Abstract
Background. Esophageal cancer is one of the most lethal tumors worldwide. The most common histological type in China is esophageal squamous cell carcinoma (ESCC), accounting for 90% of cases. Esophageal cancer occurs at a high incidence in certain areas, among which China has the highest incidence. Although various therapeutic strategies have been used in clinical treatment, the 5-year survival rate is still not satisfactory, as it is only 15–20%. The reason for the poor prognosis of ESCC is that the distant metastasis easily occurs in these tumors. However, the mechanism of metastasis has not been studied clearly.
Objectives. To investigate the function of hypoxia-inducible factor-2α (hif-2α) in ESCC.
Materials and methods. Immunohistochemistry and immunofluorescence were used to detect the expression of hif-2α in tissues and cells. Clinicopathological data from 100 ESCC patients were used to investigate the relationship between hif-2α and prognosis. Cell experiments (Cell Counting Kit-8 (CCK-8) assay and transwell migration assays) were utilized to verify the roles of hif-2α on the ESCC cells. Western blotting was used to explore the mechanism of hif-2α in ESCC. Mouse model was used to clarify the effect of hif-2α on ESCC cells in vivo.
Results. The hif-2α was overexpressed both in ESCC tissues and cells, and was related with poor prognosis in ESCC patients. The CCK-8 assay evidenced that silencing hif-2α suppressed the proliferation of ESCC cells, while transwell assay – that overexpression of hif-2α promoted the migration of ESCC cells. Western blot assay indicated that hif-2α regulated epithelial–mesenchymal transition (EMT) through Notch pathway in ESCC cells. Mouse model showed that silencing hif-2α significantly suppressed the proliferation of ESCC cells in vivo.
Conclusions. The hif-2α promotes EMT in ESCC through the Notch pathway.
Key words: esophageal squamous cell carcinoma, Notch signaling pathway, epithelial–mesenchymal transition, hypoxia-inducible factor-2α
Background
Esophageal cancer is one of the most lethal tumors worldwide. The most common histological type in China is esophageal squamous cell carcinoma (ESCC), accounting for 90% of cases.1 Esophageal cancer occurs at a high incidence in certain areas, among which China has the highest incidence.1 Although various therapeutic strategies have been used in clinical treatment, the 5-year survival rate is still not satisfactory, as it is only 15–20%.2 The reason for the poor prognosis of ESCC is that the distant metastasis easily occurs in these tumors,3 especially lymphatic metastasis.4 However, the mechanism of metastasis has not been studied clearly.
It has been reported that hypoxia can induce ESCC to undergo EMT through several mechanisms, in which hypoxia-inducible factor-1α (hif-1α) plays an important role.5 The overexpression of hif-1α correlates with lymph node metastasis and poor prognosis in patients with ESCC.6 The hif-2α is a member of the hypoxia-inducible factor family and is homologous to hif-1α. It has been reported to play important roles in several cancers.7 The hif-2α is persistently upregulated under mild hypoxia conditions and can regulate the epithelial–mesenchymal transition (EMT) process through crosstalk with Wnt/β-catenin signaling in pancreatic cancer.8 However, there have been no studies on the role of hif-2α in ESCC yet. Therefore, the role of hif-2α in ESCC is still unknown and requires further investigation.
The Notch signaling pathway is an evolutionarily conserved local cell signaling pathway that participates in a variety of cellular processes. These processes include cell differentiation, proliferation, apoptosis, adhesion, EMT, migration, and angiogenesis, and Notch signaling pathway affects embryonic development, tissue balance and whole-body immunity.9 The Notch signaling pathway has been reported to act as an oncogene in various cancers, including lung cancer, breast cancer, colorectal cancer, prostate cancer, T-cell acute lymphoblastic leukemia (T-ALL), and other malignancies.10 The Notch signaling has been proven to have crosstalk with hif-1α in some types of cancers, and this crosstalk consequently results in changes in EMT markers, such as breast cancer.11 Moreover, some studies have shown that aberrant activation of Notch signaling plays important roles in the progression of ESCC and predicts poor prognosis in ESCC patients.12 However, the specific mechanism of Notch signaling in ESCC remains unknown, especially the mechanism by which it regulates the EMT process.
Objectives
In this study, we investigated the role of hif-2α by regulating EMT in ESCC through the Notch signaling pathway and identified hif-2α as a factor that predicts poor prognosis in ESCC patients.
Materials and methods
Patients
One hundred patients who were diagnosed with ESCC and underwent esophageal cancer surgery in the Department of Thoracic Surgery at Shandong Provincial Hospital, Jinan, China, from January 2014 to December 2015, were included in this project. The inclusion and exclusion criteria were as follows:
1. ESCC confirmed by pathology after surgery;
2. chemotherapy and radiotherapy not administered to patients before surgery;
3. contraindications of surgery excluded during preoperative examination;
4. patients who died due to complications or accidents after surgery were excluded.
