Abstract
Background. Spinal cord injury (SCI), a serious damage of the central nervous system, has become an extremely important issue that threatens the health of people worldwide. The proliferation of astrocytes plays an important role in the repair of SCI, which has typical two-sided effects. The HS1-associated protein X-1 (HAX-1), plays an important role in the physiological and pathological processes of cell apoptosis, proliferation, migration, and invasion. However, the specific role and mechanism of HAX-1 in human astrocyte HA1800 are still unclear.
Objectives. To explore the effect of HAX-1 on the proliferation and apoptosis of HA1800 cells and preliminarily explore its possible underlying mechanism.
Materials and methods. The HA1800 cell lines with high- and low-expression levels of HAX-1 were established using lentiviral vector pcDNA3.1. Quantitative reverse transcription polymerase chain reaction (qRT-PCR) and western blot were employed to determine the expression of HAX-1 after transfection. Cell viability and proliferation ability were estimated using MTT and 5-Ethynyl-2’deoxyuridine (EdU) assay. The effects of HAX-1 on the HA1800 cell cycle and apoptosis were determined using flow cytometry. The BCL-2/BAX ratio and the expression of Ki67 and c-Myc in the transfected cells were detected using qRT-PCR. The Gene Expression Profiling Interactive Analysis (GEPIA) database was used to determine the relationships of HAX-1, BAX and BCL-2.
Results. The HA1800 cell lines with high and low expression of HAX-1 were obtained. The MTT, EdU and flow cytometry showed that elevated HAX-1 could inhibit the proliferation, reduce the viability and promote the apoptosis of HA1800 cells. The qRT-PCR showed that the mRNA levels of Ki67, c-Myc and the BCL-2/BAX ratio were significantly decreased in the HAX-1 high-expression group, but increased in the HAX-1 low-expression group. The results from the GEPIA database showed that HAX-1 was positively correlated with BAX and BCL-2 in the spinal cord.
Conclusions. The HAX-1 may influence the biological behavior of human HA1800 cells due to the progression of cell cycle and apoptosis associated with BCL-2/BAX.
Key words: HAX-1, HA1800, proliferation, apoptosis
Background
Damage to the central nervous system, especially spinal cord injury (SCI), is a common and serious disease resulting in physical, psychological and social disorders.1 In recent years, the morbidity and mortality of SCI have been increasing.2 Among many possible causes of SCI, traffic accidents are not negligible. In 2015, the data from many countries have shown that approx. 12–60 cases of traffic accidents per million of population occur annually.3 Spinal cord injury is classified as primary (acute phase) or secondary (recovery phase) injury, depending on the timing and mechanism of the nerve injury. The primary damage is mainly caused by a direct damage of the vertebrae, resulting in contusion or oppression of the spinal cord, and usually, the primary damage appears only at the injured site. In SCI, the neurons are directly damaged, and astrocyte and endothelial cell injuries are rare. The secondary injury may be the key factor in the damage that is difficult to repair and it results in the decline or loss of motor and sensory functions. The secondary damage occurs tens of seconds after the primary damage and lasts for several weeks. The various reactions caused by the damage appear not only at the injured and surrounding sites but also across the whole body. Some specific manifestations are ischemic dysfunction, changes in the electrolytes, accumulation of neurotransmitters, protein/lipid oxidation, DNA damage, inflammation, edema, oxidative stress necrosis, apoptosis of neurons, and glial hyperplasia.4, 5 Neuronal apoptosis is the most widely studied manifestation.6, 7 Under the stimulation of primary injury, the influence of secondary cytokines and chemical factors, reactive hyperplasia of astrocytes occurs at the injured and surrounding areas, and this process forms glial scar.8 The occurrence of this scar in the acute stage may have more positive effects, such as the limitation of the scope of injury, protection of the ischemic penumbra, maintenance of local metabolism, and immune regulation.9 However, in the subsequent recovery period, glial scar will hinder nerve repair. First, the scar in the damaged area directly acts as a physical barrier to the extension of newborn neuronal axons. Second, various cytokines secreted in the damaged area inhibit the regeneration of axons to varying degrees. Hence, astrocyte proliferation is typically double-sided for injury repair.10, 11 Effective target regulation of astrocyte proliferation at different time periods of injury may be beneficial for the regeneration of neurons and extension of axons, providing a new method for the treatment of SCI.
