Abstract
Background. Time-to-event modeling is gaining importance in drug dosage determination, particularly using pharmacometrics approaches.
Objectives. To evaluate the various time-to-event models for estimating the time to achieve a stable warfarin dose in the Bahraini population.
Materials and methods. A cross-sectional study evaluating the non-genetic and genetic covariates (single nucleotide polymorphisms (SNPs) in CYP2C9, VKORC1 and CYP4F2 genotypes) was conducted in patients receiving warfarin for at least 6 months. Time to achieving a stable dose of warfarin was defined as the duration (in days) from the day of initiating warfarin until 2 consecutive prothrombin time-international normalized ratio (PT-INR) values were observed in the therapeutic range with a gap of at least 7 days. Exponential, Gompertz, log-logistics, and Weibull models were tested, and the model with the lowest objective function value (OFV) was chosen. The covariate selection was carried out using the Wald test and OFV. A hazard ratio (HR) with a 95% confidence interval (95% CI) was estimated.
Results. A total of 218 participants were included in the study. The Weibull model was observed to have the lowest OFV (1989.82). The expected time to reach a stable dose in the population was 21.35 days. The CYP2C9 genotypes were identified as the only significant covariate. The HR (95% CI) for achieving a stable warfarin dose within 6 months of initiation for individuals with CYP2C9 *1/*2 was 0.2 (0.09, 0.3), 0.2 (0.1, 0.5) for CYP2C9 *1/*3, 0.14 (0.04, 0.6) for CYP2C9 *2/*2, 0.2 (0.03, 0.9) for CYP2C9 *2/*3, and 0.8 (0.45, 0.9) for those with the C/T genotype for CYP4F2.
Conclusions. We estimated the population time-to-event parameters for achieving a stable warfarin dose in our population and observed CYP2C9 genotypes to be the main predictor covariate followed by CYP4F2. The influence of these SNPs needs to be validated in a prospective study and an algorithm to predict a stable warfarin dose and the time to achieve it needs to be developed.
Key words: warfarin, Weibull, Gompertz, time-to-event, exponential
Introduction
Warfarin, a commonly administered oral anticoagulant, poses a lot of challenges clinically, owing to inter- and intra-individual variabilities, drug and food interactions, and the influence of genetic polymorphisms on its metabolism and pharmacodynamic target enzymes.1 Though several novel oral anticoagulants (NOACs) are more efficacious and safer, warfarin is still the preferred, cost-effective drug in most developing nations. Among genetic polymorphisms, cytochrome P450 2C9 (CYP2C9), CYP4F2 and vitamin K epoxide reductase complex subunit 1 (VKORC1) have been shown to affect the therapeutic response to warfarin.2
Pharmacometrics is the quantification of pharmacokinetic and pharmacodynamic data with the use of appropriate mathematical models to help determine dosage regimens. Pharmacometrics time-to-event modeling has recently gained importance, wherein various models can be applied to predict the median time for achieving an outcome and to identify factors that significantly affect the outcome variable.3 Commonly used models include exponential – when the hazard ratio (HR) is expected to be constant – and Weibull, Gompertz and log-logistics, when the HR is expected to change over time.4 Several clinical trials and observational studies evaluating the role of genetic and non-genetic factors included outcomes on the anticoagulation control, warfarin stable dose and bleeding episodes. However, hardly any studies assessed the time taken to reach a stable warfarin dose and the associated factors. Achieving a stable dose of warfarin is clinically important for optimized therapeutic goals so that the risk of thrombosis and/or hemorrhage can be minimized.
Previously, we conducted a warfarin pharmacogenetics study in the Bahraini population5 and in the present study, we performed a secondary analysis of the data evaluating various time-to-event models for ascertaining the time to achieve a stable warfarin dose in the studied population.
Objectives
To evaluate various time-to-event models for estimating the time to achieve a stable warfarin dose in the Bahraini population.