The tumor was completely excised in all patients, and no tumors were visible to the naked eye in the surgical field. The upper and lower resection margins were determined to be cancer cell-negative by pathology. The TNM stage was determined using the 8th edition of TNM staging criteria for esophageal cancer in 2017 according to the International Union Against Cancer.
The experimental protocol and informed consent procedure were in compliance with the Declaration of Helsinki. The study was approved by the ethics committee of the institutional ethics committee of Shandong Provincial Hospital Affiliated to Shandong First Medical University (approval No. SWYX:NO.2022-109) according to the Guide for Chinese Ethics Review Committees, and informed consent was obtained from all individual participants included in the study. Written informed consent was obtained from the patients for publication of their individual details and accompanying images in this manuscript.
Cell culture and reagents
Esophageal squamous cell carcinoma cell lines (Eca-109 and KYSE-150) were obtained from the Shanghai Institutes for Biological Sciences (Shanghai, China). The KYSE-150 cells were cultured in Roswell Park Memorial Institute (RPMI)-1640 medium (GE Healthcare Life Science, Logan, USA). The Eca-109 cells were cultured in high-glucose Dulbecco’s modified Eagle’s medium (DMEM; Gibco, Logan, USA). Both media were supplemented with 10% fetal bovine serum (FBS, Biological Industries, Kibbutz Beit Haemek, Israel). All cells were cultured at 37.0 ±0.2°C in a humidified incubator with 5.0% CO2. Before exposure to the hypoxic environment (3.0% O2), cells were cultured under normoxic conditions and grown to approx. 60% confluence. Then, the cells were cultured in a low-oxygen environment in a hypoxic incubator (Thermal Tech, Orlando, USA) for the indicated duration. Cobalt chloride (CoCl2) was purchased from Sigma-Aldrich (St. Louis, USA) and used at 50 mM concentration for 48 h. The Notch antagonist CB-103 was purchased from MCE (HY-135145; MedChem Express, Monmouth Junction, USA) and was used at 20 mM for 48 h.
Transfection
Transfections were performed using Lipofectamine 3000 (Invitrogen, Carlsbad, USA) according to the manufacturer’s instructions. All experiments were performed after the transfection medium was replaced by complete medium after 24 h of treatment. The hif-2α overexpression plasmid and silencing siRNAs were designed by and purchased from GeneChem Company (Shanghai, China).
CCK-8 assay
Prior to using Cell Counting Kit-8 (CCK-8), the cells were seeded in 96-well plates at 2000 cells per well for specific treatments. Cell viability was evaluated using CCK-8 (Dojindo, Kumamoto, Japan). After incubating the cells with CCK-8 working solution for 30 min, the absorbance was measured at 450 nm using an ELx808 microplate reader (BioTek, Santa Clara, USA). Relative cell viability was expressed as a percentage of a particular control.
Transwell assay
The tumor cell infiltration activity was evaluated by its ability to penetrate the gel matrix (Matrigel; Becton Dickinson, Franklin Lakes, USA). In short, the Matrigel solution was diluted 1:8 in FBS-free medium and filtered with 6.5 mm diameter (8-μm holes) polycarbonate in a 24-well transwell box (Corning, USA) for film coverage. All filters were filled with 40 mL of working solution, and the plates were cured at 37°C for at least 5 h. Tumor cells were seeded at a density of 2 × 105 in each chamber. They were cultured in the upper compartment of the chamber in FBS-free medium for 48 h, and the lower chamber was filled with complete medium. The filter was wiped with a cotton swab, and the cells attached to the bottom were fixed with 4% formaldehyde polyphosphate (Beyotime, Shanghai, China). After staining with crystal violet (Beyotime), the cells with greater infiltration capacity were counted. The experiment was independently repeated 3 times.
Immunohistochemistry
and immunofluorescence
The immunohistochemistry study was carried out using the streptavidin-peroxidase method. Formalin-fixed and paraffin-embedded ESCC tissue was cut into 5-μm thick sections, deparaffinized and then incubated with hydrogen peroxide. Goat serum was used at room temperature for 1 h for blocking before the primary antibody was applied. The primary antibodies used to perform immunohistochemistry were as follows: anti-hif-2a (NB100-122; Novus, St Charles, USA), anti-E-cadherin (20874-1-AP), anti-N-cadherin (22018-1-AP), and anti-vimentin (10366-1-AP). These antibodies were purchased from Proteintech (Beijing, China). The primary antibody was added to tissue slides and incubated overnight at 4°C. The next step followed the instructions of the secondary biotinylated antibody kit (Zhongshan Biotech, Guangzhou, China). The stained slides were independently evaluated by 2 observers.
