Abstract
Endometrial cancer (EC) is the most common gynecological tumor in developed countries. Nowadays, molecular biomarkers have become increasingly important in the management of EC patients, helping in early detection, risk stratification, prognosis and response to the treatment, and qualification for novel immunotherapies, especially in EC patients with metastatic disease or recurrence. When EC tumor molecular profiling is combined with the standard histopathological features such as clinical stage, histologic grading and evaluation of lymphovascular space invasion, the final therapeutic outcome may bring benefits in terms of personalized and efficient management.
Key words: endometrial cancer, molecular markers, translational researches
Background
Endometrial cancer (EC) is the most common gynecological tumor in developed countries, and the 4th most frequent malignancy in women, with 61,800 estimated new cases (7% incidence) and 12,160 deaths (4%) in the USA in 2019.1, 2 Undoubtedly, the EC incidence has been rising together with the average age and obesity in these populations. This fact determines recent translational research effort for defining EC risk groups more precisely, including molecular characterization of tumor. Combining the standard approach, based mainly on histopathological features, with new molecular technologies of EC profiling translates this growing knowledge into novel diagnostic and therapeutic approach, resulting in well-tailored personalized management of this cancer.3, 4
Endometrial cancer traditional
and novel classification systems
For many years, the Bokhman classification of 2 types endometrial carcinomas based on clinical, endocrine and epidemiological features has been used.5 Type I tumors comprise majority of the cases (60–70%), with mostly endometrioid histology, estrogen-dependent – with high sex hormones receptors expression and endometrial hyperplasia in the background, associated with obesity, diabetes and reproductive dysfunctions, but on the other hand with predominant low-grade tumor, superficial myometrial invasion, low risk for lymph nodes metastases, and better prognosis in general. Type II tumors, usually serous carcinomas, are less common, arise in a non-estrogenic environment, and are associated with an atrophic endometrium of older, postmenopausal and non-obese women without reproductive and metabolic disturbances. Such tumors mostly show high-grade differentiation with deep myometrial invasion, increased risk for lymphogenic spread and poor final outcome.6
Another traditional World Health Organization (WHO) classification of endometrial neoplasms, based on histological features, distinguishes among epithelial carcinomas endometrioid, serous, clear cell, mucinous, squamous cell, transitional cell, small cell, and undifferentiated tumors.7 Most common endometrioid adenocarcinomas can be differentiated into low-, intermediate- or high-grade tumors, whilst serous and clear cell cancers are aggressive and high-grade by definition.
The Bokhman and WHO classifications created the template for an easy-to-use, dichotomous model, practical for diagnostic, therapeutic and research approach. However, the wide implementation of this traditional classification is imperfect and sometimes misleading as biological, genetic and pathological features of endometrial carcinoma are complex and heterogeneous. There are not only overlaps between type I and II tumors, but heterogeneity is also present within each of these types.6 Furthermore, this dualistic model has been established almost 40 years ago and epidemiological characteristics of women suffering from EC has changed since that time due to widespread obesity followed by diabetes, increased use of hormone-replacement therapy, and many other multiple risk factors shared in both types.8 Most of high-grade endometrioid carcinomas exhibit clinical, histopathological and molecular patterns placing them close to type II cancers, including no association with endometrial hyperplasia and poor outcome.8, 9 By contrast, some serous carcinomas arise in the background of endometrial hyperplasia, and about 20% of them lack deep myometrial infiltration.10 Besides, the Bokhman model does not encompass such high-grade tumors as carcinosarcomas and clear cell or undifferentiated carcinomas, which are not so rare.6, 11 Likewise, endometrial cancer cases with predominant genetic predisposition, like Lynch syndrome, not associated with obesity and endometrial hyperplasia, have not been taken into account in this traditional classification.12
In recent years, next-generation sequencing (NGS) technology has changed the research approach from studying single genes or pathways into the insight of genome-wide genetic alterations in endometrial carcinomas.13 The Cancer Genome Atlas Research Network (TCGA) assessed a large group of common endometrial cancers (endometrioid, serous and mixed) with NGS tests of mutation load and copy-number aberrations combined with microsatellite instability, reverse-phase protein array and DNA methylation analysis.14 The results allowed to categorize endometrial tumors into 4 genomic clusters: 1) POLE tumors with very high mutation rates (ultramutated) and hotspot mutations in the exonuclease domain of polymerase ε (POLE), few copy-number aberrations, mutations in PTEN, PIK3R1, PIK3CA, FBXW7, KRAS, and good prognosis; 2) a microsatellite-instable group (MSI; hypermutated) characterized by microsatellite instability due to MLH1 promoter methylation, high mutations rates, few copy-number aberrations, and KRAS and PTEN mutations; 3) copy-number low cluster compromising endometrioid microsatellite-stable tumors grade 1 and 2, with low mutation rates but frequent CTNNB1 mutations; 4) copy-number high tumors (serous and some grade 3 endometrioid adenocarcinomas), characterized by extensive copy-number aberrations and low mutation rates, frequent and recurrent TP53, FBXW7, and PPP2R1A mutations, seldom PTEN and KRAS mutations, and poor outcome.6, 14 Only POLE hotspot mutations were unique for cluster 1, while other alterations and aberrations in studied genes and their pathways were expressed more or less in all 4 classes, overlapping themselves. The TCGA characterization of endometrial tumors 4 genomic classes in comparison with the traditional classification system of EC has been presented in Table 1.