Materials and methods
Study design and ethics
The analysis was carried out as a part of the warfarin pharmacogenomics study that was cross-sectional in design. The approvals from the Institutional Ethics Committee of Arabian Gulf University (Manama, Bahrain; approval No. E-024-PI-11/18), and the Research Technical Support Team of the Ministry of Health of Bahrain (approval No. AURS 301/2019) were granted. Consent was obtained from the study participants. This study was carried out in compliance with the latest Declaration of Helsinki guidelines.
Study procedure
We included patients who were enrolled in the anticoagulation clinic in a tertiary care hospital receiving warfarin for at least 6 months and with the following details available: age, gender, CHA2DS2-VASc (congestive heart failure history, age, sex, hypertension history, stroke/transient ischemic attack/thromboembolism history, vascular disease history, and diabetes history) score, HASBLED (hypertension, abnormal renal/liver function, stroke, bleeding, labile international normalized ratio (INR), elderly, and interacting drugs/alcohol) score, and SAMe-TT2R2 (sex, age, history of concomitant diseases, interacting drugs, tobacco use, and race) score. We evaluated genotypes of CYP2C9, VKORC1 and CYP4F2 using reverse transcription polymerase chain reaction (RT-PCR), as described previously.5 Statins, proton pump inhibitors, carbamazepine, and amiodarone were considered as drugs with potential interaction with warfarin. The following genotypes were considered wild-type: *1/*1 for CYP2C9, C/C for VKORC1 and C/C for CYP4F2. Therapeutic prothrombin time (PT)-INR categories were defined as either between 2.5 and 3.5 for those with heart valve replacement, or between 2 and 3 for others.
Outcome variable
Time to achieve a stable dose of warfarin was defined as the duration (in days) from the day of initiating warfarin until 2 consecutive PT-INR values were observed in the therapeutic range with a gap of at least 7 days.6
Time-to-event modeling
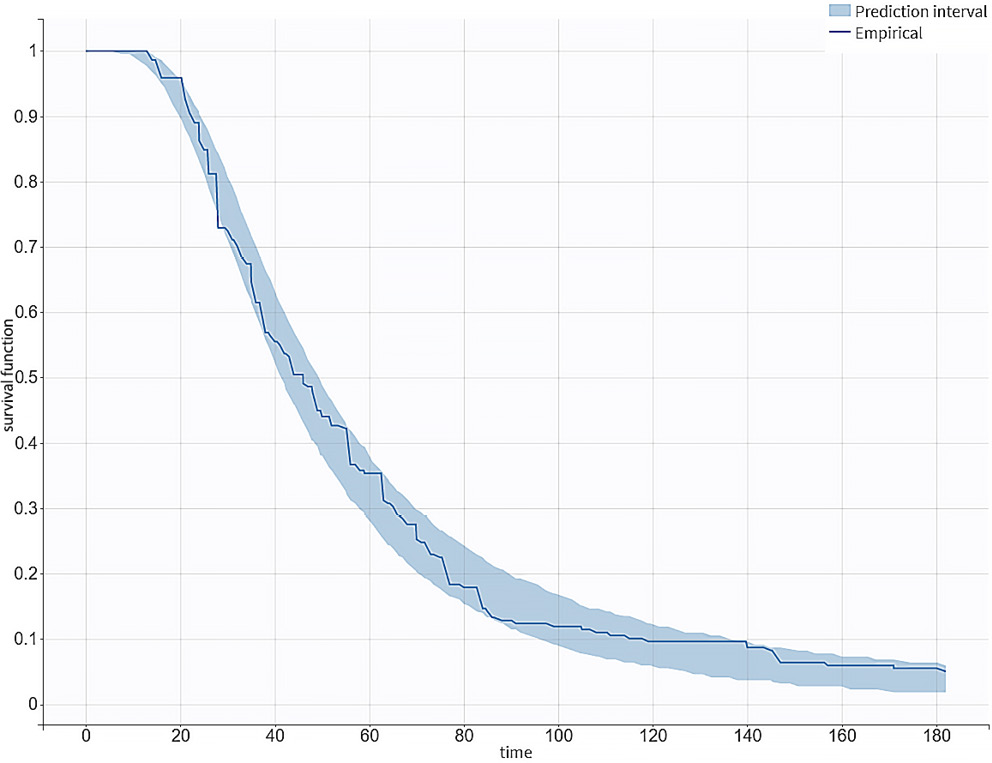
Time to achieve a stable warfarin dose was the outcome variable with the following covariates: age, gender, CHA2DS2-VASc score, HASBLED score, SAMe-TT2R2 score, presence of any drugs with potential interaction, PT-INR categories (either between 2.5 and 3.5 or between 2 and 3), and genotypes of CYP2C9, VKORC1 and CYP4F2. We explored the following base models: exponential, Gompertz, log-logistics, and Weibull. The model with the lowest (minimum) objective function value (OFV) was chosen. Both the Wald test and a decline in OFV by at least 3.84 points were used as criteria for defining significant covariates.7 The HRs were calculated for the significant covariates using standard formulae.8 A visual predictive check (VPC) was used for evaluating the model fit.
