Abstract
Background. Since the beginning of the coronavirus disease (COVID-19) pandemic, numerous infections have been observed with various symptoms and degrees of severity. Not all patients have had a confirmation of infection made using reverse transcription polymerase chain reaction (RT-PCR) or antigen tests. It has been observed that some people, including convalescents or those without knowledge of a past infection, perform serological tests to detect anti-severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) antibodies.
Objectives. We aimed to evaluate the levels of anti-SARS-CoV-2 immunoglobulin G (IgG) antibodies in a cohort of convalescents and in individuals not previously infected, who were willing to get vaccinated. We also aimed to assess several socio-clinical factors associated with participants’ humoral responses.
Materials and methods. We recruited 298 individuals from the region of Lower Silesia who were willing to get vaccinated for SARS-CoV-2. The participants were divided into 2 groups: convalescents (group I) and participants without a past infection (group II). Several seropositive individuals in group II were identified, and they were transferred to group I, resulting in a final distribution of 171 individuals in group I and 127 individuals in group II. For serological testing, the QuantiVac anti-SARS-CoV-2 (IgG) enzyme-linked immunosorbent assay (ELISA) was used.
Results. The results showed the presence of anti-SARS-CoV-2 IgG antibodies in participants from group I, with an average number of 190.3 IU/mL. Twenty-three participants (13.45%) did not have a detectable level of antibodies despite a previous SARS-CoV-2 infection. In 21 participants (12.28%), antibodies were detected despite no previous symptoms of infection (average level: 145.0 IU/mL).
Conclusions. Older participants were more likely to experience a symptomatic SARS-CoV-2 infection, and the severity of the symptoms was related to higher antibody titers seen later after COVID-19. Numerous individuals from group II were unaware of past SARS-CoV-2 infections. In several participants, antibodies were not detected despite a previous infection.
Key words: antibodies, serology, seronegative, SARS-CoV-2, asymptomatic
Background
The first information about severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) emerged at the beginning of 2020. At present, over 12.88 billion vaccine doses have been administered worldwide, with 67.9% of the world’s population having received at least 1 dose. In Poland, over 57 million vaccines were administered in the 2 years since their introduction.1 However, we are still facing new infections in everyday clinical practice. Therefore, we are continuously in need of information regarding this topic.2
Serological tests have been used to detect antibodies produced as a result of infection. Severe disease, compared to mild disease, correlates with persistently higher antibody levels.3 There is also a small group of people, mainly those with mild/asymptomatic infections, who do not produce antibodies.4 However, it should be noted that the detection of the persistence of antibodies can vary depending on the assay used.5
Objectives
We present preliminary data on a cohort of participants qualified to receive vaccination against SARS-CoV-2. This is the 1st part of a larger study evaluating the humoral immune response to vaccination against coronavirus disease (COVID-19). The 2nd part of the analysis, entitled “How humoral response and side effects depend on the type of vaccine and past SARS-CoV-2 infection” has also been published (Vaccines. 2022:10(7):1042) and is available online (https://doi.org/10.3390/vaccines10071042).
The main aim of this study is to evaluate the levels of anti-SARS-CoV-2 immunoglobulin G (IgG) antibodies in a cohort of convalescents and not previously infected individuals from the Lower Silesia region (Poland) who were willing to get vaccinated.
Materials and methods
The inclusion criteria were: age >18 years, providing written informed consent to participate in the study and a willingness to get vaccinated. Two groups of participants were selected for the study: COVID-19 convalescents (group I) and naïve participants (group II). The exclusion criteria were: the presence of diabetes, any cancer detected within the last 5 years, chronic kidney, liver or lung diseases, acquired immunodeficiency syndrome (AIDS), or immunosuppression for any other reason.
Before each blood draw, the participants were asked to complete a questionnaire. The questions concerned previous SARS-CoV-2 infections and whether it was confirmed through testing, as well as general wellbeing, persistence of COVID-19 symptoms, adverse vaccine reactions, chronic diseases, and allergic reactions to drugs, substances and foods.