With the use of a system named yeast two-hybrid, Suzuki et al. discovered that HS1-associated protein X-1 (HAX-1) has an intracellular antiapoptotic effect and is homologous to the antiapoptotic protein – BCL2.12, 13 The HAX-1 mainly interacts with the hematopoietic cell-specific substrate protein 1 (HS1) and functions as a protein with molecular weight of 35 kD. The HAX-1 is the substrate of serine/tyrosine kinase located on the human chromosome 1q21.3, encoding 279 amino acid fragments.13 This protein consists of a transmembrane domain, an acidic domain, and a proline, glutamic acid, serine and threonine (PEST) sequence.14 The programmed pseudogenes are found on the human X chromosome, and similar pseudogenes are found in the rat and mouse genomes. The HAX-1 analogs have been found in many other species, including zebrafish (mackerel), rather than in more primitive organisms, such as nematodes and new rod-shaped nematodes. Also, the comparison and analysis of the mRNA expression data of human, rat and mouse HAX-115 have shown that HAX-1 is widely expressed in various tissues and organs of mammals (highly expressed in the skeletal muscle, cardiac muscle, colon, and other tissues), but lowly expressed in the nervous system and kidney.16 As an antiapoptotic protein, HAX-1 has been confirmed to participate in the regulation of apoptosis through several signal pathways.17, 18, 19 In the mitochondrial-related caspase apoptosis pathway, HAX-1 could inhibit the activation of caspase-3 and caspase-9 and then inhibit apoptosis. The HAX-1 also plays an antiapoptotic role by regulating the calcium ion homeostasis and improving calcium overload. In the serine protease Omi/HtrA2 signal transduction pathway in the mitochondria, HAX-1 is the substrate protein of Omi/HtrA2. The Omi/HtrA2 plays an enzymatic cleavage role in the apoptosis pathway. This effect can degrade HAX-1, thereby producing an antiapoptotic effect. In addition, HAX-1 interacts with the 2-pore channel of the endolysosome, which reduces the permeability of the lysosomal membrane and the release of enzymes, exerting an antiapoptotic effect. The HAX-1 also exerts antiapoptotic effects through the antioxidative stress response. The HAX-1 can affect the Ak1t/MDM2/p53 axis and the expression of p21, Bax and p53 proteins to participate in cell apoptosis. The HAX-1 can interact with the heat shock protein 90 (Hsp90) to affect the IRE-1 signal transduction, in order to regulate cell apoptosis.20 However, studies have shown that HAX-1 may act not only as an antiapoptotic (human v1 isoform) but also as a proapoptotic (human v4 isoform) regulator through homodimerization or heterodimerization in cardiac cells.21, 22 The HAX1 is involved in many important physiological and pathological processes, such as the regulation of cell apoptosis, cell proliferation, motility, endocytosis, and mRNA transport.23, 24 Recent reports have shown that HAX-1 levels are elevated in various tumors, such as those in the esophagus, colorectal, nasopharynx, throat, prostate, ovary, breast, skin, and lymphoma.24, 25, 26, 27, 28 In addition, by collecting clinical case studies, Deng et al. showed that HAX-1 was also expressed in gliomas and might be positively correlated with tumor malignancy. The role of HAX-1 in promoting tumor proliferation might be achieved through estrogen-related apoptosis pathways. However, whether a direct effect exists remains to be confirmed.29 In the neurons, HAX-1 can interact through Racl and contraction to affect the formation of neuronal cells and regulate the apoptosis and migration ability of tumor cells. Upregulated HAX-1 has been detected during brain damage caused by trauma or epilepsy.30, 31 The HAX-1 protein level was increased significantly in neurons after traumatic brain injury (TBI) but it was not expressed within glial cell population and microglia in the brain cortex. However, whether different subtypes of HAX-1 play distinct roles in the brain, especially in the astrocytes after TBI, has not been studied yet.31 All the aforementioned studies showed that HAX-1 might be related to the regulation of astrocyte proliferation.
Apoptosis plays a key role in secondary SCI.32 Lu et al. performed experiments on the effect of HAX-1 on neuronal apoptosis after SCI in rats. The results showed that HAX-1 expression was mainly upregulated in the local neurons after SCI but almost not expressed in the normal astrocytes. In addition, the upregulated HAX-1 was negatively correlated with neuronal apoptosis. The downregulation of HAX-1 in the reactive astrocytes in the experiments in vitro changed the cell viability of the primary cultured wild-type rat spinal cord astrocytes. Thus, HAX-1 might be related to the reduction in astrocyte proliferation.23 These results suggested that HAX-1, as a multifunctional protein, may be involved in the proliferation of astrocytes after SCI. However, the experimental results failed to further verify the specific possible mechanism of activity, and whether HAX-1 had a certain regulatory effect on cell migration was not observed.23 In conclusion, we speculate that HAX-1 may play a regulatory role in the growth, proliferation and migration of astrocytes. In this study, we constructed in vitro human HA1800 cell lines with high and low HAX-1 expression to verify the function of HAX-1 on the proliferation and apoptosis of human astrocytes. We also preliminarily explored the possible signal pathways to provide a certain experimental basis for clinically improving and regulating the formation of glial scars caused by injury and promoting the repair of nerve injury.
Objectives
We examined the specific effects of HAX-1 expression on the proliferation, viability and apoptosis of human HA1800 cells by upregulating and downregulating its expression. We preliminarily explored the effects of HAX-1 on the cell proliferation and apoptosis-related factors to further clarify its possible underlying mechanism.
Materials and methods
Establishment of HA1800 cell lines with stable high and low HAX-1 expression
Lentiviral transfection was used to construct stable transfected HA1800 cell lines. The coding sequences of HAX-1 and HAX-1 siRNAs were synthesized by RiboBio Biotech Company (Guangzhou, China). The interference sequences were as follows: HAX-1 siRNA1#, 5ʹGTACGAGATTTCAATAGCA3ʹ; HAX-1 siRNA2#, 5ʹGGATACGTTTCCACGATAA3ʹ; HAX-1 siRNA3#, 5ʹGGATACGTTTCCACGATAA3ʹ; and NC-siRNA, 5ʹUUCUCCGAACGUUGACACACGUdTdT3ʹ. The synthesized HAX-1 and HAX-1 siRNAs were integrated into the lentiviral vector pcDNA3.1 (+) (Invitrogen, Carlsbad, USA) vector, using BamHI and EcoRI (Thermo Fisher Scientific, Waltham, USA). The plasmids were confirmed by sequencing and then transfected into DH5a for amplification, screening of positive clones, identification, and sequencing, in order to obtain the recombinant retroviral vector. The plasmids were extracted according to the instructions provided by the manufacturer of Endo-free Plasmid Mini Kit I (Omega, Stanford, USA). Finally, plasmids with high HAX-1 expression and low HAX-1 expression (1#, 2# and 3#) were obtained.
Human HA1800 cells were purchased from the Shanghai GeneChem, Co., Ltd. (Shanghai, China). The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Thermo-Life, Waltham, USA) with fetal bovine serum (FBS, 10%; Thermo Fisher Scientific), penicillin (100 μg/mL; Beyotime, Shanghai, China) and streptomycin (Beyotime). The cultivation environment was 37°C with 5% CO2. Approximately 1 × 105 cells were plated per well in a six-well plate. The following day, when the cell density reached 80% of the well, the plasmids were transfected with Lipofectamine™ 2000 (Invitrogen), according to the manufacturer’s instructions. The empty lentiviral vector was used as the control group for the high-HAX-1 expression group, and NC-RNAi was used as the negative control (NC) group for the low-expression groups.
