Abstract
Background. (−)-Epigallocatechin-3-gallate (EGCG) is an active constituent of green tea, whose efficacy on chemoprevention and chemotherapy has been extensively researched. Its anticancer potency with low toxicity and easy administration allows for its widespread use. Bortezomib, a proteasome inhibitor, has a significant influence on multiple myeloma (MM) in chemotherapy. Previous studies about the role of EGCG in the antitumor effect induced by bortezomib remain controversial.
Objectives. In our study, the effect of EGCG on the antitumor activity of bortezomib was investigated in myeloma cell lines U266 and RPMI8226, and the underlying mechanism was explored.
Materials and methods. The effect of EGCG on the antiproliferative and pro-apoptotic activities of bortezomib were investigated in myeloma cells using MTT assay and cell cycle analysis, respectively. The Wnt/β-catenin signaling pathway and related proteins were involved in this antagonistic effect and measured with western blot assay.
Results. Our results showed the inhibitory activity of EGCG on myeloma cells in a time- and dose-dependent manner. The EGCG neutralized the antiproliferative and pro-apoptotic effect induced by bortezomib by activating Wnt/β-catenin signaling pathway with the accumulation of β-catenin. An increase of the downstream target proteins as c-Myc and cyclin D1 was also observed. was also observed. These findings demonstrated the antagonistic role of EGCG in the antitumor effect of bortezomib likely through the activated Wnt/β-catenin signaling pathway and the upregulated target proteins.
Conclusions. When bortezomib is involved in the MM chemotherapy, the consumption of green tea should be avoided in order to maintain the biological efficacy of bortezomib.
Key words: multiple myeloma, bortezomib, Wnt/β-catenin, apoptosis, (−)-epigallocatechin-3-gallate
Background
Multiple myeloma (MM) is one of the most common hematological malignancies, characterized by abnormal and out-of-control proliferation of plasma cells in bone marrow and monoclonal immunoglobulins, or immunoglobulin chain secretion in blood and/or urine in large amounts.1 Although highly efficient therapeutic agents are constantly emerging to improve the prognosis and the quality of life in a much better way than before, MM remains incurable.2, 3 At the same time, notable adverse effects of drugs and disease recurrence due to drug resistance became the key problems in treatment failure.4 Therefore, it is paramount to develop novel therapeutic strategies with high efficiency and low toxicity.
Green tea is recognized as one of the favorite beverages for people of all ages worldwide, bringing health benefits and reducing the risk of diseases. (−)-Epigallocatechin-3-gallate (EGCG), the main active ingredient of catechins in green tea, exhibits antioxidative, anti-inflammation, as well as antimicrobial and antitumor activity.5, 6, 7 Recently, many studies have identified the antiproliferative role of EGCG on carcinomas such as osteosarcoma, lung cancer, gastric cancer, melanoma, and so on; it may be involved in cell cycle modulation, induction of apoptosis and inhibition of angiogenesis.8, 9, 10, 11 However, the antitumor activity of EGCG on MM cells, especially when combined with proteasome inhibitors, as well as the related mechanism, is still unknown. Researchers considered that EGCG could counteract the antitumor effects of boronic acid-based proteasome inhibitors. Conversely, the synergistic effect of EGCG and proteasome inhibitors on growth inhibition on myeloma cells was also identified by other researchers.
The Wingless-Int (Wnt)/β-catenin signaling pathway plays an important role in regulating the cell proliferation and differentiation in various human cancers.12, 13 In MM, recent studies indicated that Wnt/β-catenin signaling pathway is activated by Wnt regulatory components, including deletion of tumor suppressor and overexpression of the co-transcriptional activator.14 Evidence provided by Sukhdeo et al. demonstrated that a small molecular compound, PKF 115-584, downregulates the Wnt/β-catenin signaling pathway, resulting in suppressing tumor growth in vitro and in vivo.15 Zhao et al. found that miR-30-5p, one of the tumor suppressor microRNAs, is downregulated in MM and further enhances the expression of BCL9.16 The BCL9, which is a coactivator for transcription in the Wnt signaling pathway, promotes myeloma cells proliferation, migration and invasion. The reproducibility of miR-30-5p as a therapeutic approach can reduce tumor loading and metastasis in vivo. These findings imply that Wnt/β-catenin signaling pathway is on the way to become a candidate for targeted therapy in MM.