This novel, molecular classification system of endometrial cancer has a great potential to develop better prognostic accuracy and provide predictive information.6 The integration of genomic markers of an individual, endometrial tumor with its clinicopathological features has become an important tool for clinical decision-making, especially in the context of escalation or de-escalation of clinical management and introducing new targeted immunotherapies.
Clinical management recommendations for EC patients based on integrated traditional pathologic and novel molecular features
The results reported by TCGA were retrospectively confirmed by analyzing data from PORTEC-1 and PORTEC-2 trials.15 Molecular subgroups were combined with other significant EC biomarkers such as L1CAM, PTEN, β-catenin, ARID1a, estrogen and progesterone expression, and lymphovascular space invasion. Integration of prognostic molecular alterations with established clinicopathological factors resulted in a stronger model with improved risk prognostication in high–intermediate risk cohort stratified into 3 subgroups of 50% (favorable), 35% (intermediate) and 15% (unfavorable) features. Another retrospective analysis (PORTEC-3 trial) of a cohort of high-risk EC patients randomized for adjuvant chemoradiotherapy compared to radiotherapy alone investigated the prognostic value of molecular EC classification, concluding that incorporation of this classification into risk stratification is essential. Patients with p53abnormal (p53abn) mutation should be referred for adjuvant chemotherapy whilst high-risk POLE-mutated patients had much better outcome; therefore, de-escalation of adjuvant treatment should be considered.16 While most EC cases can be classified based on a single TCGA classifier (POLE exonuclease domain mutations – POLEmut, MMR deficiency – MMRd, p53 abnormal – p53abn), a small but clinically relevant group of tumors harbor more than 1 molecular classifying feature and are referred to as ‘multiple-classifier’ ECs.17 This small population (3% of all ECs) consists of MMRd-p53abn, POLEmut-p53abn and MMRd-POLEmut-p53abn subgroups, and shows much better 5-year recurrence-free survival than single-classifier p53abn (94.1% compared to 70.8% for stage I), and thus could be de-escalated from the aggressive treatment, similarly to the single-classifier POLEmut EC.
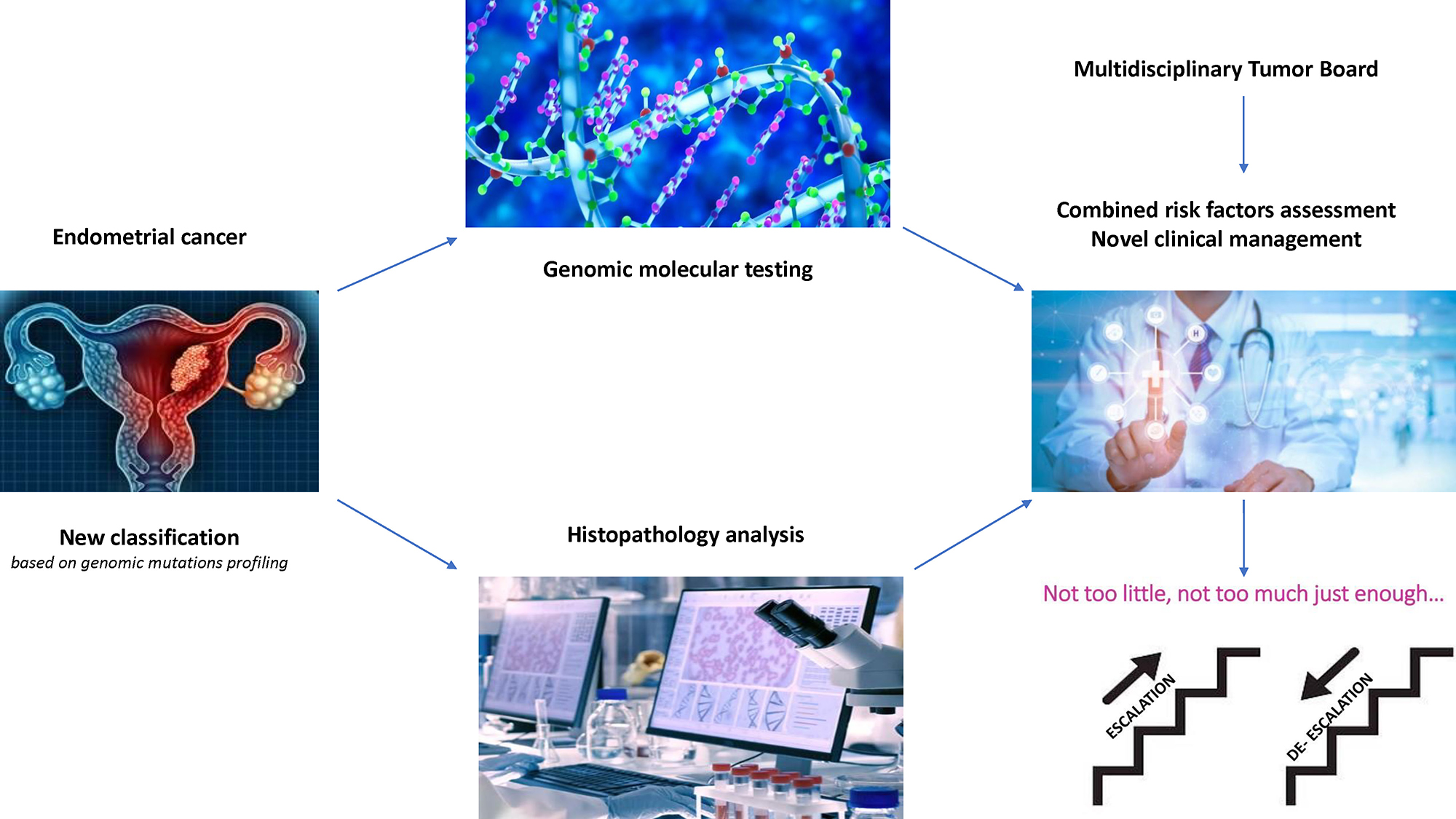
Based on these recent translational studies, the newest, 5th edition of WHO classification of tumors of female genital tract has included molecular markers.18 Furthermore, the National Comprehensive Cancer Network (NCCN) in its newest edition of Clinical Practice Guidelines in Oncology (2022) recommends molecular analysis of the endometrial tumor that could have an impact on the management of metastatic and recurrent cases presenting mismatch repair deficiency (dMMR/MSI-H tumors) regarding the therapeutic use of efficient immunotherapy (checkpoint inhibitors such as pembrolizumab, dostarlimab or nivolumab).19 However, introducing molecular biomarkers into the clinical practice may be difficult and depends on availability of resources and the multidisciplinary team of each hospital dealing with the endometrial cancer management. Therefore, some immunohistochemical surrogates have been proposed to be combined with genomics and thus enable classifying each case into 1 from 4 clusters of the molecular classification.19, 20 Such molecular and histologic evaluation algorithm of each endometrial cancer case has been introduced in author’s institution since the beginning of 2022 and is presented in Figure 1.