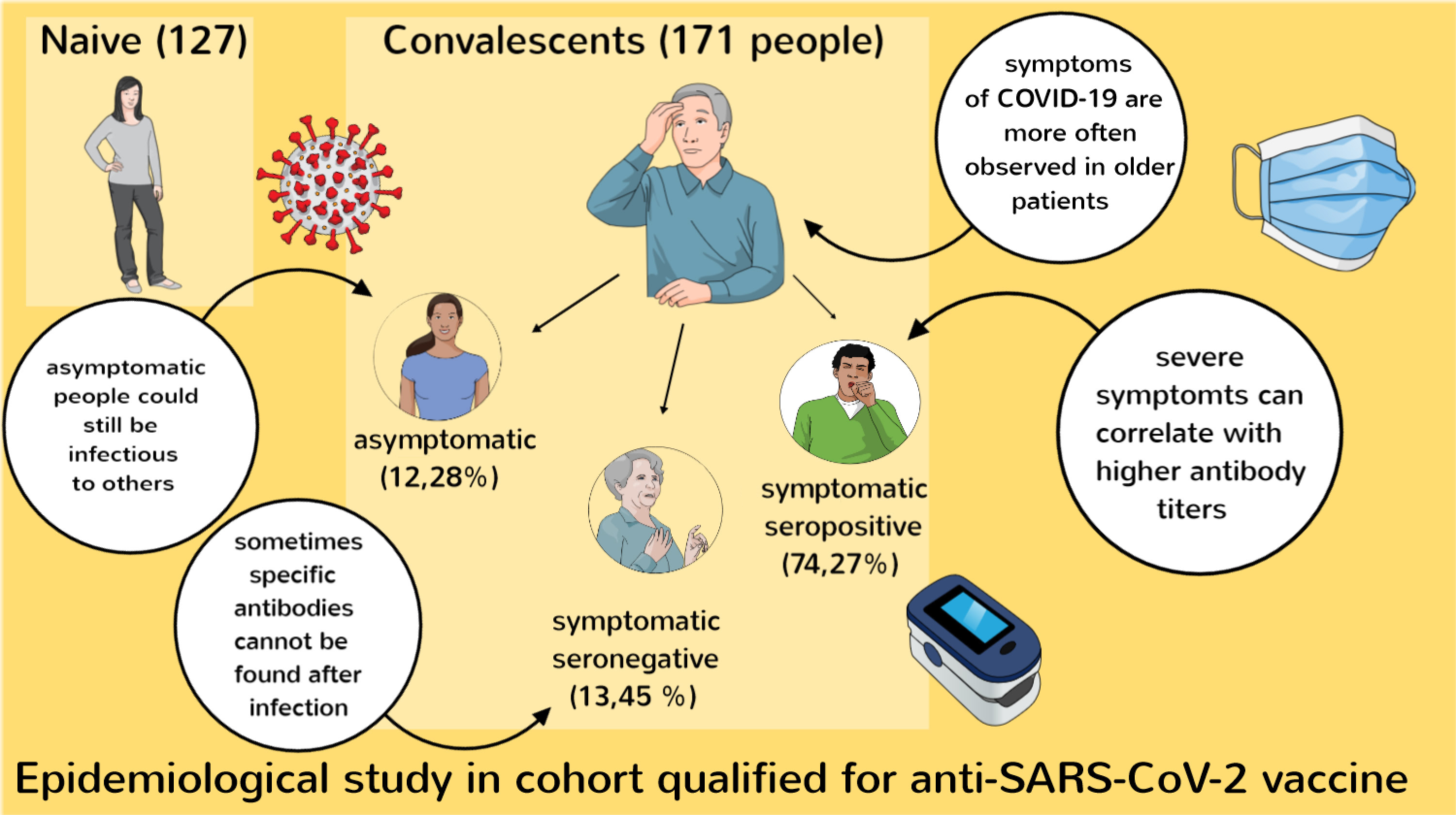
Overall, 298 participants were included in the study. After receiving results showing that 21 supposedly naïve participants had current IgG antibodies, we decided to analyze them together with the convalescent group, which resulted in the final division: group I (COVID-19 convalescents, n = 171) and group II (naïve participants, n = 127).
This single-center study was conducted from February 20, 2021, to May 19, 2021. The participants were inhabitants of the Lower Silesia region, aged 21–69 years, and were of both sexes. The participants were recruited by announcements in the local media. Due to changes in the registration rules and participants’ individual contraindications at the moment, the interval between taking the blood sample for testing and the actual vaccination date varied from 1 day to 6 weeks, usually approx. 1 week (mean: 2.00, interquartile range (IQR): 0.25–6.00, standard deviation (SD): 8.54). Before vaccination, all participants were tested for the presence of anti-SARS-CoV-2 IgG antibodies.
