Abstract
Background. The complex clinical status of modern day patients hospitalized due to acute heart failure (AHF) results from their advanced age, comorbidities, frailty, heart failure symptoms (including massive swelling of the lower limb), and dramatic reduction of exercise tolerance. Hence, there is a need to implement physiotherapeutic procedures as early as possible, aiming to both accelerate the restoration of clinical stabilization and prevent post-hospital disability.
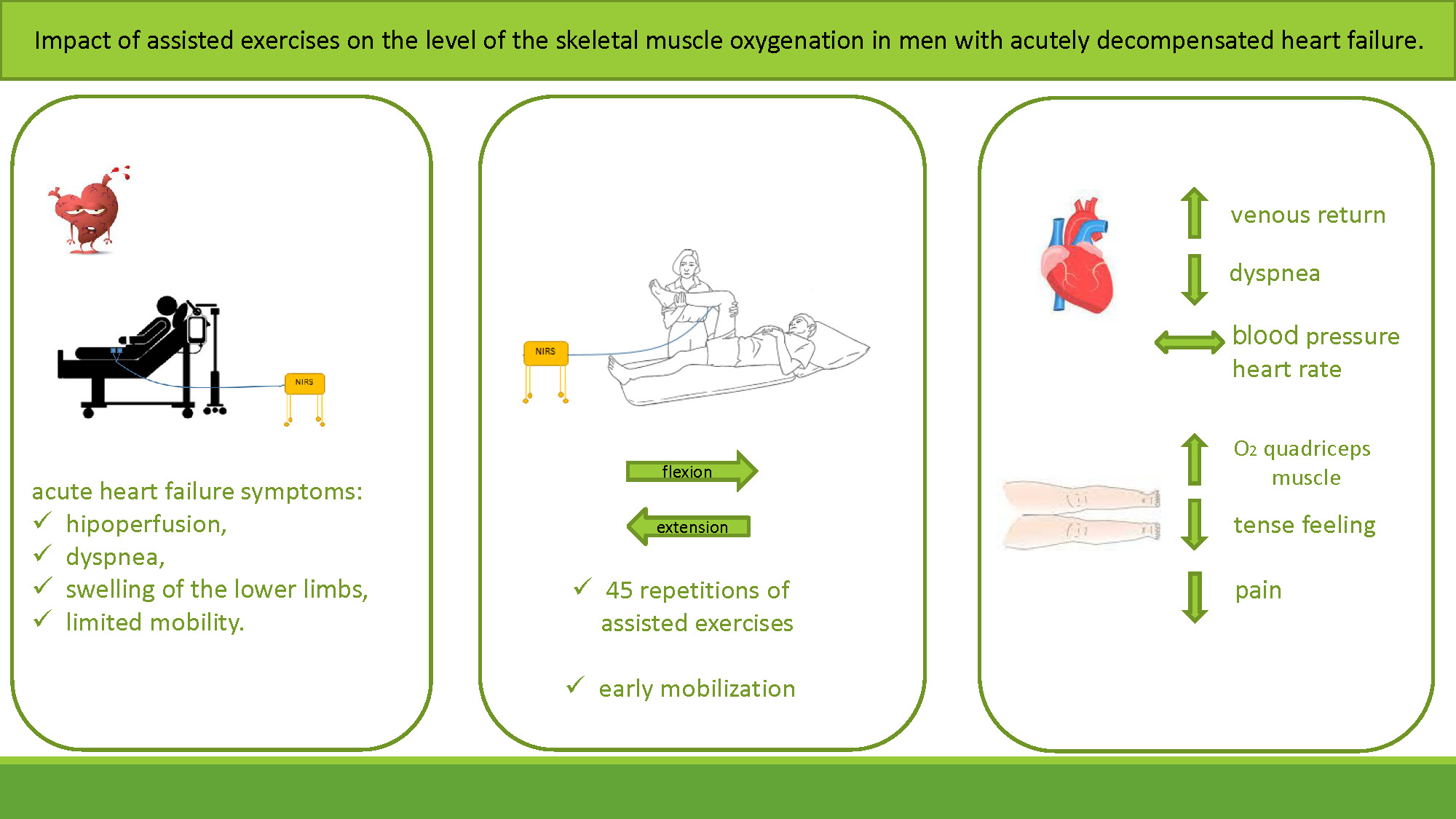
Objectives. We investigated whether assisted lower limb exercises have an impact on perfusion and oxygenation in skeletal muscle and if they are feasible in patients with AHF.