Before immunofluorescence staining was performed, 4% polyphosphate formaldehyde was used to fix the cells for 15 min. After incubation with 1% Triton for 30 min, the cells were blocked with goat serum for 1 h at room temperature. Then, the cells were incubated overnight at 4°C with rabbit anti-hif-2α antibody (NB100-122; Novus) and incubated with secondary antibodies conjugated to fluorescent dyes for 1 h at room temperature in the dark. After counterstaining with 4′,6-diamidino-2-phenylindole (DAPI) (C0065; Solarbio, Beijing, China) for 5 min, the cells were observed under an inversion fluorescence microscope.
Protein extraction and western blotting
Mouse model
The Eca-109 cells transfected with siRNA and negative-control lentivirus were used to investigate the effect of hif-2α on tumor progression in vitro. Sixteen 5-week-old female BALB/c nude mice were randomly divided into 2 groups, with 8 mice in each group. The negative control (NC)-siRNA and siRNA cells (1 × 106) were independently injected subcutaneously into the flanks of mice. Then, the tumor sizes were measured every 5 days and weighed after 20 days. The following formula was used to calculate the volume of the tumor: V = L × W2/2, in which L is the largest and W is the smallest diameter. The protocol was approved by the ethics committee of the Provincial Hospital affiliated with Shandong First Medical University, Jinan, China (approval No. LCYJ:NO.2019-164).
Statistical analyses
The statistical data are presented as the mean ± standard deviation (SD). Tests of normality and homogeneity of variance were performed before the analysis of differences (Supplementary Table 1). The analysis of differences between the 2 groups was performed using Student’s t test and multiple groups were compared using one-way analysis of variance (ANOVA) if the data were normally distributed and had uniform variance. Otherwise, Mann–Whitney test was used to analyze differences between the 2 groups and multiple groups were compared using Welch’s test. Regarding the post hoc analysis among multiple groups, Bonferroni’s test was used if the data had uniform variance. Otherwise, Tamhane’s T2 test was used. The relationships between hif-2α and clinicopathological information displayed