Plasma samples were collected using heparin, centrifuged, and stored in aliquots at −70°C for later use. The QuantiVac anti-SARS-CoV-2 (IgG) enzyme-linked immunosorbent assay (ELISA) (EUROIMMUN, Lübeck, Germany) was used for quantitative detection of anti-SARS-CoV-2 antibodies by means of 6-point calibration curve.
The ELISAs were performed and the results were evaluated as recommended by the manufacturer. Samples with an absorbance higher than the absorbance of the highest standard (386 international units (IU)/mL) were diluted and retested. The assay was standardized against the First WHO International Standard for anti-SARS-CoV-2 immunoglobin (NIBSC 20/136) and the quantitative results are given in standardized units (IU/mL).
Ethical approval
This study received approval from the Bioethics Committee of Wroclaw Medical University, Poland (approval No. 51/2021). The study was performed in accordance with the Declaration of Helsinki and the principles of good clinical practice with respect to the rights and dignity of participants.
Statistical analyses
Counts, percentages, means, medians, SDs, ranges, and lower and upper quartiles are reported where appropriate. The normality of the distributions was tested with the Shapiro–Wilk test.
Statistical significance between means for different groups was calculated using the non-parametric Kruskal–Wallis test, followed by Dunn’s post hoc tests with Bonferroni correction. Statistical significance between frequencies was calculated using the χ2 test.
A value of p < 0.05 was required to reject the null hypothesis. Statistical analyses were performed using the Statistica v. 13.3 (StatSoft Inc., Tulsa, USA) software package.
Results
All 298 participants were tested for anti-SARS-CoV-2 IgG antibodies, creating 2 groups: I (COVID-19 convalescents, n = 171) and II (naïve participants, n = 127). Table 1, Table 2 present characteristics of the groups.
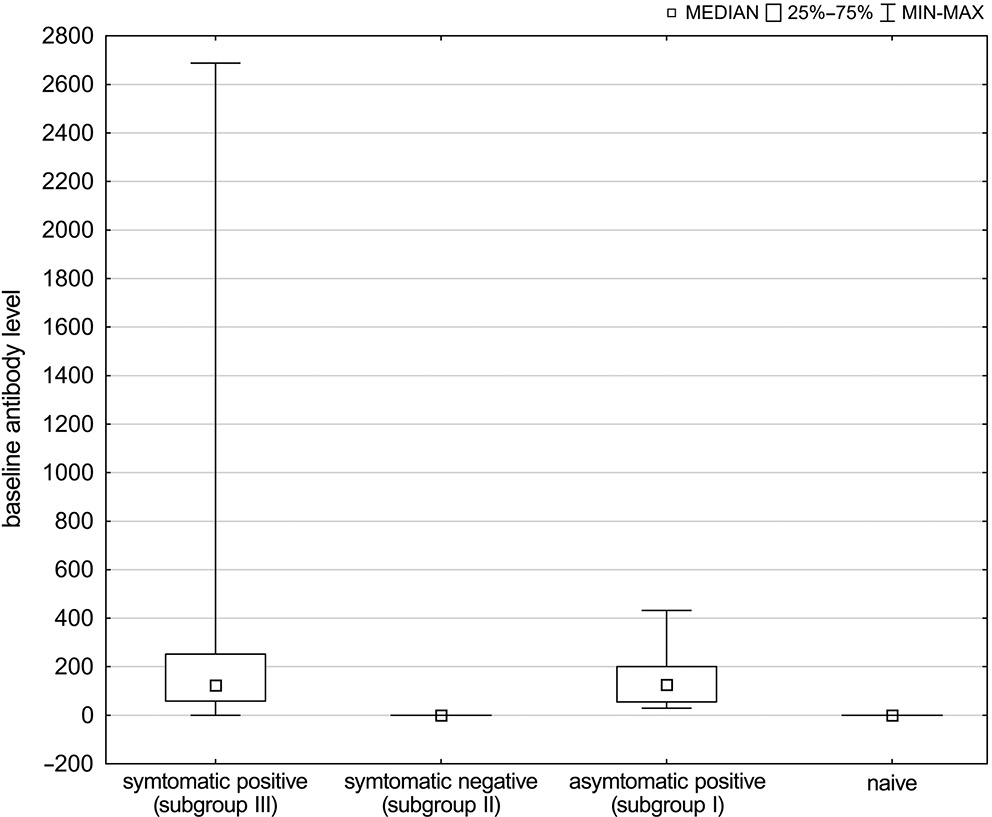
After testing the blood samples, the results showed that anti-SARS-CoV-2 IgG antibodies were present in participants from group I with a median of 105.6 (38.4–198.4) IU/mL. In group II, no spike antibodies were found (0 IU/mL). The detailed data are presented in Figure 1.