Materials and methods. We examined 34 men (age: 66 ±11 years; left ventricular ejection fraction (LVEF): 34 ±11%; clinical presentation: 31 wet-warm and 3 wet-cold). The intervention (carried out on the 2nd day of hospitalization) included: 1) a 3-minute rest period; 2) an exercise phase (45 repetitions of assisted flexion and extension of the lower limb; and 3) a 10-minute relaxation period. We analyzed blood pressure (BP), heart rate (HR), respiratory rate (RR), tissue oxygenation (reflected by oxygen saturation measured with a pulse oximeter), and changes in peripheral tissue perfusion (reflected by the tissue oxygenation index (TOI) measured with near-infrared spectroscopy (NIRS)).
Results. The hemodynamic parameters (both ∆HR and ∆systolic BP) and oxygen saturation did not change (all p > 0.05), whereas the RR declined (p < 0.001). The exercises improved venous outflow (reflected by decreased oxygenated, deoxygenated and total hemoglobin, all p < 0.05) and increased peripheral tissue perfusion, as reflected by the TOI (p < 0.05). The patients reported relief and lack of dyspnea during and after the assisted exercises.
Conclusions. The physiotherapeutic intervention improved both venous outflow and muscle oxygenation in men with AHF. The presented protocol was safe, feasible and well-tolerated, and resulted in relief for the patients. We believe that such procedures might be recommended for the initial period of rehabilitation in this challenging subgroup of patients.
Key words: physiotherapy, heart failure, near-infrared spectroscopy, skeletal muscle oxygenation
Background
Morbidity and mortality due to heart failure increase every year, affecting more than 10% of people over the age of 70.1 A fundamental aspect of heart failure is periods of exacerbation, known as cardiac decompensation or acute heart failure (AHF).2 Efficient therapeutic options for patients with AHF have been evolving over the last 2 decades and have resulted in an extension of life expectancy and significant changes to their clinical profile compared to 20 years ago. Therefore, patients hospitalized for AHF are now older, have more comorbidities, such as frailty syndrome, and a worse prognosis.3, 4, 5
It is noteworthy that hospitalization of patients due to AHF may have additional negative consequences, such as the so-called “post-hospital syndrome”, which is associated with an increased risk of disability.6, 7 Studies have demonstrated that up to 60% of AHF patients develop hospital-acquired disability, which is associated with a worse clinical outcome.8
Hospitalization due to AHF is associated with extended time spent lying in bed, especially for patients who are unable to walk independently. As a result, some experience a new major physical disability following hospital discharge.9, 10
The data suggest that exercise and early rehabilitation protocols applied during acute hospitalization may prevent functional and cognitive decline in older patients. Additionally, these treatments are associated with shorter hospital stay and lower costs.11, 12, 13 It is very likely that physiotherapeutic interventions implemented as early as possible during hospitalization due to AHF may mitigate these unfavorable outcomes.10, 14, 15, 16 However, cardiac physiotherapy implemented early in AHF has not been well established. It is generally accepted that such patients require special physiotherapeutic care.17, 18
In AHF, the patient’s clinical picture can vary greatly. One major symptom is massive swelling of the lower limbs, which limits the patient’s mobility or even makes it impossible to move, forcing the patient to remain in a lying or sitting position. Additionally, AHF is associated with an increased level of fatigue and dyspnea associated with fear of exacerbation of symptoms during physical exercise.18
Having these considerations in mind, we selected assisted exercises for being investigated in this study. Importantly, there is currently no available literature describing the feasibility and effects of assisted exercises in patients in the acute stages of heart failure.
Objectives
The aim of this study was to evaluate the feasibility of assisted lower limb exercises and to examine their impact on the levels of perfusion and oxygenation in the skeletal muscles of patients with AHF. We decided to analyze this at 3 levels: level related to the patient’s subjective perception, hemodynamic level (heart rate (HR) and blood pressure (BP) response) and tissue level (i.e., tissue oxygenation).
