Abstract
Background. Blood concentration of galectin-3 (Gal-3) – a biomarker of fibrosis useful in diagnostics and prognostication in heart failure (HF) – is known to be elevated in patients with renal impairment, a condition which often accompanies cardiac insufficiency.
Objectives.To investigate the effect of moderately reduced renal function (estimated glomerular filtration rate (eGFR) 30–60 mL/min/1.73 m2) on the diagnostic and prognostic utility of circulating Gal-3 in patients with HF with preserved ejection fraction (HFpEF).
Materials and methods. Clinical, biochemical and echocardiographic variables were collected at baseline in 154 patients with HFpEF: 101 with normal and 53 with moderately reduced renal function, who were followed up for 48 (24–60) months for HF hospitalization and cardiovascular (CV) death (composite endpoint).
Results. Patients with moderately impaired renal function were characterized by higher age, Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) risk score, Gal-3, B-type natriuretic peptide (BNP) and New York Heart Association (NYHA) class, lower hemoglobin, and more advanced left ventricular diastolic dysfunction. Older age, female sex and deeper impairment of renal performance were determinants of higher Gal-3 blood concentrations. Lower exercise capacity (lower peak VO2) was associated with higher Gal-3 level and more pronounced renal impairment. Multivariable regression analysis demonstrated the significance of renal dysfunction as a determinant of lower exercise capacity and revealed a significant interaction between Gal-3 and eGFR with respect to peak VO2. The addition of Gal-3 to the prognostic models based on clinical data improved their predictive power for the study endpoint. The Kaplan–Meier analysis revealed that the presence of moderately reduced renal function with eGFR 30–60 mL/min/1.73 m2 did not enhance the increased risk of adverse outcome associated with Gal-3 above the median.
Conclusions. In patients with HFpEF, the coexistence of moderate renal dysfunction does not deteriorate the prognostic usefulness of circulating Gal-3. However, renal impairment modifies the association between Gal-3 and exercise capacity, which supports the need to adjust for kidney function when interpreting the contribution of Gal-3 to exercise intolerance in this population.
Key words: prognosis, galectin-3, renal dysfunction, heart failure with preserved ejection fraction
Background
Galectin-3 (Gal-3), a member of the beta-galactoside-binding lectin family, is a multifunctional protein secreted by activated macrophages, fibroblasts and vascular cells, and involved in the regulation of pro-inflammatory and profibrotic signaling.1 The pathophysiological relevance of Gal-3 for the heart has been documented in animal investigations showing the promotion of myocardial fibrosis by exogenous Gal-3,2 as well as the regression of fibrotic changes in response to Gal-3 inhibition.3 Given that cardiac fibrosis and inflammation play major roles in the development of heart failure (HF), especially the category with preserved ejection fraction (HFpEF), Gal-3 has emerged as an attractive biomarker in diagnostics and prognostication in this disease entity.4
Accumulating evidence from experimental and human studies indicates that Gal-3 can be useful in the diagnostic and prognostic strategies both in the preclinical and clinical phase of HF.2, 5 Circulating Gal-3 levels have been found to be associated with cardiovascular and all-cause mortality in HF population and general population, with exercise intolerance and treatment effects of angiotensin receptor blockers and statins.6, 7, 8, 9, 10 However, Gal-3 is not a cardiac-specific marker. Chiefly, decreased renal function has been shown to contribute to Gal-3 overexpression in the blood.11 This interference may provide a confounding effect, decreasing the diagnostic and prognostic usefulness of Gal-3 since renal impairment frequently accompanies HF.12
According to the existing evidence, advanced kidney compromise with estimated glomerular filtration rate (eGFR) <30 mL/min/m2 is associated with a significant increase in circulating Gal-3 of noncardiac origin.13 However, data reporting the impact of the coexistence of a lesser degree of renal insufficiency on the accuracy of Gal-3 as a cardiac marker are scarce.
Objectives
In view of this, we sought to investigate the effect of moderately reduced renal function (eGFR 30–60 mL/min/
1.73 m2) on the diagnostic and prognostic utility of circulating Gal-3 in patients with exertional dyspnea categorized as having HFpEF.