Objectives
The objective of this research was to investigate the effect and underlying mechanism of EGCG on the antitumor activity of bortezomib in myeloma cells.
Materials and methods
Materials
The EGCG (Mw 458.4, purity ≥95%; Sigma-Aldrich, St. Louis, USA) was dissolved in water for storage at 2–8°C. Bortezomib (Velcade; Millennium Predictive Medicine, Inc., Cambridge, USA) was dissolved in saline at 10 mM for storage at −20°C. Rabbit antibody against β-catenin, mouse antibody against cyclin D1, mouse antibody against c-Myc, mouse antibody against β-actin, and fluorescein isothiocyanate (FITC)-conjugated secondary antibody were all purchased from Santa Cruz Biotechnology (Santa Cruz, USA).
Cell lines and cultures
Human MM cell lines RPMI8226 and U266 were provided by Hematology Institute of Zhejiang University, Zhejiang, China, and normal hematopoietic cells were obtained from peripheral blood of healthy volunteers who signed informed consent. Mononuclear cells were harvested using density gradient centrifugation at 3500 rpm for 20 min with Ficoll-Hypaque Solution (Sigma-Aldrich). All cells were cultivated in RPMI-1640 (Gibco, Waltham, USA) supplemented with 10% fetal bovine serum (FBS; Invitrogen, Waltham, USA) and penicillin-streptomycin solution (100 U/mL penicillin and 0.1 mg/mL streptomycin; Siji Qing Biotech, Hangzhou, China), and grown at 37°C in a humidified incubator with 5% carbon dioxide atmosphere.
Cell viability assay
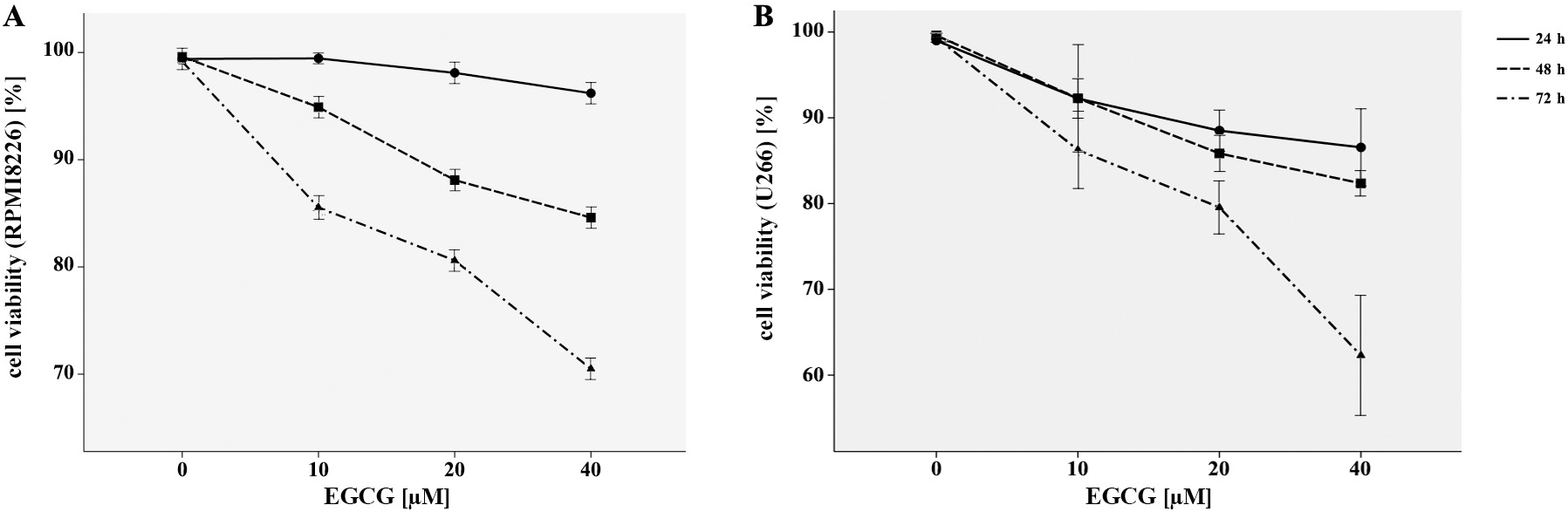
The RPMI8226 and U266 cells were grown with 1 × 104 cells per well in 96-well plates and incubated with increasing concentrations of EGCG as 10 μM, 20 μM and 40 μM for 24 h, 48 h and 72 h, respectively. Then, 20 μL of 5 mg/mL 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl-tetrazolium bromide (MTT; Sangon Biotech, Shanghai, China) was added for another 4 h. The MTT formazan crystals were then dissolved with Dulbecco’s modified Eagle’s medium (DMEM) and their optical density (OD) was examined at 570 nm. All the experiments were performed in triplicate independently.