Materials and methods
Study design and participants
The study included patients with AHF. Acute heart failure was defined according to the current 2021 European Society of Cardiology (ESC) Guidelines2 as a rapid or gradual onset of symptoms and/or signs of heart failure requiring unplanned hospitalization of the patient. The use of an intravenous loop diuretic and/or vasodilator was not necessary to participate in this study.
This study was registered at Clinicaltrials.gov (ID No. NCT03102164). All study participants provided written informed consent to participate in the study.
Patients were excluded if they met any of the following exclusion criteria: acute coronary syndrome, bacterial infection, pre-existing chronic respiratory failure, need for invasive mechanical ventilation, significant arrhythmia (arrhythmia that was hemodynamically unstable and/or needed urgent targeted treatment) or conductivity disorders, anemia (hemoglobin <9 g%), active neoplastic process, liver injury (aspartate aminotransferase (AST) or alanine aminotransferase (ALT) more than 3 times the reference level), or chronic kidney failure with creatinine clearance <30 mL/min. Patients with acute deep vein thrombosis or significant orthopedic diseases of the lower limb were also excluded. Finally, patients with massive swelling and/or shortness of breath making it impossible to lie down were excluded.
Procedure and exercise settings
Patients were recruited into the study by doctors who exercised medical supervision of the rehabilitation process. A total of 34 men with AHF participated in the study. The clinical characteristics of the study group are presented in Table 1.
The tests were carried out in the patient’s room at the cardiology department on their 2nd day of hospitalization. No noninvasive ventilation was used during the study.
The study protocol was divided into 3 stages:
1) Recording of the resting parameters (3 min), during which the accuracy of measurement and recordings were simultaneously monitored.
2) Assisted exercise phase. Assisted exercise refers to exercise in which a physiotherapist alone facilitates the movement by relieving the exercised limb. The movement is performed with a minimal use of the patient’s muscle strength.19 During this phase, the physiotherapist performed 45 repetitions of assisted exercise, involving flexion of the studied lower limb at the knee and hip joints. For the hip joint, the angle of flexion was in the 45–90° range, and the knee joint was expected to be at an obtuse angle of 90–100° (heel raised above the knee). The exercise duration was approx. 1.5 min.
3) Relaxation phase. Relaxation phase comprised of a 10-minute measurement. Care was taken to ensure the patient lay still during this time and does not make any movements.
Variables and data sources
The impact of the exercises on the following factors was assessed:
1) Subjective feelings of the patient. After the exercise, the patient was asked to provide information (feedback) as to whether the exercise: affected their discomfort levels, tissue tension and lower limb pain (unchanged ↔/increased ↑/decreased ↓); provoked or increased their feeling of breathlessness (yes/no).
2) Hemodynamic parameters. Each examination was conducted with supervision and monitoring of the following hemodynamic parameters: HR, BP and respiratory rate (RR).
3) Tissue oxygenation. In addition to standard saturation assessment measured with a pulse oximeter, near-infrared spectroscopy (NIRS) was used to assess tissue oxygenation. This was important considering the reduced peripheral perfusion and frequent swelling of the lower limbs experienced by patients with AHF. Near-infrared spectroscopy is a tool used for continuous noninvasive monitoring of changes in peripheral tissue perfusion.20 Near-infrared spectroscopy is based on the absorption of near-infrared photons by chromophores (e.g., hemoglobin). Differences in the intensity of NIRS absorption by oxygenated and reduced hemoglobin were analyzed using algorithms based on the Beer–Lambert law. The NIRS light emitted by the sensor’s diode penetrated locally into the tissue underneath, and the hemoglobin level in the microcirculation was measured with 2 detectors.21, 22, 23 The photosensor was placed on the subject’s quadriceps muscle, 1/3 of the way to the proximal lateral head. The distance between the sensors in the photosensor was 4 cm; thus, the sensor’s penetration depth was up to 4 cm. Commonly, NIRS is used to check the dynamic balance between the delivery and extraction of oxygen in the tissue during surgery or exercise.20 We used NIRS to assess leg muscle perfusion and to indirectly determine venous outflow during the assisted exercises. The following parameters were measured and subsequently analyzed: tissue oxygenation index (TOI, i.e., the percentage ratio of oxygenated hemoglobin to total hemoglobin) and changes in oxygenated hemoglobin (ΔO2Hb), deoxygenated hemoglobin (ΔHHb) and total hemoglobin (Δtotal Hb).