Materials and methods
Patient selection
In this study, we enrolled 154 patients who were diagnosed at a tertiary cardiology center between 2012 and 2015 due to exertional dyspnea, and fulfilled the HFpEF criteria in place at the time of recruitment14: 1) signs of HF classified as New York Heart Association (NYHA) functional class II or III; 2) exercise capacity below normal (<100% of age- and sex-predicted normal ranges for peak oxygen consumption; 3) preserved left ventricle ejection fraction (>50%); and 4) evidence of the left ventricular diastolic dysfunction (LVDD). The exclusion criteria included: 1) atrial fibrillation or flutter (irregular heart rhythm decreases the accuracy of echocardiographic evaluation); 2) significant ischemic heart disease (defined by the presence of atherosclerotic lesions on coronary angiography or inducible ischemia during exercise testing); 3) moderate or severe valvular heart disease; 4) obesity (body mass index (BMI) >36 kg/m2); 5) anemia (hemoglobin (Hb) <11 g/dL); and 6) suspected or confirmed pulmonic, liver, rheumatic, oncological, skeletal, or kidney (creatinine eGFR < 30 mL/min/1.73 m2) disease. The study cohort was divided into 2 subgroups: with eGFR > 60 mL/min/1.73 m2 (n = 101) and with eGFR 30–60 mL/min/1.73 m2 (n = 53).
The study protocol comprised medical interview and examination, cardiopulmonary exercise testing, echocardiography, and assessment of blood biomarkers: serum Gal-3 and plasma B-type natriuretic peptide (BNP).
All enrollees provided written informed consent following obtaining full information on the essence of the study. Investigations were approved by the institutional review board (Wroclaw Medical University Bioethics Committee approval No. 731/2018) and were conducted in accordance with the Declaration of Helsinki.
Outcome
Enrollees were followed up over a median period of 48 (24–60) months for the composite endpoint of HF hospitalization (defined as hospital admission due to HF worsening requiring intensification of diuretic therapy) or cardiovascular (CV) death. The event rate was verified through systematic contact with patients or their authorized relatives. A standard clinical history was taken, including information about risk factors, etiology and treatment.
Standard equipment (Vivid e9; General Electric Medical Systems, Milwaukee, Wisconsin) with a phased array 2.5-MHz multifrequency transducer was used for echocardiographic imaging. Ultrasound data were saved in the digital format on a secure server and analyzed offline.
Cardiopulmonary exercise testing
and blood assays
A symptom-limited exercise testing was performed on a treadmill, according to Bruce protocol, with standard ECG and blood pressure monitoring. Ventilation, oxygen uptake and carbon dioxide production were monitored continuously, and peak oxygen uptake (peak VO2) was computed as the average oxygen consumption during the last 30 s of exercise. Peripheral venous blood samples were drawn between 8:00 and 9:00 AM after 30 min of rest in the supine position, and subsequently frozen at −70°C. Serum Gal-3 levels were estimated using enzyme-linked immunosorbent assay (ELISA) kits from BioVendor, Inc. (Brno, Czech Republic). Intra- and inter-assay coefficients of variation were 6.4% and 11.4%, respectively. Serum BNP levels were quantified using commercially available fluorescence immunoassay from Biosite Diagnostics Inc. (San Diego, USA).