Examination of HAX-1 mRNA
and protein expression
Quantitative reverse transcription polymerase chain reaction (qRT-PCR) was performed to examine the HAX-1 mRNA expression. After the HA1800 cells were transfected with the plasmids (HAX-1, pcDNA3.1; NC and HAX-1 siRNAs (1#, 2#, and 3#)), the total RNA of each group of cells was extracted (UNIQ-10 Spin Column RNA Purification kit; Sangon Biotech Co., Ltd., Shanghai, China). The first-strand cDNA was synthesized (RevertAid First Strand cDNA Synthesis kit (Ferments; Thermo Fisher Scientific)) using the prepared total RNA (1 µg) and then analyzed in the Step One Plus™ real-time PCR system (Thermo Fisher Scientific), using the AceQ qPCR SYBR Green Master Mix (Vazyme, Piscataway, USA). The GAPDH was chosen as the control reference gene. The primers were as follows: HAX-1 (183 bp) forward, 5ʹ-GGGGTCTTGGAGAGTGATG-3ʹ, and reverse, 5ʹ-CTGGGGCTGTAGAACCG-3ʹ; GAPDH (131 bp) forward, 5ʹ-GAAGGTCGGAGTCAACGGAT-3ʹ, and reverse, 5ʹ-TCCCGTTCTCAGCCATGTAGTT-3ʹ. The cycling parameters were as follows: 95°C for 5 min; 40 cycles of 95°C for 10 s and 60°C for 30 s. The 2–ΔΔCq method was used to calculate the expression levels.
Western blot was utilized to check the HAX-1 protein expression. The total proteins of the cultured groups of cells were extracted using the Tissue or Cell Total Protein Extraction kit (Sangon Biotech Co., Ltd.) and measured using the Enhanced BCA Protein Assay Kit (Beyotime, Haimen, China). Then, 10% sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) was used to load and separate the total protein (30 µg). After electrophoresis, the proteins were transferred to polyvinylidene fluoride membranes, and the membranes were blocked with skim milk (5%) for 1 h at room temperature. Then, the primary antibodies were incubated overnight at 4°C and washed thrice with 1× Tris-buffered saline and Tween-20 (TBST) solution (0.1% Tween; Sangon Biotech Co., Ltd.) (5 min each instance). The secondary antibody was incubated with the membranes for 4 h at room temperature. The following antibodies were used: rabbit anti-β-actin (dilution, 1:1000; cat. No. ab8227; Abcam, Cambridge, UK); rabbit anti-HAX-1 (1:500; cat. No. ab137613; Abcam); horseradish peroxidase-conjugated goat anti-rabbit as the secondary antibody (1:3000; cat. No. ab205718; Abcam). Then, the membranes were washed thrice with TBST (5 min each instance) and detected using enhanced chemiluminescence reagent (cat. No. E411; Vazyme, Nanjing, China). Finally, the membranes were scanned using Chemidoc XRS system (Bio-Rad Laboratories, Inc., Hercules, USA), and the expression levels of the proteins were calculated using Image Lab software (Bio-Rad Laboratories, Inc.).
The best interfering group, referred to as HAX-1 siRNA, was chosen. Thus, the groups utilized for subsequent experiments contained pcDNA3.1-HA1800, HAX-1-HA1800, NC-HA1800, and HAX-1 siRNA-HA1800.
MTT assay
Cells from each group were incubated in 96-well plates for 24 h and were added with MTT (0.5 mg/mL; Beyotime). After incubation at 37°C for 3 h, the cells were added with dimethyl sulfoxide (DMSO) (150 μL). After incubation for 15 min, the absorbance was measured at 490 nm with a spectrophotometer (Tecan, Männedorf, Austria).
5-Ethynyl-2ʹ-deoxyuridine (EdU) assay
Cells from each group were incubated in a 6-well plate for 24 h and treated with EdU (50 µM) reagent. Then, the cells were treated with Apollo-567 reaction mixture for 30 min and washed thrice with phosphate-buffered saline (PBS). Finally, the cells were counterstained with Hoechst (dilution, 1:1000) for 10 min at room temperature for nuclear staining. The EVOS® FL imaging system (Thermo Fisher Scientific) was used to observe and count the cells. All materials in this assay were purchased from RiboBio Biotech Company.
Cell cycle analysis
The cells were incubated, harvested, washed twice with PBS, and then fixed on ice-cold 75% ethanol for 30 min. After being washed twice successively with citrate phosphate buffer and PBS, the cells were treated with PBS containing 100 μg/mL RNase A for 30 min at 37°C and then incubated in PBS containing 100 μg/mL propidium iodide for 30 min at room temperature. Finally, flow cytometry (Becton Dickinson Biosciences, Franklin Lakes, USA) was used to ensure the cell cycle distribution of each sample (over 10,000 cells). The data were analyzed using ModFit software (Becton Dickinson Biosciences). The experiment was performed in triplicate.
Measurement of Ki67, c-Myc, BCL-2, and Bax
The qRT-PCR was utilized to analyze the expression of Ki67, c-Myc, BCL-2, and Bax in the cells from each group. The specific method is as above. The primers applied are presented in Table 1.
GEPIA analysis
The The Gene Expression Profiling Interactive Analysis (GEPIA)33 (http://gepia.cancer-pku.cn/index.html) database was used to analyze the relationships between HAX-1 and BAX, as well as between HAX-1 and BCL-2, in the spinal cord.
Statistical analyses
The IBM Statistical Package for the Social Sciences (SPSS) software v. 22.0 (IBM Corp., Armonk, USA) was applied to count the data, which are presented as mean ± standard deviation (SD) (each experiment was repeated at least thrice, independently). Two sided Student’s t-test (for parametric data set, meaning the data was suitable for parametric test) was used for double comparison and Pearson’s correlation was applied to assess correlation. One-way analysis of variance (ANOVA) with Bonferroni’s post test (for parametric data set, meaning the data suitable for parametric test) was performed for multiple comparisons. A value of p < 0.05 was considered statistically significant. Graphs were drawn in GraphPad Prism software v. 5.0 (GraphPad Software, San Diego, USA).
