Abstract
Background. The first-line obinutuzumab-based immunochemotherapy improves the outcome of patients with follicular lymphoma (FL) compared with rituximab-based regimens. However, infusion-related reactions occur in almost half of patients during the 1st obinutuzumab administration.
Objectives. The study aimed to evaluate the early effectiveness and safety of obinutuzumab-based induction regimens in a real-world setting.
Materials and methods. Outcomes of patients diagnosed with FL and treated with obinutuzumab between January 2020 and September 2021 were analyzed.
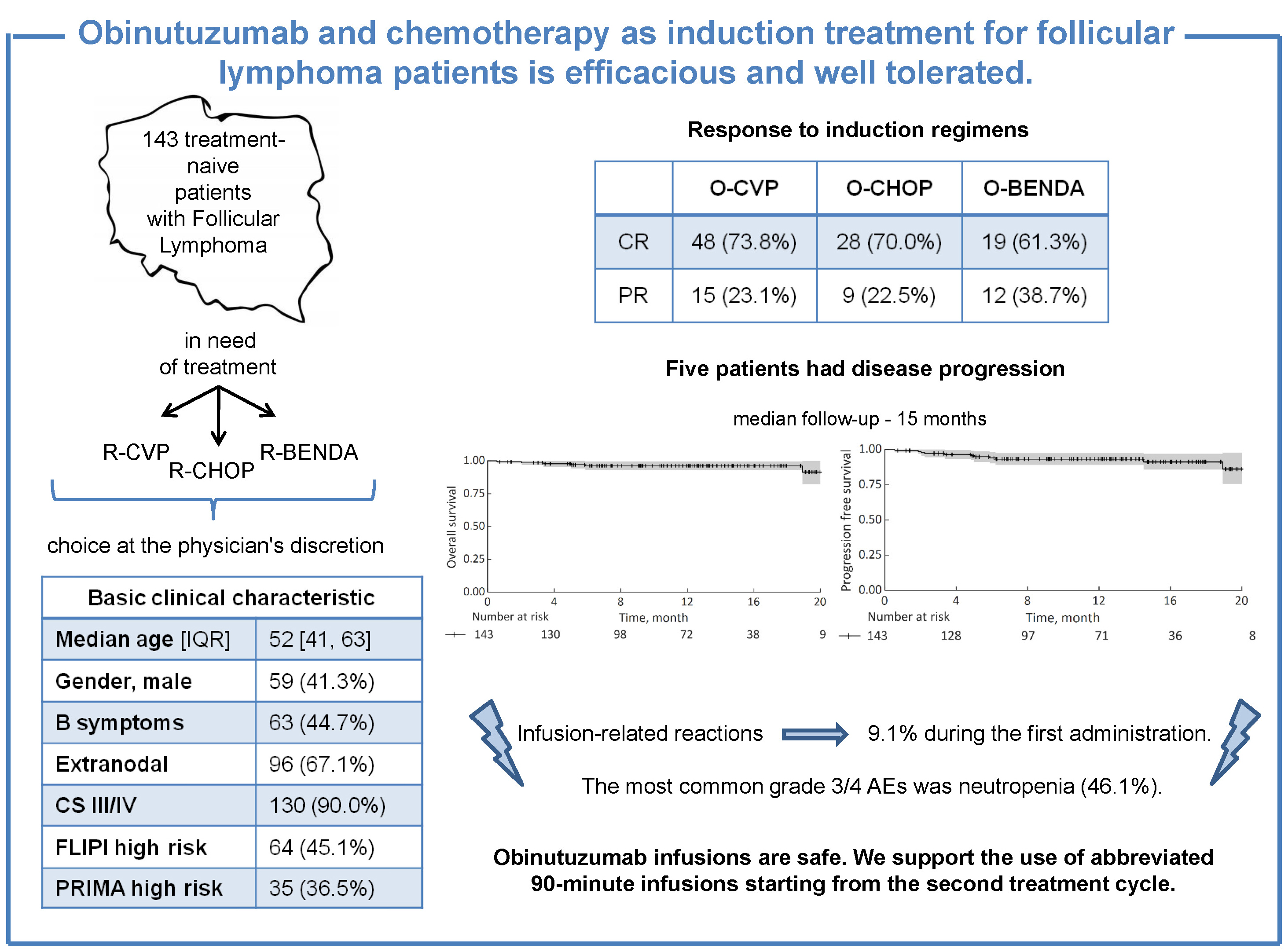
Results. The study group included 143 treatment-naïve patients with FL. The median age was 52 years (range: 28–89 years); 45.1% of patients had a high-risk disease as assessed using the Follicular Lymphoma International Prognostic Index (FLIPI). Induction chemotherapy included: O-CVP (obinutuzumab, cyclophosphamide, vincristine, prednisolone) in 49.0% of patients, O-CHOP (O-CVP plus doxorubicin) in 28.7% and O-BENDA (obinutuzumab, bendamustine) in 22.4%. Complete response (CR) and partial response (PR) rates were 69.9% and 26.5%, respectively. There was no difference in response rates between different regimens (p = 0.309). Maintenance was started in 115 patients (85.2%). In the 1st cycle, obinutuzumab was administered as a single 1000-milligram infusion in 47.9% of patients, whereas in 52.1%, initial infusions were split over 2 days (100 mg/900 mg). Infusion-related reactions were reported only during the 1st administration of obinutuzumab in 9.1% of patients, with a similar incidence in those receiving the total dose on a single day or split over 2 days (p = 0.458). The most common adverse events were hematological. Five patients died from coronavirus disease 2019 (COVID-19).
Conclusions. The early responses to induction regimens and adverse events profile were similar for every type of induction treatment. The infusion-related reactions were rare and limited to the 1st dose of obinutuzumab.
Key words: follicular lymphoma, chemotherapy, obinutuzumab, induction treatment
Background
Follicular lymphoma (FL) is the 2nd most common type of B-cell non-Hodgkin lymphoma. The median age at diagnosis is 65 years, and the median overall survival is 20 years.1 There are still controversies about the most efficient first-line treatment for FL and the time when it should be initiated. The watch-and-wait strategy is recommended for advanced FL without symptoms or organ impairment.2, 3