Risk stratification
Based on the age, sex, presence of diabetes, and chronic obstructive pulmonary disease (COPD), current smoking status, duration of HF, HF functional class, use of β-blockers and angiotensin converting enzyme inhibitors/angiotensin receptor blockers, systolic blood pressure (SBP), BMI, serum creatinine, and left ventricular ejection fraction, the Meta-Analysis Global Group in Chronic Heart Failure (MAGGIC) risk score was calculated.15
Statistical analyses
Data are presented as mean ± standard deviation (SD) for normally distributed variables, as median (interquartile range (IQR)) for skewed variables (Gal-3, BNP), and as counts and percentages for categorical variables. Before being analyzed, the BNP was subjected to logarithmic transformation and Gal-3 to Box–Cox transformation. Between-group comparisons were performed with an unpaired two-sided Student’s t-test. Associations between variables were evaluated using univariable and stepwise multiple regression analysis. The interaction between Gal-3 and eGFR with respect to prediction of exercise capacity was tested using a general linear model. Nested Cox regression models were built to evaluate the incremental value of BNP, eGFR and Gal-3 for prediction of the composite endpoint. In the 1st analysis, the base model included the MAGGIC risk score, and BNP and Gal-3 were successively added in the next steps. In the 2nd analysis, eGFR and Gal-3 were consecutively added to patient age. The change in overall log-likelihood ratio χ2 value was used to estimate the increase in predictive power after the addition of subsequent parameter. To evaluate model performance, the Harrell’s C-statistic was used. Univariable and multivariable Cox proportional hazards models were used to assess the associations of analyzed parameters with the study endpoint. Event-free survival curves were estimated using the Kaplan–Meier method, and differences were assessed with the log-rank test. All calculations were carried out using standard statistical software (STATISTICA v. 13; TIBICO Software Inc., Palo Alto, USA). The level of statistical significance was set at p-value <0.05.
Results
Patient characteristics
A total of 154 patients were finally enrolled into the study and divided into 2 subgroups: with moderately reduced renal function (eGFR 30–60 mL/min/1.73 m2 (n = 53)) and normal renal function (eGFR > 60 mL/min/1.73 m2 (n = 101)). The demographic and clinical profile of the studied population presented in Table 1 was typical of HFpEF, with the predominance of female sex, hypertension and overweight/obesity. There were significant differences between groups: the subgroup with reduced renal function was characterized by older age, higher MAGGIC risk score and Gal-3 and BNP concentration, lower hemoglobin level, more advanced exertional dyspnea as expressed by NYHA class, and lower exercise capacity as assessed by peak oxygen uptake. Echocardiographic characteristics of study population are presented in Table 1. Patients with reduced renal function demonstrated abnormalities of left ventricle (LV) diastolic parameters: peak early diastolic tissue velocity of the mitral annulus (e’) and the ratio of peak early diastolic mitral inflow velocity of the mitral annulus (E) to e’. There were no significant differences in other demographic, clinical and echocardiographic variables between both subgroups.
Determinants of Gal-3 concentration in HFpEF population
Univariable and multivariable linear regression analysis revealed that older age, female sex and deeper impairment of renal function were significant independent determinants of higher Gal-3 concentration in the studied population (Table 2).
Gal-3 and renal function as determinants of exercise capacity in HFpEF population
Univariable linear regression analysis demonstrated significant associations of Gal-3 and eGFR with exercise capacity expressed by peak VO2. Multivariable linear regression model revealed the presence of significant interaction between Gal-3 and eGFR with respect to peak VO2 (Table 3).
Prognostic utility of Gal-3 in the prediction of HF hospitalization and CV death in HFpEF patients
Univariable associations of demographic and clinical predictors of the study composite endpoint (HF hospitalization or CV death) in Cox proportional hazards analysis are presented in Table 4. A higher risk of the adverse outcome significantly correlates with higher Gal-3 and BNP concentration, more advanced age, higher MAGGIC risk score, and more severe renal function impairment.
