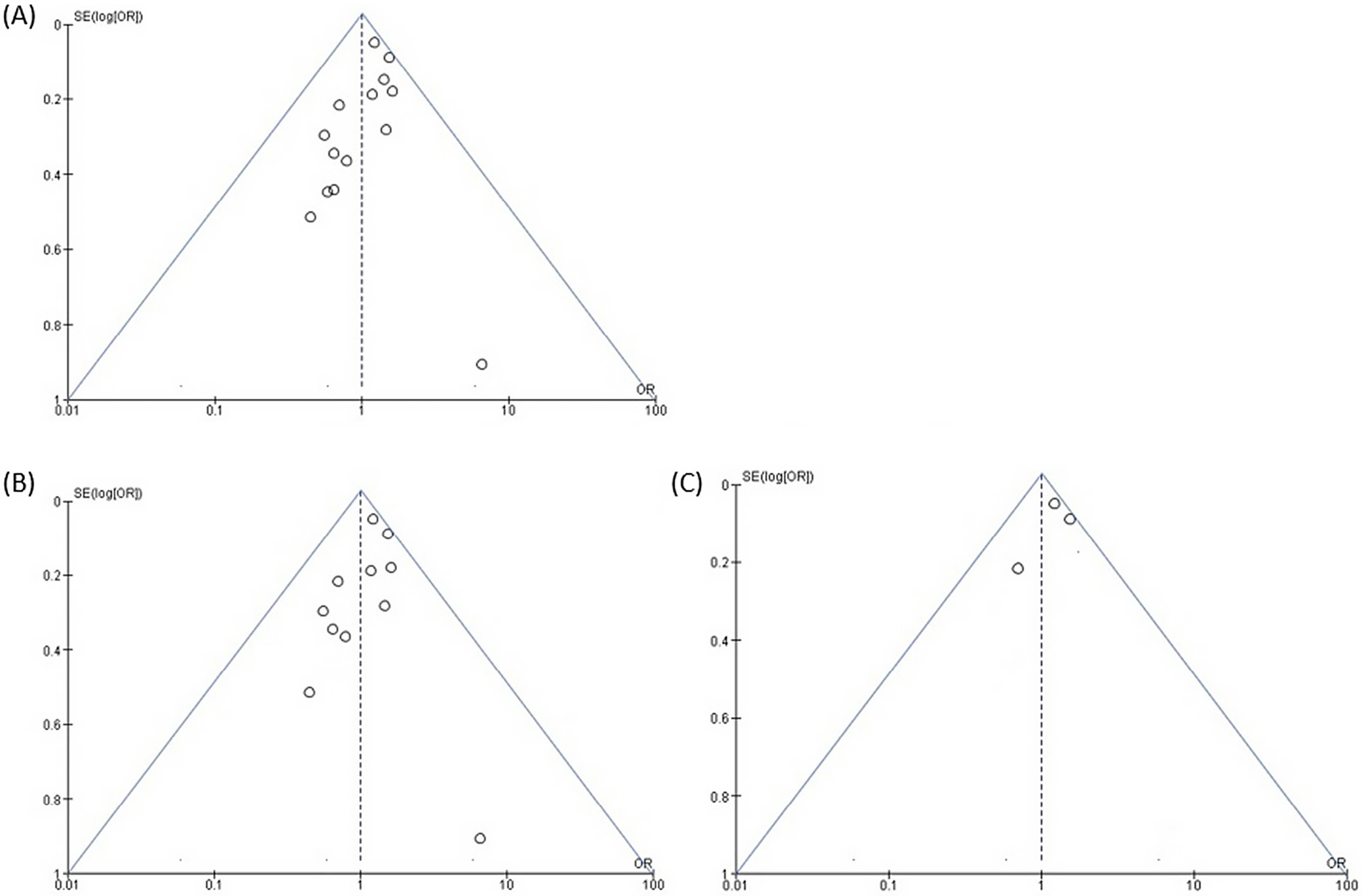
A predesigned questionnaire was used to conduct a risk of bias assessment for each of the 14 studies, and the findings are presented in Table 2. There was a low risk of bias in 10 studies and a moderate risk in 3 studies due to the randomization procedure and bias in selecting the data reported. As can be seen in the risk of bias summary (Figure 2) and risk of bias graph (Figure 3), only 1 publication had a significantly high risk due to bias in selecting the results reported. The funnel plot for publication bias is presented in Figure 4. The symmetrically shaped funnel plot for the overall effect size, specifically the OR of neostigmine compared to the control group, is shown in Figure 