Results
High and low expression of HAX-1 was established in the HA1800 cells
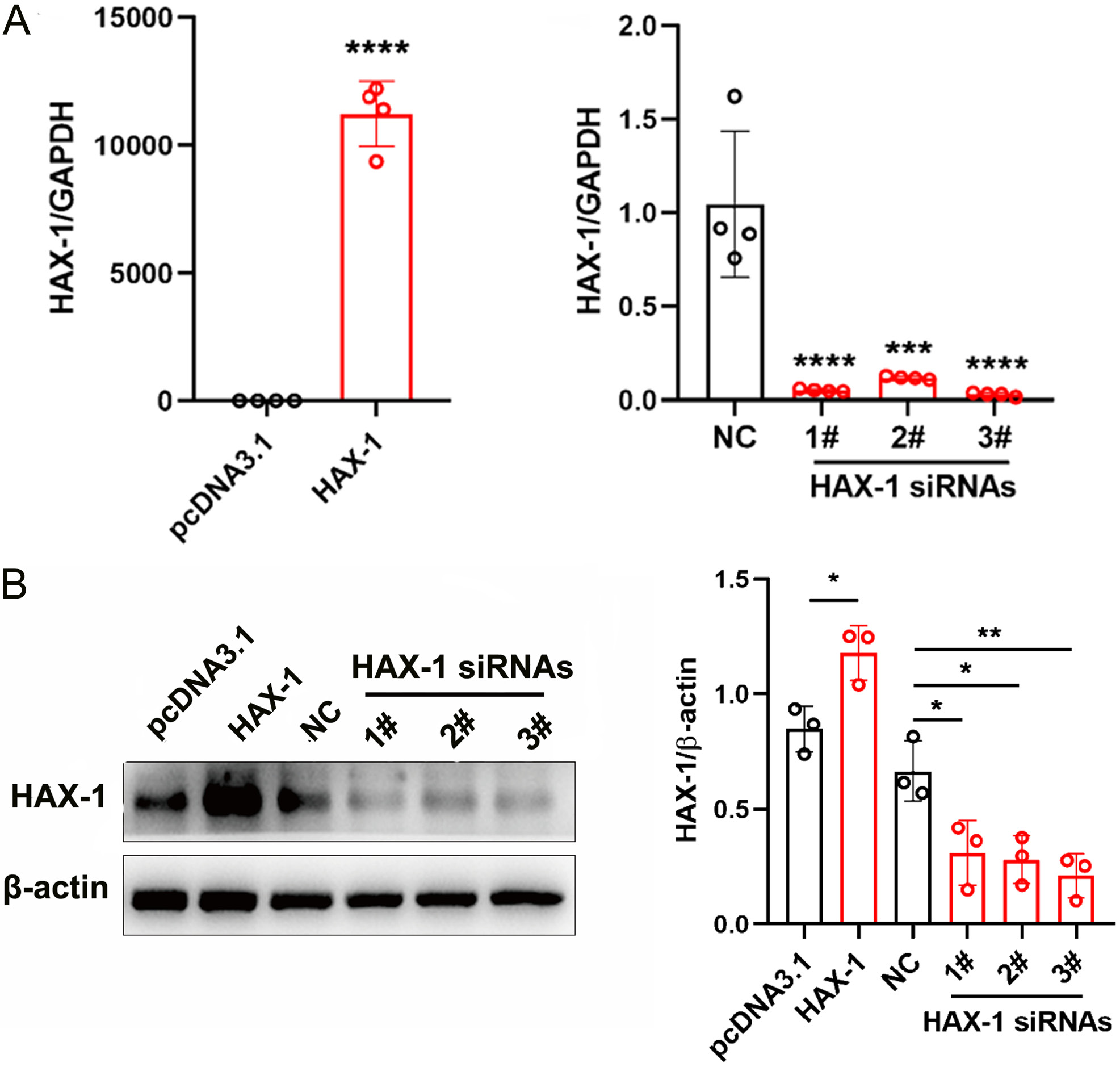
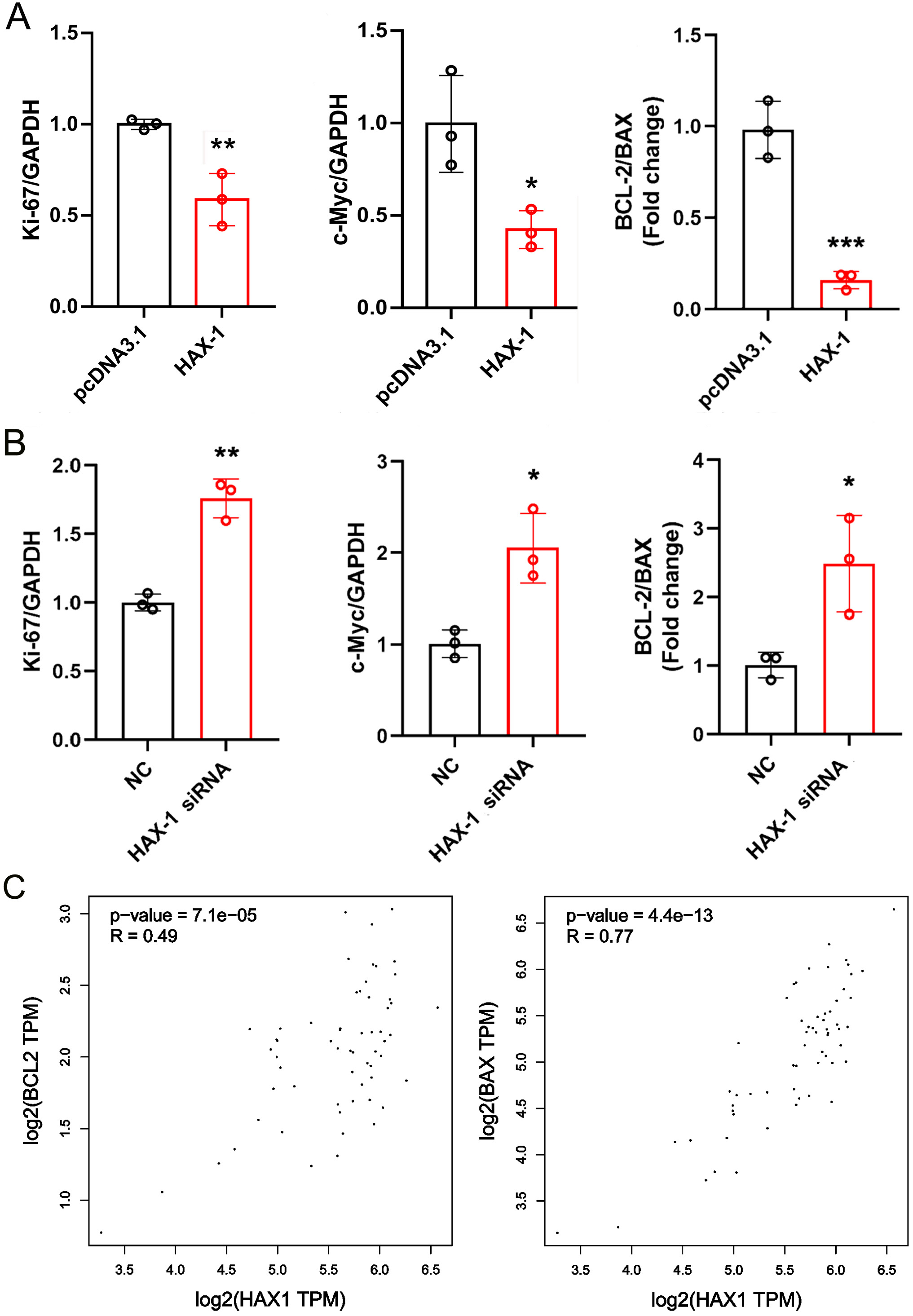
The HA1800 cells with either high or low expression of HAX-1 were established. The results from the qRT-PCR and western blot indicated that the mRNA and protein expression of HAX-1 were significantly upregulated in the high-HAX-1 expression group, which was transfected with pcDNA3.1-HAX-1 (p < 0.05; t-test; t = 17.41) (Figure 1), compared with that in the control group (pcDNA3.1 transfected with empty lentiviral vector). The HAX-1 was obviously downregulated in the low-HAX-1 expression group, which was transfected with HAX-1 siRNAs, compared with that in the NC group (p < 0.05, p < 0.05, p < 0.05; ANOVA post tests; F (qRT-PCR) = 25.38, F (western blot) = 8.741) (Figure 1). The HAX-1 siRNA3# was the best interfering group and was chosen for subsequent experiments.