The Monolix v. 2021R1 (http://lixoft.com/products/monolix/; Lixoft SAS, Antony, France) was used for modeling the outcome variable.
Results
Demographics
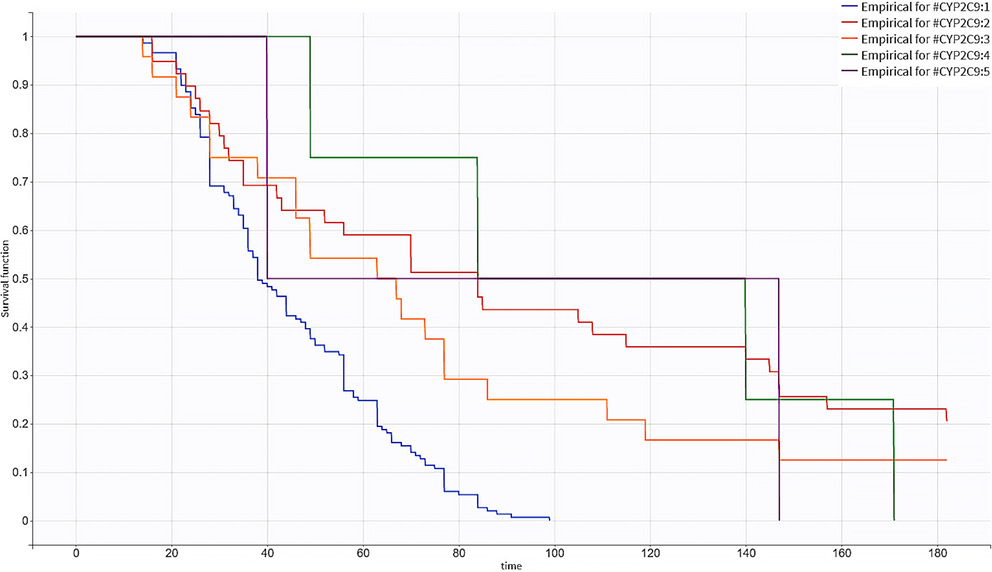
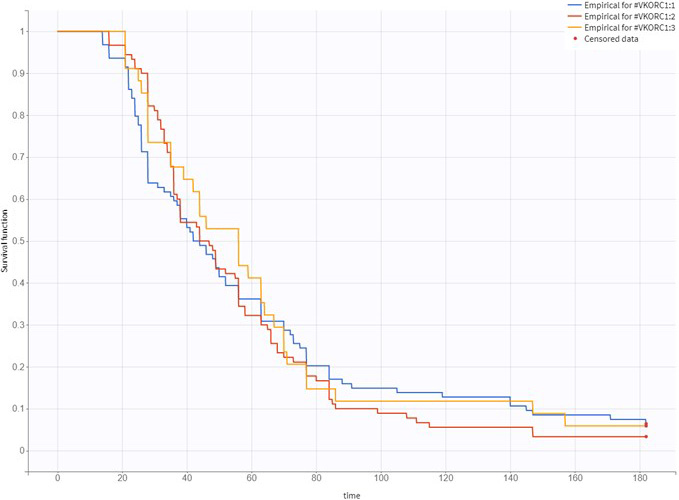
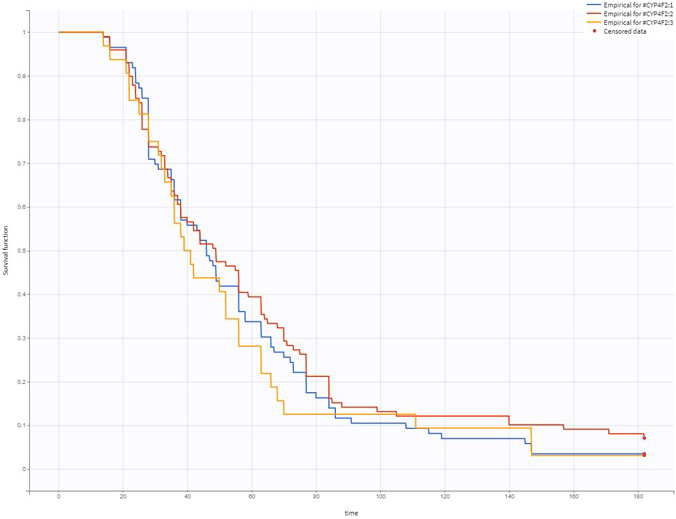
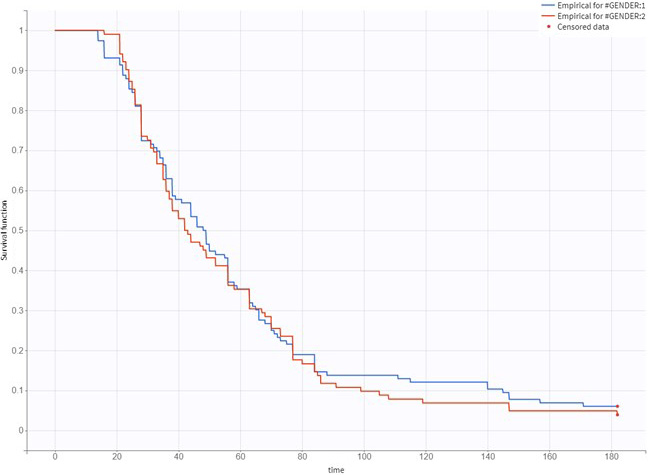
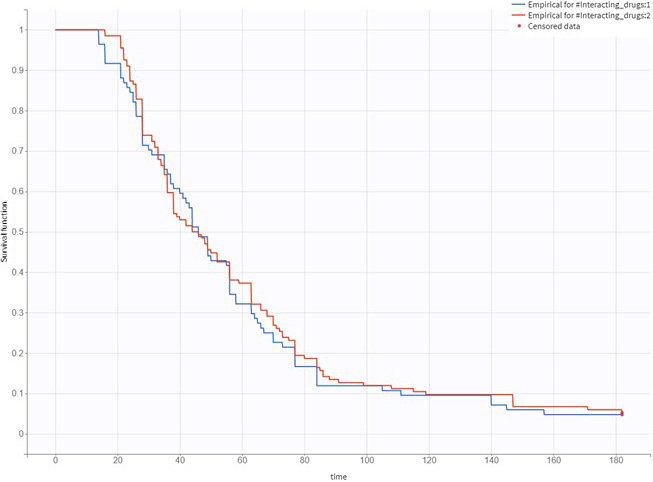
The study involved 218 participants. Table 1 shows a summary of their demographic characteristics. Among the participants, 206 achieved a stable dose of warfarin within 6 months, with the median (range) time required for achieving the stable dose being 44 (14–171) days. The Kaplan–Meier curve for achieving a stable warfarin dose as stratified by CYP2C9 genotype is depicted in Figure 1, and the curves according to gender, potentially interacting drugs, and CYP4F2 and VKORC1 genotypes are depicted in Figure 2, Figure 3, Figure 4, Figure 5.