The newest European Society of Gynecological Society/European Society for Radiotherapy and Oncology/European Society of Pathology (ESGO/ESTRO/ESP) 2021 guidelines for the management of patients with EC recommend using molecular classification in EC patients to define a prognostic risk group and to choose the best possible therapeutic strategy.21 These comprehensive guidelines are a very good example of decision-making for escalation or de-escalation management of endometrial cancer patients depending on the presence or absence of molecular biomarkers. For instance, stage I–II patients with pathogenic POLE mutation may omit the adjuvant treatment. On the other hand, EC patients staged I–IVA with myometrial invasion and no residual disease but with p53abn mutation expressed should undergo external beam radiotherapy (EBRT) with concurrent and adjuvant chemotherapy.
To date, other sophisticated molecular studies in EC patients have been conducted in order to obtain better translational impact on clinical practice. Under the auspices of the National Cancer Institute’s Clinical Proteomic Tumor Analysis Consortium (CPTAC), multi-omic characterization of EC samples and appropriate normal tissues from a prospective cohort of 95 EC patients were extensively studied. Integrated measurements of DNA, RNA, proteins, and post-translational modifications (phosphorylation and acetylation) were used to identify novel regulatory relationships and potential pathways for identifying therapeutic targets. This analysis revealed possible new consequences of perturbations to the p53 and Wnt/β-catenin pathways, identified a potential role for circRNAs in the epithelial–mesenchymal transition, and provided new information about proteomic markers of clinical and genomic tumor subgroups, including relationships to known druggable pathways.22
Another recent analysis used the Immuno-oncology panel and the Target 96 Oncology III panel to detect 92 specific cancer-related serum protein in early-stage EC cancer patients. The authors generated an algorithm based on multivariate logistic regression model that was able to discriminate early EC type I patients from controls with high specificity and sensitivity (83.7% and 97.6%, respectively) thanks to the analysis of Gal-1, Gal-9, MMP7, COL9A1, and FASLG serum levels.23
Conclusions
The diagnostic and clinical landscape in EC has changed a lot in recent years. The translational studies on genomics and proteomics have a practical impact on management of this cancer. Novel molecular biomarkers are essential for stratifying the risk of each EC patient and creating a novel diagnostic tool for optimal therapeutic scenario and cancer surveillance regimens after the treatment completion. The use of new immunotherapies has been based on molecular-integrated risk profile to determine the treatment efficacy in patients with advanced and metastatic disease. However, further prospective studies are necessary to develop complete and commonly used molecular profiling that should be useful for tailoring the best individual EC management.