Abstract
Background. Favipiravir is very effective in the treatment of many viral infections, especially at high doses. It was used at such doses to treat coronavirus disease 2019 (COVID-19) during the pandemic. However, liver damage was reported in patients undergoing such treatment.
Objectives. This study aimed to investigate the effects of low and high doses of favipiravir on the liver of rats, using biochemical and histopathological methods.
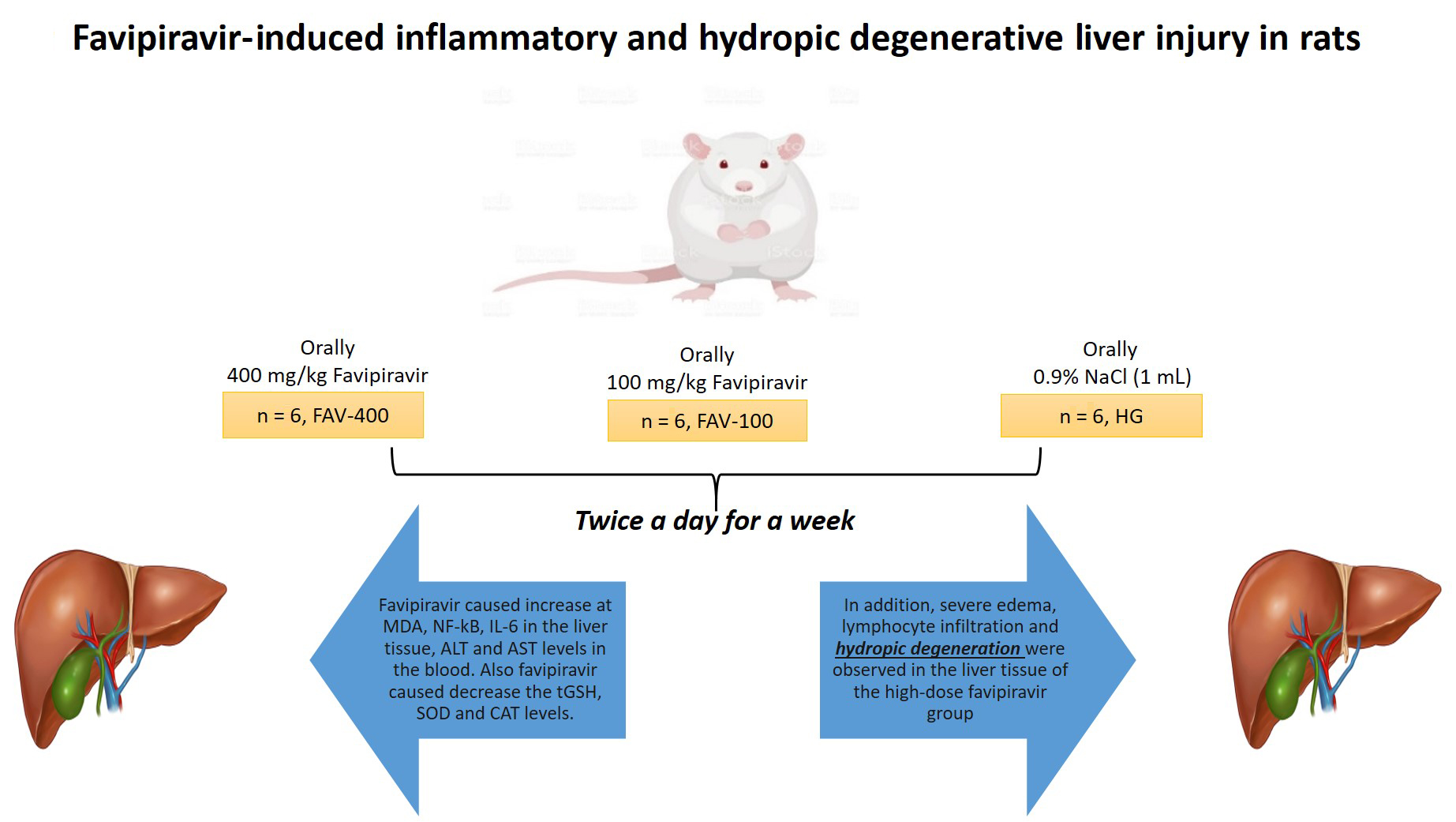
Materials and methods. Wistar albino rats were allocated to one of 3 groups, namely a healthy group (HG), a 100 mg/kg favipiravir (FAV-100) group and a 400 mg/kg favipiravir (FAV-400) group. Favipiravir was administered orally at 100 mg/kg and 400 mg/kg doses to the FAV-100 (n = 6) and FAV-400 (n = 6) groups, respectively. Distilled water was administered orally (1 mL) using the same method to the HG (n = 6). This procedure was repeated twice a day for 1 week. At the end of this period, the animals were euthanized with a high dose of thiopental anesthesia (50 mg/kg) and their liver tissues were removed.
