Abstract
Background. There is no consensus regarding the standard treatment method for anorectal abscesses accompanied by anal fistulas and complex anal fistulas. Simultaneous surgical treatment of the underlying anal fistula with anorectal abscess drainage is controversial due to incontinence problems.
Objectives. We aimed to investigate the effectiveness of the loose seton method for the treatment of chronic anal fistulas and acute anorectal abscesses accompanied by anal fistula.
Materials and methods. In this retrospective study, 114 patients who were operated on in our clinic due to chronic anal fistulas and anorectal abscesses with an applied loose seton between 2020 and 2022 were included in the study. The patients were divided into 2 groups: those with chronic complex anal fistula and those with anorectal abscess accompanied by anal fistula. The groups were compared in terms of their continence status, rate of recurrence, recurrent abscess formation, postoperative pain scores, duration of operation, and demographic characteristics.
Results. Of the patients included in the study, 78 had a complex chronic anal fistula, and 36 had an anorectal abscess accompanied by an anal fistula. There were no differences between the demographic characteristics of the 2 groups. The mean seton dissociation time was 6.8 (3–19) months. Gas or stool leakage was not observed in patients during the mean follow-up period of 18 (6–30) months. There was no difference in postoperative continence levels between the 2 groups. No recurrent fistulas were observed in patients during the follow-up period. Recurrent abscesses were observed in 5 (13.9%) patients in the anorectal abscess group. Abscesses due to insufficient drainage were observed in 2 (2.6%) patients in the chronic fistula group. There was no significant difference in operation time between the 2 groups.
Conclusions. A loose seton can be a safe and effective method for the treatment of abscesses. It is a painless surgical method that produces good results in the treatment of all types of abscesses.
Key words: fecal incontinence, anorectal abscess, anal fistula, loose seton
Background
Anal fistulas are tracts that usually develop due to cryptoglandular abscesses that extend from the anal canal or distal rectum to the skin.1 Patients with anal fistulas present with complaints of intermittent perianal discharge, contamination, pain, and swelling. These fistulas occur at a rate of 1.2–2.8/10,000.2 In addition to physical examinations, endoanal ultrasonography (USG) and pelvic magnetic resonance imaging (MRI) are used for diagnosis. Contrast-enhanced MRIs are accepted as the gold standard, as they allow for detailed visualization of the anal sphincter anatomy and fistula map.3 Based on the normal muscular anatomy of the pelvic floor, fistulas are classified as intersphincteric, transsphincteric, suprasphincteric, or extrasphincteric.4
Loose setons are routinely used in surgical operations. The probability of regeneration after seton loss is high. Notably, the incidence of seton loss in patients is unknown. However, it has been reported that the rate of seton loss is high in patients with complex anal fistulas.5 In a pilot study, the clinical advantages of using loose setons to prevent the recurrence of acute anorectal abscesses were demonstrated.6 However, the treatment of patients with high anal fistulas remains challenging.
Short- and long-term benefits have been observed with the use of loose setons.7 It has been reported that loose seton use is beneficial in patients with transsphincteric anal fistulas. The preservation of an anatomical loss of the external sphincter function is important with this technique.8 In a study comparing the effectiveness of the traditional seton cutting method with the loose combined cutting method, it was reported that the loose combined cutting technique produced more successful and reliable results in the treatment of suprasphincteric anal fistulas.9 The treatment of loosely located anal fistulas over a 3-year period has also been investigated retrospectively, and it was reported that this technique is the most appropriate, as it is both reliable and less costly.10 The impact of the use of chronic loose and non-cutting seton types on the quality of life in patients with fistulized perianal Crohn’s disease has also been investigated.11 It has been reported that patients with this condition respond well to a seton technique combined with infliximab therapy, and outcomes are good no matter if early or late application is used.12 Similarly, seton drainage in combination with infliximab treatment was observed to be an effective method for the closure of a fistula in 75% of patients with Crohn’s disease.13
In surgical practice, fistulas are categorized as simple or complex based on their relationship with the anal sphincter complex. Fistulas where fistulotomy can safely be performed without the risk of incontinence are classified as simple fistulas, while others are classified as complex.5 Complex anal fistulas include transsphincteric fistulas involving more than 30% of the external anal sphincter, suprasphincteric fistulas, extrasphincteric fistulas, horseshoe fistulas, fistulas with multiple tracts, and fistulas coexisting with an abscess.14 Notably, 15–40% of complex anal fistulas are associated with abscesses.15, 16 Anal abscesses occur when an infection in the anal glands in the intersphincteric space spreads to the surrounding spaces,1 and 40% of these turn into anal fistulas. Abscesses and fistulas should be considered acute and chronic stages of the same disease because of their pathophysiological, etiological and anatomical relationships.17 Therefore, treatment strategies for these conditions should be evaluated similarly. In both diseases, the goal is to reduce the recurrence rate without affecting continence, while eliminating the painful condition and improving the quality of life.18
To date, many surgical methods have been described for both diseases. However, the reported success rates for most methods are controversial. Moreover, there is currently no standard treatment modality.19
Objectives
We aimed to investigate the surgical results of the loose seton method for the treatment of acute anorectal abscesses accompanied by anal fistulas and chronic anal fistulas.