The research was carried out as part of the Sonata 6 UMO-2013/11/D/NZ7/00922 research grant awarded by the National Science Centre, Kraków, Poland. It was conducted in accordance with the Declaration of Helsinki and approved by the Bioethics Committee of Wroclaw Medical University, Poland (approval No. 204/2015).
Statistical analyses
Continuous variables with normal distribution were described using mean ± standard deviation (M ±SD). Variables with skewed distribution were described as medians with upper and lower quartiles. Categorical variables were given as numbers and percentages. In the case of a nonparametric test for 2 trials of dependent variables, a Wilcoxon signed-rank test was used to analyze changes in the hemodynamic parameters before and after the exercise. The differences between particular phases of exercise were analyzed using the Friedman’s analysis of variance (ANOVA) with determination of the Kendall’s compliance factor. A value of p < 0.05 was considered statistically significant. Statistical analyses were performed using Statistica v. 12 (StatSoft, Tulsa, USA).
Results
The assisted exercises were well tolerated by the patients. After the series of 45 repetitions of the assisted exercise, BP and HR remained unchanged, while a decrease in RR was observed (Table 2). No adverse symptoms were reported during the exercises or directly after (Table 3).
The exercises did not change the saturation levels (p = 0.11); their value remained at 95% (93–96%). The percentage changes in selected oxygenation parameters of the quadriceps muscle during the various phases of testing are presented in the Figures. Figure 1, Figure 2, Figure 3, Figure 4 present the values of changes in the ∆O2Hb, ∆HHb, ∆total Hb, and TOI at the following test phases: at rest, during the assisted exercise, and after the 1st and 10th min of relaxation following exercise completion.
A decrease in the level of oxygenated hemoglobin was achieved as a result of the assisted exercises. In the 1st minute of relaxation, an upward trend was observed, only to return to the resting value in the final phase (Figure 1). A significant decrease in the levels of deoxygenated hemoglobin and total hemoglobin was achieved as a result of the assisted exercises. In the 1st minute of relaxation, an upward trend was observed, only to return to the resting value in the final phase (Figure 2, Figure 3). After performing the assisted exercises, a slight increase in oxygenation in the quadriceps muscle of the thigh was observed in the later phase of rest (p < 0.03) (Figure 4).
Discussion
The increased frequency of peripheral swelling and resting dyspnea during a decompensated heart failure episode significantly limits the individual’s ability to independently perform basic activities. Consequently, the management of AHF by a physiotherapist remains an ongoing challenge.
This research demonstrated that the assisted exercises studied were safe and accepted by AHF patients. Also, no adverse symptoms were observed. The majority of patients reported relief due to reduced discomfort and lower tissue tension in the lower limb. We also observed a decrease in the oxygenated hemoglobin, deoxygenated hemoglobin and total hemoglobin levels, accompanied by a slight increase in oxygenation in the quadriceps muscle of the thigh in the later phase of rest.
Most patients were of the warm-wet profile (i.e., with peripheral stagnation and lower limb swelling), which is a group of patients to which these exercises are dedicated.
Additionally, we showed that the assisted exercises applied in this study are safe because they do not adversely affect the hemodynamic parameters (no changes in HR and RR). Unfavorable tendencies of decreased systolic BP or HR changes were not observed; these have an adverse impact on the prognosis in AHF.24, 25 In evaluating the supply and use of oxygen by the skeletal muscles, we used NIRS, whose suitability has previously been demonstrated.26, 27, 28 Near-infrared spectroscopy has commonly been used to evaluate the dynamic balance between the delivery and extraction of oxygen in the tissue during surgery or exercise.20, 29
In a study by Belardinelli et al., changes in skeletal muscle oxygen saturation were evaluated during incremental exercise, both in patients with chronic heart failure (CHF) and in healthy subjects. It was observed that patients with CHF had reduced muscle oxygenation during exercise at any level of intensity compared to the control group. Additionally, patients with CHF had earlier acceleration in muscle deoxygenation, indicating a premature onset of anaerobic metabolism.30, 31 Although the abovementioned publications evaluated lower limb skeletal muscle oxygenation in patients with heart failure, several disparities prevent us from comparing them with our results. The study by Belardinelli et al. involved patients with stable heart failure (New York Heart Association (NYHA) class II), whereas our project included subjects with AHF (NYHA class IV). Moreover, the studies differ in the type of exercise analyzed: dynamic exercise with a constant or increasing workload of several minutes on an ergometer cannot be compared with assisted exercises performed in clinically unstable patients. Assisted exercises last approx. 2 min and are performed by a physiotherapist, with minimal work performed by the patient.30 We were unable to identify any studies in the available literature that had evaluated the impact of assisted exercises on the level of skeletal muscle oxygenation in patients with AHF. Thus, our results cannot be directly compared with the results of other studies.