High HAX-1 expression reduced the HA1800 cell viability
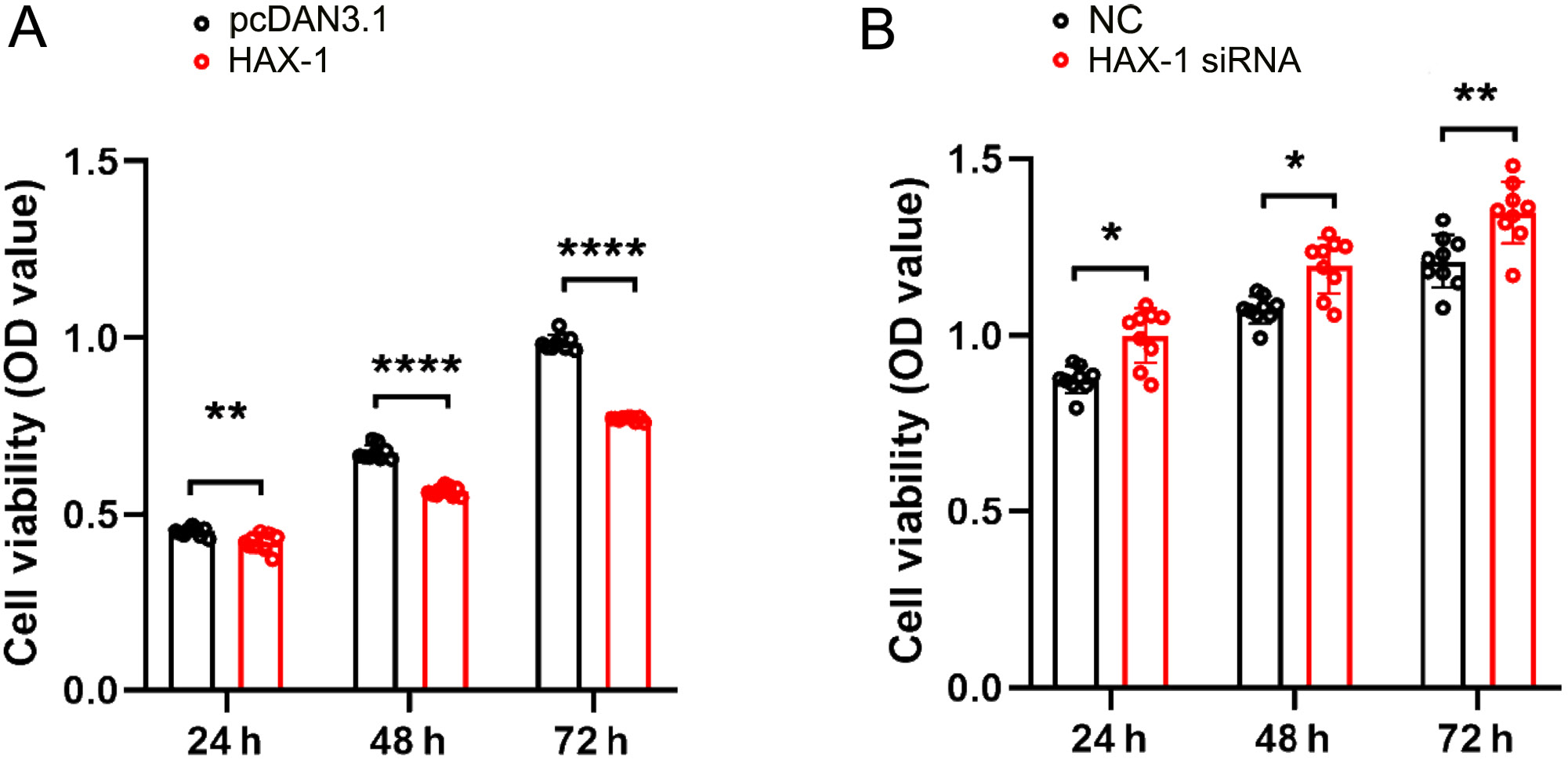
The MTT assay was performed to determine the influence of HAX-1 on the cell viability of the HA1800 cells. The results showed that the viability of the high-HAX-1 expression group (HAX-1 transfected with pcDNA3.1-HAX-1) cells was significantly reduced, compared with that of the control group (pcDNA3.1) (p < 0.05; ANOVA; F = 981.2) (Figure 2A), but was markedly enhanced in the low-HAX-1 expression group (HAX-1 siRNA transfected with HAX-1 siRNA3#) compared with the NC group (p < 0.05; ANOVA; F = 79.14) (Figure 2B). These results indicated that the elevation of HAX-1 may play a key role in reducing the HA1800 cell viability.