Materials and methods
Patient follow-up
A total of 114 patients who were operated on in our clinic for complex chronic anal fistulas and anorectal abscesses with the loose seton application between 2020 and 2022 were included in the study. Patient data were examined retrospectively using patient files. Of the 114 patients, 78 had a complex chronic anal fistula, and 36 had an anorectal abscess accompanied by an anal fistula. With the exception of 12 patients who were operated on for anorectal abscesses under emergency conditions, all patients were evaluated using contrast-enhanced pelvic MRI, in addition to a physical examination in the preoperative period. Patients with abscesses who underwent only incision and drainage for an anorectal abscess or those whose internal fistula os could not be detected during the operation were excluded from the study. Of the patients with complex fistulas included in the study, 3 were suprasphincteric, 2 were extrasphincteric, 2 were horseshoe fistulas, and the remaining were transsphincteric. Patients who underwent fistulotomy due to intersphincteric and anocutaneous fistulas in chronic anal fistula cases and patients with a history of inflammatory bowel disease were not included in the study.
A signed consent form was obtained from all patients. Our study was approved by the Ethics Committee of Memorial Şişli Hospital (Istanbul, Turkey) (approval No. July 5, 2022/004).
Classification of fistulas
Fistulas are typically classified as described by Parks et al.,4 Standard Practice Task Force (2005), St. James’s University Hospital (2000), and Garg et al.20 These classifications are summarized in Table 1. In the current study, the oldest and most widely used classification system (Parks et al.4) was utilized.
Surgical technique
With the exception of 12 patients who were operated on for anal abscesses under emergency conditions, all patients were operated on under elective conditions. In the elective patients, a preoperative rectal enema was performed and bowel cleansing was achieved. Five patients were operated on under general anesthesia and the remaining patients were operated on under spinal anesthesia. Patients were operated on in the jackknife or lithotomy position, depending upon the location of the fistula.
In patients with a chronic fistula, diluted methylene blue was administered via the fistula tracts. After the internal os was detected, the fistula tract was passed with the help of a stylet. The external fistula os was excised all around up to the external sphincter border and the material was sent to the pathology laboratory. The loose seton was attached to the internal end of the stylet and passed through the tract. It was tied with silk sutures so that the sphincter was not compressed.
In patients with an anorectal abscess, an anoscope was inserted into the anal canal to check whether there was spontaneous abscess drainage from the internal os. With the help of electrocautery, an incision was made on the abscess pouch and the purulent fluid and debris were drained. Subsequently, an attempt was made to find the internal os by applying diluted hydrogen peroxide to the abscess pouch. In patients whose internal os could not be detected, the procedure was terminated and no additional procedures were performed. These patients were excluded from the study. In patients where the internal os could be found, it was passed with the help of a stylet. The same procedure was applied in fistula patients.
In addition to the vein strap used as a seton material, in several patients, a circular piece cut from the thickest part of a surgical glove sleeve was used. When adequate progression could not be observed, the vascular slings were replaced with the material prepared from surgical latex gloves. A seton was also applied in cases of low transsphincteric fistulas. Fistulotomy was not performed on any patient operated on for an anorectal abscess. Patients who underwent fistulotomy due to intersphincteric, subcutaneous-mucosal fistulas were excluded from the study. A seton was applied in all patients with an anorectal abscess who had an internal os to facilitate drainage of the abscess. Operation time was recorded for each patient.
Postoperative patient follow-up
In the postoperative period, the patients received monthly follow-ups subsequent to 1st- and 2nd-week follow-ups. During the monthly follow-ups, seton materials that had loosened and sagged excessively were tied with silk sutures and shortened to prevent compression of the sphincter. Weakened seton materials were replaced end-to-end with new materials.