The purpose of using NIRS in our study was not to assess oxygen extraction and metabolism, but blood flow and fluid distribution in the venous circulation, as well as to evaluate perfusion changes in the lower limb muscles under the influence of elevation and assisted movement. The NIRS tissue oxygen saturation measurement is made on a mixture of both venous and arterial blood with a mean ratio of 70% venous to 30% arterial. It has been empirically determined that the same mean ratio of 70% venous to 30% arterial blood volume applies to the whole body.32 This suggests that increased venous outflow from the peripheral tissues leads to a reduction in peripheral stasis, which may contribute to relief of the right ventricle. We can also assume that an increased venous outflow results in a better outflow of metabolites (such as H+, lactate, and prostaglandins PGE2 and PGF1), which may in turn positively affect a response from the ergoreceptors.33 In the pathogenesis of heart failure symptoms, an increased response from the ergoreceptors is of major importance. It causes increased stimulation of the sympathetic nervous system, increased peripheral resistance, excessive ventilation, and increased dyspnea.34
In our study, we observed increased venous outflow as a result of the assisted exercises with elevation of the lower limbs taken into account; however, the exercises did not (as one might expected) increase dyspnea in patients. On the contrary, the RR decreased. Based on these results, we can assume that the increased outflow of venous blood may contribute to an improvement in outflow of metabolites, reduction of the ergoreflex, and, consequently, reduction of sympathetic activation and dyspnea.
Patients with CHF have a decreased functional efficiency due to age, comorbidities and skeletal muscle dysfunction.35, 36, 37 Additionally, frailty syndrome, a consequence of decompensation, hospitalization and bed rest, leads to a sharp worsening of functional deficits, disease progression and a poorer prognosis.4, 5 In order to increase their chances of returning to baseline functional physical fitness or to minimize the deficits resulting from AHF, it is necessary to start the rehabilitation as soon as possible.15, 38
The assumption of our research was to commence physical exercises, making it the first step as an introduction to the rehabilitation program of patients who are weak and bed-bound, helping them regain independence and mobility. Providing a uniform rehabilitation program and simple recommendations during an AHF episode is not possible. This is due to the varied clinical picture and diverse decompensation etiologies of the underlying disease or comorbidities.5, 39, 40
Patients with AHF are often placed in intensive cardiac supervision wards where their rehabilitation commences; this resembles early mobilization of patients staying in the intensive care unit (ICU). It involves a gradual increase in functional activities, beginning with passive and active assisted exercises while the patient is lying in bed, through active exercises in a sitting and standing position, progressing to walking in one spot, and then gait walking.16, 41, 42, 43 Taking into account the absolute contraindications to exercise, the physiotherapist should develop an individualized, monitored and supervised rehabilitation program.17, 40, 44
When choosing exercises for our research, the following principle was adopted: the more severe the heart failure, the lower the intensity and duration of exercise, and the higher the exercise frequency. Assisted exercises have been included in the Polish “HF model” of cardiac rehabilitation of patients with AHF.40
In order to prevent progressive impairment of numerous body functions, Massimo Piepoli, an expert on heart failure and then the Chairman of the European Guidelines on cardiovascular disease prevention in clinical practice, has been recommending early mobilization of patients for years. Following a period of immediate threat and clinical instability, he encourages simple and slow movements performed as soon as possible.17 Piepoli emphasizes the important role of early activation of patients after stable clinical AHF status has been achieved. Exercises should be individualized based on simple movements, engaging body weight only.17, 37
Researchers have demonstrated the safety and efficacy of multidomain rehabilitation, which improves physical function and shortens rehospitalization. In their studies, they included independent and mobile patients who could walk at least 4 m at enrollment, were functionally independent before admission and were expected to be discharged from hospital.13, 14 In the REHAB-HF project and in a multi-center study by Kitzman et al., the 30-minute rehabilitation sessions commenced in the hospital during AHF and continued at home and in the outpatient clinic for the next 12 weeks.14, 45, 46
Our research stems from practice and from the daily needs of those working with elderly patients with disabilities who are bed-bound in hospital with severe disease symptoms. It was challenging to decide which exercises to start off with, and how to perform them or to estimate which groups of patients will benefit from them the most. In this project, we wanted to determine whether early activation is safe, if it has a positive impact on the patient’s well-being, and, above all, whether it has an objective, measurable impact on the oxidation of skeletal muscles during a decompensated heart failure stage.