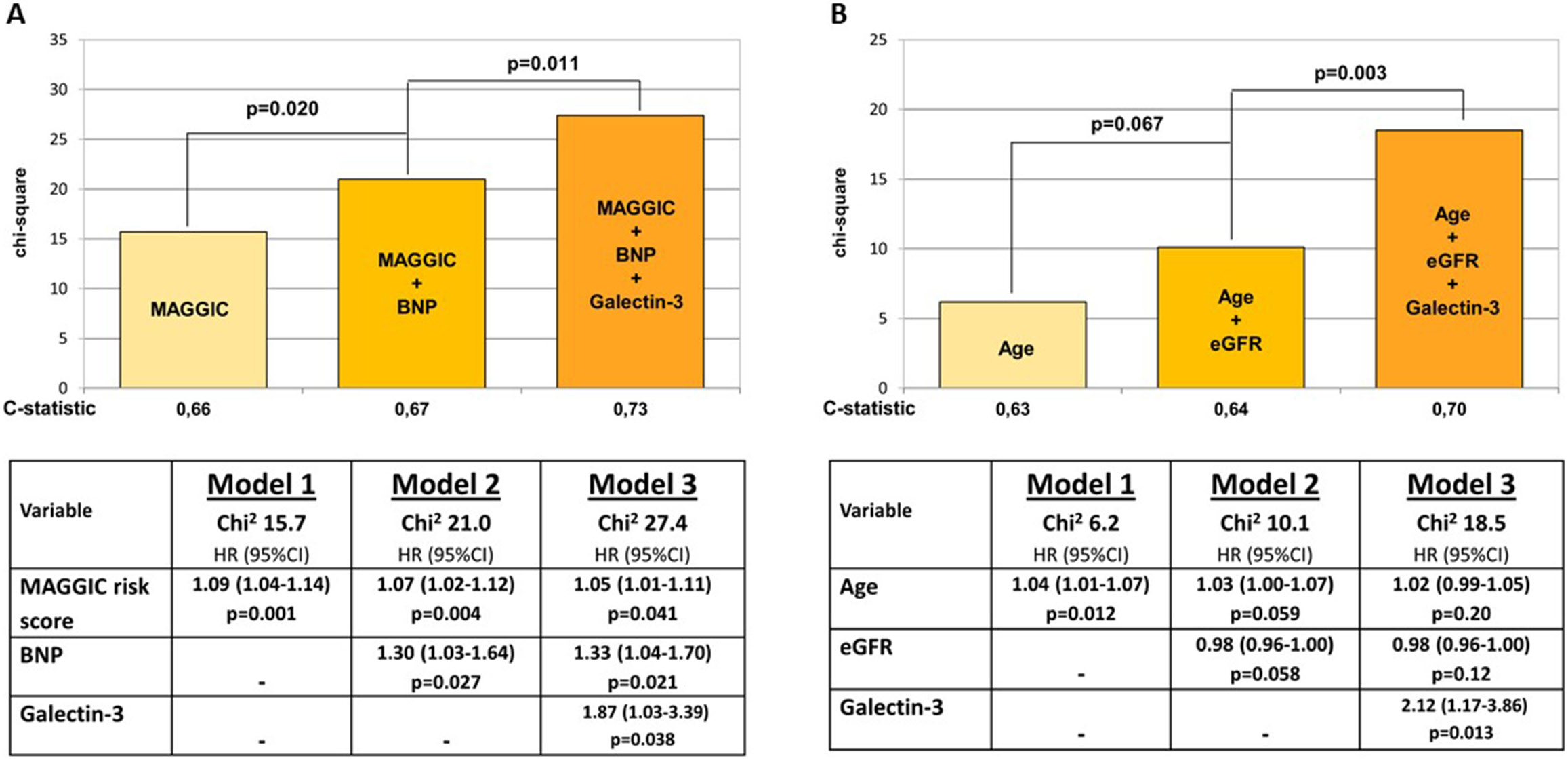
In the sequential Cox analysis presented in Figure 1A, the addition of Gal-3 to the model including MAGGIC risk score and BNP improved the predictive power for HF hospitalization or CV death. Analogically, the addition of Gal-3 to the model including patient age and eGFR provided an incremental predictive benefit for the composite study endpoint (Figure 1B).