In patients whose setons had progressed to the anocutaneous level during follow-up, the seton was removed by fistulotomy under local anesthesia in an outpatient setting. In 8 patients, the seton spontaneously ruptured at the anocutaneous level. No interventions were performed on these patients. Patients were asked about their continence levels during follow-up. Postoperative continence levels were evaluated after the seton dissociation process was completed, using the Wexner incontinence score. This scoring system cross-tabulates frequencies and different anal incontinence presentations (solid/liquid/gas/wears pad/need for lifestyle alterations) and sums the returned score for a total of 0–20 (where 0 = perfect continence and 20 = complete incontinence; Table 2).21
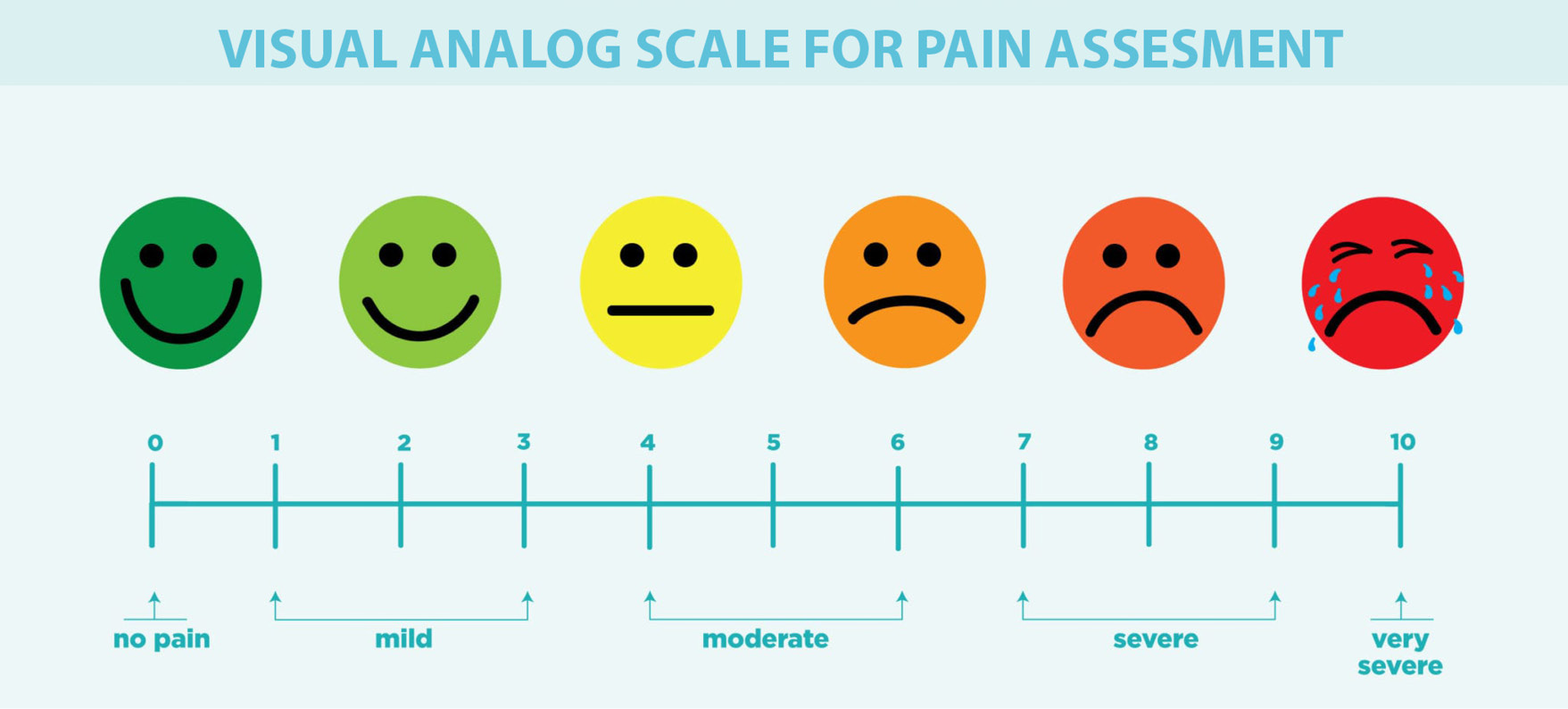
The patients were assessed for recurrence during follow-up. Postoperative pain was assessed using the visual analogue scale (VAS) 24 h after the operation. On this scale, a score of 0 indicates no pain and a score of 10 indicates very severe pain. Scores <3 were interpreted as mild pain, 3–6 were considered to indicate mild–moderate pain, and >6 indicated severe pain (Figure 1).