Time-to-event model
The Weibull model was found to have the lowest OFV among the base models (Table 2). The CYP2C9 improved the base model, followed by the addition of the CYP4F2 genotype. Population time-to-event parameters are listed in Table 3. The Wald statistics revealed a significant prolongation of time to achieve a stable warfarin dose with CYP2C9 genotypes and a shortened time with CYP4F2 genotypes. The model used for evaluating the effect of significant genotype covariates is outlined as follows.
The HR (95% confidence interval (95% CI)) for achieving a stable warfarin dose within 6 months of initiation for individuals with CYP2C9 *1/*2 was 0.2 (0.09, 0.3), 0.2 (0.1, 0.5) for CYP2C9 *1/*3, 0.14 (0.04, 0.6) for CYP2C9 *2/*2, and 0.2 (0.03, 0.9) for those with CYP2C9 *2/*3. Similarly, the HR (95% CI) for the CYP4F2 C/T genotype was 0.8 (0.45, 0.9), but it was not significant for the T/T genotype (1.4 (0.9, 1.3)) when compared to the C/C genotype.
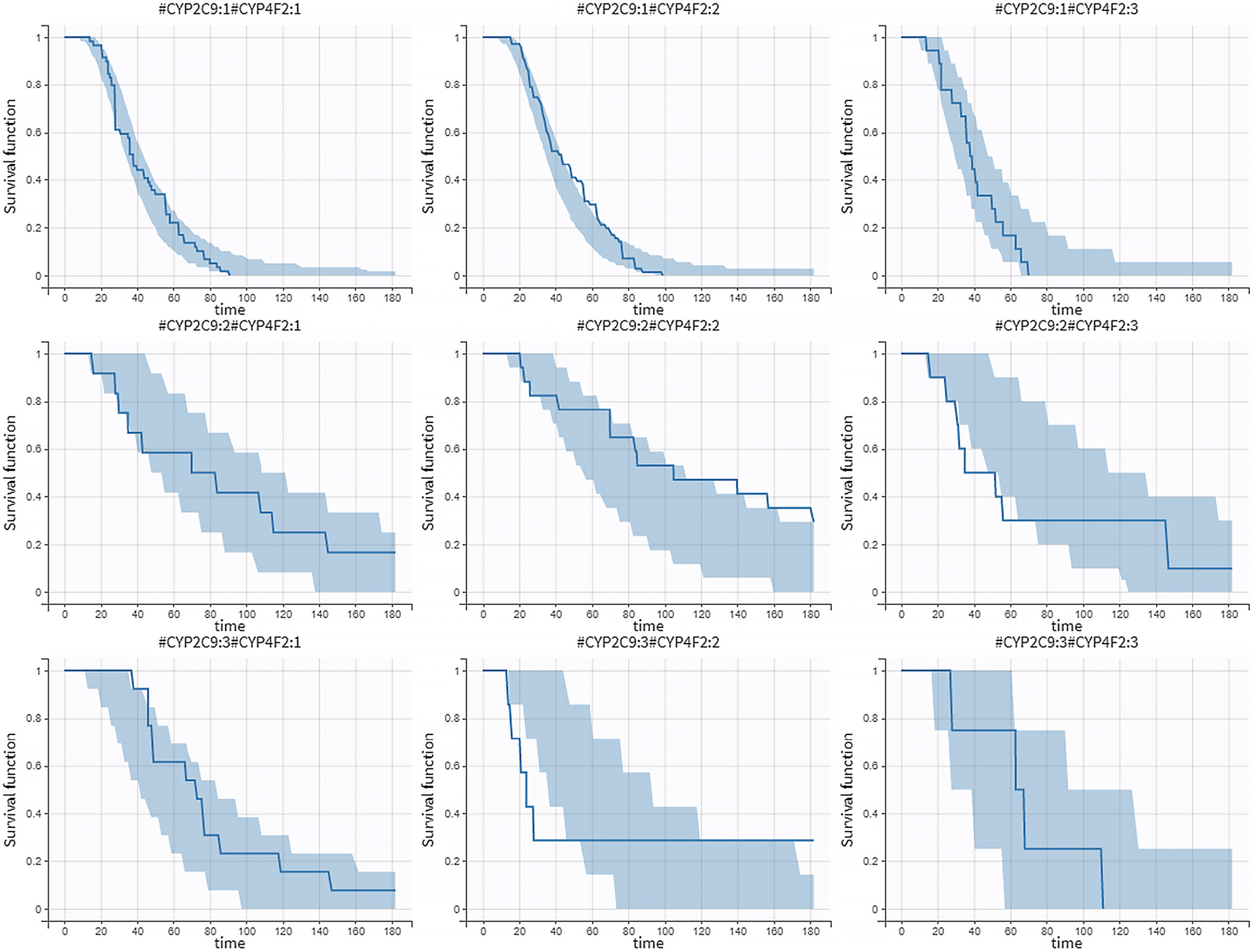
The VPC revealed a good fit of the model to the observed data (Figure 6). Those with CYP2C9 *1/*1 and CYP4F2 C/C genotypes were observed to have a shorter time to achieve a stable warfarin dose compared to other genotype categories (Figure 7).