4A. The results indicate a low probability of publication bias, as evidenced by a significant p-value of 0.463 for Egger’s test.37 The symmetrically shaped funnel plot for the percentage reduction in POUR in neostigmine groups (NG) compared to control groups (Cg) is depicted in Figure 4B. The plot indicates a low probability of publication bias, with a significant p-value of 0.385 for Egger’s test. The results in Figure 4C indicate the volume of urine excreted, with a statistically significant p-value of 0.241.37
Primary study outcomes
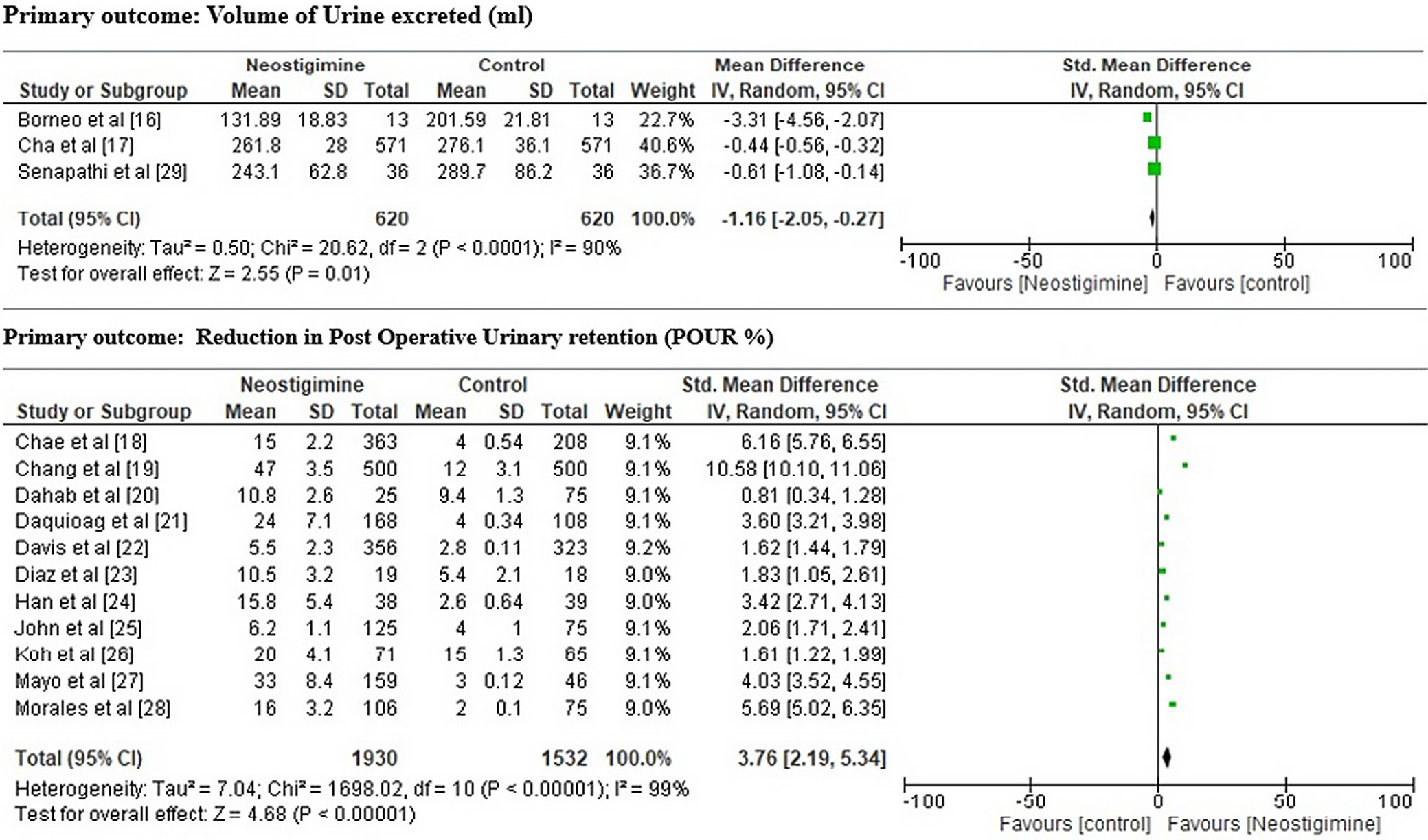
Table 3 displays the findings of the primary outcomes, including the volume of urine expelled [mL], reported in 3 studies,16, 17, 29 and the reduction in POUR, reported in 11 studies.18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28