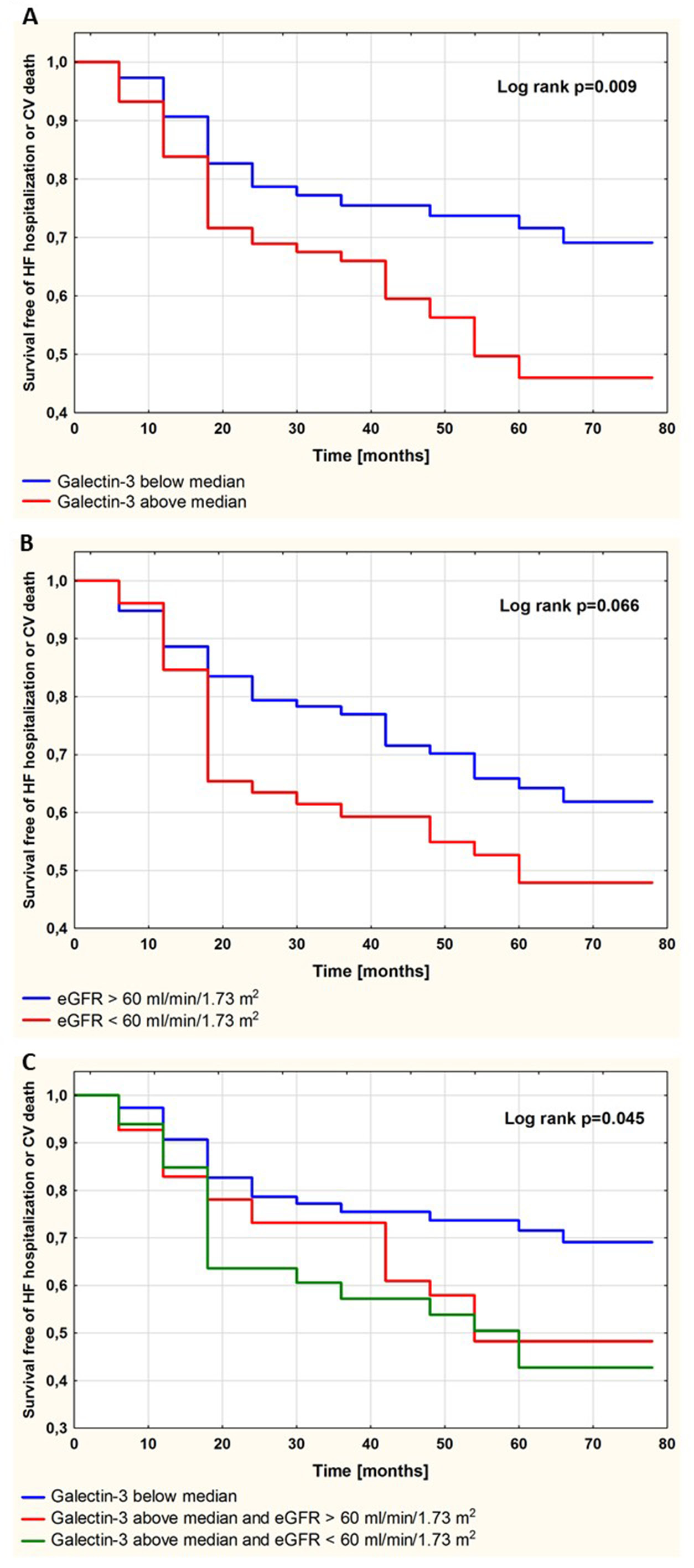
Survival free of the study endpoint is shown in Figure 2. Patients with Gal-3 concentration above the median (1.30 ng/mL) were at higher risk for HF hospitalization or CV death in comparison with patients with Gal-3 concentration below the median (p = 0.009; Figure 2A). There was a trend towards a higher incidence of the study endpoint in patients with eGFR 30–60 mL/min/1.73 m2 compared to patients with normal renal function (p = 0.066; Figure 2B). The increased risk for adverse outcome associated with higher (above the median) Gal-3 was not amplified by the coexistence of moderate renal impairment (eGFR 30–60 mL/min/1.73 m2; p = 0.60; Figure 2C).
Discussion
The major findings of this study are that in patients with exertional dyspnea and possible HFpEF, Gal-3 provides prognostic information that is independent from and incremental to clinical data and BNP, and the value of this marker in predicting adverse outcome is similar between subjects with normal and moderately reduced (eGFR 30–60 mL/min/1.73 m2) renal function. The association between Gal-3 and exercise capacity is modified by renal function; therefore, kidney performance should be taken into account when using Gal-3 for defining the pathophysiologic phenotype of exercise intolerance in this population.