When describing our findings, we would like to underline the fact that, among the participants of group I, there can be found 3 specific subgroups based on antibody levels (Figure 1). One consists of participants without knowledge or symptoms of a previous COVID-19 infection but with positive antibody results (n = 21; 12.28% of group I, 7.05% of all participants). The 2nd group includes participants with a proven previous infection but with no antibodies found in the first blood sample (n = 23; 13.45% of I group, 7.72% of all participants). The 3rd group consists of seropositive convalescents (n = 127) with a median age of 45 (40.0–52.0) years, a maximum age of 69 years, and a median number of antibodies of 123.2 (58.7–252.8) IU/mL. In this subgroup, the time between sample collection and symptoms of the infection varied between 18 days and 7 months.
Subgroup I participants without knowledge of a previous infection were aged from 24 to 50 years, with antibody levels between 28.8 IU/mL and 432 IU/mL (median: 126.4 IU/mL, range: 54.4–2000.0 IU/mL). After receiving the serological results, we confirmed that the participants were not able to recall any possible COVID-19 manifestations.
Subgroup II includes participants with undetectable antibody levels despite the fact that they had a previous SARS-CoV-2 infection and were aged from 23 to 63 years (n = 23). The time between COVID-19 and blood sampling varied from 2.5 months to 6.5 months.
Discussion
Assuming that 4 main COVID-19 waves in 2020 and 2021 can be distinguished, the current study covers the 2nd (most convalescent participants suffered from a SARS-CoV-2 infection between October and December 2020) and 3rd wave (when we collected blood samples).
A precise analysis of group I (divided into: subgroup I – convalescents, asymptomatic; subgroup II – symptomatic, seronegative; and subgroup III – seropositive, symptomatic) showed that there was a significant age difference. In subgroup III, the age was significantly higher than in subgroup II (median 45.0 (40.0–52.0) years compared to 43.0 (33.0–46.0) years p = 0.0271, Mann–Whitney U test, U = 1038.5) and than with group II (45.0 (40.0–52.0) compared to 43.0 (39.0–47.0) p = 0.0228, Mann–Whitney U test, U = 6675,5). There was no such age difference between subgroups III and I ((45.0 (40.0–52.0) years compared to 42.0 (39.0–46.0) years, p = 0.0983, Mann–Whitney U test, U = 1032.5)). We also analyzed these data using the Scheffé’s test (mean square error (MSE) = 57057, degrees of freedom (df) = 290.00), which showed that, with regard to age, the antibody results differed between subgroup III and group II (p = 0.00000), and between subgroups II and III (p = 0.000452). These results are consistent with an earlier meta-analysis, which concluded that elderly or older participants (age ≥50 years) are at a higher risk of severe disease course.6
Furthermore, the asymptomatic subgroup may be an important link in the transmission of the virus. These participants took part in our study in mid-April–mid-May, so it is possible that they were infected during the so-called 3rd wave. It has been established that individuals without symptoms are capable of infecting others.7, 8 A similar situation occurs when participants are isolated with a delay due to developing symptoms 1–2 days after becoming infectious.9
The level of antibodies in group I differed across individuals, starting from 27.2 IU/mL, which is a borderline titer (blood sample taken 6.5 months after infection), and going up to 2688.2 IU/mL (blood sample taken 2.5 months after infection). The average titer was 190.3 IU/mL. These differences in titer may be explained by the varying times between SARS-CoV-2 infection and testing. It has been observed that spike antibody levels are relatively stable for several months before waning.10 In addition, antibody levels are better sustained in participants with severe symptoms.11 Our participants with borderline titers had no or mild symptoms, whereas the participant with the highest result was hospitalized due to the severity of COVID-19.12 These findings, together with the theory that some people are serological non-responders,13 can also partially explain why a subgroup of 23 participants (subgroup II) did not have a detectable level of antibodies.
Limitations
There are several limitations to the current study. First, there are uneven sample sizes due to the presence of asymptomatic convalescents. In addition, there were irregular time intervals between taking blood samples and receiving first vaccine dose.
Conclusions
The symptoms of COVID-19 were more often observed in older participants, and the severity of the disease can correlate with higher antibody titers seen later after COVID-19 compared to a mild infection. One should be aware that numerous participants without symptoms of past SARS-CoV-2 infection can transmit the virus to other people. Serological data are not an unambiguous evidence of a past infection.