Discussion
Key findings from the study
This pharmacometrics-based modeling study was carried out to evaluate the time needed to achieve a stable warfarin dose in 218 patients. We established the population-specific parameters associated with time to reach a stable warfarin dose, and observed that CYP2C9 and CYP4F2 genotypes are the predominant predictor covariates. Those with CYP2C9*1/*1 and CYP4F2C/C genotypes were observed to achieve the stable warfarin dose much earlier, and those with genetic polymorphisms in CYP2C9 were less likely to reach the stable dose at 6 months.
Comparison with existing literature
This is the first study evaluating the time needed to reach a stable warfarin dose using pharmacometrics time-to-event modeling. Combined CYP2C9 and VKORC1 polymorphisms are recommended by the US Food and Drug Administration (FDA) for determining the appropriate warfarin dose.2 We observed that those with the wild-type CYP2C9 genotype achieved the stable dose sooner compared to those with genetic polymorphisms. This finding reiterates the importance of determining the CYP2C9 genotype as soon as possible when a patient has been identified as requiring warfarin therapy, so that genotype-based warfarin dosing can be initiated to aid in achieving the stable dose sooner, particularly when genetic polymorphisms are identified. The existing standard of care is to initiate warfarin at a fixed dose (5 mg/day) and titrate based on the therapeutic response. In the same population, we have observed that genetic polymorphisms in CYP2C9 require lower warfarin doses for achieving therapeutic PT-INR.5 Nearly 31.8% of the population was observed to carry at least 1 functionally variant CYP2C9 allele that necessitates a lower warfarin dose to be administered for achieving an optimal PT-INR value. Even then, those with at least 1 single nucleotide polymorphism (SNP) in CYP2C9 were observed to take a significantly longer time to achieve a stable dose. Such candidates are at an increased risk of thromboembolic attacks for bleeding episodes until a stable dose is achieved, and thus need either intense monitoring or serve as likely candidates for receiving a NOAC.
Limitations
The study is limited by a lack of external validation of the model using data from prospective patients.
Conclusions
In this study, we have estimated the time-to-event parameters for achieving a stable warfarin dose in Bahraini population. The CYP2C9 has been observed to be the main predictor covariate, followed by the CYP4F2 genotype. Those with at least one SNP in CYP2C9 were observed to have a significantly lower likelihood of achieving a stable warfarin dose, as follows: CYP2C9 *1/*2: 0.2 (0.09, 0.3), CYP2C9 *1/*3: 0.2 (0.1, 0.5), CYP2C9 *2/*2: 0.14 (0.04, 0.6), and for those with CYP2C9 *2/*3: 0.2 (0.03, 0.9). There is a need to validate the influence of these SNPs in a prospective study and develop an algorithm predicting a stable warfarin dose and time to achieve it.
Data availability statement
The data are available from the corresponding author upon reasonable request.