Galectin-3 is implicated in the fibrotic and inflammatory processes, and its expression is increased in damaged and remodeling tissues, irrespective of the affected organ and etiology of injury. The contribution to the blood Gal-3 pool can come from a variety of sources of increased fibrosis. Accordingly, the coexistence of multiple organ diseases, one of the most frequent of which is cardiorenal syndrome, constrains the diagnostic ability of circulating Gal-3 to track specific pathologies, as well as specific organ-related outcomes.
The clearance of Gal-3 is thought to be largely hepatic; however, renal excretion of this protein also appears to play a role.1, 16 Some data indicate that the urinary elimination of Gal-3 is impaired in HF even if the kidney function is normal.11 It has been postulated that the impact of renal disease on HF worsening may in part be mediated by the Gal-3 pathways that aggravate cardiac remodeling and dysfunction.17, 18, 19
The prognostic significance of Gal-3 has been demonstrated both in acute and chronic HF across a wide spectrum of kidney function.20, 21, 22, 23 However, in multivariate adjustments including renal impairment, the significance of independent predictive contribution from Gal-3 was lost in some studies.13, 24, 25 Interestingly, the association of higher Gal-3 with adverse outcome in predialysis cohorts was preserved for all-cause mortality but not for HF progression, which might suggest that in patients with more severe renal dysfunction, the prognostic profile of Gal-3 becomes less cardiac-oriented.26
The commonly believed notion that in the setting of advanced chronic kidney disease (CKD) Gal-3 is not a useful marker of cardiac injury, does not exclude the possibility that a lesser degree of renal impairment can be more neutral in terms of limiting the diagnostic utility of Gal-3 in cardiovascular disease (CVD). The current study demonstrated that in patients with exertional dyspnea satisfying the former criteria for HFpEF and having only a moderately reduced kidney function, circulating Gal-3 is a robust prognostic marker, providing predictive information independent from demographic and clinical data, including renal function status. It is worth emphasizing that the prognostic significance of Gal-3 was corroborated after adjustment for the MAGGIC risk score – a widely approved risk-stratifying tool in HF, including several risk-associated variables.
The incremental value of Gal-3 in predicting adverse outcome was evident despite the association of this biomarker with eGFR, which, however, was weaker than that reported in prior studies including subjects with more advanced renal impairment. Thus, our results support the use of Gal-3 in the prognostic assessment of patients suspected of having HFpEF in the absence of severe kidney dysfunction.
The association between Gal-3 and exercise capacity has a pathophysiological basis. Higher Gal-3 reflects more intense myocardial fibrosis, which is a substrate for increased LV stiffness, leading to LV filling pressure elevation and pulmonary congestion. Abnormal LV filling is exaggerated by exercise, which is clinically evidenced as exertional dyspnea. On the other hand, renal disease can contribute to myocardial dysfunction through a number of mechanisms, including changes in neurohormonal, metabolic and pro-inflammatory milieu, as well as in cardiac loading conditions.
This study revealed that both Gal-3 and eGFR were associated with peak oxygen consumption. The presence of significant interaction between these predictors in multivariable analysis suggests the interplay of fibrosis and renal impairment in their effect on exercise capacity. This finding indicates that the role of Gal-3 in the pathophysiology of exercise intolerance should be considered in the context of kidney function.
Limitations
First, the analysis in this paper was limited to the demographic, clinical and laboratory factors. The contribution of echocardiographic parameters to the discussed aspects was presented in the previous papers analyzing the currently studied population.27, 28 Second, the clinical profile of this study population, with exclusions of atrial fibrillation, coronary artery disease (CAD) and COPD, may constrain the extrapolation of our findings to other patient cohorts with exercise intolerance, suspected of HFpEF. Third, the enrollment of patients from a single academic center might limit the generalizability of our results.
Conclusions
In patients with exertional dyspnea classified as HFpEF, the coexistence of moderately reduced renal function does not deteriorate the prognostic usefulness of circulating Gal-3. However, renal impairment modifies the association between Gal-3 and exercise capacity, which supports the need to adjust for kidney function when interpreting the contribution of Gal-3 to exercise intolerance in this population.