in Table 1 were calculated using Fisher’s test. The information of Kaplan–Meier curve is presented in Supplementary Table 2. The multivariate Cox regression and verification of assumption of proportional hazard (Supplementary Table 3) were performed using IBM SPSS software v. 24.0 (IBM Corp., Armonk, USA). In the multivariate Cox regression, age, gender, hif-2α, maximal dimension of tumor, lymph node metastasis, and TNM stage were explanatory variables, while response variables were survival time and outcome status. Differences were considered to be significant if the p-value <0.05. The IBM SPSS v. 24.0 and GraphPad Prism v. 7 (GrahPad Software, San Diego, USA) statistical software were used to perform all statistical calculations and to construct graphs.
Results
Hif-2α is overexpressed in ESCC
and relates with poor prognosis
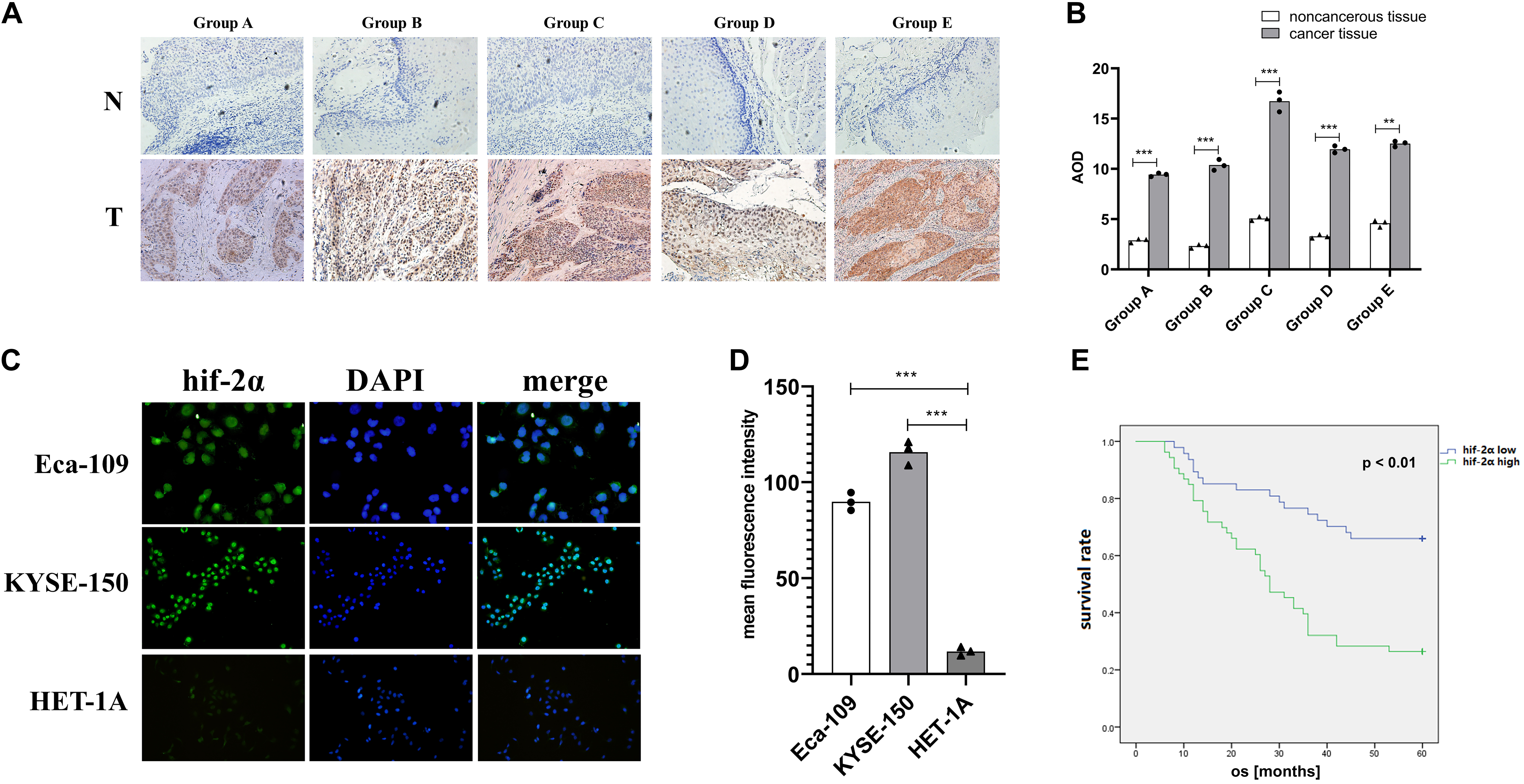
To investigate whether hif-2α is overexpressed in ESCC tissues and cell lines, immunohistochemistry and immunofluorescence were performed. Immunohistochemistry showed that ESCC tissues expressed high levels of hif-2α, but hif-2α was hardly detected in normal esophageal epithelial cells (Figure 1A,B). In addition, the results show that hif-2α is mainly aggregated in the nucleus, which is consistent with the transcription factor role of hif-2α. Immunofluorescence results suggested that hif-2α could be detected in the ESCC cell lines Eca-109 and KYSE-150 under normoxia conditions and was located mainly in the nucleus (Figure 1C,D), which is consistent with the immunohistochemistry results.
The relationship between hif-2α expression levels and clinical factors in ESCC patients was also studied. The expression level of hif-2α was divided into high and low groups according to the H-score. The overexpression of hif-2α was significantly related with lymph node metastasis (p = 0.009) and tumor stage (p = 0.018) (Table 1). In addition, the hif-2α expression level was a predicting factor for prognosis (hazard ratio (HR): 4.538; 95% confidence interval (95% CI): [1.426; 14.44], p = 0.01) (Table 2). Moreover, lymph node metastasis was also indicated to be a prognostic factor (HR: 2.411; 95% CI: [1.284; 4.527], p = 0.006) (Table 2). To investigate the relationship between the hif-2α expression level and overall survival (OS) of patients with ESCC, a Kaplan–Meier curve was constructed, and the results showed that patients with a low expression level of hif-2α had a greatly improved OS rate (p = 0.015) (Figure 1E).