The standard of care for initial therapy of patients with FL in need of treatment is rituximab with one of the following regimens of chemotherapy: CVP (cyclophosphamide, vincristine and prednisone), CHOP (CVP with doxorubicin) or BENDA (bendamustine), with rituximab with CVP (R-CVP) representing the least toxic and R-BENDA the most active regimen.3, 4
The type II anti-CD20 antibody, obinutuzumab, when combined with chemotherapy, resulted in prolonged progression-free survival (PFS) compared to rituximab with chemotherapy in a randomized Gallium study, although grade 3 or 4 neutropenia, thrombocytopenia and infusion-related reactions (IRRs) were more frequent.5, 6 Effective measures for reducing incidence of IRRs are generally adopted, including splitting the dose of the 1st infusion, slowing down infusion rates and routine use of pre-medications.7
Objectives
The aim of the study was to evaluate the efficacy and adverse events, especially IRRs, following obinutuzumab-based induction therapy in patients with FL in need of treatment.
Materials and methods
The study included patients aged ≥18 years diagnosed with FL in need of treatment. The study was conducted in the Polish Lymphoma Research Group (PLRG) sites (participating health centers). Clinical and laboratory characteristics of the patients were identified retrospectively in electronic records of 14 contributing PLRG sites. The Independent Ethics Committee of the Maria Sklodowska-Curie National Research Institute of Oncology in Warsaw approved the study (approval No. 84/2021), which was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice Guidelines.
All patients included in the study had a histological diagnosis of FL based on the World Health Organization (WHO) classification.8 Patients were treated with obinutuzumab in combination with CVP, CHOP or BENDA at the physician’s discretion. Obinutuzumab was administrated at a fixed dose of 1000 mg, in up to eight 21-day cycles, in combination with CVP (O-CVP), up to six 21-day cycles with CHOP (O-CHOP), followed by 2 additional cycles of obinutuzumab monotherapy or up to six 28-day cycles in combination with bendamustine (O-BENDA). In the 1st cycle, obinutuzumab was administered on days 1, 8 and 15, then only on the 1st day of every next cycle.9 However, physicians modify the treatment schedule, e.g., split the 1st dose over 2 days instead of 1 in individual cases. Patients received standard premedication before each infusion. Intravenous infusions of obinutuzumab were administered at the standard recommended infusion rates.9 Seventeen patients received corticosteroid dose the day before obinutuzumab at 1 participating center.