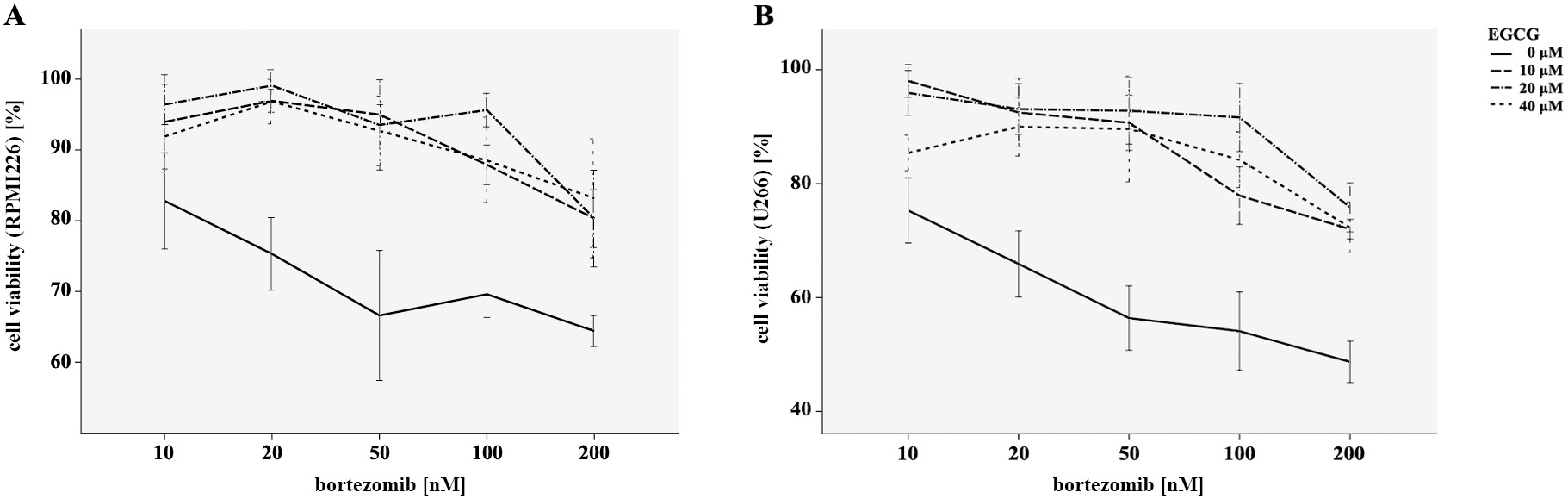
To detect the effect of EGCG combined with bortezomib on cell growth, U266 and RPMI8226 cells were incubated with EGCG (10 μM, 20 μM and 40 μM) alone and in combination with bortezomib (10 nM, 20 nM, 50 nM, 100 nM, and 200 nM) for 24 h. The MTT assay was used to determine the cell viability as described above.