During AHF, patients often experience massive swelling associated with an increased tension of the skin integument of the entire lower limb, occasionally expanding the skin pores through which serous fluid may seep out. These symptoms are often accompanied by pain and discomfort, which hinder or prevent independent, active movements of the lower limb. With these ailments and our own experiences in mind, assisted exercises were selected. These types of exercises do not require effort from the patient or significant involvement of their own muscles. Furthermore, during the early period of the disease, assisted exercises intend to gradually introduce other physiotherapeutic activities to the exercise therapy program with greater patient involvement. The additional benefit of assisted exercises is the reduction of risks involving thromboembolism and the reduction or prevention of edema.47, 48 Peripheral edema of the lower extremities occurs in more than 50% of patients with stable heart failure and in about 75% of patients with decompensated heart failure.49, 50 The role of assisted exercises was to enhance flow in the venous vascular bed by raising the heel and assisting the outflow of accumulated fluid, which can reduce stasis of the peripheral circulation.
When assessing oxygenation of the quadriceps muscle as a reaction to the exercises and in the rest phase 10 min after the end of the exercise, we observed a slight increase in the TOI. This is certainly a beneficial effect that requires further in-depth research. We presume that the improvement in venous outflow contributed to an increase in the arteriovenous difference and increased blood flow and oxygenation throughout the muscles.
The assisted lower limb movements also helped reduce the pain connected with swelling, making it more pleasant for the patient. This result may have been attained because the fluid pressure exerted on the peripheral tissue was reduced. This was reflected in the feeling of reduced discomfort due to the relieved skin tension reported by the patients. This may have the added benefit of reducing reluctance to exercise. The beneficial effects of early activation of the patient as a way of preventing intensification of the symptoms of FS, post-hospital syndrome or rehospitalization should not be underestimated.
Limitations
Our research is not free from limitations, and this needs to be acknowledged. First, the study included a relatively small number of patients. This is due to the unique nature of our study population, which is comprised of elderly patients with many different chronic conditions. Due to the small study group, we decided not to include both genders to avoid heterogeneity within our sample, which could introduce another limitation. However, we felt that gender diversity was particularly important in the context of our study in terms of the effects of hormones on muscle tissue and, moreover, significant differences in muscle mass, strength and endurance. Another limitation is the lack of a control group. It is due to the fact that, at this stage of the research, we focused on the effects of exercise on hemodynamic parameters and muscle oxygenation, while the observed changes and their dynamics were compared to resting values. The methods used for diagnosis and assessment of pulmonary congestion have had limited accuracy and reliability. We endeavor to eliminate these limitations by continuing this research in the future.
Conclusions
We have demonstrated that assisted exercises of the lower limb are safe because they do not burden the circulatory system. This is because they did not cause changes in BP, HR and oxygen saturation or increase dyspnea; therefore, they can be performed during the initial rehabilitation period of male patients hospitalized for AHF. Furthermore, the presented data suggest that such physiotherapeutic interventions are beneficial, particularly in patients with peripheral congestion and lower limb edema, as they improve venous outflow and muscle oxygenation. Finally, assisted exercises helped reduce the severe and uncomfortable symptoms of AHF, such as feelings of tension and pain in the lower extremities, that limit the patient’s mobility.