Results. Favipiravir caused an increase in malondialdehyde (MDA), nuclear factor kappa B (NF-κB) and interleukin 6 (IL-6) levels in the liver tissue, as well as elevated alanine aminotransaminase (ALT) and aspartate aminotransferase (AST) levels in the blood. Moreover, favipiravir caused a decrease in total glutathione (tGSH), superoxide dismutase (SOD) and catalase (CAT) levels. In addition, severe edema, lymphocyte infiltration and hydropic degeneration were observed in the liver tissue of the FAV-400.
Conclusions. High-dose favipiravir caused more significant oxidative and inflammatory damage in the liver tissue of rats than low-dose favipiravir.
Key words: inflammation, rat, hydropic degeneration, favipiravir
Background
Favipiravir is an antiviral drug that has been tested for use against coronavirus disease 2019 (COVID-19).1 It has also been found to be effective in treating influenza and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).2 Favipiravir is effective against RNA viruses such as human rhinovirus, respiratory syncytial virus, metapneumovirus, and parainfluenza, and has been considered a candidate for the treatment of hantavirus-induced infections.3 The drug is converted to the active favipiravir-ribofuranosyl-50-triphosphate metabolite through phosphorylation and ribosylation in tissues.4 The antiviral effects of favipiravir depend on blocking the inward and outward movements of viruses by the drug.5 The action is achieved through the inhibition of RNA-dependent RNA polymerase (RdRp).6 Favipiravir has been found to be effective against these infections at high doses.7 Two doses of 2400–3000 mg every 12 h have been recommended for the treatment of COVID-19, followed by 1200–1800 mg every 12 h.8, 9 However, favipiravir can cause toxic effects at high doses.10
The most frequent toxic effects of favipiravir are diarrhea, nephrotoxicity, increased serum uric acid and transaminase levels, decreased white blood cell count and neutrophil level, nausea, vomiting, abdominal pain, skin rash, itching, delirium, hallucinations, and convulsions.11, 12 Moreover, liver damage has been reported in patients undergoing favipiravir treatment.13 This was also shown experimentally, with favipiravir causing hepatic dysfunction and vacuolization in hepatocytes.13, 14 Furthermore, favipiravir treatment was shown to cause an increase in liver enzymes and oxidative and histopathological damage in rats. Recent study reported that endogenous oxidants and antioxidants such as superoxide dismutase (SOD), catalase (CAT), malondialdehyde (MDA), and total glutathione (tGSH) played a critical role in the pathogenesis of favipiravir hepatotoxicity.15 These data indicated that oxidative stress was responsible for favipiravir-induced hepatotoxicity. However, no studies investigated the effects of favipiravir on the liver of animals treated with low and high doses of this drug.
Objectives
The purpose of the study was to examine the biochemical and histopathological effects of different doses of favipiravir on rat liver.
Materials and methods
Animals
Rats were obtained from Medical Experimental Application and Research Center (Erzincan Binali Yıldırım University, Erzincan, Turkey). In total, 18 albino male Wistar rats weighing 280–290 g were included in the study. Before the commencement of the study, the rats were housed at room temperature (22°C) in a 12-hour light/dark cycle. The animals were fed with normal tap water and pellet food. The procedures were approved by the Animal Experiments Local Ethics Committee (September 29, 2022, meeting No. 09/46, approval No. E-85748827-050.01.04-204261)
Chemicals
The thiopental sodium was procured from a commercial supplier (IE Ulagay, Istanbul, Turkey), and the favipiravir was obtained from the Education and Research Hospital (Erzincan, Turkey) affiliated with the Ministry of Health.
Animal groups
The rats were randomly assigned to one of 3 groups (n = 6/group), namely a healthy group (HG), a 100 mg/kg favipiravir (FAV-100) group and a 400 mg/kg favipiravir (FAV-400) group.
Experimental procedure
Favipiravir was given orally at doses of 100 mg/kg or 400 mg/kg to the FAV-100 and FAV-400 animal groups, respectively. Distilled water was administered orally (1 mL) to the HG using the same method. This practice was continued twice a day for 1 week. At the end of the 1st week, the rats were euthanized with a high dose of thiopental anesthesia (50 mg/kg), and their liver tissues were excised. The MDA, tGSH, SOD, and CAT levels in liver tissues were measured. In addition, serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities were determined in blood samples taken from the rats before they were euthanized. The liver tissues were assessed biochemically and histopathologically. All experimental results were analyzed by comparing the groups.