HAX-1 regulated the HA1800 cell proliferation
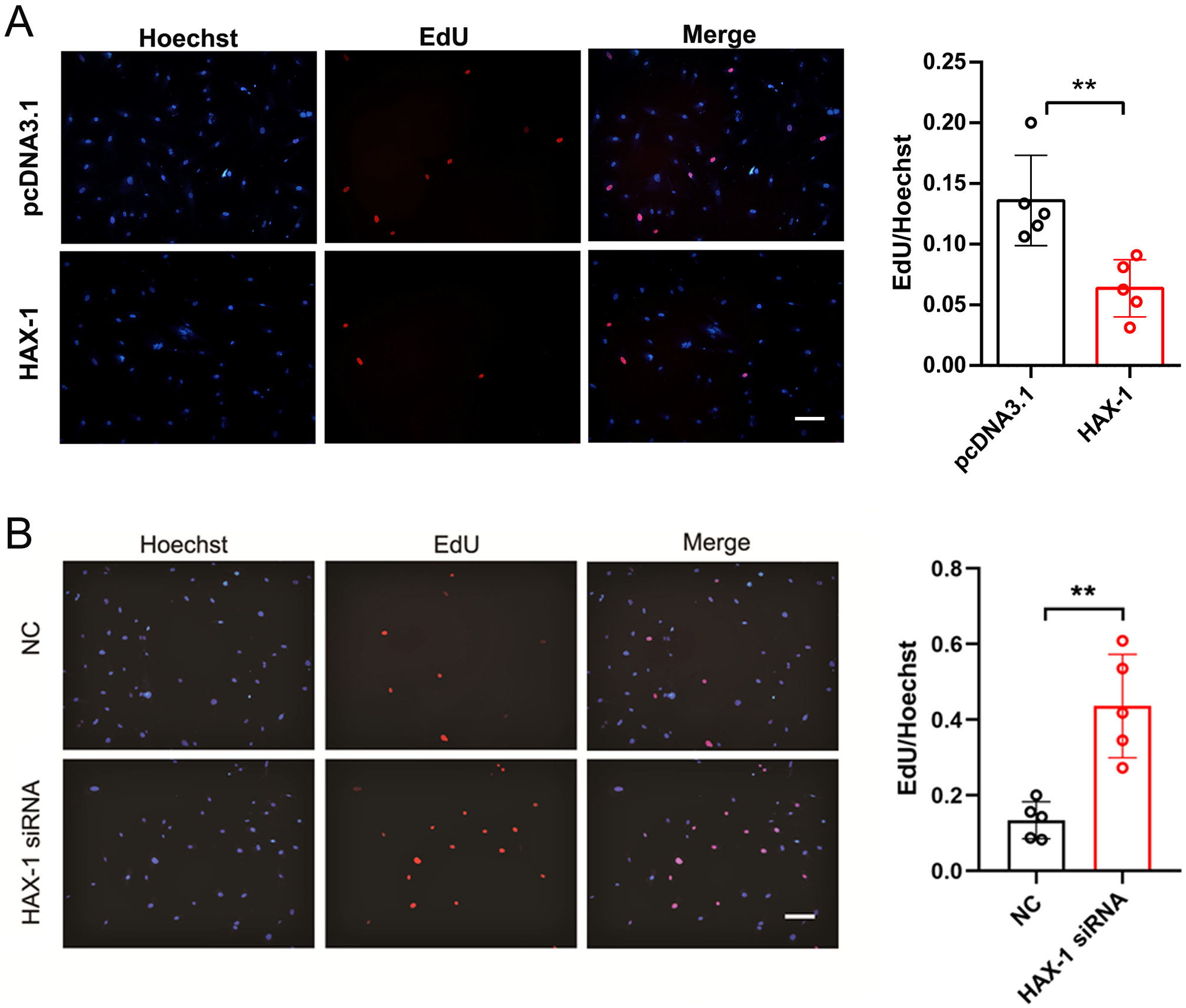
The EdU proliferation assay was applied to explore the influence of HAX-1 on the HA1800 cells. The proliferation rate of the cell lines in the HAX-1 group was decreased significantly compared with those in the pcDNA3.1 group (p < 0.01; t-test; t = 3.676) (Figure 3A), and the proliferation rate of the cell lines in the HAX-1 siRNA group was increased, compared with that in the NC group (p < 0.01, t-test; t = 4.648) (Figure 3B). These results indicated that the elevation of HAX-1 may cause slower proliferation of HA1800 cells.