Hypoxia induces EMT and hif-2α overexpression in ESCC
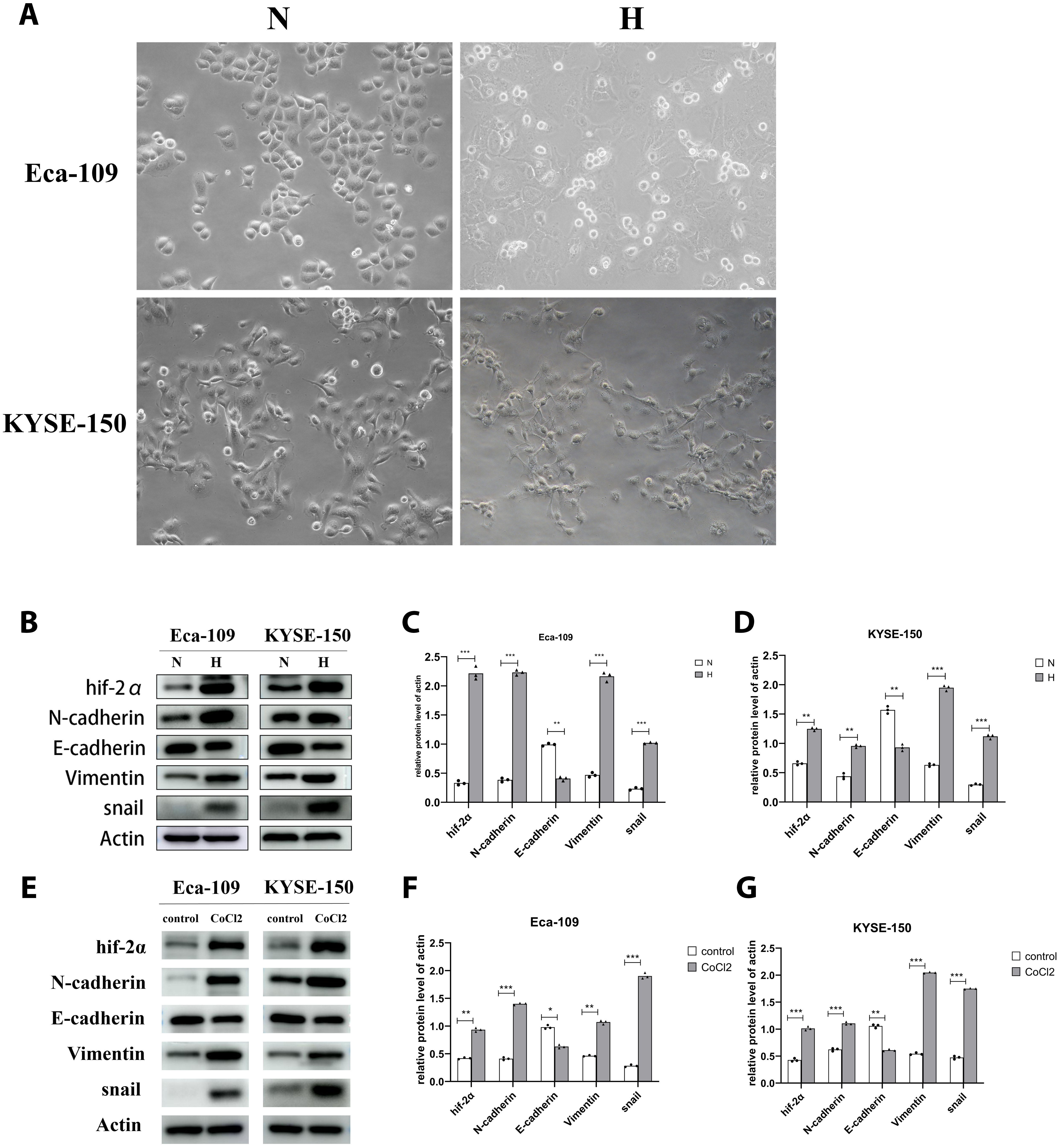
Two esophageal cancer cell lines were used to mimic hypoxia-induced EMT. As expected, morphological changes of the cells were observed after treatment with hypoxic conditions for 48 h (Figure 2A). These findings were accompanied by a decreased expression of E-cadherin and increased expression of hif-2α, N-cadherin, vimentin, and Snail (Figure 2B–D). We also used CoCl2 to mimic hypoxia.13 The expression of E-cadherin was decreased, and the expression of N-cadherin, vimentin and Snail was increased (Figure 2E–G).
Hif-2α promotes the proliferation and invasion of esophageal cells
The hif-2α has been reported to promote cell proliferation and invasion in different tumors, but the role of hif-2α in esophageal cancer cells has not yet been studied. To investigate whether hif-2α could promote proliferation and invasion in esophageal cells, CCK-8 and transwell assays were performed. The CCK-8 assay showed that the optical density (OD) value (450 nm) of the hif-2α-silenced group was significantly decreased at 24 h, 48 h, 72 h, and 96 h in both Eca-109 and KYSE-150 ESCC cells (Figure 3A). Moreover, silencing hif-2α resulted in a strong inhibitory effect on proliferation in Eca-109 cells. For the transwell assay, compared to the control and NC-siRNA groups, the number of hif-2α-silenced ESCC cells entering the transwell membrane was significantly reduced (Figure 3B,C). This finding suggests that the invasion capability of ESCC cells was suppressed when hif-2α was silenced. In contrast, the hif-2α-overexpressing group showed significantly increased cell numbers penetrating the membrane compared to the control and negative control to overexpression (NC-OE) groups (Figure 3D,E). This finding indicates that the overexpression of hif-2α enhances the invasion ability of ESCC cells. These results suggest that hif-2α plays an important role in the proliferation and invasion of ESCC cells.