Cell cycle analysis
and apoptosis detection
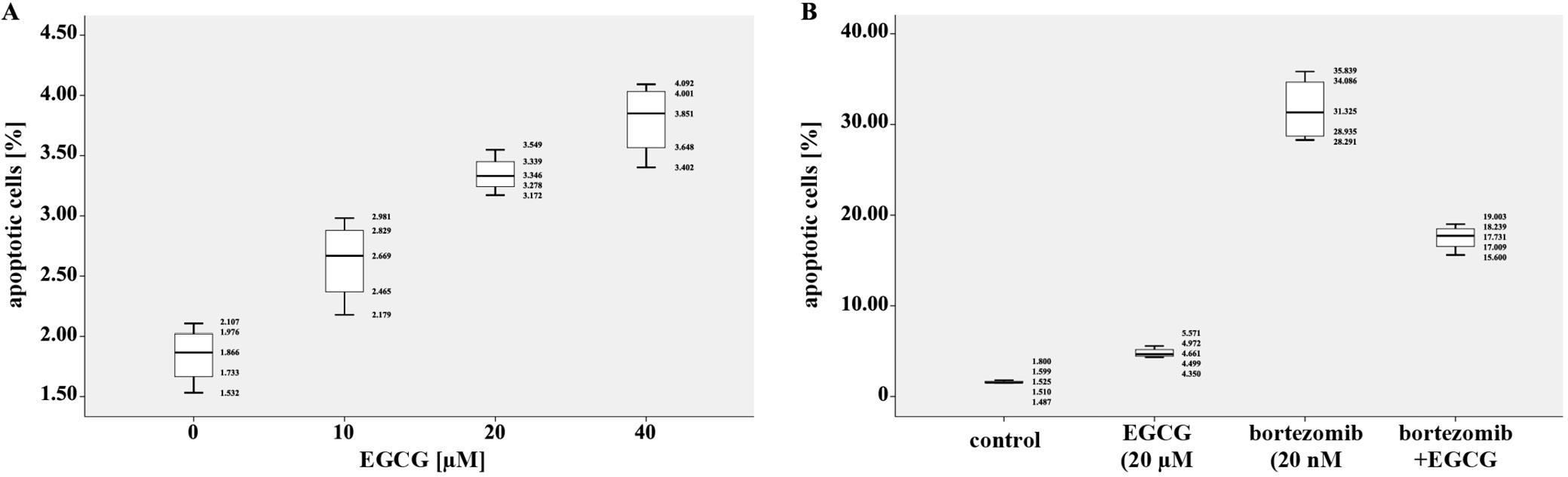
The RPMI8226 cells were incubated with 5 × 105 cells per well in 6-well plates with EGCG (20 μM) for 24 h at a concentration of 0 μM, 10 μM, 20 μM, and 40 μM. The RPMI8226 cells were incubated with the combination of EGCG (20 μM) and bortezomib (20 nM) for 24 h. Then, the cells were collected and fixed using 70% cold ethanol overnight, and stained with 50 μg/mL propidium iodide (PI; Sigma-Aldrich) for 30 min at 4°C. The FACScan flow cytometry (Becton Dickinson Biosciences, San Jose, USA) was used to analyze the cell cycle distribution. Double staining with Anexin-V fluorescein (AV; Sigma-Aldrich) and PI were used to identify the cell apoptosis. After incubation in the dark at room temperature for 15 min, flow cytometry method (FCM) could quantitatively detect the apoptotic cells.
Western blotting assay
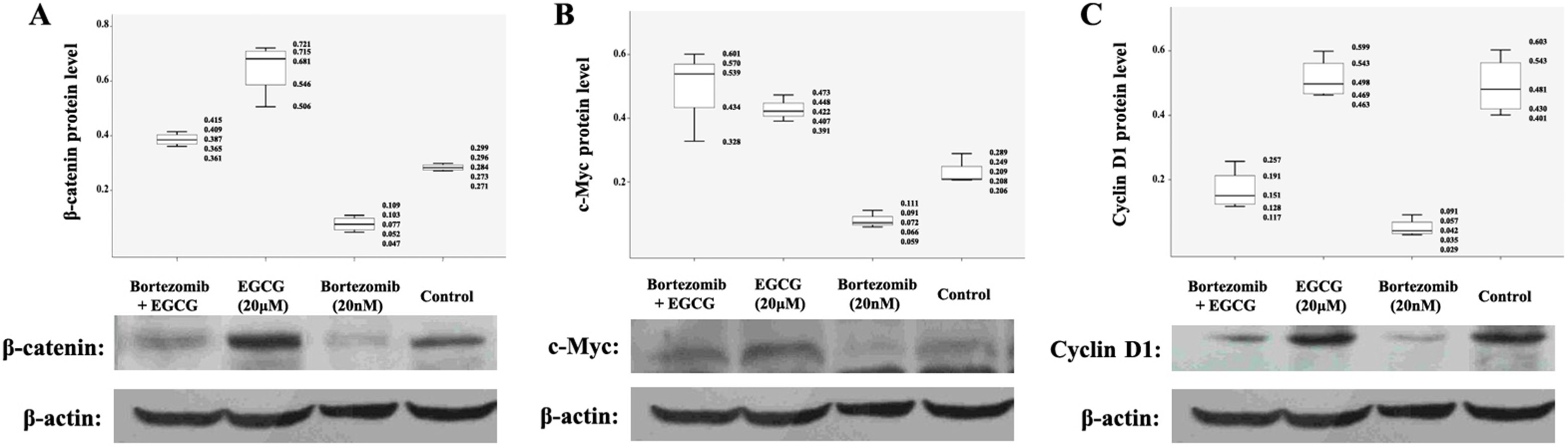
The RPMI8226 cells were treated with EGCG (0 μM, 10 μM, 20 μM, and 40 μM) alone and with the combination of EGCG (20 μM) and bortezomib (20 nM) for 24 h. These cells were then collected and dissolved in protein extraction reagent (Pierce Chemical, Rockford, USA). The total cellular protein was extracted, separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene difluoride (PVDF) membranes (Bio-Rad, Hercules, USA). Tris-buffered-saline (TBS) with 5% skim milk and 0.1% Tween-20 was supplied to block the nonspecific antibodies binding on the membranes for 2–3 h. After that, primary antibodies were added and allowed to culture overnight with the cells at 4°C. The membranes were washed 3 times with TBS mentioned above and secondary antibodies were used at room temperature for 1 h. The electrochemiluminescence (ECL) kit (Pierce Chemical) was applied to detect the specific immunoreactive bands, according to the manufacturer’s protocols.