Biochemical analyses
Malondialdehyde, glutathione, superoxide dismutase, catalase, and protein determination in tissue
The concentration of SOD, tGSH and MDA in liver tissues of the experimental animals was evaluated using commercially available enzyme-linked immunosorbent assay (ELISA) kits (cat. No. 706002, 703002 and 10009055, respectively) according to the kit instructions (Cayman Chemical Company, Ann Arbor, USA). The determination of CAT was performed according to the method described by Góth.16 The protein analysis was performed according to the Bradford method.17
Nuclear factor kappa B and interleukin 6 analysis in tissues
The samples were weighed and all tissues was separated, snap-frozen using liquid nitrogen, and homogenized with a mortar. After the samples were melted, they were stored at 2–8°C as a standard. Phosphate-buffered saline (PBS) (pH 7.4, 1/10 w/v) was added to the samples, which were then vortexed for 10 s and centrifuged for 20 min at 10,000 × g. Nuclear factor kappa B (NF-κB; pg/L) was assayed using a rat NF-κB ELISA immunoassay kit (Shanghai Sunred Biological Technology Co., Ltd., Shanghai, China) and the levels of interleukin 6 (IL-6; ng/L) were assayed using a commercial ELISA kit (Hangzhou Eastbiopharm Co., Ltd., Hangzhou, China).
Alanine aminotransferase and aspartate aminotransferase analysis in tissues
Serum AST and ALT activities were measured with commercially available kits (Roche Diagnostics GmbH, Mannheim, Germany) to assess liver function. Samples were analyzed spectrophotometrically using a cobas® 8000 auto-analyzer (Roche Diagnostics GmbH).
Histopathological analyses
Sections of liver tissue were stained with hematoxylin and eosin (H&E) and examined using an Olympus BX51 fluorescence microscope (Olympus Corp., Tokyo, Japan). Histopathological severity of damage was evaluated as normal (0), mild injury (1), moderate injury (2), or severe injury (3).
Statistical analyses
The experimental results are presented as mean ± standard deviation (M ±SD) and 95% confidence interval (95% CI) of the mean. The Shapiro–Wilk test was used to determine whether the data were distributed normally, and the homogeneity of variances was determined with the Levene’s test. Since the data were normally distributed, one-way analysis of variance (ANOVA) was chosen for statistical analysis. Subsequently, the Tukey’s honest significant difference (HSD) and Games–Howell post hoc tests were performed after ANOVA. GraphPad Prism v. 8 software (GraphPad Software, San Diego, USA) was used to create the images. Biochemical findings were analyzed with IBM SPSS v. 25.0 software (IBM Corp., Armonk, USA). A value of p < 0.05 was considered statistically significant.
Results
Biochemical findings
Oxidative and antioxidant findings in liver tissue
Favipiravir increased MDA levels more significantly at a dose of 400 mg/kg and decreased tGSH levels more significantly at a dose of 100 mg/kg (Figure 1). Also, favipiravir decreased SOD and CAT activity more significantly at a dose of 400 mg/kg compared to a dose of 100 mg/kg (Figure 2).
Nuclear factor kappa B and interleukin 6 findings in liver tissues
We found that levels of NF-kB and IL-6 in liver tissue were significantly increased at a favipiravir dose of 400 mg/kg compared to the 100 mg/kg dose (Figure 3).
Alanine aminotransferase and aspartate aminotransferase findings in the blood
Favipiravir significantly increased ALT and AST activity in comparison to the HG (p < 0.001). The ALT and AST activities were higher in the FAV-400 group than in the FAV-100 group (p < 0.001; Figure 4). Biochemical parameters are presented in Table 1.
Histopathological findings
As shown in Figure 5A, no microscopic pathological findings were found in the HG. However, moderate edema and lymphocyte infiltration were found in the FAV-100 group (Figure 5B). Moreover, severe edema, lymphocyte infiltration and hydropic degeneration were observed in the FAV-400 group (Figure 5C).