Hif-2α regulates EMT through
the Notch pathway
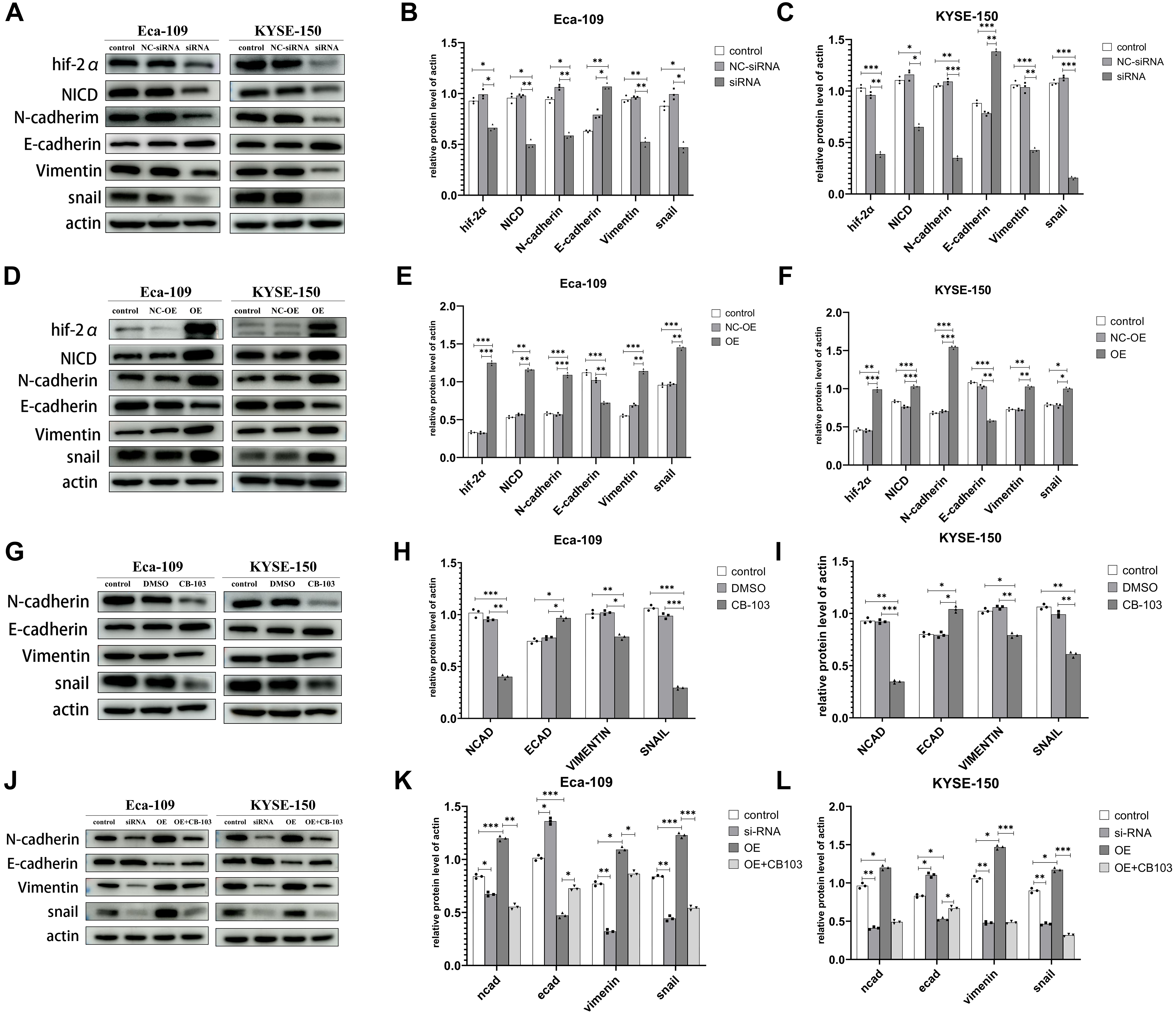
To determine whether hif-2α could regulate EMT in ESCC cells, western blot assay was performed to detect the protein levels of EMT markers. Compared to the control and NC-siRNA groups, the hif-2α-silenced group showed increased levels of E-cadherin and decreased expression levels of N-cadherin, vimentin and Snail in both Eca-109 and KYSE-150 ESCC cells (Figure 4A–C). Furthermore, the hif-2α overexpression group demonstrated the opposite results (Figure 4D–F). In addition, the expression level of NICD, which is the active form of Notch and functions as a transcriptional regulator, showed the same trend as hif-2α (Figure 4A,D). These results show that both EMT and Notch could be regulated by hif-2α. Next, the inhibitor CB-103 was used to study whether the Notch pathway could regulate EMT. The CB-103 is a specific and highly effective inhibitor of the Notch pathway. The results showed that CB-103 could significantly reverse the EMT process in ESCC cells (Figure 4G–I). Furthermore, a retrial experiment was carried out to investigate whether hif-2α regulated EMT in ESCC cells through the Notch pathway. The EMT process was reversed in hif-2α-overexpressing ESCC cells after the Notch pathway inhibitor was used (Figure 4J–L). These results suggest that hif-2α could regulate EMT through the Notch pathway in ESCC cells.