Statistical analyses
All analyses were conducted using IBM SPSS v. 20.0 (IBM Corp., Armonk, USA) and all data were demonstrated as mean ± standard deviation (SD). The difference of cell viability was analyzed with two-way repeated-measure analysis of variance (ANOVA) with 2 factors – time and drug concentration. The effects of 2 different drugs on cell viability was analyzed with two-way ANOVA. The discrepancies of the percentage of apoptotic cells and the protein levels between groups were analyzed with one-way ANOVA. The pairwise comparisons between each 2 groups were based on the post hoc analysis of least significant difference (LSD). Values of p < 0.05 were considered statistically significant.
Results
EGCG inhibits MM cells proliferation
The MTT assay was applied to explore the effects of EGCG on the viability of myeloma cell lines as RPMI8226 and U266. As demonstrated in Figure 1A,B, EGCG played a significantly inhibitory role on proliferation of both RPMI8226 and U266 cells in a time- and dose-dependent manner, when compared with the control ones. Our results indicated that EGCG had the antiproliferative effect on MM cells without cytotoxic effects on normal hematopoietic cells.
EGCG neutralizes the growth inhibiting effect of bortezomib on MM cells
As was shown in our data, the inhibitory influence of bortezomib on the myeloma cells proliferation was dose-dependent – it increased with concentration. To further investigate the effect of EGCG combined with bortezomib, RPMI8226 and U266 cells were incubated with EGCG at a concentration of 10 μM, 20 μM and 40 μM and bortezomib (10–200 nM) for 24 h in vitro. Figure 2 shows that the myeloma cell viability was significantly increased under the combination of 2 drugs when measured with MTT assay, as compared with the group treated with bortezomib alone. These results demonstrated that EGCG could minimize the antitumor efficacy of bortezomib on myeloma cells.
EGCG neutralizes the apoptosis of MM cells induced by bortezomib
The percentages of apoptotic cells were analyzed after RPMI8226 cells were treated with EGCG, at a concentration of 10 μM, 20 μM and 40 μM alone and with the combination of 2 drugs (20 μM of EGCG and 20 μM of bortezomib) for 24 h. As shown in Figure 3A, cell apoptosis increased significantly along with the rise in concentration of EGCG, as compared with the controls. Additionally, the combination of EGCG and bortezomib obviously reduced cell apoptosis when compared with bortezomib alone, which demonstrated that EGCG reversed the cell apoptosis induced by bortezomib.
Wnt/β-catenin signaling pathway is activated when EGCG combined with bortezomib
To further explore the mechanism of blocking the antiproliferative and pro-apoptotic effect of bortezomib on MM cells by ECCG, the proteins related to Wnt/β-catenin signaling pathway were investigated using western blot assay. As mentioned in Figure 4, β-catenin was largely unchanged when RPMI8226 cells were exposed to EGCG (10 μM, 20 μM and 40 μM) alone. Furthermore, β-catenin was downregulated when exposed to bortezomib alone, which could be highly expressed when EGCG and bortezomib were added together. Similar results were also observed in the protein levels of downstream target genes like c-Myc and cyclin D1, which are responsible for promoting cell proliferation and inhibiting cell apoptosis. The upregulated expressions of c-Myc and cyclin D1 were significant when RPMI8226 cells were exposed to the combination of 2 (EGCG and bortezomib), compared with the exposure to bortezomib alone. Our results indicate that EGCG has an antagonistic effect on the antitumor effect of bortezomib through the cumulation of β-catenin and activation of downstream proteins.