The response to treatment was evaluated with positron emission tomography/computed tomography (PET/CT) in 81.3% of patients (n = 109/134), and with computed tomography (CT) in 18.7% of patients (n = 25/134), according to response criteria for non-Hodgkin’s lymphomas or revised response criteria when PET/CT was available.10, 11 Adverse events (AEs) were described and graded according to the National Cancer Institute Common Terminology Criteria for AEs (NCI-CTCAE v. 5).12
Statistical analyses
Descriptive statistics was used to evaluate variables including demographic, pathologic, clinical, laboratory, type of treatment, response to the therapy, and deaths of patients receiving obinutuzumab-based immunochemotherapy regimens. The χ2 test or the exact Fisher’s test was used to compare categorical variables. Survival curves were estimated using the Kaplan–Meier method, and log-rank test was used to compare time-to-event distributions. Statistical analyses were performed using Matlab v. 2021a (https://www.mathworks.com) and R v. 4.2.0 (R Foundation for Statistical Computing, Vienna, Austria).13 The χ2 test, the Fisher’s exact test and the Kaplan–Meier method were performed using ‘chisq.test’, ‘fisher.test’ and ‘survfit’ functions from R, whereas figures were plotted using Matlab. Effects were considered statistically significant for p < 0.05.
Results
A total of 143 consecutive patients who started treatment after obinutuzumab was made available for use in Poland on January 1, 2020, were included in the analysis. The last analyzed patients began treatment in September 2021. The characteristics of patients are presented in Table 1. According to the Groupe d’Étude des Lymphomes Folliculaires (GELF) criteria, the median tumor burden was 2 (range: 0–7). Four patients with GELF = 0 received treatment on their explicit request.
The O-CVP regimen was used across the analyzed group the most often (49.0%). The majority of patients responded to the induction treatment. Progression of disease (PD) was reported at the end of immunochemotherapy in less than 3% of patients. We did not find differences in response rates between used regimens (Fisher’s exact test, p = 0.309; Table 2). Clinical and laboratory characteristics of patients treated according to different regimens are presented in Table 1.
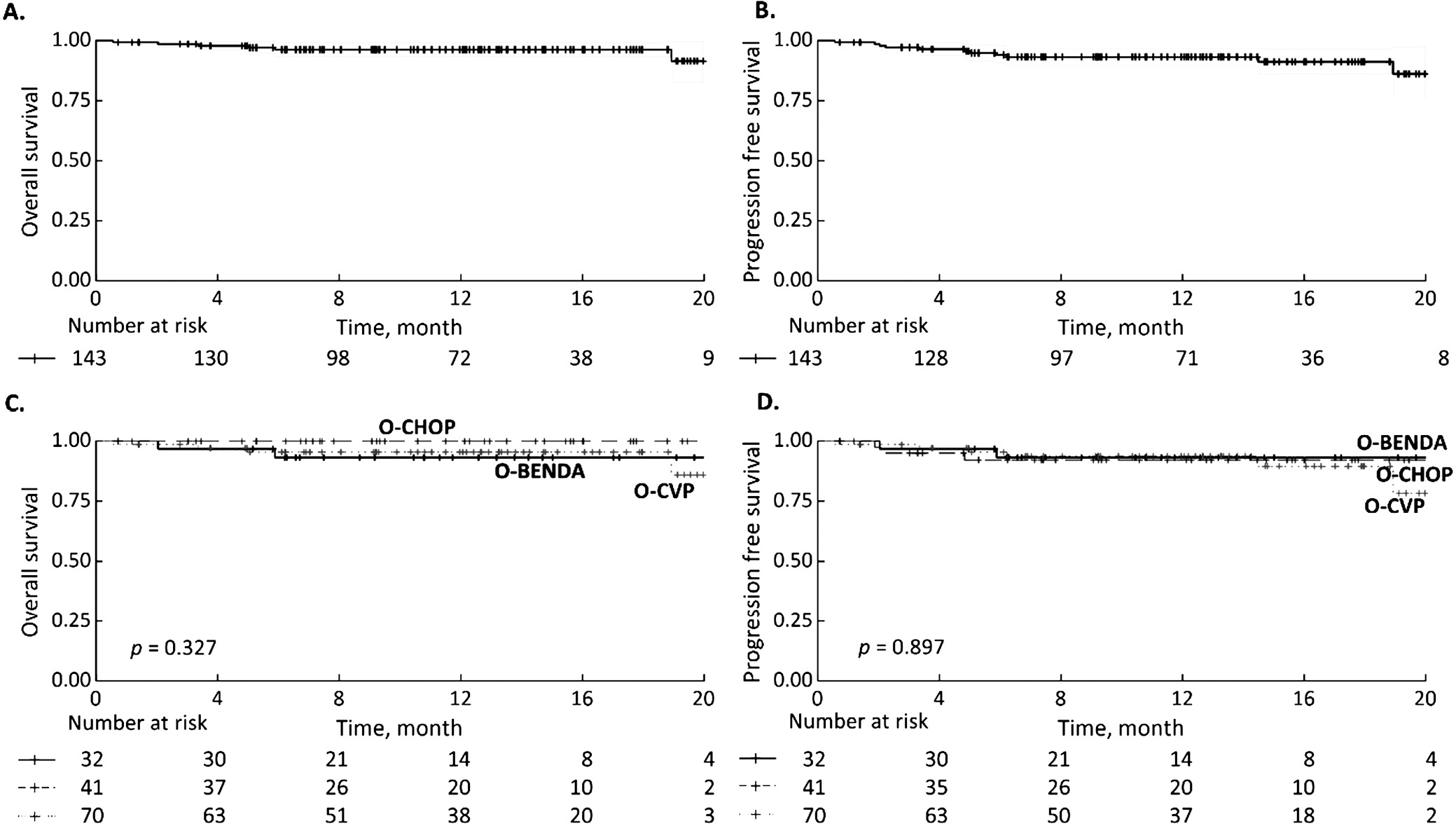