Silencing hif-2α suppressed tumor growth in a mouse model
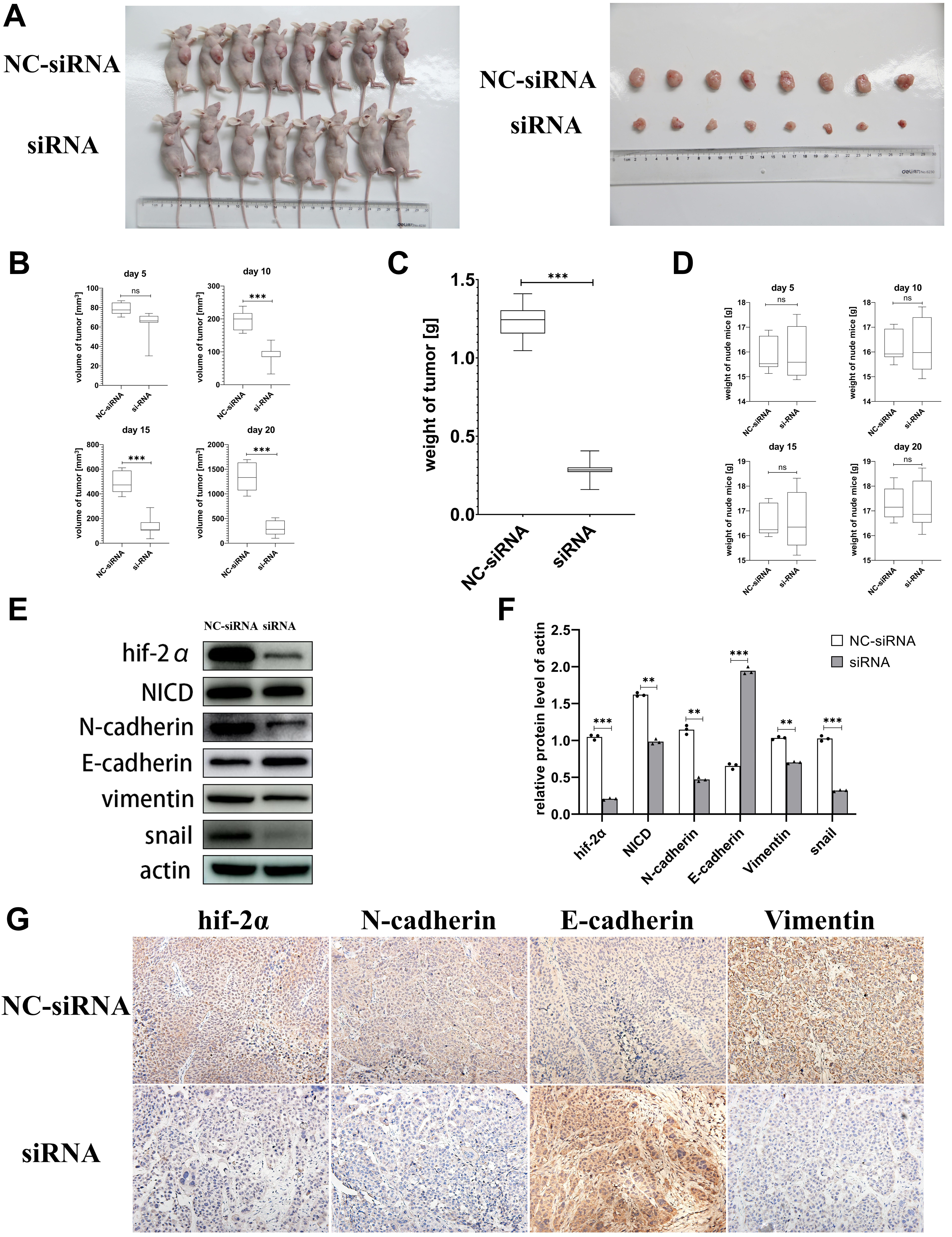
A xenograft mouse model was used to confirm the effect of hif-2α in vivo. Compared to the NC group, the tumor volume and weight of the siRNA group were markedly decreased (Figure 5A–C), and no significant difference in the body weight of nude mice was observed (Figure 5D). Besides the comparison of tumor volume between groups, comparisons within groups were also performed (Supplementary Fig. 1). This result suggests that inhibiting hif-2α significantly suppresses the growth of tumors without any apparent impacts on mouse growth. Immunohistochemistry and western blot analyses were performed using xenograft tumors to confirm that hif-2α could regulate EMT through the Notch pathway (Figure 5E–G). The results showed that hif-2α deficiency could significantly suppress Notch signaling and reverse EMT in vivo.
Discussion
Surgical resection is still considered the first line of treatment for early and local ESCC.14 However, ESCC is generally resistant to conventional therapeutic agents. In addition, the newest ESCC inhibitors are clinically ineffective. Therefore, finding new biological targets that may be useful in improving the prognosis of patients with ESCC has been a central issue in recent years.