Heterogeneity analysis of the experimental and control groups
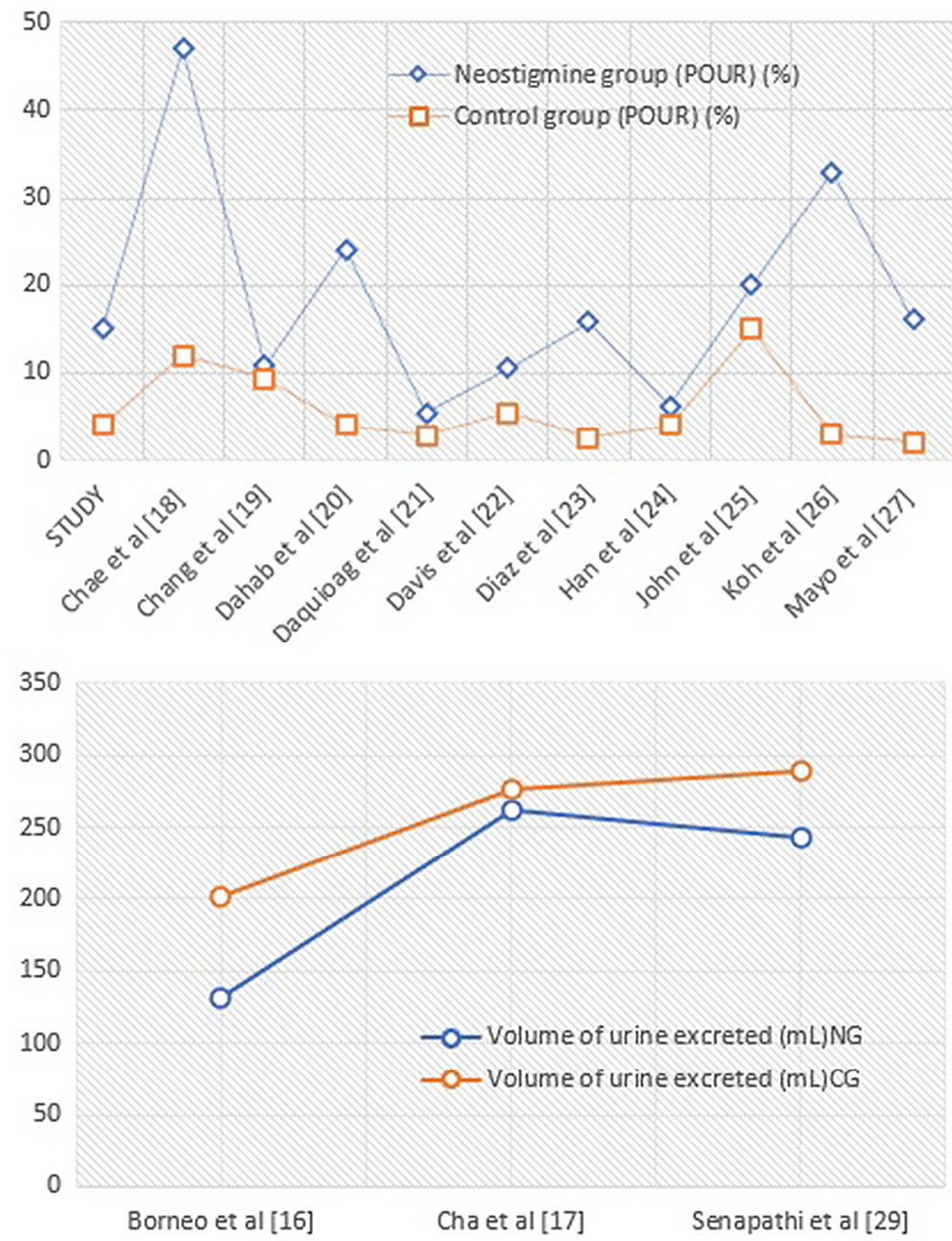
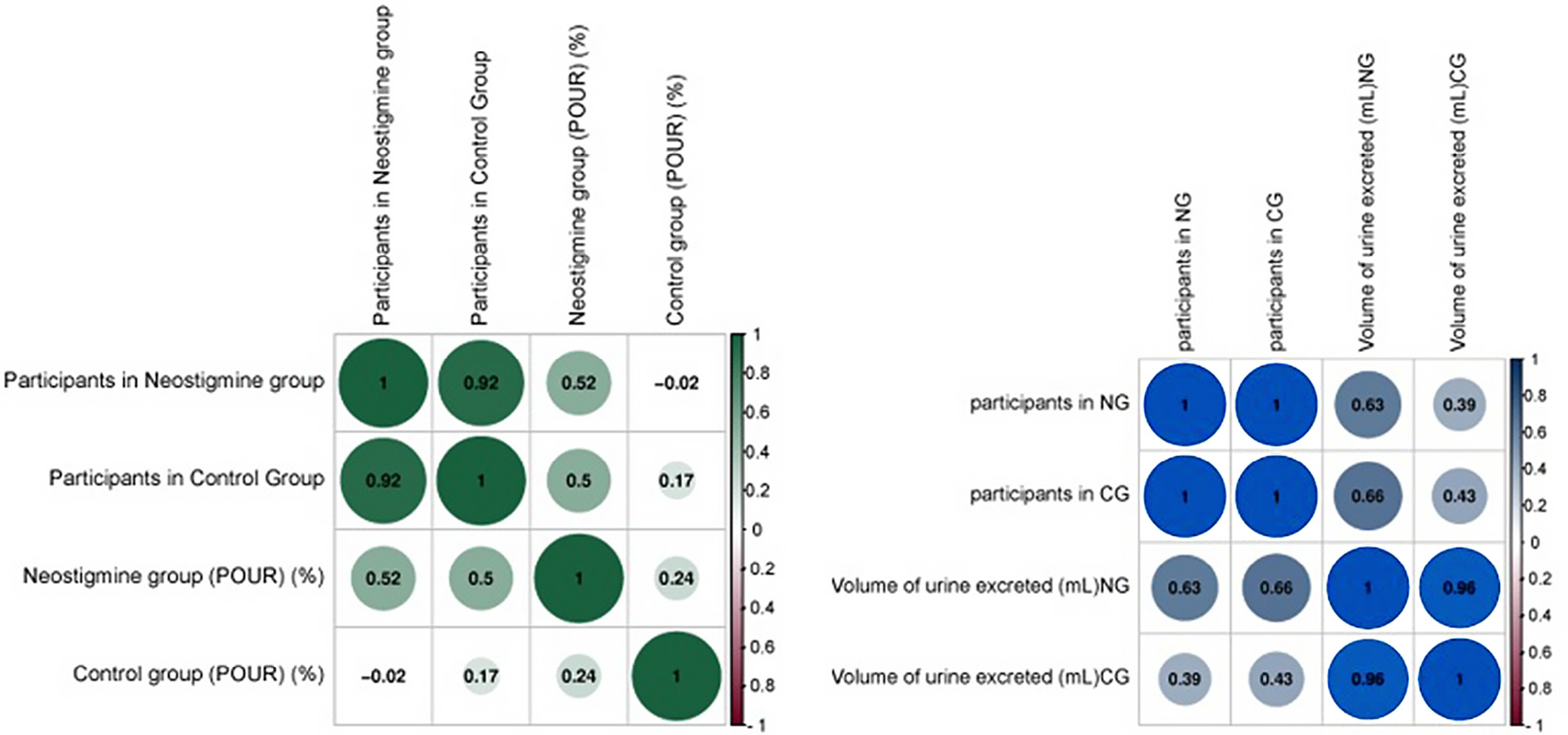
This meta-analysis compared differences in urine retention efficiency rates between experimental and control groups using heterogeneity analysis. The overall result demonstrated that the NG had a greater effective rate than the CG (OR = 1.70, 95% CI: 1.11–2.60, Tau2 = 0.58, χ2 = 257, degrees of freedom (df) = 13, overall effect p < 0.05, I2 = 95%, in the random effects model), as shown in Figure 5. The findings of the subgroup analysis revealed that neostigmine performed noticeably better than the conventional treatments typically used for urinary retention. Figure 6 highlights that the neostigmine group had a lower residual urine volume (MD = –1.16, 95% CI: –2.05––0.27, overall p < 0.05, I2 = 90%) and a more substantial POUR reduction than controls (standardized MD (SMD) = 3.76, 95% CI: 2.19–5.34, overall p < 0.001 I2 = 99%). Figure 7 displays comparative scatter plots indicating that the NG had a higher percentage of participants with POUR improvements and a lower volume of urine retention than the CG. Similarly, the correlation plot depicted in Figure 8 demonstrates a noteworthy higher POUR and residual urine volume decrease in the NG than in the CG. All of these results were statistically significant, with p < 0.05.