The median follow-up was 15 months and the median number of treatment cycles was 6. Maintenance treatment was started in 115 patients (85.2%) at the discretion of treating physicians. We did not find differences between regimens regarding progression-free survival (PFS; log-rank test, p = 0.327) and overall survival (OS; log-rank test, p = 0.897) (Figure 1). During the follow-up, 5 patients exhibited disease progression. No lymphoma-related deaths were reported. Five patients died from coronavirus disease 2019 (COVID-19) during the induction therapy of lymphoma.
The 1st obinutuzumab dose was administered as a single infusion on day 1 of the 1st cycle in 68 patients (47.9%) or split in 2 infusions in 74 patients (52.1%), i.e., 100 mg on day 1 and 900 mg on day 2 of the 1st cycle. The majority of patients (n = 136, 95.1%) received all planned infusions in the 1st cycle. Doses were omitted in 7 patients due to COVID-19 (n = 4), neutropenia (n = 2) or pneumonia with neutropenia (n = 1). In 27 patients (19.3%), doses were postponed. In 11 patients, day 8 dose was delayed, and in 20 – day 15 dose. Delays occurred due to grade 3 or 4 neutropenia (n = 18), admission restrictions due to the pandemic (n = 5), infections other than COVID-19 (n = 4), COVID-19 (n = 1), and other reasons (n = 3).
The IRRs were reported during the 1st administration of obinutuzumab only, and occurred in 13 patients (9.1%) (grade 1 or 2 in 11 patients and grade 3 in 2 patients). A similar proportion of patients receiving the 1st infusion on a single day or split over 2 days experienced IRRs – 11.8% and 6.8%, respectively (χ2 test statistics = 0.551, df = 1, p = 0.458). A median duration of the 1st infusion measured in 69 patients was 4 h and 52 min (range: 1.30–9.45).
The most common grade 3 to 4 AEs in patients receiving obinutuzumab-containing regimens was neutropenia (46.1%). Granulocyte-colony stimulating factor (G-CSF) was used in 94 patients (65.7%), including 69 patients (48.2%) who received G-CSF as a primary prophylaxis of neutropenic fever. Grade 3 or 4 thrombocytopenia occurred in 4 patients (2.8%). The COVID-19 infection occurred in 19 patients (13.3%), and other infections (pneumonia, upper respiratory infections, zoster, contagious impetigo) in 6 patients (4.2%). The incidence of AEs was similar across chemotherapy regimens used.