High HAX-1 expression inhibited the HA1800 cell cycle progression
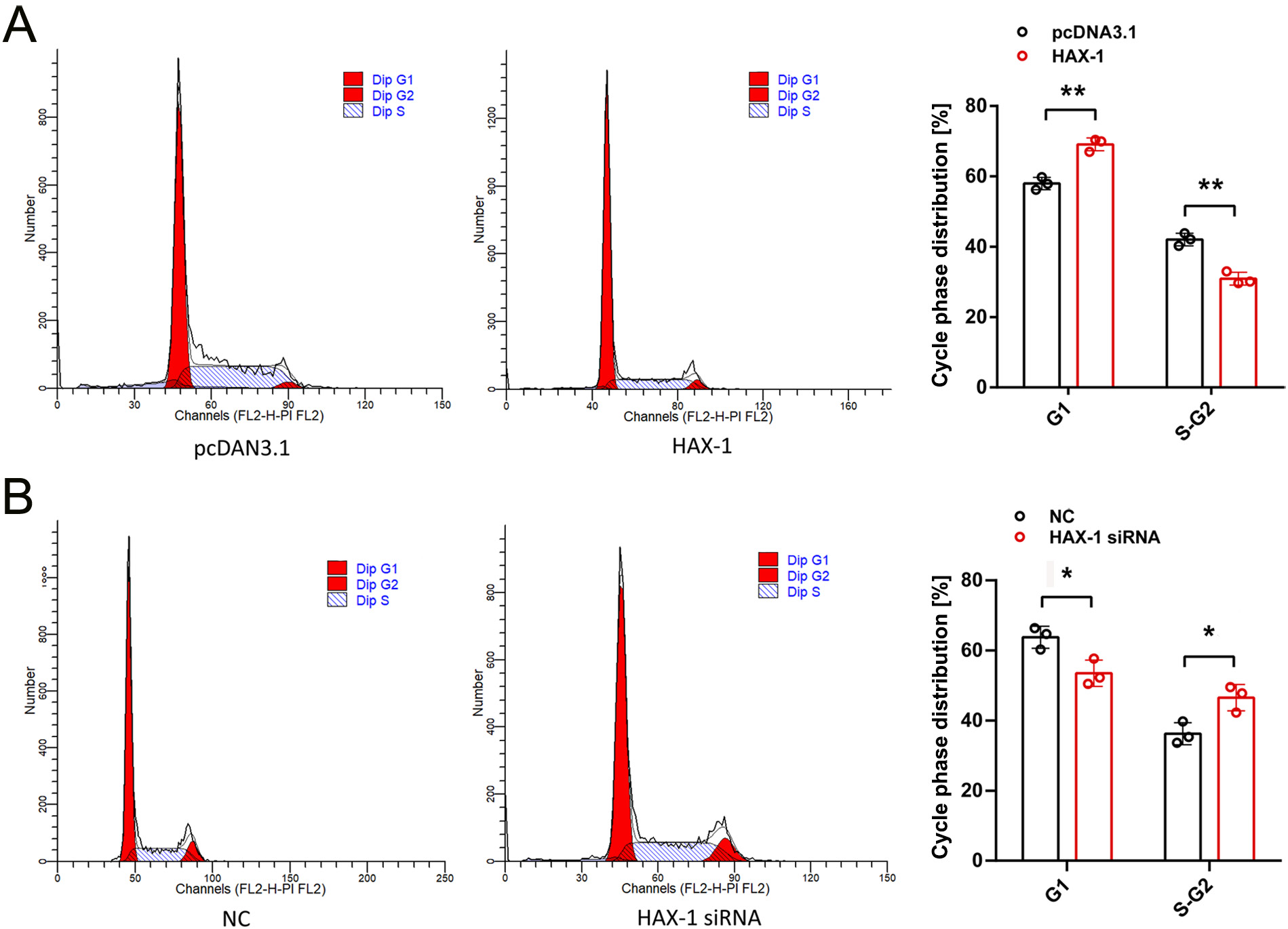
Flow cytometry (TCM) was used to evaluate the cell cycle distribution of the HA1800 cells. The results showed more HA1800 cells in the G1 phase and less HA1800 cells in the S and G2 phases in the HAX-1 group, compared with those in the pcDNA3.1 group (p < 0.01; t-test; t = 7.545) (Figure 4A). However, contrasting results were obtained from the HAX-1 siRNA group compared with the NC group (p < 0.05; t-test; t = 3.632) (Figure 4B), demonstrating that the elevation of HAX-1 may suppress HA1800 cell cycle progression.
HAX-1 participated in regulating the expression of genes related to cell proliferation and apoptosis
To study the possible mechanism of the expression of HAX-1 regulating the proliferation of HA1800 cells, the qRT-PCR analysis was used to examine the expressions of Ki67, c-Myc, BCL-2, and BAX. The results indicated that, compared with the pcDNA3.1 group, the mRNA expression of Ki67 and c-Myc, and the BCL-2/BAX ratio were all significantly lower in the HAX-1 group (p < 0.01; t-test; t = 4.889, p < 0.05; t-test; t = 3.524, p < 0.005; t-test; t = 8.759) (Figure 5A). By contrast, the expression of Ki67 mRNA and c-Myc, and the BCL-2/BAX ratio were all significantly higher in the HAX-1 siRNA group (p < 0.01; t-test; t = 8.498, p < 0.05; t-test; t = 4.296, p < 0.05; t-test; t = 3.51) (Figure 5B) than in the NC group. Thus, high HAX-1 expression may inhibit the proliferation of human HA1800 cells and promote the apoptosis.
The results from the GEPIA online database displayed that BCL-2 and BAX were positively correlated with the HAX-1 in the spinal cord (both p < 0.001, Pearson’s correlation analysis) (Figure 5C). Thus, BCL-2/BAX might constitute a possible mechanism for the effect of HAX-1 in HA1800.