Discussion
This meta-analysis investigated the effectiveness of neostigmine as a POUR treatment method. A total of 14 studies, comprising 4196 participants, were included in the analysis. The primary indicators considered were the effective urinary retention rate, voided urine volume and POUR reduction. The results indicate that neostigmine is a viable therapeutic option for addressing POUR.
The regulation of bladder function is thought to be significantly influenced by the parasympathetic nervous system. The system is responsible for urination through detrusor muscle contraction and sphincter muscle relaxation, though it remains inactive during bladder filling.38, 39 Neostigmine is a potent acetylcholinesterase inhibitor that exhibits dual functionality by reducing cholinesterase activity and enhancing acetylcholine efficacy. The drug achieves this through dose-dependent stimulation of the detrusor muscle of the bladder, resulting in direct contractions that ultimately lead to an augmentation in micturition frequency and urine volume output.40, 41
Several preventative strategies targeting POUR have been published recently. Due to a limited understanding of the efficacy of different treatments and concerns surrounding their potential side effects, there is currently no established protocol for preventing this particular ailment.42 However, reports suggest that neostigmine could be a potential medication for POUR. The plasma half-life of neostigmine following intravenous administration is variable, ranging from 47 min to 60 min, with a mean value of 53 min. Clinical manifestations of neostigmine are typically observed within 20–30 min of intramuscular administration, with a duration of action ranging from 2.5 h to 4 h.43 According to Zhong et al., epidural neostigmine administration (at doses of 1 g/kg, 2 g/kg or 4 g/kg) in combination with lidocaine produced a dose-independent analgesic effect that persisted for 8 h after knee surgery, in contrast to the patients who received only lidocaine and did not display concomitant escalation in adverse reactions.44 Similarly, Hassanin et al. conducted a blinded RCT to investigate the impact of neostigmine supplementation with bupivacaine on ultrasound-guided supraclavicular brachial plexus block in forearm surgeries.45 The study findings revealed that the neostigmine group exhibited statistically significant reductions in visual analogue scale (VAS) scores compared to the control group at 1 h, 2 h, 4 h, and 6 h. Nevertheless, it is advisable to only employ this approach in cases where surgical procedures extend beyond 4 h. Thus, neostigmine exhibits promising prospects as a viable pharmaceutical agent for surgical procedures of diverse durations.
Prior meta-analyses carried out by Sirisreetreerux et al.46 and Jackson et al.47 demonstrated neostigmine efficacy, a medication classified as either a parasympathomimetic or a reversible cholinesterase inhibitor, in the management and prevention of POUR. Nonetheless, it is imperative to subject these medications to RCTs with substantial sample sizes to determine their clinical efficacy and patient acceptability.
In a recent systematic review and meta-analysis conducted by Cao et al., the neostigmine group exhibited a higher rate of effectiveness in treating urine retention compared to traditional Chinese treatments and physical therapy.48 The authors derived an OR of 7.47 (95% CI: 4.10–13.59, p < 0.001) to support their findings. Neostigmine has been found to effectively mitigate POUR symptoms. The results of our study align with previous meta-analyses and support the utilization of neostigmine as a viable intervention for addressing POUR.
Limitations
This study had several limitations. Indeed, it is probable that the “small study effect”, which occurs when most studies have extremely small sample sizes, skewed the results. The problem stems from the paucity of available reports. As such, more research is required on a larger sample size to reach robust scientific conclusions. Any future evaluations must account for new data, especially on adverse incidents or issues. Due to the low number of studies published on this subject up to this point, any future study and analysis must incorporate a greater number of articles that feature research from many countries.
Conclusions
According to the findings of this meta-analysis, neostigmine may be related to decreased POUR incidence and could effectively manage POUR symptoms with enhanced therapeutic effects. However, validating the impact of the interventions evaluated in this meta-analysis and designing an effective treatment and prevention plan for surgical patients at risk of developing POUR requires further analysis of large, robust and properly designed RCTs.
Data availability statement
All data generated or analyzed during this study are included in this article. Further inquiries should be directed to the corresponding author.