Discussion
The promising effects produced by EGCG against the development of various cancers have received a great deal of attention for decades. In hematological malignancy like MM, EGCG also exhibits remarkable antitumor effect properties and the underlying mechanism has been investigated. Masood et al. identified the efficacy of EGCG on growth arrest and apoptosis promotion in MM cells in vitro and in vivo, and they found that the 67-kDa laminin receptor1 (LR1) played a crucial part in mediating the biological activity of EGCG.17 Furthermore, Zhou et al. confirmed the enhancer of zeste homolog2 (EZH2), which was a member of Pc-G family, to be inactivated in the antitumor effect of EGCG on MM cells.18 The mitochondrial apoptosis pathway was also involved. MST-312 is an another modified derivative of EGCG which was named as telomerase inhibitor and induces apoptosis of myeloma cells via modulating apoptotic-related genes.
As EGCG can be administered orally, researchers had high hopes for augmenting the antitumor outcome of chemotherapy in clinical use when EGCG was added. Unexpectedly, Encouse et al. discovered that EGCG effectively blocked myeloma cells death induced with bortezomib in vitro and in vivo.20 This antagonistic function was performed via a direct reaction with the boronic acid group of bortezomib, which resulted in the discontinuation of endoplasmic reticulum stress induction and inactivation of caspase-7. Qing et al. demonstrated that when the concentration of EGCG increased, the combination of EGCG and bortezomib played a synergistic role in inhibiting cell proliferation and inducing apoptosis, and that the NF-κB pathway was involved.21 These results implied that various concentrations of EGCG had completely opposite effects on the anticancer role of bortezomib, and the underlying mechanism was still not clear and required further investigation.
In our study, the proliferation inhibition and apoptosis-inducing effect of EGCG on MM cells was demonstrated. The EGCG played an antagonistic role in growth inhibition induced by bortezomib. Moreover, we found that the number of apoptotic cells was decreased significantly in combination with bortezomib when compared with the effect of bortezomib alone. Especially with the concentrations of bortezomib of 20 nM and EGCG also of 20 μM, the combination of these 2 drugs maximized the impact of antagonism. Based on this result, we chose these concentrations to study the underlying mechanism further. As it is commonly known, the canonical Wnt signaling pathway, which is one of the most essential regulators for embryogenesis and homeostasis, is conserved evolutionarily in mammals.22, 23 Abnormal inactivation of Wnt/β-catenin signaling pathway is closely correlated with tumorigenesis.24 When β-catenin is accumulated in the cell nucleus, its binding to the TCF/LEFs makes the downstream target genes upregulate.25 Our study demonstrated that significant accumulation of β-catenin was found when EGCG and bortezomib were combined as compared to the effect of bortezomib alone; such accumulation subsequently activated the downstream target genes as c-Myc and cyclin-D1, and finally induced the growth, proliferation and differentiation of tumor cells. These results indicate that EGCG exerts an antagonistic effect on the antitumor effect of bortezomib in MM cells, probably by activating Wnt/β-catenin signaling pathway.
Limitations
Our study has some limitations. Although the inhibitory role of EGCG on the antiproliferative and pro-apoptotic effect of bortezomib in myeloma cells was verified, we also found that the combination of bortezomib (20 nM) and EGCG (20 μM) maximized the antagonism, which was not in a dose-dependent relationship, and the mechanism was still unclear. Moreover, whether other crucial signaling pathways also took part in this antagonistic function requires further investigations.
Conclusions
Our results illustrate that EGCG had the anti-proliferative and pro-apoptotic effects on myeloma cells. Furthermore, it neutralized the antiproliferative and pro-apoptotic effects of bortezomib on myeloma cells. The underlying mechanism was probably involved in the activation of Wnt/β-catenin signaling pathway. In summary, we further identified the antagonistic effect of EGCG in combination with bortezomib, and provided another insight into the underlying mechanism. As a widely consumed drink throughout the world, green tea, in which EGCG is the most abundant and active ingredient, should be avoided by MM patients regularly receiving the chemotherapy including bortezomib.