Statistical analyses
The SPSS v. 11.5 (SPSS Inc., Chicago, USA) was used for data analyses. Mean ± standard deviation (M ±SD) and median (minimum–maximum) are used as descriptors for quantitative variables, and the number of patients (percentage) is presented for qualitative variables. The differences in quantitative variables between the 2 categories were examined using the Mann–Whitney U test because the assumption of a normal distribution was not met. Fisher’s exact tests were used to examine the relationships between 2 qualitative variables. Results were considered statistically significant if the p-value was <0.05.
Results
There was no statistically significant difference in patient gender between the 2 groups (χ2 = 1.380, p = 0.334). Mean age and operation times were significantly higher in the anorectal abscess group (z = −2.471, p = 0.013 and z = −3.224, p = 0.001, respectively; Table 3).
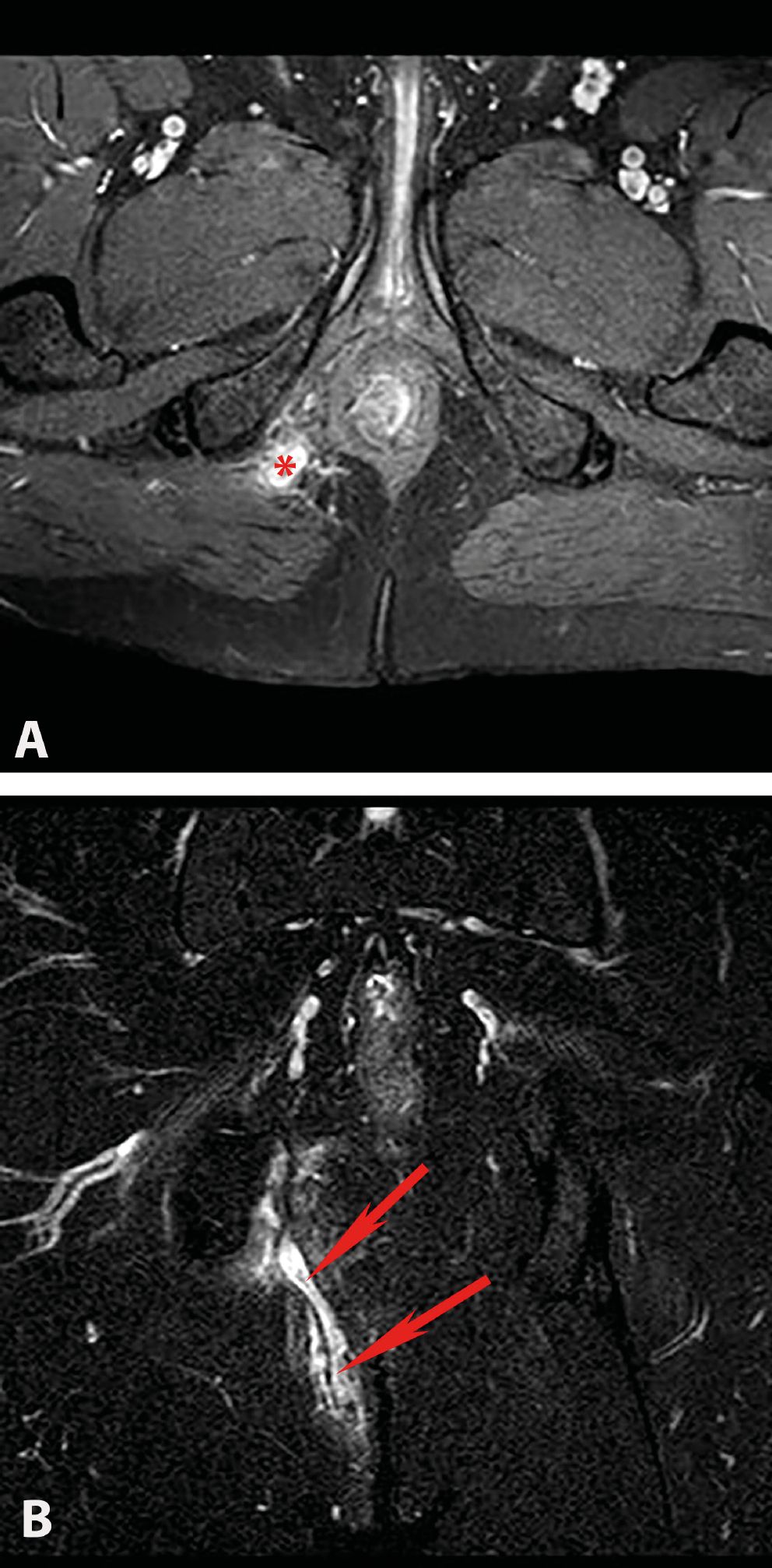
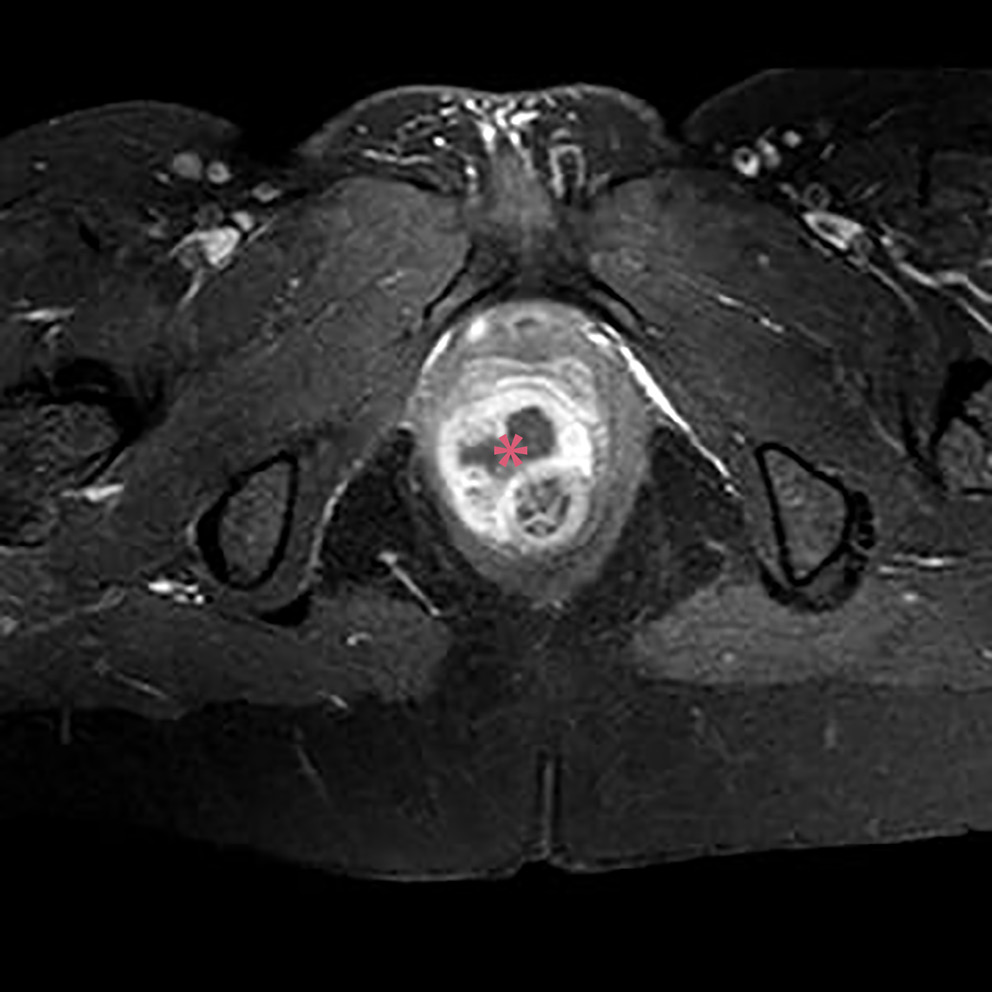
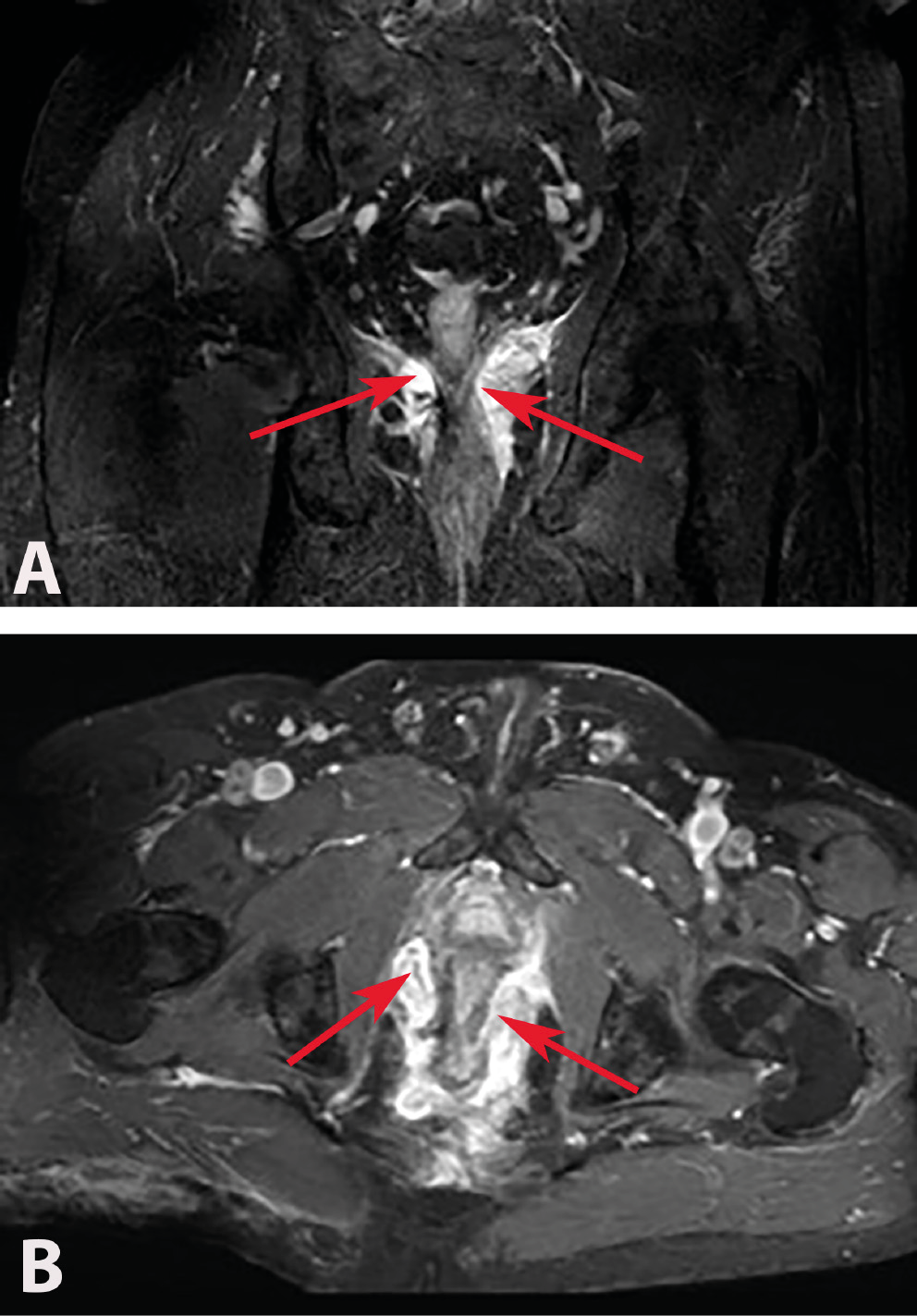
Of the 114 patients included in the study, 78 had complex chronic anal fistulas, and 36 had anorectal abscesses accompanied by anal fistulas. As a result of the MRIs performed in the anorectal abscess group, perianal abscesses were detected in 14 (58%) patients (Figure 2A,B), ischiorectal abscesses in 6 (25%) patients (Figure 3A,B) and intersphincteric abscesses in 4 (17%) patients (Figure 4). Suprasphincteric fistulas were observed in 3 patients, extrasphincteric fistulas were found in 2 patients, horseshoe fistulas were present in 2 patients (Figure 5A,B), and transsphincteric fistulas were observed in the remaining patients with complex fistulas. Thirteen patients had 2 external fistula ora and 4 patients had 3 external fistula ora. Eight patients in the fistula group had an abscess pouch associated with the fistula tract. These findings are consistent with the literature.
The mean time to seton removal was 6.8 (3–19) months. In patients whose setons progressed to the anocutaneous level during the follow-up period, the seton was removed by performing a fistulotomy under local anesthesia at the outpatient clinic. In 8 patients, the seton spontaneously ruptured and fell to the anocutaneous level. No interventions were performed on these patients.
In the early postoperative period, liquid stool and gas leakage from the seton area were observed in 3 patients. In these patients, continence improved in the 3rd month of follow-up after the fistula tract matured. Neither gas nor stool leakage was observed in any patient in the chronic fistula or anorectal abscess groups on the course of long-term follow-up after seton removal. There were no statistically significant differences in postoperative continence levels between the groups.