In recent decades, hypoxia-inducible factor (HIF) has been widely and comprehensively researched. It is a key regulator in cells responsible for preventing damage from hypoxia, which is mainly regulated by the oxygen level in cells.15 Under normoxic conditions, HIF is modified by HIF-specific prolyl hydroxylases (PHDs), which lead to its degradation by von Hippel–Lindau tumor suppressors (pVHLs).16 When the cell is under hypoxic conditions, PHDs are inactivated and the VHL-dependent degradation pathway is blocked. This results in the accumulation of HIF and the activation of downstream genes. As a member of HIF, hif-2α has been reported to be associated with a poor prognosis in various cancers, such as non-small-cell lung carcinoma (NSCLC), breast cancer, colorectal cancer, hepatocellular cancer, and glioblastoma.7 However, the role of hif-2α in ESCC has not yet been studied. In this study, we identified hif-2α as a key molecule that regulates EMT, and we revealed that it can be a prognostic factor in ESCC patients. We found that hif-2α was overexpressed in both ESCC tissues and cell lines. The overexpression of hif-2α in tumor tissue might be due to the hypoxic tumor microenvironment, but the mechanism by which hif-2α is overexpressed under normoxia in ESCC cell lines remains unclear. A possible hypothesis could be that the transcription is increased or that the degradation pathway of hif-2α is impacted. However, these hypotheses require further investigation. In addition, we discovered the function of hif-2α in promoting proliferation and metastasis in ESCC. Both the in vitro and in vivo experiments verified the role of hif-2α in promoting the growth of ESCC cells. The function of hif-2α in promoting metastasis and regulating the expression of EMT markers was also confirmed in our investigation. In addition, we confirmed that hif-2α was associated with poor prognosis in patients with ESCC, characterized by a lower 5-year survival rate and a higher incidence of lymph node metastasis.
The Notch signaling pathway has been studied for over 100 years; it was first described by Thomas Hunt Morgan in 1917.9 Previous studies have proven that Notch functions as an oncogene in many types of cancers, and the mechanism of how the Notch signaling pathway works has been clearly interpreted.17 The transduction process of the Notch signaling pathway mainly includes the classical pathway that relies on CSL and CSL-independent transduction pathways.18 The classical pathway of Notch signaling is triggered by the interaction between the Notch ligand and the receptor. Then, the Notch receptor releases the active form of the Notch protein, that is, the intracellular segment of the ICN (NICD).19 The NICD enters the nucleus, where it acts as a transcription regulator. In recent decades, it has been confirmed that Notch signaling plays important roles in regulating the EMT process in different cancers, including esophageal cancer.20 The crosstalk between signaling pathways in cells is an extremely complicated network, which is not an exception for Notch signaling. It has been reported that both hif-1α and hif-2α can regulate Notch signaling in different cancers.21, 22 However, the relationship between HIF signaling and Notch signaling in ESCC remains unclear. In this study, Notch signaling was found to be downstream of hif-2α. We observed that the expression of NICD, the active form of Notch, could be regulated by hif-2α. As a modulator of EMT, NICD can enter the nucleus and regulate the transcription of downstream genes. Both EMT markers and NICD can be regulated by hif-2α, but whether EMT is regulated by hif-2α through the Notch signaling pathway remains unknown. Therefore, we carried out a retrieval experiment, and the results verified our speculation that Notch is the intermediary of hif-2α in regulating EMT in ESCC. As an oncogene, hif-2α is a potential target in cancer therapy, and a specific inhibitor of hif-2α has been proven to be efficient in clear cell renal cell carcinoma.23 According to this study, hif-2α plays an important role in the progression of ESCC. Thus, hif-2α inhibitors may be a new treatment target in ESCC therapy strategies and may improve the prognosis of ESCC patients in the future.
Limitations
This study still has some limitations in spite of clarifying the function of hif-2α in ESCC. For instance, we confirmed that Notch signaling pathway was downstream of hif-2α, but the in-depth mechanism of how hif-2α regulate Notch signaling pathway, by pre-translational or post-translational modification, was not clear. Furthermore, we confirmed that hif-2α could enhance the invasion ability of ESCC in vitro, but the experiment investigating whether hif-2α could promote metastasis of ESCC in vivo was not conducted because of the limitations of experimental conditions. Therefore, more in-depth and extensive research is still needed.
Conclusions
In general, this project illuminated a protumor role of hif-2α in esophageal cancer. The hif-2α enhanced the proliferation and invasion ability of ESCC cells and promoted EMT through the Notch signaling pathway. In addition, we identified hif-2α as an indicator of poor prognosis in ESCC patients and found that it was associated with shorter OS and lymph node metastasis. Hence, hif-2α may become a potential target for treating esophageal cancer and might serve as a new strategy to prolong the survival time of ESCC patients.
Supplementary data availability
The Supplementary material is available at doi:10.5281/zenodo.6316427. The Supplementary data consist of:
Supplementary Table 1. Verification of assumption for Student’s t test and ANOVA analysis.
Supplementary Table 2. Information of Kaplan–Meier analysis.
Supplementary Table 3. Verification of proportional-hazard assumption for COX model.
Supplementary Fig. 1. Comparison of tumor volume within NC-siRNA and siRNA groups.