Discussion
Favipiravir has been used at high doses for the treatment of COVID-19 as its antiviral efficacy is known to be greater at such doses.18 In this study, the effects of low and high doses of favipiravir on the liver were analyzed biochemically and histopathologically. The biochemical tests revealed that a high dose of favipiravir significantly increased MDA, NF-kB, IL-6, ALT, and AST levels when compared to the low dose. A high dose also decreased the levels of antioxidants, including tGSH, SOD and CAT. The MDA is known to be one of the most important indicators of oxidative damage and is the final product of lipid peroxidation (LPO),19 a process that is associated with tissue damage.20
In healthy tissues, a small portion of oxygen is converted to reactive oxygen species (ROS) during metabolism in the mitochondria and other cellular compartments. The primary ROS include hydrogen peroxide (H2O2), superoxide radical (O2⋅−) and hydroxyl radical (OH•).19 Excessive ROS formation causes inflammatory diseases,21 oxidative stress, and damage at a molecular and cellular level.22 Results of this study, as well as information derived from other studies, indicate that favipiravir causes oxidative damage at low and high doses. Indeed, recent study showed that favipiravir caused oxidative damage by increasing MDA levels in liver tissue.15 Another study demonstrated that side effects increased as the dose of favipiravir increased, which is in agreement with our biochemical findings.10
Antioxidants are known to protect liver tissue from oxidative damage.23 Therefore, we performed antioxidant measurements to analyze liver injury. The tGSH levels in the liver tissue of animals treated with a 400 mg/kg dose of favipiravir were found to be significantly lower than in those treated with a 100 mg/kg dose. The GSH is a tripeptide that includes L-glutamate, L-cysteine and glycine, and it is one of the most remarkable antioxidants.24 Indeed, in living tissues, GSH detoxifies ROS reactions and protects cells from ROS toxicity.25 The decrease in tGSH levels in the liver tissue of animals administered favipiravir indicated that the oxidant/antioxidant balance had changed in favor of oxidants, which was determined to be oxidative stress.26 Similar results were found in another study, in which it was observed that the GSH levels of cells decreased after favipiravir treatment.27
Other parameters used to evaluate oxidative stress included SOD and CAT levels.28 Normal levels of SOD and CAT help maintain tissue integrity and function by neutralizing overproduced ROS.29 Our experimental results showed a decrease in the levels of the enzymatic antioxidants SOD and CAT in groups treated with favipiravir. Similarly, Kara et al. showed that SOD and CAT levels decreased in lung and liver tissue groups treated with favipiravir compared to healthy tissues,15 which is consistent with our experimental results.
The levels of the pro-inflammatory cytokines NF-κB and IL-6 were found to be increased in the liver tissue of animals treated with favipiravir. The NF-κB plays an important role in mediating the cellular response to damage, stress and inflammation.30 According to previous studies, favipiravir increased NF-κB and IL-6 levels in the liver,15 and increased NF-κB in primary intervertebral disc tissue cell cultures.31 At the same time, Zhao et al. also reported that favipiravir increased levels of IL-6.32 In other words, favipiravir plays an important role in inflammation by causing an increase in pro-inflammatory cytokine production.
Increased serum transaminase, an important indicator of hepatocellular damage, occurs in 8% of the population.33 In this study, favipiravir significantly increased ALT and AST levels at a dose of 100 mg/kg and 400 mg/kg. Bayram et al. found that serum ALT and AST levels were elevated in patients treated with favipiravir.34 Another study showed that ALT and AST levels increased in patients administered favipiravir for the treatment of COVID-19, which caused hepatotoxicity.13 Furthermore, a study by Izci Cetinkaya et al. showed a significant increase in ALT and AST levels in patients treated with favipiravir.35
The biochemical results obtained from the liver tissues of animals in this study aligned with the histopathological findings. Severe edema (grade 3), lymphocyte infiltration and hydropic degeneration were observed in the liver tissues of the FAV-400 group, which also had high oxidant and low antioxidant levels. However, only moderate edema (grade 2) and lymphocyte infiltration were observed in the FAV-100 group. These results indicate that increasing the dose of favipiravir increased hepatic edema, lymphocyte infiltration and hydropic degeneration. It has been reported that hydropic degeneration is associated with oxidative liver injury.36 In addition, it was shown that increased inflammatory cells and pro-inflammatory mediators were associated with ROS production.37
Limitations
The study did not investigate the effects of favipiravir in combination with other drugs in the treatment of COVID-19 or other infections. This should be clarified in future studies. Due to a very small sample size used, it is impossible to reliably verify the statistical test assumptions; therefore, the calculated p-values need to be interpreted with caution.
Conclusions
In conclusion, high dose of favipiravir caused significantly more oxidative and inflammatory injury in liver tissue compared to the low dose. Indeed, animals administered a high dose (400 mg/kg) of favipiravir had higher levels of oxidants and pro-inflammatory cytokines, as well as hydropic degeneration of the liver. This is the first study to show hydropic degeneration of the liver with the use of favipiravir. Therefore, hydropic degeneration may be associated with the excessive production of oxidants and pro-inflammatory cytokines. Due to its adverse effects on the liver, close and frequent follow-up of liver bioparameters during favipiravir therapy would be beneficial for predicting the occurrence of complications.