In our study, the median follow-up period was 24 (1–41) months. No fistula recurrence was observed in either the chronic fistula group or the abscess group during the follow-up period. Recurrent abscesses were observed in 5 (13.9%) patients in the anorectal abscess group. In addition, abscesses were observed in 2 (2.6%) patients in the chronic fistula group. These results were statistically significant (χ2 = 1.380, p = 0.031). Setons were renewed by applying drainage in these patients.
The anorectal abscess group had statistically significantly higher pain scores 24 h after the surgery (z = −8.235, p < 0.001). These findings are summarized in Table 3.
There was no significant difference in pre- and post-operative Wexner continence scores between the anorectal abscess and chronic anal fistula groups (0.03 ±0.17 and 0.03 ±0.16, respectively, p = 0.947). In the preoperative evaluation, a Wexner continence score above 0 was rarely seen, with only 1 (2.8%) patient with a gas leak in the anorectal abscess group and only 2 (2.6%) patients with gas leaks in the chronic anal fistula group. Postoperative evaluation results were the same as the preoperative evaluation results (Table 3).
Discussion
The most important factor for reducing recurrence is revealing the internal os. In the case of an anorectal abscess, it may be difficult to locate the internal os due to edema and debris in the tissues. It has been reported that the internal fistula os can be found in 83% of anorectal abscess cases.22 In our sample, the internal os was found in 45% of patients with anal abscesses. Patients in whom the internal os had not been found were excluded from the current study. Simultaneous fistula treatment carries a risk of incontinence at different rates depending on the surgical method chosen. The coexistence of anal fistulas is seen in approx. 1/3 of anorectal abscess cases.23
Garg et al. reported that an inaccurate identification of or an inability to find the internal opening is the most important cause of fistula recurrence.24 An inability to determine the exact position of the internal os complicates the management of fistulas. A protocol to localize the internal os in patients where it cannot be clearly found was recently described by Garg et al.24 According to this protocol, attempts are made to localize the internal os with preoperative clinical examination (maximum hardening point), MRI evaluation, intraoperative examination under anesthesia, and injection of povidone-iodine solution through the external opening. If the internal os of the fistula cannot be clearly found, a 3-step protocol is followed. First, MR images are re-evaluated by the surgical team. Second, it is assumed that the internal opening is at the location where the fistula is closest to the internal sphincter. Third, if there is a horseshoe fistula and the internal opening cannot be clearly found, the internal opening is assumed to be on the midline. If the horseshoe fistula is located posteriorly, the internal opening is assumed to be on the posterior midline, while in anterior horseshoe fistulas, it is assumed to be on the anterior midline, and it is recommended to manage fistulas accordingly.25 Thus, the Garg protocol effectively helps to find the internal os of fistulas.
In patients receiving only incision and drainage of the abscess, the procedure can result in insufficient drainage in high abscesses, resulting in a recurrence rate of 44% and the formation of anal fistulas.26 Along with drainage of the anorectal abscess, simultaneous surgery for the fistula is useful in preventing recurrent abscess formation and subsequent fistula surgery. Meta-analyses have shown that anal fistula surgery during abscess drainage reduces the presence of persistent abscesses and fistulas, recurrence, and repetitive surgery.27 In our study, abscess recurrence was observed in 5 (13.38%) patients in the anorectal abscess group and 2 (2.56%) patients in the chronic fistula group. It seems that the recurrence was due to the insufficient drainage of deep abscesses. Setons were renewed by applying drainage to these patients. A low recurrence rate suggests that a seton is effective in reducing the rate of recurrent abscesses.