Discussion
As one of the diseases with high probability to cause human disability, SCI is a global medical problem. However, no reliable treatment for this disease is currently available.1, 4 The regeneration of the damaged axons after injury is very limited, resulting in a subsequent permanent dysfunction. Patients are disabled due to SCI caused by various reasons, leading to serious personal and social problems and heavy economic burden to patients and their families.1, 34 After SCI, the astrocytes become hypertrophic and proliferate, forming a dense network of astrocyte processes at the lesion site. This process builds a physical and biochemical barrier for axon regeneration.7 Therefore, the inhibition of the activation and scar formation of the astrocytes will create a favorable environment for axon regeneration to promote recovery after SCI.10
Recent studies have shown that HAX-1 exists in numerous tissues and participates in or mediates the formation of cytoskeleton components by interacting with some proteins. This molecule also regulates the proliferation, adhesion and metastasis of many tumor cells.25, 35, 36 The HAX-1 has been preferentially located in the mitochondria in the cell, and in a small part, on the endoplasmic reticulum and nuclear membrane, which was primarily inferred to be related to cell apoptosis.37 However, some studies have demonstrated that the effect of HAX-1 on apoptosis was controversial. Human and rat HAX-1 genes have been severely spliced, resulting in at least 7 different variants, mainly due to the differences in the NH2 ends.38 Different splice variants of HAX-1 have contrasting roles in regulating the apoptosis. The prototypical variant 1 has an antiapoptotic function, whereas HAX-1 variant 2 promotes cell death and abrogates the protective effect of variant 1.21 The HAX-1 subtypes play different roles in apoptosis due to heterodimerization and homodimerization.22 Based on this, HAX-1 has also been found to have an impact on cellular behaviors, such as the proliferation, migration and invasion of tumor cells (in particular, colorectal and nasopharyngeal cancer cell lines).39, 40, 41 Qian et al. studied the expression and role of HAX-1 in cervical cancer and found that in the tumor cells, HAX-1 had an inhibitory effect on apoptosis and could promote the cell proliferation by reducing the formation of oxygen free radicals as well as protecting mitochondria through the P53 pathway. However, the effect of HAX-1 on cell migration is unknown.42 A study on the effect of HAX-1 on the biological behavior of endothelial precursor cells showed that HAX-1 inhibited cell apoptosis and promoted proliferation of these cells by regulating the Akt signaling pathway and changing the interaction between Akt1 and HSP90.43 The important role of HAX-1 in cell protection might also be related to the inhibition of mitochondrial and endoplasmic reticulum apoptosis pathways.37 The HAX-1 also had certain effects on the nervous system. Studies have shown that HAX-1 had a neuroprotective effect against ischemic neuronal injury, and apoptosis after cerebral ischemia was induced by the downregulation of HAX-1 by NOX2-produced reactive oxygen species (ROS).17 In addition, the lack of expression of HAX-1 had been associated with severe congenital neutropenia and the delayed and abnormal neural development.44, 45, 46 The HAX-1 might partially contribute to the formation of Lewy bodies and be associated with the occurrence and development of Parkinson’s disease and dementia with Lewy bodies.47 The HAX-1 expression was upregulated after hippocampal injury or traumatic brain injury induced by seizures.30 In the current study, we successfully established HA1800 cell lines with high or low expression of HAX-1 to investigate the influence of HAX-1 on the proliferation and apoptosis of human astrocyte. The results showed that the cell activity and proliferation ability of HA1800 cells in the high-HAX-1 expression group were significantly weakened, and the cell cycle progress was decelerated, contrary to the results obtained from the low-HAX-1 expression group.
The Ki-67 is a nuclear protein that is also known as MKI-67, because it is encoded by the MKI-67 gene, which is related to ribosomal RNA transcription and can be used as a marker of cell proliferation. The expression level of Ki67 is positively correlated with the activity of cell proliferation.48 The C-Myc gene is one of the important members of the Myc proto-oncogene family. It is a translocation gene and an adjustable gene (regulated by various substances), which can promote cell division and make cells proliferate indefinitely and achieve immortalization. The level of C-Myc expression is closely related to the proliferation ability of cells.49 The BCL-2 gene family plays an important role in regulating cell apoptosis. The BCL-2 and BAX belong to the BCL-2 family and play, respectively, an antiapoptotic and a proapoptotic effect on cells. The BAX has an antagonistic effect on BCL-2, which may lead to the loss of the proapoptotic effect of the BCL-2 protein. The BCL-2/BAX ratio determines cell survival or death.50 The results of this study showed that, in the low-HAX-1 expression HA1800 group, the expressions of Ki67 and c-Myc were upregulated, and the BCL-2/BAX ratio was increased. Contrasting results were obtained from the high-HAX-1 expression HA1800 group. Combined with the results from the GEPIA, these findings suggested that HAX-1 expression was closely related to the proliferation and apoptosis of human astrocytes, and the mechanism may be related to the BCL-2/BAX signaling pathway. Therefore, the level of HAX-1 at different stages of the SCI may be increased or decreased according to the double-sided role of glial cells in the process. The promotion of the repair of SCI may be achieved by silencing HAX-1 in the acute phase (promoting the formation of glial scars) and strengthening HAX-1 in the recovery phase (inhibiting the formation of glial scars). However, the specific mechanism and the involved signal pathway of HAX-1 in different injury stages need further studies and discussion.
Limitations
In this study, the specific role of HAX-1 in the signaling pathway have not been clarified, which is a limitation of this study.
Conclusions
This study shows that the high expression of HAX-1 could inhibit the proliferation, reduce cell viability and promote cell apoptosis of human HA1800 cells. In addition, the expression of Ki67 and c-Myc, and the ratio of BCL-2/BAX decreased in the high-HAX-1 expression group but significantly increased in the low-HAX-1 expression group. These results indicated that HAX-1 may promote the apoptosis of human HA1800 cells through the BCL-2/BAX signaling pathway, thereby inhibiting the cell proliferation. Further study on the specific role of HAX-1 in the signaling pathway should be performed.