Discussion
Follicular lymphoma patients in this real-world study were younger than FL patients in general population in Poland (median age: 52 years compared to 61 years, respectively),14 and younger than patients treated in Gallium study (median: age 59 years) that established superiority of obinutuzumab over rituximab (both combined with chemotherapy). Briefly, 45.1% and 36.5% of patients had high-risk FL according to Follicular Lymphoma International Prognostic Index (FLIPI) and PRIMA prognostic index (PRIMA PI) assessment, respectively. These patient characteristics may suggest a preferred choice of obinutuzumab for younger patients with more aggressive disease.
In our study, we focused on evaluation of response and safety. The median follow-up time was 15 months. We presented objective response rate of 96.3%. The limitations of our analysis are retrospective design and small group of patients, but our overall response rate (ORR) is superior to the 88.5% of ORR in the Gallium study.5
The choice of chemotherapy regimen combined with the antibody was consistent with the European Society for Medical Oncology (ESMO) guidelines and practice in the particular health center.3 So far, it is not clear which of the recommended induction chemotherapy regimens is the most effective for FL – CVP, CHOP or bendamustine – but except for Gallium trial, they were analyzed in combination with rituximab. In FOLL05 trial, response rates after induction therapies R-CVP, R-CHOP, R-FM (rituximab, fludarabine, mitoxantron) were similar (88%, 93% and 91%, respectively, p = 0.247), but improved PFS of R-CHOP and R-FM over R-CVP were demonstrated.15 The PLRG4 trial compared R-CVP to R-CHOP followed by rituximab maintenance for the treatment of indolent lymphoma in need of induction therapy (including FL). Overall response rate (ORR) in the R-CVP arm was 97.1%, and in the R-CHOP arm – 94.5% (p = 0.218). No differences is PFS or OS were demonstrated.16
The Gallium study was not designed to compare differences within chemotherapy groups combined with rituximab or obinutuzumab, but ORR and PFS were superior with obinutuzumab to rituximab, with consistent effects across chemotherapy regimens.5 In any of these trials, OS difference was demonstrated.
The events of special interest for safety evaluation were IRRs, which usually occur during the 1st administration of obinutuzumab.9 In a phase IV GAZELLE study, where safety of 90-minute infusion of obinutuzumab from C2 was evaluated in 101 patients,17 IRRs were observed in the 1st cycle of treatment with a standard infusion duration. A single patient experienced grade >3 IRRs beyond the 1st cycle. In our study, the rate of IRRs was even lower and they occurred only during the 1st obinutuzumab infusion. We assume that this phenomenon can be associated with the underreporting of adverse events in retrospective studies. The initial dose of obinutuzumab was split in 2 in half of the patients; however, the incidence of IRRs was similar no matter if the dose was split or not. An amendment of the CLL11 study protocol changing the administration of the 1st obinutuzumab infusion to patients with chronic lymphocytic leukemia (CLL) from 1 day to 2 days resulted in a decreasing grade 3 or 4 IRRs to 20%.18 The practice adapted from the CLL therapy probably does not need to be extended to patients with FL. The occurrence of IRRs in FL and CLL patients beyond the 1st infusion are only occasional; thus, shortened duration of obinutuzumab infusions starting from the 2nd can be used as a routine, and since October 2021, obinutuzumab is approved for induction treatment of FL.9, 14
Neutropenia was the most common AE that required therapeutic use of G-CSF and delay of subsequent cycles of treatment. Unfortunately, 5 patients died from COVID-19; there were no other deaths in this group of patients. Therefore, the protection of patients requiring treatment with anti-CD20 antibodies against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a high priority, with measures including appropriate vaccination and/or pre-exposure treatment with anti-SARS-CoV-2 antibodies, as well as early treatment with anti-viral agents in case the infection occurs.
Conclusions
The first-line treatment of follicular lymphoma patients with obinutuzumab combined with cytotoxic chemotherapy is efficacious and well-tolerated. The response to treatment with 3 different chemotherapy regimens was similar. Low grade and incidence of infusion-related reactions that exclusively occurred in the 1st administration of obinutuzumab support the use of abbreviated 90-minute infusions starting from the 2nd treatment cycle.