The 2017 German S3 guidelines recommend primary fistulotomy for superficial fistulas involving a small portion of the anal sphincter. The same guidelines recommend postponing definitive surgery to a later date in patients with high fistulas or fistulas where it is not clear how much of the sphincter is involved.28 In contrast, in a meta-analysis that included 479 patients who had only abscess drainage or fistula surgery with abscess drainage, there was no statistically significant difference in incontinence in patients who underwent fistula surgery performed simultaneously with abscess drainage.29 However, results vary according to the chosen surgical method. Recent studies have shown that even in fistulas with a high acute anorectal abscess, definitive fistula surgery can be carried out with excellent results by performing sphincter-sparing procedures.30
Many methods have been developed to prevent incontinence in patients with complex fistulas. In a study by Garg et al., in a series of 1250 patients, 4 different surgical techniques were applied and a 98.6% recovery rate was reported for simple fistulas that underwent fistulotomy without affecting continence. The healing rate was reported to be 90.6% in fistulas accompanied by abscess and 94.5% in fistulas without abscess, and the difference was not statistically significant (p = 0.057).31 In this study, the same sphincter-sparing surgery (transanal opening of the intersphincteric space (TROPIS)) was applied to high fistulas with accompanying anorectal abscesses and chronic complex fistulas without abscesses. In the TROPIS procedure, the intersphincteric space is opened from the anal canal. The internal sphincter and mucosa are incised and the internal os of the fistula is deroofed. Thus, in this procedure, infected crypt glands and the internal opening of the fistula are destroyed and left open for secondary healing. Moreover, the external sphincter is preserved, minimizing the risk of incontinence. Healing rates in the acute anorectal abscess and chronic fistula groups were 87% (100 of 115) and 88% (168 of 191), respectively; thus, there was no difference in healing rate between the groups. However, the author emphasized that long-term results and a larger patient series are needed.32
In our surgical method, we used a loose seton in patients with a complex fistula or an anorectal abscess accompanied by a complex fistula. It is thought that setons facilitate the drainage of the associated abscess and create a local inflammatory reaction to provide resolution of the fistula pathway.32 Setons used in the treatment of complex anal fistulas are divided into cutting and loose setons. Cutting setons create compression necrosis as periodic tightening is applied. Due to the painful tightening process, cutting setons have disadvantages, such as patient incompatibility and continence problems.33, 34
In the literature, solid stool incontinence, liquid stool incontinence and gas incontinence were reported in 2.3%, 8.5% and 36% of patients, respectively, who underwent cutting seton due to complex fistulas.35 Due to the disadvantages mentioned above, loose setons are often used instead of cutting setons. Loose setons also provide more effective drainage, facilitating drainage of the associated abscess and promoting resolution of the fistula tract by creating a local inflammatory reaction without causing compression necrosis.36 Many materials are used as loose seton material. Vascular slings, Penrose drains, rubber bands, or latex gloves are most commonly used.37 In our study, we used a seton prepared by cutting a thick strip from the arm of a latex surgical glove. The prepared seton was formed by passing the material through the fistula area in a single layer, ensuring that the seton was not tight, and tying it with silk sutures. Using a similar material, Mentes et al. reported a 100% recovery rate after 3 months. In their study, incontinence worsening of 20% was reported in patients treated with an elastic seton compared to the initial scores, but this difference was not statistically significant.37
Seton application in a similar fashion has been reported in the literature but gas and stool incontinence was not monitored in any patient.38 Similarly, in the study by Vrzgula et al., a loose seton was used in 14 out of 99 patients who were operated on for anal abscesses. In this study, no relapses were observed, and no solid or liquid stool leakage was reported in any patient.39
During the follow-up period, we did not observe gas or stool leakage in any of the patients in either group. The absence of clinical solid and liquid stool incontinence in any of the patients during the follow-up period in our series indicates that elastic seton application significantly contributes to the preservation of continence. However, a recurrent abscess was observed in 5 (13.9%) patients in the anorectal abscess group during the follow-up period. In addition, an abscess was observed in 2 (2.6%) patients in the chronic fistula group due to insufficient drainage. Drainage was applied to these patients and their setons were renewed.
We evaluated the patients’ pre- and postoperative continence levels with the Wexner incontinence score. Gas or stool leakage was not observed in any patient in the chronic fistula or anorectal abscess groups during the follow-up period. There was no difference in pre- or postoperative continence scores between the groups. Our findings provide evidence that the seton method is a safe treatment option that can be used in both complex anal fistulas and perianal abscesses, providing a high recovery rate without affecting continence.
Limitations
The current study has a number of limitations. First, the number of cases included in the study is relatively limited. In addition, this is a single-center study. The Wexner continence scale was used to evaluate incontinence. The definitive method for assessing the anal sphincter complex is to take preoperative and postoperative anal manometry measurements. However, preoperative anal manometry measurement is not possible in patients with acute anal abscess due to pain. Therefore, manometry was not used in our study. Despite these limitations, the results obtained in our study show that the loose seton method is safe.
Conclusions
It is emerging that acute anorectal abscessed fistulas, including high complex fistulas, can be definitely treated by performing sphincter-sparing procedures at the initial surgery. We believe that using a loose seton is a safe and effective method for abscess treatment because, during patient follow-up, we observed a painless and continuous process with good results in the treatment of all types of abscesses.